Preparation of sodium alginate-carboxymethyl cellulose-graphene oxide composite aerogel for adsorption of Pb(II) ion
-
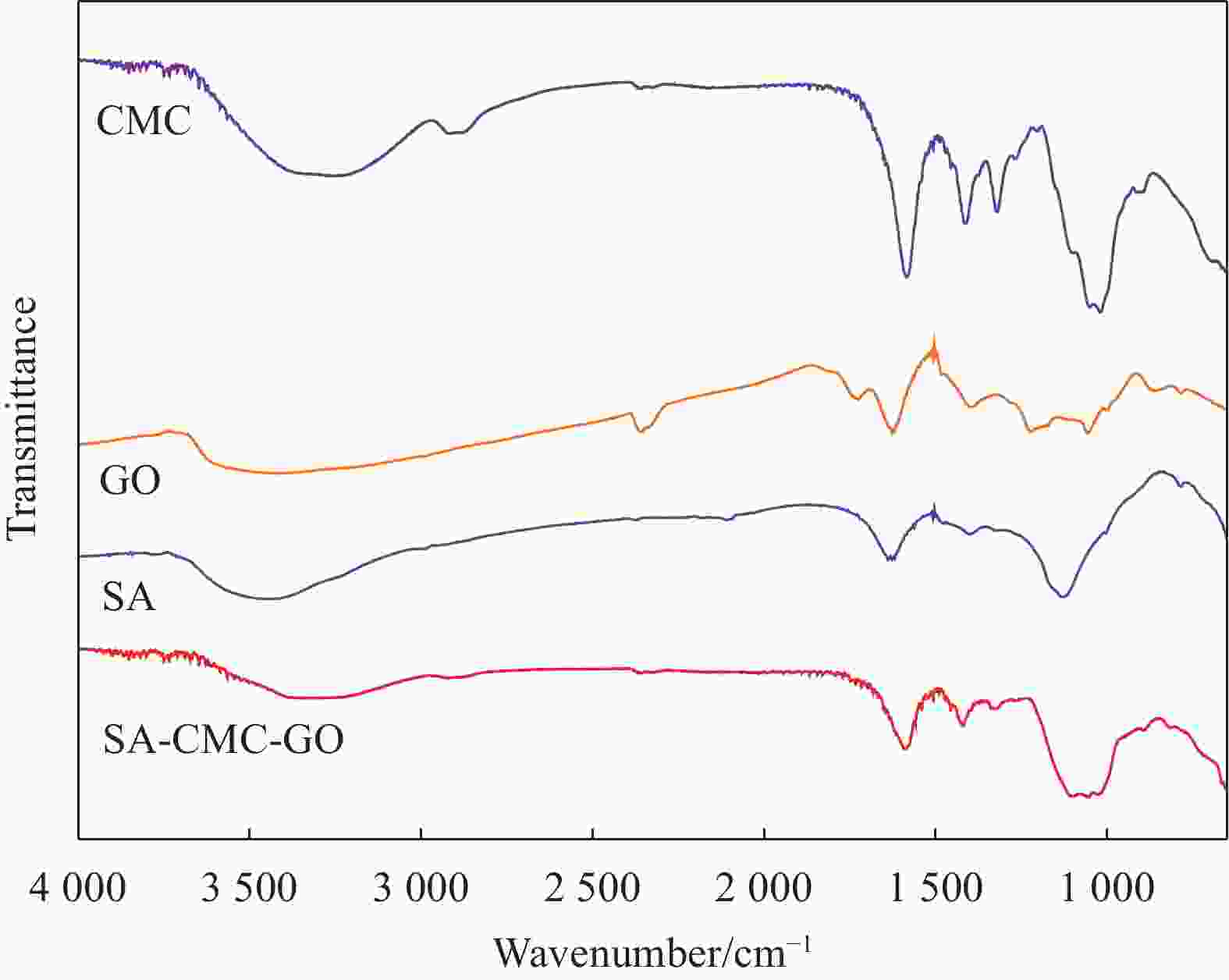
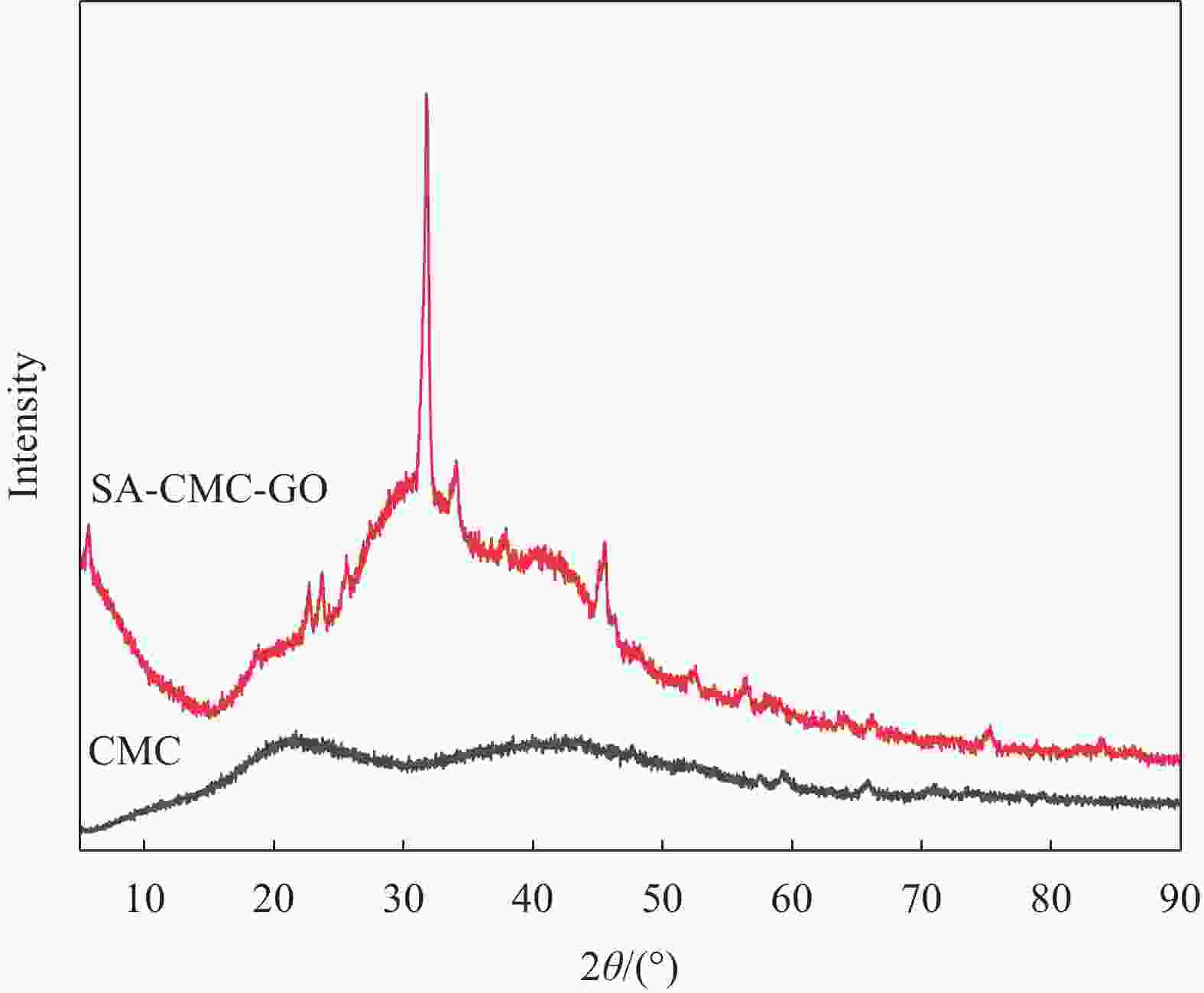
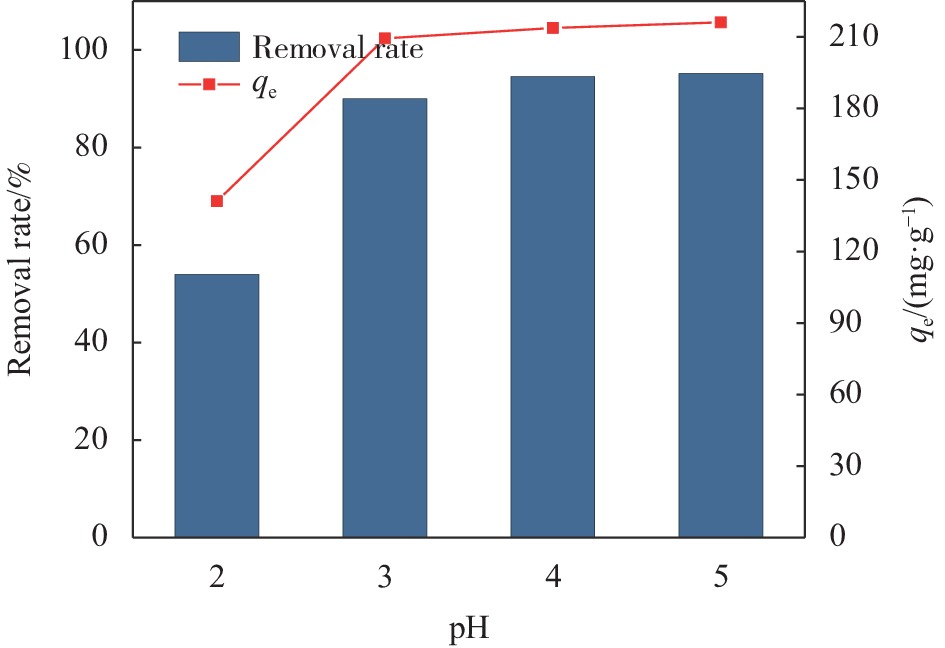
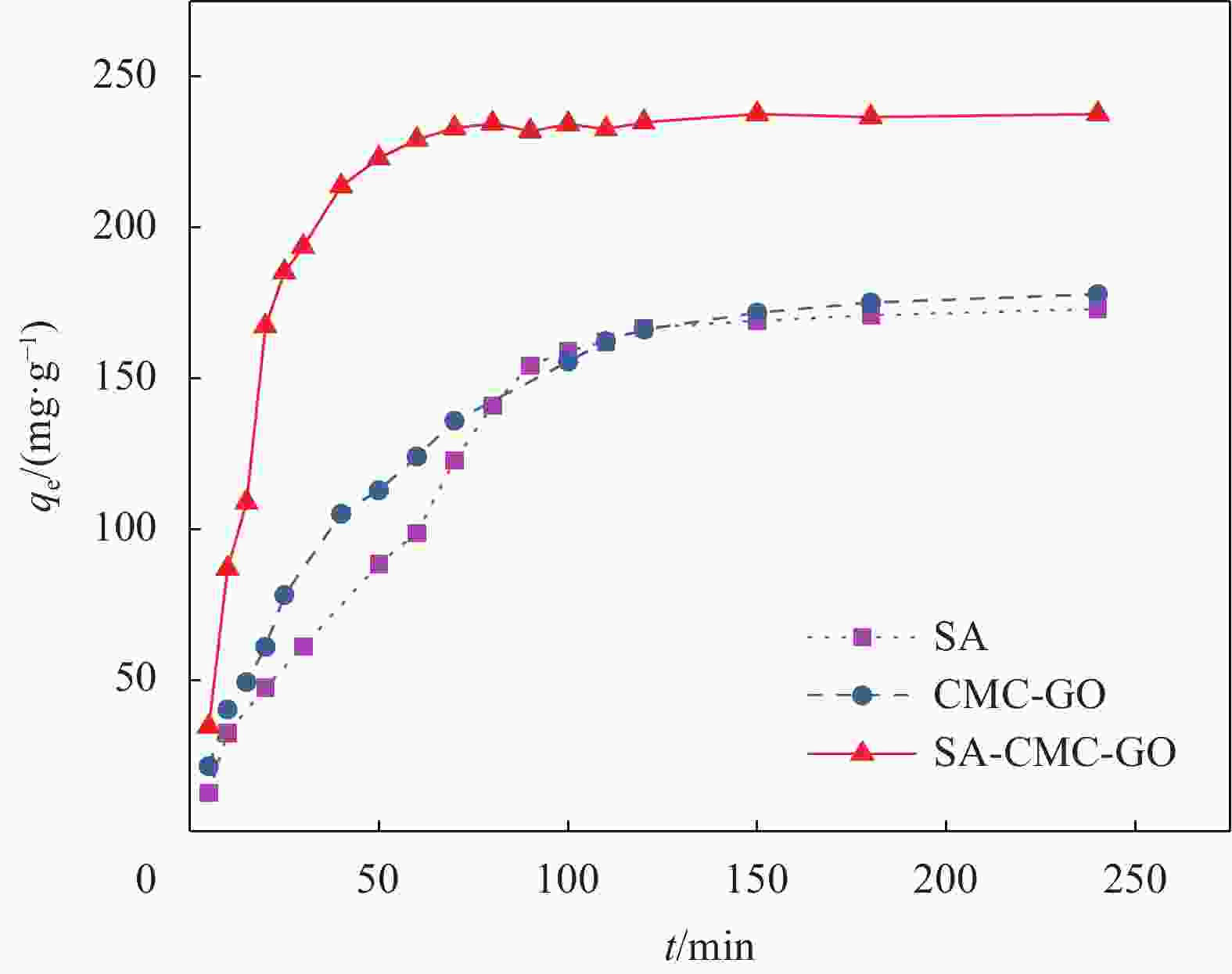
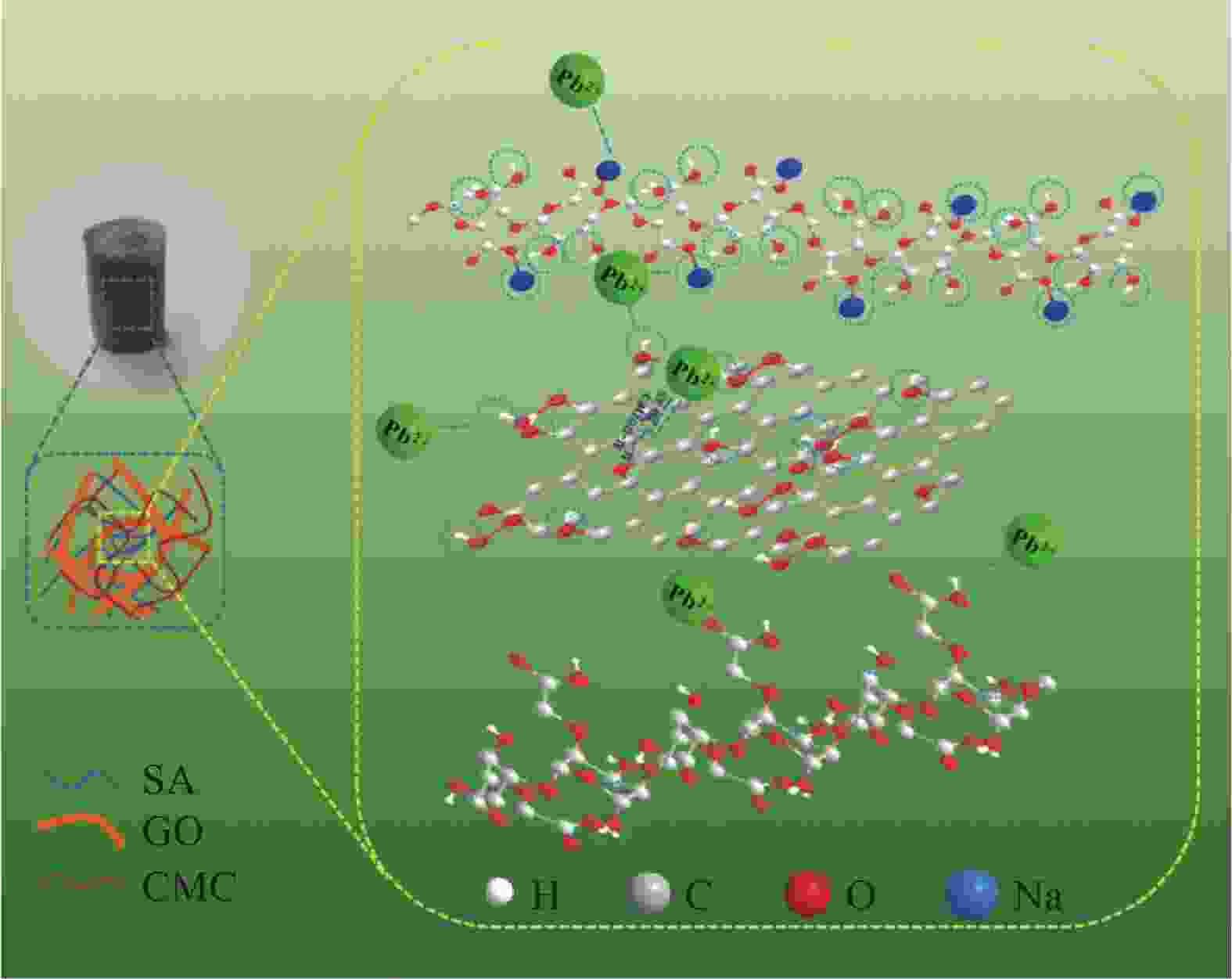
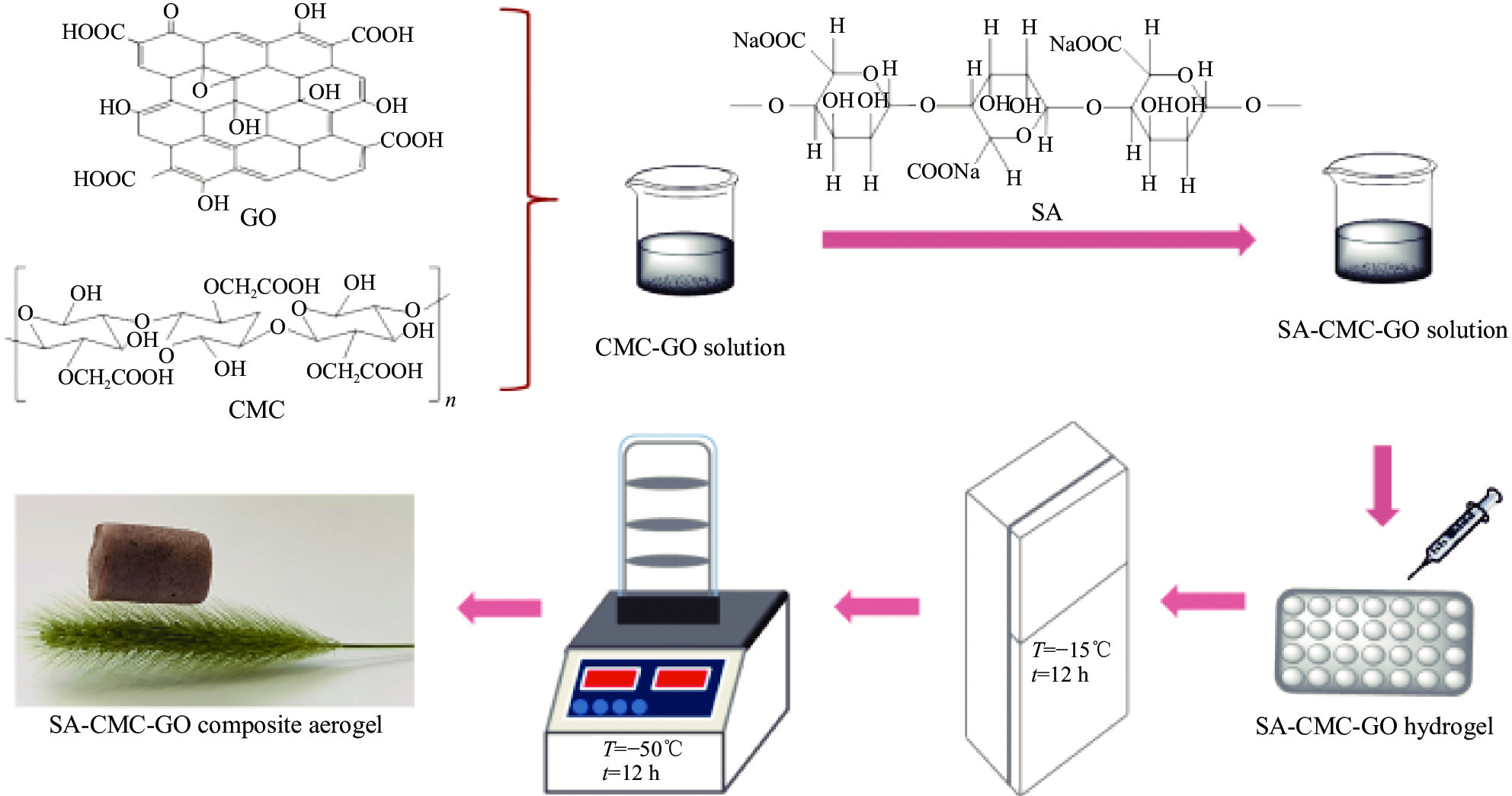
摘要: 目前,开发具有优异吸附性能、可持续使用和绿色环保的吸附剂仍然是水污染治理领域的焦点问题。因此,本文以海藻酸钠(SA)、羧甲基纤维素(CMC)和氧化石墨烯(GO)为原料,通过简单的溶胶-凝胶法结合冷冻干燥,构建了具有三维多孔网络结构的SA-CMC-GO复合气凝胶。利用SEM、FTIR、XRD等对SA-CMC-GO复合气凝胶的微观形貌、官能团结构等进行表征分析。以水中Pb2+为吸附对象,通过一系列间歇吸附实验,探究了各种因素(pH、介质温度、接触时间等)对吸附剂去除水体中Pb2+的影响。结果表明:在pH值2~5的范围内,复合气凝胶对Pb2+的吸附量随着pH的升高而升高;复合气凝胶对于Pb2+的吸附过程属于自发放热过程并遵循Langmuir吸附等温模型,其最大吸附量为272.5 mg·g−1;动力学研究表明,SA-CMC-GO复合气凝胶对Pb2+具有较快的吸附速率,可在60 min内达平衡并符合准二级动力学模型;此外,经过5次吸附-脱附试验,复合气凝胶仍对Pb2+保持较高的吸附性能。SA-CMC-GO复合气凝胶可以作为一种高效、快速的吸附剂用于从水体中去除Pb2+。Abstract: Exploiting adsorbents with excellent adsorption activity, good durability and environment friendly is still the core focus of water pollution treatment. Herein, in this study, sodium alginate (SA), carboxymethyl cellulose (CMC), and graphene oxide (GO) were used as raw materials to frame a SA-CMC-GO composite aerogel with a 3D network structure by a sol-gel and freeze-drying method. The functional group structure and microstructure of SA-CMC-GO composite aerogel were tested and analyzed by SEM, FTIR and XRD. Various parameters affecting the removal of Pb2+ such as pH, temperature and contact time were optimized by using a series of batch adsorption experiments. The results show that the adsorption amount of Pb2+ by the composite aerogel increases with the increase of pH=2-5. The adsorption process is a spontaneous exothermic process and the experimental data of the adsorption process are more fitted to Langmuir isotherm, the theoretical maximum adsorption capacity of Pb2+ on SA-CMC-GO composite aerogel is 272.5 mg·g−1. Adsorption kinetics studies indicate the adsorption of Pb2+ by the SA-CMC-GO composite aerogel shows rapid uptake rates and reaches equilibrium within 60 min. The pseudo-second-order kinetic model coincides with the adsorption behavior of the composite aerogel. Furthermore, the composite aerogel exhibited better reusability for five adsorption and desorption cycles with highly adsorption properties. The results imply that the new SA-CMC-GO composite aerogel could be potentially applied as an effective and rapid adsorbent for Pb2+ removal from aqueous solutions.
-
Key words:
- aerogel /
- sodium alginate /
- carboxymethyl cellulose /
- graphene oxide /
- adsorption
-
表 1 元素的原子分数
Table 1. Atomic fraction of the element
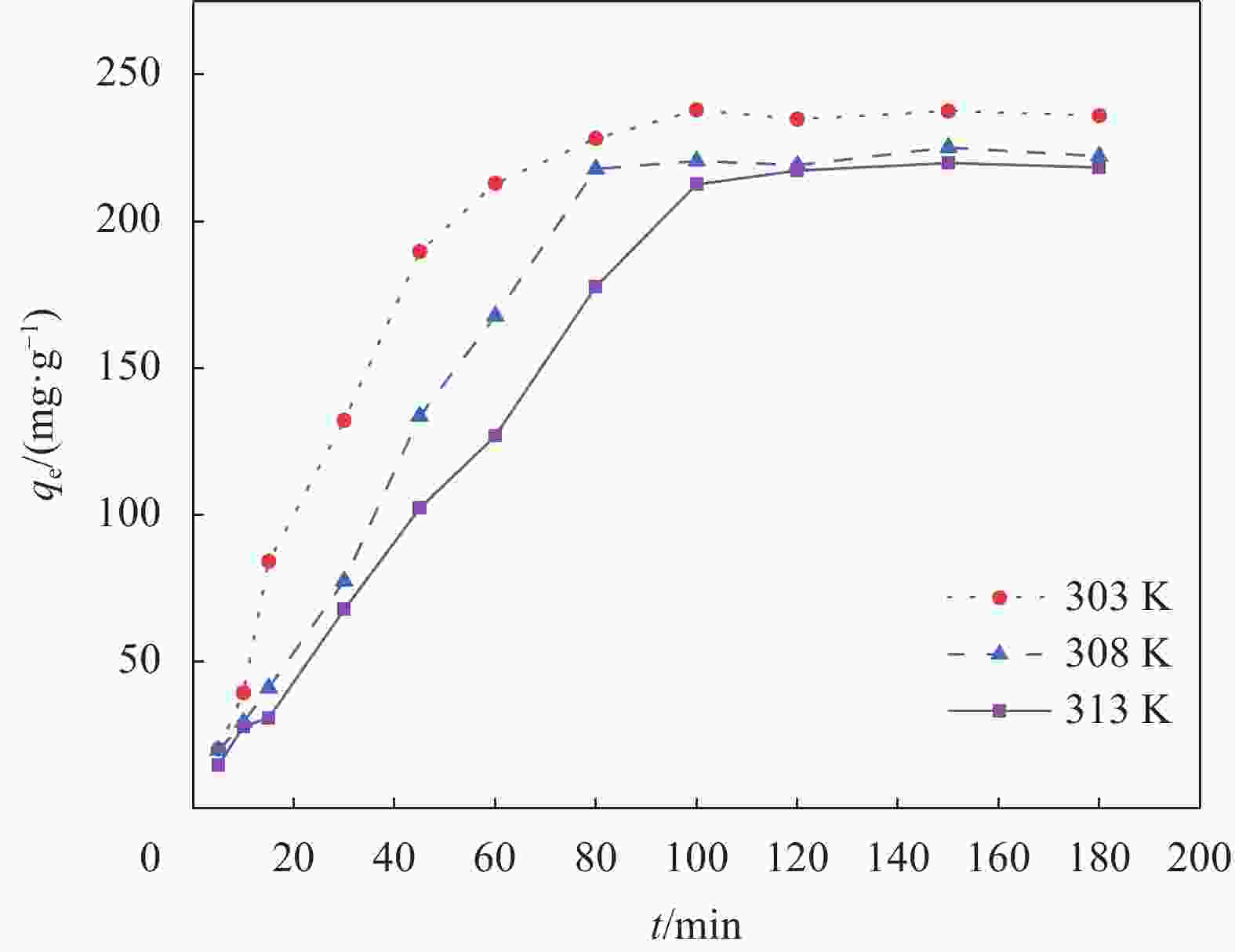
Atomic fraction/at% C O Na C—C C—O C=O C—O COO− — 65.55 10.63 4.88 19.23 1.83 1.87 表 2 不同吸附剂对Pb2+的平衡吸附时间
Table 2. Equilibrium adsorption time of Pb2+ by different adsorbents
Adsorbent Time/min Ref. DGO/CMC 550 [3] GO/CMC 600 [21] NSC 150 [25] Cell@PEI 240 [26] NPCS-PEI 120 [27] SA-CMC-GO 60 This study Notes: DGO—Functionalized graphene oxide; NSC—Nano-cellulose/sodium alginate/carboxymethyl chitosan aerogel; Cell@PEI—Amino-modified cellulose aerogel; NPCS-PEI—N-methylene phosphonic acid chitosan. 表 3 SA-CMC-GO复合气凝胶对Pb2+的吸附热力学相关参数
Table 3. Thermodynamically relevant parameters for the adsorption of Pb2+ by SA-CMC-GO composite aerogel
T/K ΔG/(kJ·mol−1) ΔS/(kJ·mol−1·K−1) ΔH/(kJ·mol−1) 303 −8.297
−0.08298
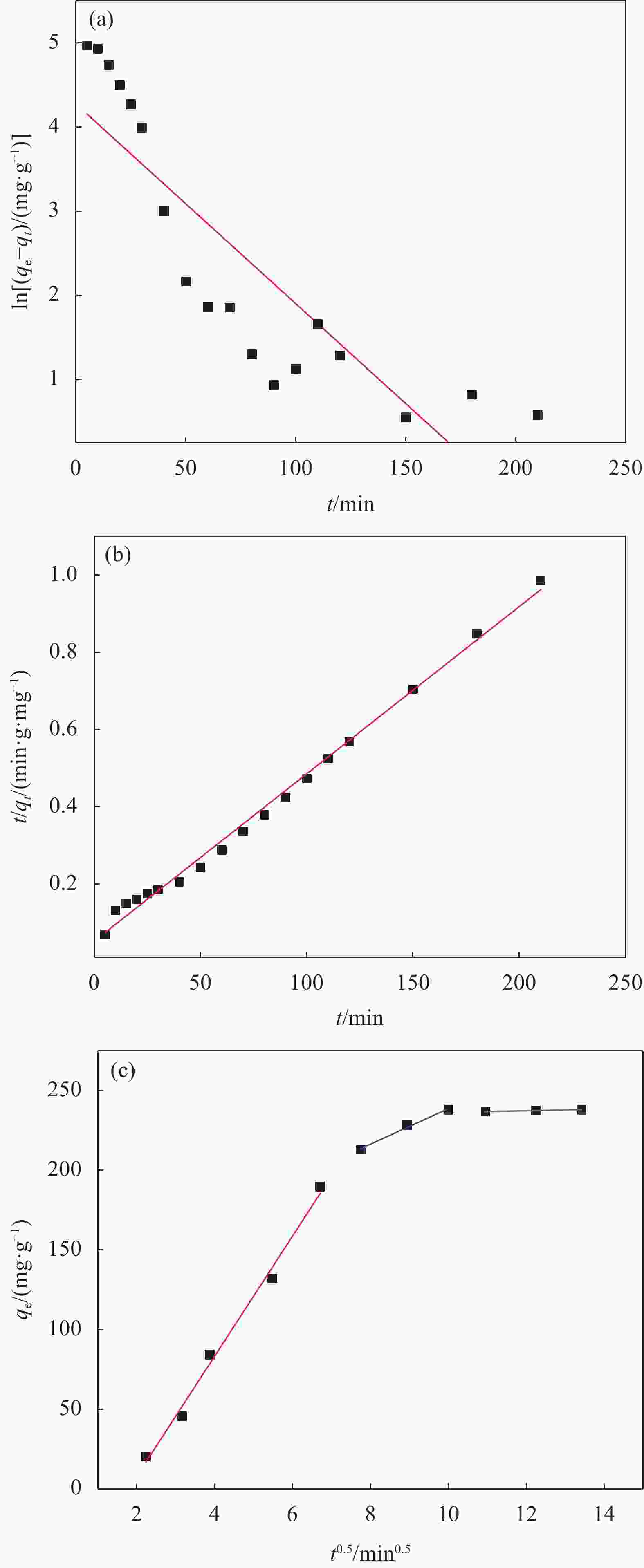
−33.44308 −7.882 313 −7.467 Notes: T—Temperature; ΔH—Enthalpy change; ΔS—Entropy change; ΔG—Gibbs free energy change. 表 4 SA-CMC-GO复合气凝胶对Pb2+的吸附动力学拟合参数
Table 4. Fitting parameters for the kinetics of Pb2+ adsorption by SA-CMC-GO composite aerogel
Pseudo-first-order kinetic model Pseudo-second-order kinetic model qe/(mg·g−1) k1/min−1 R2 qe/(mg·g−1) k2/(g·mg−1·min−1) R2 71.71 0.02374 0.7546 230.9 3.576×10−4 0.9942 Notes: R2—Linear correlation coefficient; k1—Pseudo-first-order kinetic constant; k2—Pseudo-second-order kinetic constant. 表 5 SA-CMC-GO复合气凝胶吸附Pb2+的粒子内扩散模型拟合参数
Table 5. Fitting parameters for the intra-particle diffusion model for Pb2+ adsorption by SA-CMC-GO composite aerogel
k1/
(mg·g−1·min0.5)R12 k2/
(mg·g−1·min0.5)R22 k3/
(mg·g−1·min0.5)R32 37.70 0.9920 11.13 0.9917 0.5009 0.9923 Note: ki—Intra-particle diffusion rate constant, i=1, 2, 3. 表 6 SA-CMC-GO复合气凝胶吸附Pb2+的等温线吸附Langmuir模型和Freundlich模型参数
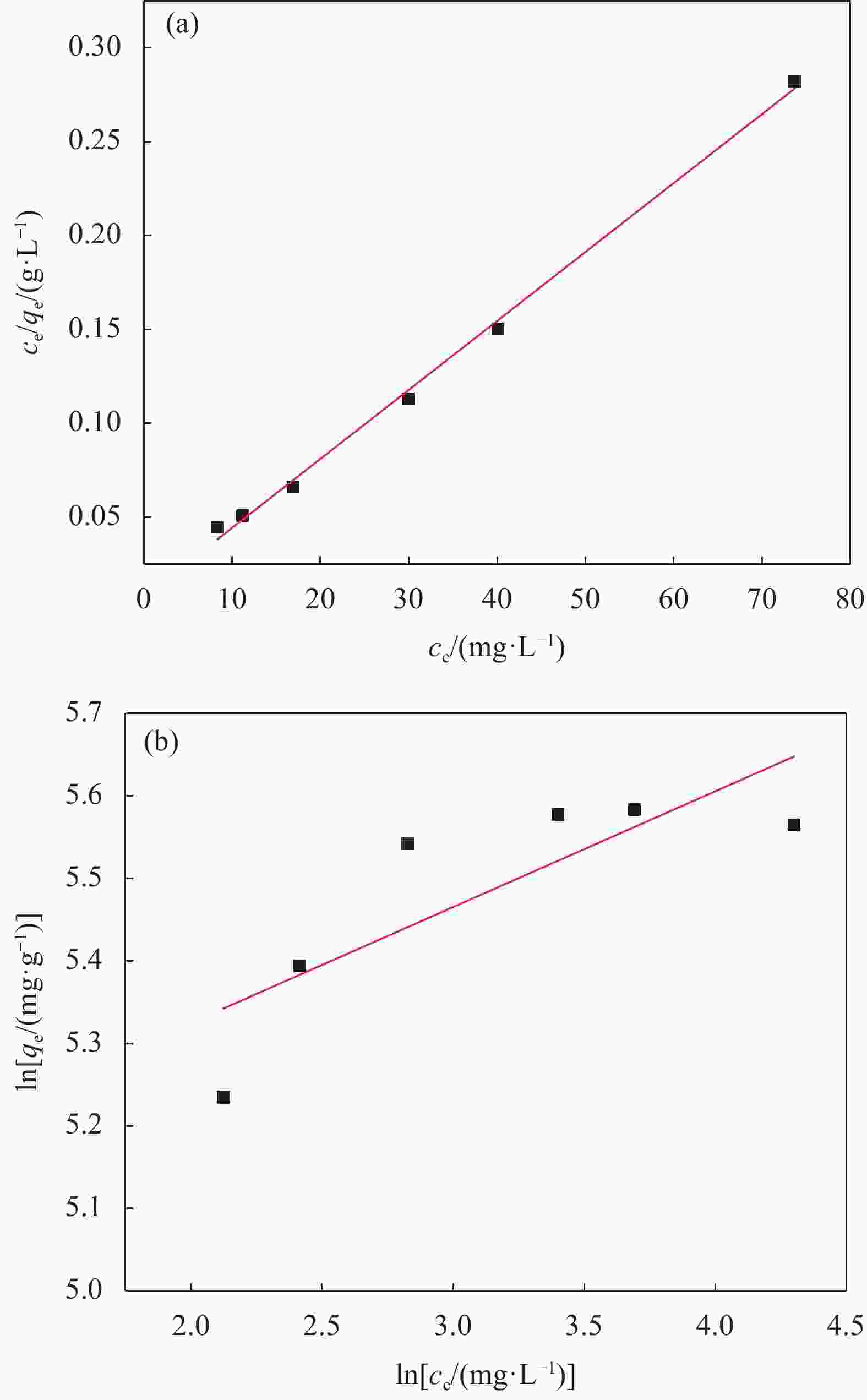
Table 6. Isothermal adsorption parameters of SA-CMC-GO composite aerogel for Pb2+ adsorption by Langmuir model and Freundlich model
Langmuir model Freundlich model qe/
(mg·g−1)KL R2 KF n R2 272.5 0.4809 0.9974 155.1 7.125 0.6719 Notes: KL—Langmuir adsorption coefficient; KF—Freundlich adsorption coefficient; n—Adsorption strength constant. -
[1] YANG W X, HAN Y, LI C H, et al. Shapeable three-dimensional CMC aerogels decorated with Ni/Co-MOF for rapid and highly efficient tetracycline hydrochloride removal[J]. Chemical Engineering Journal,2019,375:122076. doi: 10.1016/j.cej.2019.122076 [2] ZHANG S Y, HAN X S, CAI H Z, et al. Aramid nanofibers/WS2 nanosheets co-assembled aerogels for efficient and stable Pb (II) adsorption in harsh environments[J]. Chemical Engineering Journal,2022,450:138268. doi: 10.1016/j.cej.2022.138268 [3] LUO J Q, FAN C J, ZHOU X D. Functionalized graphene oxide/carboxymethyl chitosan composite aerogels with strong compressive strength for water purification[J]. Journal of Applied Polymer Science,2020,138(12):50065-50079. [4] 李继丰, 闫文静, 方婷, 等. C6位羧基纤维素制备及其对Cu2+吸附性能[J]. 复合材料学报, 2022, 39(3):1280-1290.LI Jifeng, YAN Wenjing, FANG Ting, et al. Preparation of C6 carboxylic cellulose and adsorption for Cu2+[J]. Acta Materiae Compositae Sinica,2022,39(3):1280-1290(in Chinese). [5] LIU Q, LI S S, YU H H, et al. Covalently crosslinked zirconium-based metal-organic framework aerogel monolith with ultralow-density and highly efficient Pb(II) removal[J]. Journal of Colloid and Interface Science,2020,561:211-219. doi: 10.1016/j.jcis.2019.11.074 [6] 徐晓燕, 张鹏, 朱静, 等. 水环境中天然有机物对纳米颗粒吸附铅和镉的不同作用[J]. 环境化学, 2021, 40(2):571-582. doi: 10.7524/j.issn.0254-6108.2020052501XU Xiaoyan, ZHANG Peng, ZHU Jing, et al. The varying roles of natural organic matters on nanoparticles adsorbing Cd2+ and Pb2+ in water environment[J]. Environmental Chemistry,2021,40(2):571-582(in Chinese). doi: 10.7524/j.issn.0254-6108.2020052501 [7] 牛乙涛, 包国庆, 吴纯鑫, 等. 功能化纳米复合材料Fe3O4@SiO2-3-氨丙基三甲氧基硅烷的制备及其对Pb(II)的吸附[J]. 复合材料学报, 2023, 40(6): 3350-3365.NIU Yitao, BAO Guoqing, WU Chunxin, et al. Preparation of functionalized nanocomposites Fe3O4@SiO2-3-aminopropyltrimethoxysilane and its adsorption to Pb(II)[J]. Acta Materiae Compositae Sinica, 2023, 40(6): 3350-3365(in Chinese). [8] JIAO G J, MA J L, ZHANG J Q, et al. High-efficiency capture and removal of phosphate from wastewater by 3D hierarchical functional biomass-derived carbon aerogel[J]. Science of the Total Environment,2022,827:154343. doi: 10.1016/j.scitotenv.2022.154343 [9] MO L T, TAN Y, SHEN Y L, et al. Highly compressible nanocellulose aerogels with a cellular structure for high-performance adsorption of Cu(II)[J]. Chemosphere,2022,291:132887. doi: 10.1016/j.chemosphere.2021.132887 [10] LEI C Y, WEN F B, CHEN J M, et al. Mussel-inspired synthesis of magnetic carboxymethyl chitosan aerogel for removal cationic and anionic dyes from aqueous solution[J]. Polymer,2021,213:123316. doi: 10.1016/j.polymer.2020.123316 [11] GE X S, SHAN Y N, WU L, et al. High-strength and morphology-controlled aerogel based on carboxymethyl cellulose and graphene oxide[J]. Carbohydrate Polymers,2018,197:277-283. doi: 10.1016/j.carbpol.2018.06.014 [12] QIANG X H, GUO X, SU H X, et al. In situ nanoarchitectonics of magnesium hydroxide particles for property regulation of carboxymethyl cellulose/poly(vinyl alcohol) aerogels[J]. RSC Advances,2021,11(56):35197-35204. doi: 10.1039/D1RA06556D [13] 翟红侠, 赵越, 李超凡, 等. 氨基改性SiO2气凝胶去除Cu(II)的性能与机制[J]. 复合材料学报, 2023, 40(8): 3981-3992.ZHAI Hongxia, ZHAO Yue, LI Chaofan, et al. Performance and mechanism of the amine-modified silica aerogel for the removal of Cu(II)[J]. Acta Materiae Compositae Sinica, 2023, 40(8): 3981-3992(in Chinese). [14] 张春梅, 杨婷婷, 陆桂花, 等. 纳米纤维素/壳聚糖气凝胶对六价铬的吸附性能[J]. 功能材料, 2022, 53(10):10180-10184.ZHANG Chunmei, YANG Tingting, LU Guihua, et al. Adsorption properties of cellulose nanocrystalline/chitosan aerogels for hexavalent chromium[J]. Journal of Functional Materials,2022,53(10):10180-10184(in Chinese). [15] HAN X H, LIANG J C, FUKUDA S, et al. Sodium alginate-silica composite aerogels from rice husk ash for efficient absorption of organic pollutants[J]. Biomass and Bioenergy,2022,159:106424. doi: 10.1016/j.biombioe.2022.106424 [16] DONG K Q, XU K J, WEI N S, et al. Three-dimensional porous sodium alginate/gellan gum environmentally friendly aerogel: Preparation, characterization, adsorption, and kinetics studies[J]. Chemical Engineering Research and Design,2022,179:227-236. doi: 10.1016/j.cherd.2022.01.027 [17] GAO C, WANG X L, AN Q D, et al. Synergistic preparation of modified alginate aerogel with melamine/chitosan for efficiently selective adsorption of lead ions[J]. Carbohydrate Polymers,2021,256:117564. doi: 10.1016/j.carbpol.2020.117564 [18] KONG Y, ZHUANG Y, HAN K, et al. Enhanced tetracycline adsorption using alginate-graphene-ZIF67 aerogel[J]. Colloids and Surfaces A,2020,588:124360. doi: 10.1016/j.colsurfa.2019.124360 [19] CHEN P, XIE F W, TANG F Z, et al. Glycerol plasticisation of chitosan/carboxymethyl cellulose composites: Role of interactions in determining structure and properties[J]. International Journal of Biological Macromolecules,2020,163:683-693. doi: 10.1016/j.ijbiomac.2020.07.004 [20] ELTAWEIL A S, ELGARHY G S, EL-SUBRUITI G M, et al. Carboxymethyl cellulose/carboxylated graphene oxide composite microbeads for efficient adsorption of cationic methylene blue dye[J]. International Journal of Biological Macromolecules,2020,154:307-318. doi: 10.1016/j.ijbiomac.2020.03.122 [21] LUO J Q, FAN C J, XIAO Z, et al. Novel graphene oxide/carboxymethyl chitosan aerogels via vacuum-assisted self-assembly for heavy metal adsorption capacity[J]. Colloids and Surfaces A,2019,578:123584. doi: 10.1016/j.colsurfa.2019.123584 [22] LI J J, TAN S C, XU Z Y. Anisotropic nanocellulose aerogel loaded with modified UiO-66 as efficient adsorbent for heavy metal ions removal[J]. Nanomaterials,2020,10(6):1114. doi: 10.3390/nano10061114 [23] XU W L, CHEN S, ZHU Y N, et al. Preparation of hyperelastic graphene/carboxymethyl cellulose composite aerogels by ambient pressure drying and its adsorption applications[J]. Journal of Materials Science,2020,55(24):10543-10557. doi: 10.1007/s10853-020-04720-5 [24] LIU P, CHEN M G, XIONG C G, et al. Flexible and highly sensitive graphene/carboxymethyl cellulose films for bending sensing[J]. Journal of Materials Science: Materials in Electronics,2020,31(17):14118-14127. doi: 10.1007/s10854-020-03966-8 [25] LI W Q, ZHANG L P, HU D, et al. A mesoporous nanocellulose/sodium alginate/carboxymethyl-chitosan gel beads for efficient adsorption of Cu2+ and Pb2+[J]. International Journal of Biological Macromolecules,2021,187:922-930. doi: 10.1016/j.ijbiomac.2021.07.181 [26] 李琦琪, 杨桂芳, 刘以凡, 等. 氨基改性纤维素气凝胶吸附Pb2+的研究[J]. 纤维素科学与技术, 2022, 30(1):34-46.LI Qiqi, YANG Guifang, LIU Yifan, et al. Adsorption behavior of Pb2+ on amino-modified cellulose aerogel[J]. Journal of Cellulose Science and Technology,2022,30(1):34-46(in Chinese). [27] LIU T, GOU S H, HE Y, et al. N-methylene phosphonic chitosan aerogels for efficient capture of Cu2+ and Pb2+ from aqueous environment[J]. Carbohydrate Polymers,2021,269:118355. doi: 10.1016/j.carbpol.2021.118355 [28] 张宏伟, 谢鸿, 肖欣荣, 等. 不同氧化程度氧化石墨烯/聚乙烯醇气凝胶对亚甲基蓝的吸附[J]. 复合材料学报, 2021, 38(9):2788-2795. doi: 10.13801/j.cnki.fhclxb.20201203.002ZHANG Hongwei, XIE Hong, XIAO Xinrong, et al. Adsorption of methylene blue by graphene oxide/polyvinyl alcohol aerogels with different oxidation degrees[J]. Acta Materiae Compositae Sinica,2021,38(9):2788-2795(in Chinese). doi: 10.13801/j.cnki.fhclxb.20201203.002 [29] YANG P, YANG L, WANG Y, et al. An indole-based aerogel for enhanced removal of heavy metals from water via the synergistic effects of complexation and cation-π interactions[J]. Journal of Materials Chemistry A,2019,7(2):531-539. doi: 10.1039/C8TA07326K [30] WANG Z G, SONG L, WANG Y Q, et al. Lightweight UiO-66/cellulose aerogels constructed through self-crosslinking strategy for adsorption applications[J]. Chemical Engineering Journal,2019,371:138-144. doi: 10.1016/j.cej.2019.04.022 [31] HOSSEINI H, ZIRAKJOU A, MCCLEMENTS D J, et al. Removal of methylene blue from wastewater using ternary nanocomposite aerogel systems: Carboxymethyl cellulose grafted by polyacrylic acid and decorated with graphene oxide[J]. Journal of Hazardous Materials,2022,421:126752. doi: 10.1016/j.jhazmat.2021.126752 [32] ZHOU Y Q, GAO Y, WANG H L, et al. Versatile 3D reduced graphene oxide/poly(amino-phosphonic acid) aerogel derived from waste acrylic fibers as an efficient adsorbent for water purification[J]. Science of the Total Environment,2021,776:145973. doi: 10.1016/j.scitotenv.2021.145973 [33] XIANG C, WANG C, GUO R H, et al. Synthesis of carboxymethyl cellulose-reduced graphene oxide aerogel for efficient removal of organic liquids and dyes[J]. Journal of Materials Science,2019,54(2):1872-1883. doi: 10.1007/s10853-018-2900-5 -






 下载:
下载: