Preparation and photocatalytic performance of polyvinyl alcohol /oxygen-doped carbon nitride composite nanofiber films
-
摘要:
大量有机污染物对水循环造成严重污染,同时耐药细菌对人类健康构成严重威胁。光催化纳米材料已成为抗菌领域的研究热点。本文通过尿素和甲酸热聚合法烧制成氧掺杂氮化碳(O-CN),优选比例的O-CN与聚乙烯醇(PVA)溶液混合,通过静电纺丝技术成功制备了PVA/O-CN复合纳米纤维膜。对O-CN和PVA/O-CN复合膜进行形貌和结构表征以及光催化抑菌和有机污染物去除性能的分析。结果表明,O原子部分代替CN中三嗪环结构中N的位置,O-CN的可见光吸收能力和电子-空穴对分离率较CN有所提高, PVA/O-CN-0.6复合纳米纤维膜对大肠杆菌和金黄色葡萄球菌有较好的抑制作用,抑菌率可达96%和93.7%。另外,PVA/O-CN-0.6复合纳米纤维膜对染料具有良好的去除性能,PVA/O-CN-0.6在4 h内对亚甲基蓝(MB)的去除率达到了97.7%。此外,该膜具有良好的热稳定性和优异的力学性能,在水净化及抑菌领域具有很大的应用潜力。
Abstract:A large number of organic pollutants caused serious pollution to the water cycle, and drug-resistant bacteria posed a serious threat to human health. Photocatalytic nanomaterials have become a research hotspot in the field of antibacteria. In this paper, oxygen-doped carbon nitride (O-CN) was prepared by hot polymerization with urea and formic acid. The optimal proportion of O-CN was mixed with polyvinyl alcohol (PVA) solution, and the PVA/O-CN composite nanofiber film was successfully prepared by electrospinning technology. The micro-morphology, structure, photocatalytic bacteriostatic properties and organic pollutant removal properties of O-CN and PVA/O-CN composite films were studied. The results show that O atom can replace the N position in the triazine ring structure of CN, and the light absorption capacity and the separation rate of electron-hole improve compared with that of CN. PVA/O-CN-0.6 composite nanofiber membrane has the best inhibition effect on E. coli and S. aureus, and the inhibition rates are 96% and 93.7%, respectively. In addition, PVA/O-CN-0.6 composite nanofiber membrane has a good performance in dye removal, and the removal rate of methylene blue (MB) by PVA/O-CN-0.6 reaches 97.7% within 4 h. Moreover, the film with excellent mechanical properties, has great application potential in many fields.
-
Janus材料因其独特的结构或组成的不对称性展现出很多特殊的功能,近年来为人们所广泛关注[1-4]。目前,Janus材料在乳化剂[5]、自驱动马达[6]、药物释放[7]、催化[8]和柔性可穿戴[9]等众多领域都展现出巨大的应用前景。自1991年被诺贝尔奖获得者De Gennes[1]首次提出以来,经过30多年的发展,已经有多种不同组成或形貌结构的Janus材料被合成出来,从组成来分类,主要有聚合物/聚合物型、无机物/无机物型及聚合物/无机物型;从形貌来区分,包括颗粒状[10]、棒状[11]、片状[12]、锥状[13]及雪人状[14]等。
不同于传统Janus材料,Janus中空球在空间结构上中心对称,其非对称性体现在内外表面的化学组成和性质上,这种独特的结构使其可以用作特殊微容器,用于分离富集、传输和受限反应等[15-19]。但是,研究发现,完整致密的壳层会限制微球和环境之间的物质传输[15]。因此,为了提高Janus微球和环境之间的物质传输速率,引入了贯穿壳层的孔道,即Janus笼。目前,已经有多种功能Janus笼被制备出来,作为高效传输的微反应容器,在水处理、催化,环境响应富集释放等方向展现出一定的应用前景[20-23]。
课题组在之前的工作中,最早提出了乳液界面溶胶凝胶法制备二氧化硅Janus中空球的方法[24]。进而在此基础上通过两种表面活性剂在乳液界面发生微相分离在中空球表面形成通孔,同时引入磁性纳米粒子并在中空球内腔接枝温敏性聚合物,得到热敏性的聚合物-无机物复合功能Janus笼[22]。但是,在之前的研究中也发现,Janus笼的表面孔径很难调控,孔径也只能限制在50 nm以下,如果想要通过增加致孔表面活性剂的量得到更大孔径的笼,不仅孔径变化不大,而且还会导致壳层局部呈不连续结构,在表面形成二氧化硅小颗粒,进而破坏球形结构,无法得到完整的中空球。
基于这些考虑,在此提出了新的研究思路,先用烷基和末端带卤素基团的硅烷偶联剂代替带活性双键的硅烷偶联剂,壳层依旧引入磁性纳米颗粒,先制备出支撑性良好、孔径大范围可调的无机Janus笼,进而利用可控自由基聚合代替自由基聚合,在更温和的反应条件下在内腔接枝pH响应性聚合物,得到支撑性好、孔径大范围可调的功能有机/无机功能复合Janus笼,并验证了其可以在改变环境pH的条件下实现对油相的可控吸收和释放,以及在磁操控下实现装载物的定向传输,有望用于药物装载和体内靶向释放等领域。
1. 实验材料及方法
1.1 原材料
无水三氯化铁(FeCl3,分析纯)、油酸钠(分析纯)、油酸(分析纯)、吐温80(Tween 80,化学纯)、十二烷基硫酸钠(SDS,化学纯)、铜粉(分析纯)和52#石蜡(熔点50~52℃)购自国药集团化学试剂有限公司;3-氨丙基三乙氧基硅烷(APTES,98%)、香豆素-6 (98%)、荧光素5-异硫氰酸酯(FITC,95%)、三乙胺(99%)、正辛基三乙氧基硅烷(98%)和2-溴-2-甲基丙酰溴(98%)购自北京百灵威科技有限公司;正硅酸乙酯(分析纯)、甲醇(分析纯)、正癸烷(分析纯)和甲苯(分析纯)购自西陇科学股份有限公司;1, 1-双十八烷基-3, 3, 3, 3-四甲基吲哚羰花青高氯酸盐(dil-C18,98%)和甲基丙烯酸二乙氨基乙酯(DEAEMA,>98.5%)购自西格玛奥德里奇(上海)贸易有限公司(Sigma-Aldrich);三[2-(二甲氨基)乙基]胺(Me6TREN,97%)购自梯希爱(上海)化成工业发展有限公司(TCI);预水解的苯乙烯马来酸酐共聚物水溶液(HSMA,10wt%)为自制。
1.2 实验方法
1.2.1 样品制备
(1) 磁性无机Janus笼的制备。取0.3 g合成的油溶性磁性Fe3O4颗粒[25]、1.1 g APTES、0.88 g正辛基三乙氧基硅烷和5.2 g正硅酸乙酯加入到25 g正癸烷中混合均匀作为油相;将15 mL 10wt%的HSMA的水溶液加入75 mL水中,用2 mol/L HCl调pH为3~4。然后再加入0.03 g Tween 80,混合均匀,作为水相。混合水油两相,用高速剪切机(Fluko FA25,德国Fluko)在12000 r/min的速度下剪切乳化3 min。将剪切所得乳液转移至三口瓶中,70℃反应12 h,得到磁性无机Janus笼。
(2) 末端Br基硅烷偶联剂的合成。将4.31 g APTES和2.55 g三乙胺(TEA)加入到20 mL甲苯中,通氮气30 min。再将5.95 g溴代异丁酰溴(BiBB)溶解于10 mL甲苯中,加入到恒压漏斗中。在冰浴条件下,缓慢的将溴代异丁酰溴甲苯溶液缓慢滴加到体系中,磁力搅拌过夜后,离心除去下层白色沉淀后,旋蒸除去甲苯,即得到末端为溴基的硅烷偶联剂。
(3) pH响应性磁性复合Janus笼的制备。将10 mg 铜粉和50 mg上述步骤(1)合成的磁性无机Janus笼分散到36 mL水中,通氮气30 min。将0.2 g DEAEMA和30 μL配体Me6TREN分散到18 mL甲醇中,通氮气30 min后和水相混合,在50℃水浴中反应12 h。磁分离,用乙醇洗涤3次,真空干燥,得到内表面接枝有PDEAEMA的磁性聚合物/无机磁性复合Janus笼。
1.2.2 复合Janus笼pH响应性吸释实验
(1) 复合Janus笼对油相的pH响应性吸释和磁分离。在0.2 g甲苯中加入少量油溶性荧光染料dil-C18染色,在样品瓶中将10 mg磁性复合Janus笼分散在5 g水中,再加入染色甲苯,调节pH至5,此时水油两相明显分层,再调节pH至9,振荡吸附,甲苯层消失,用磁铁吸附,吸附有甲苯的复合Janus笼即在磁铁一侧的瓶壁处富集,将pH调回5,振荡静置后用磁铁吸附,甲苯层重新出现;同样在0.2 g甲苯中加入少量油溶性荧光染料dil-C18染色,在样品瓶中将10 mg磁性复合Janus笼分散在5 g水中,再加入染色甲苯,调节pH至5,此时水油两相明显分层,保持pH值不变,振荡后用磁铁吸附,甲苯无法被复合Janus笼吸附。
(2) 复合Janus笼在荧光显微镜下的pH响应性吸释:将50 mg磁性复合Janus笼分散在10 g水中,加入水溶性荧光燃料FITC进行染色标记。将0.5 g加入少量香豆素-6荧光染料染色的甲苯加入到溶解有10 mg表面活性剂SDS的10 g水中,超声乳化得到水包油乳液。将FITC标记的复合Janus笼水分散液加入到乳液中,调节pH至9左右,避光搅拌,分别在磁分离复合Janus笼前后在荧光显微镜(Olympus IX83,日本Olympus)下观察;再调节pH至5左右,避光搅拌,同样分别在磁分离复合Janus笼前后在荧光显微镜下观察。
2. 结果与讨论
2.1 磁性无机Janus笼的制备、孔径调节与表征
在之前制备复合Janus笼时,课题组是将硅烷前驱体和聚合物单体同时溶解在油相中,一步得到有机/无机复合Janus多孔球[22]。但是多孔球的孔径只能在50 nm左右且难以调节,对应的致孔表面活性剂Tween 80在水相的浓度在0.22wt%以下,当Tween 80浓度继续升高时,会使多孔球外表面生成SiO2小颗粒,进而破坏球形结构。
本文用正辛基三乙氧基硅烷代替含活性双键的硅烷偶联剂作为球壳内侧接枝亲油基团的硅前驱体,同时将制备的油溶性Fe3O4纳米颗粒分散在油相中,得到外侧为氨基,内侧为烷基的磁性无机Janus笼。样品分别在扫描电子显微镜(SEM,带有元素分析 EDX的Quanta FEG 250,美国 FEI)和透射电子显微镜(TEM,JEM-1011,日本 JEOL)下进行观察,如图1所示。Janus笼的直径约为1~3 μm,孔径约为200~400 nm (图1(a)、图1(b)),同时不同于内侧接枝活性双键的无机Janus笼,即使没有聚合物的支撑,内侧为正辛基硅烷修饰的Janus笼也不会塌缩。进而对无机Janus笼进行超声粉碎,可以得到表面具有孔道的磁性Janus片(图1(c))。对得到的Janus片进行包埋切片,在TEM下观察,如图1(d)所示,可以看到磁性纳米粒子镶嵌在壳层中。
调节致孔表面活性剂Tween 80的加入量,即可对Janus笼球壳表面的通孔孔径进行调节。如图2所示,Tween 80在水相的浓度分别为0.01wt% (图2(a))、0.02wt% (图2(b))、0.04wt% (图2(c))和0.08wt% (图2(d))时,其表面孔径可从40 nm左右增加到约1 μm。
![]() 图 1 内侧修饰正辛基的磁性无机Janus笼的SEM图像 (a) 和TEM图像 (b);由磁性无机Janus笼破碎得到的Janus多孔片的SEM图像 (c) 和甲基丙烯酸甲酯(MMA)包埋切片后的TEM图像 (d)Figure 1. SEM image (a) and TEM image (b) of magnetic organic Janus cage with octyl group grafted onto interior surface; SEM image (c) of Janus porous nanosheets crushed from magnetic inorganic Janus cage and cross section TEM image (d) of slice of magnetic inorganic Janus cage after embedding in methyl methacrylate (MMA) and section
图 1 内侧修饰正辛基的磁性无机Janus笼的SEM图像 (a) 和TEM图像 (b);由磁性无机Janus笼破碎得到的Janus多孔片的SEM图像 (c) 和甲基丙烯酸甲酯(MMA)包埋切片后的TEM图像 (d)Figure 1. SEM image (a) and TEM image (b) of magnetic organic Janus cage with octyl group grafted onto interior surface; SEM image (c) of Janus porous nanosheets crushed from magnetic inorganic Janus cage and cross section TEM image (d) of slice of magnetic inorganic Janus cage after embedding in methyl methacrylate (MMA) and section在之前的工作中,即使Tween 80的含量提高近10倍,孔径的大小变化也不明显[22],相比之下,本文孔径对Tween 80的量变化更敏感。这是由于在之前工作中,由于无机/有机壳层是一锅法制备,因而在油相中加入了大量的聚合物单体,其和Tween 80相容性比烷烃溶剂要差,使Tween 80在界面的微相分区面积较小,即使浓度增加,对分区面积影响也不大,因而难以对孔径进行大范围调节,继续增加Tween 80的浓度只能破坏壳层。而本文先构建无机壳层,油相中只添加了硅前驱体,同时用烷基硅烷偶联剂代替带有活性双键的硅前驱体,使油相和Tween 80非极性部分的基团极性相近,使Tween 80微区可以在界面更加铺展,因而能够通过改变Tween 80的量来实现孔径的大范围调节。为了进一步验证,在不改变其他条件,仅仅不添加聚合物单体的情况下重复之前的制备过程,如图3所示,腔内不一步接枝聚合物时,制备的无机笼的孔径明显变大且孔径对Tween 80的含量更加敏感,但是失去聚合物的支撑,无机笼壳层本身支撑性差,呈塌缩状态。
![]() 图 3 (a) 水相中Tween 80添加量为0.22wt%制备的无机/有机复合Janus笼的SEM图像;(b) 水相中Tween 80添加量为0.04wt%制备的无机笼的SEM图像Figure 3. (a) SEM image of the polymer/inorganic composite Janus cages synthesized at 0.22wt% of Tween 80 in aqueous phase; (b) SEM image of inorganic Janus cages synthesized at 0.04wt% of Tween 80 without addition of monomer in aqueous phase
图 3 (a) 水相中Tween 80添加量为0.22wt%制备的无机/有机复合Janus笼的SEM图像;(b) 水相中Tween 80添加量为0.04wt%制备的无机笼的SEM图像Figure 3. (a) SEM image of the polymer/inorganic composite Janus cages synthesized at 0.22wt% of Tween 80 in aqueous phase; (b) SEM image of inorganic Janus cages synthesized at 0.04wt% of Tween 80 without addition of monomer in aqueous phase2.2 磁性pH响应性聚合物/无机复合Janus笼的制备
研究发现,用带烷基的硅前驱体替代带活性双键的硅前驱体能够得到支撑性良好的无机笼,但是单纯的烷基链修饰内侧使得无机笼没有活性位点去接枝功能性的聚合物,限制了Janus笼的应用。考虑到自由基聚合难以控制,本工作基于“铜媒介”可控自由基聚合法(CuCRP)代替自由基聚合,在笼内侧接枝了pH响应性聚合物[26]。
为了在无机Janus笼内表面接枝聚合物,先要在内侧修饰上卤素基团。通过氨丙基三乙氧基硅烷和溴代异丁酰溴在等物质的量的条件下进行反应,制备出末端为溴基的硅烷偶联剂。对产物进行核磁共振(1H NMR,Bruker Avance III 400 HD,德国 Bruker)表征,各个H原子峰已在图4(a)中标注,其中,化学位移在6.96×10−6的峰对应着酰胺上与N相连的H,且积分结果为1 ,证明末端为溴基的硅烷偶联剂成功合成。再以摩尔比1∶1的比例将合成的末端Br基硅烷偶联剂和正辛基三乙氧基硅烷一起加入油相作为亲油一侧的修饰基团,得到内表面接枝Br基的Janus笼。如图4(b)所示,对Janus笼进行EDX元素分析,证明Br的成功引入。
![]() 图 4 (a) 合成的末端带Br基的硅烷偶联剂的核磁共振图谱;(b) 内表面接枝Br基的磁性无机Janus笼的SEM图像及内嵌EDX图谱(方框为元素分析所选区域)Figure 4. (a) 1H NMR spectrum of synthesized silane end with Br group; (b) SEM image and inset EDX spectrum of magnetic inorganic Janus cage with Br group grafted onto the interior surface (Box is the selected area for element analysis)
图 4 (a) 合成的末端带Br基的硅烷偶联剂的核磁共振图谱;(b) 内表面接枝Br基的磁性无机Janus笼的SEM图像及内嵌EDX图谱(方框为元素分析所选区域)Figure 4. (a) 1H NMR spectrum of synthesized silane end with Br group; (b) SEM image and inset EDX spectrum of magnetic inorganic Janus cage with Br group grafted onto the interior surface (Box is the selected area for element analysis)成功在壳层内侧引入溴基以后,采用“铜媒介”可控自由基聚合法(CuCRP)制备pH响应性Janus笼[26],将磁性无机Janus笼分散在水相,pH响应性单体DEAEMA和配体分散在甲醇中,混溶后加入铜粉作为催化剂,壳层内侧的溴基引发聚合,接枝上pH响应性聚合物PDEAEMA,即得到pH响应的磁性聚合物/无机复合Janus笼。其反应体系温和简单,在水和甲醇混溶体系就可以聚合油溶性单体,更加清洁无污染,反应体系耐氧性高。在扫描电镜下观察,其形貌未有明显变化(图5(a))。将接枝功能聚合物的复合Janus笼用预聚物甲基丙烯酸甲酯(MMA)包埋,再进行切片并在TEM下观察,如图5(b)所示,发现Janus笼壳层内出现衬度较低的一层,说明PDEAEMA仅接枝在Janus笼的内表面(为了便于观察,包埋切片的Janus笼未复合磁性纳米颗粒)。将复合Janus笼超声破碎得到复合多孔片,发现内侧接枝聚合物之后变得粗糙(图5(c)),用HF刻蚀除去无机层,得到聚合物笼(图5(d))。
对复合Janus笼分别进行红外图谱(FTIR,Bruker EQUINOX 55,德国 Bruker)和热失重(TGA, Perkin-Elmer Pyris 1, 美 国 Perkin-Elmer)分析。其红外图谱如图6(a)所示,接枝聚合物后的Janus笼(曲线2)对比于无机磁性Janus笼(曲线1),在1725 cm−1出现新的特征峰,对应着聚合物PDEAEMA的酯基峰。此外,1050~1150 cm−1对应着二氧化硅Si—O—Si的非对称伸缩振动峰,1633 cm−1对应的为氨基的特征峰,595 cm−1对应着的Fe—O特征峰。通过TGA分析可测得复合Janus笼中聚合物的含量(图6(b))。图中曲线1为无机磁性Janus笼的热失重曲线,其残余63.1wt%,失重的原因主要为有机基团热解和硅前驱体进一步缩合造成。而曲线2为接枝聚合物后的复合Janus笼的曲线,其热解后残余36.8wt%,因而计算出复合Janus笼中聚合物质量含量为41.7wt%。
![]() 图 5 (a) 内表面接枝聚甲基丙烯酸二乙氨基乙酯(PDEAEMA)的磁性无机/有机复合Janus笼的SEM图像;(b) 未复合磁性纳米颗粒的内表面接枝PDEAEMA无机/有机复合Janus笼切片的TEM图像;(c) 复合Janus笼破碎后的多孔片的SEM图像;(d) 用HF酸刻蚀除去无机壳层后的有机笼的SEM图像Figure 5. (a) SEM image of magnetic composite Janus cage with polydiethylaminoethyl methacrylate (PDEAEMA) grafted onto interior surface; (b) Cross-section TEM image of composite Janus cage with PDEAEMA grafted onto interior surface (without Fe3O4 nanoparticles); (c) SEM image of Janus porous nanosheets crushed from composite Janus cage; (d) SEM image of organic cage obtained by etching inorganic shell with HF
图 5 (a) 内表面接枝聚甲基丙烯酸二乙氨基乙酯(PDEAEMA)的磁性无机/有机复合Janus笼的SEM图像;(b) 未复合磁性纳米颗粒的内表面接枝PDEAEMA无机/有机复合Janus笼切片的TEM图像;(c) 复合Janus笼破碎后的多孔片的SEM图像;(d) 用HF酸刻蚀除去无机壳层后的有机笼的SEM图像Figure 5. (a) SEM image of magnetic composite Janus cage with polydiethylaminoethyl methacrylate (PDEAEMA) grafted onto interior surface; (b) Cross-section TEM image of composite Janus cage with PDEAEMA grafted onto interior surface (without Fe3O4 nanoparticles); (c) SEM image of Janus porous nanosheets crushed from composite Janus cage; (d) SEM image of organic cage obtained by etching inorganic shell with HF![]() 图 6 (a) 磁性无机Janus笼(曲线1)和磁性有机/无机复合Janus笼(曲线2)的FTIR图谱;(b) 磁性无机Janus笼(曲线1)和磁性有机/无机复合Janus笼(曲线2)的在空气中的热失重曲线Figure 6. (a) FTIR spectra of magnetic inorganic Janus cage ( curve 1) and magnetic composite Janus cage (curve 2); (b) Thermogravimetric analysis (TGA) curves in air of magnetic inorganic Janus cage (curve 1) and magnetic composite Janus cage (curve 2)
图 6 (a) 磁性无机Janus笼(曲线1)和磁性有机/无机复合Janus笼(曲线2)的FTIR图谱;(b) 磁性无机Janus笼(曲线1)和磁性有机/无机复合Janus笼(曲线2)的在空气中的热失重曲线Figure 6. (a) FTIR spectra of magnetic inorganic Janus cage ( curve 1) and magnetic composite Janus cage (curve 2); (b) Thermogravimetric analysis (TGA) curves in air of magnetic inorganic Janus cage (curve 1) and magnetic composite Janus cage (curve 2)2.3 磁性pH响应性聚合物/无机复合Janus笼用于油水分离、传输和响应性吸释
PDEAEMA为pH响应性聚合物,pKa约为7.2,当环境pH值大于其pKa时,PDEAEMA表现为疏水,反之则为亲水。Janus笼接枝pH响应性聚合物后,可以通过调节pH值来改变Janus笼内部的亲疏水环境,实现磁操控的pH响应性油水分离,进而可用于药物在体内的靶向释放等。
首先对复合Janus笼进行磁操控pH响应性吸释和磁分离实验,如图7所示。先将油溶性荧光染料dil-C18加入到少量甲苯中以便后续观察,将甲苯加入到分散有复合Janus笼的水中并调节水相pH值为5,两者不互溶形成界限分明的两相(图7(a))。再将水相pH值调至9,振荡吸附,甲苯和水相分层消失,体系呈浑浊的均相,再用磁铁进行磁分离,发现不再出现两相分层,而是甲苯都随着复合Janus笼被吸附在瓶壁上(图7(b))。将pH调回5,振荡后静置,再用磁铁进行磁分离,发现油水分相重新出现,当环境变为酸性时,油相重新被Janus笼释放出来(图7(c))。作为对照,保持水相pH值为5不变,重复上述实验,发现油相无法在酸性条件下被复合笼吸入(图7(d)、图7(e))。由此,复合Janus笼可对环境中的油进行pH响应性的吸释并可在磁操控下定向运输。
![]() 图 7 内表面接枝PDEAEMA的磁性复合Janus笼对油相的pH响应性及磁操控分离实验:(a) 分散有复合Janus笼的水(下层)和甲苯(上层)分相照片,甲苯中加入1, 1-双十八烷基-3, 3, 3, 3-四甲基吲哚羰花青高氯酸盐 (dil-C18)染料以便于观察,水相pH值为5;(b) 调节pH至9,振荡后磁分离吸收甲苯的复合Janus笼的照片;(c) 调节pH至5,振荡静置后磁铁吸附复合Janus笼的照片;(d) 分散有复合Janus笼的水(下层)和经dil-C18染色的甲苯(上层)分相照片,水相pH值为5;(e) 保持pH为5不变,振荡静置后磁铁吸附复合Janus笼的照片Figure 7. pH-responsive absorption and release of the oil and magnetic manipulation of magnetic composite Janus cage with PDEAEMA grafted onto interior surface: (a) Immiscible mixture of toluene (top)/aqueous dispersion of composite Janus cage (bottom), oil soluble dye 1, 1'-dioctadecyl-3, 3, 3', 3'-tetramethylindodicarbocyanine perchlorate (dil-C18) is added in toluene and the pH value of aqueous dispersion is 5; (b) pH value is modulated to 9 and oil contained magnetic Janus cages are collected by magnets after vibration; (c) pH value is modulated back to 5 and magnetic Janus cages are collected by magnets after vibration with the release of dyed oil; (d) Immiscible mixture of toluene (top)/aqueous dispersion of composite Janus cage (bottom), oil soluble dye dil-C18 is added in toluene and the pH value of aqueous dispersion is 5; (e) pH value remains to be 5 and magnetic Janus cages are collected by magnets after vibration while dyed oil remains unchanged
图 7 内表面接枝PDEAEMA的磁性复合Janus笼对油相的pH响应性及磁操控分离实验:(a) 分散有复合Janus笼的水(下层)和甲苯(上层)分相照片,甲苯中加入1, 1-双十八烷基-3, 3, 3, 3-四甲基吲哚羰花青高氯酸盐 (dil-C18)染料以便于观察,水相pH值为5;(b) 调节pH至9,振荡后磁分离吸收甲苯的复合Janus笼的照片;(c) 调节pH至5,振荡静置后磁铁吸附复合Janus笼的照片;(d) 分散有复合Janus笼的水(下层)和经dil-C18染色的甲苯(上层)分相照片,水相pH值为5;(e) 保持pH为5不变,振荡静置后磁铁吸附复合Janus笼的照片Figure 7. pH-responsive absorption and release of the oil and magnetic manipulation of magnetic composite Janus cage with PDEAEMA grafted onto interior surface: (a) Immiscible mixture of toluene (top)/aqueous dispersion of composite Janus cage (bottom), oil soluble dye 1, 1'-dioctadecyl-3, 3, 3', 3'-tetramethylindodicarbocyanine perchlorate (dil-C18) is added in toluene and the pH value of aqueous dispersion is 5; (b) pH value is modulated to 9 and oil contained magnetic Janus cages are collected by magnets after vibration; (c) pH value is modulated back to 5 and magnetic Janus cages are collected by magnets after vibration with the release of dyed oil; (d) Immiscible mixture of toluene (top)/aqueous dispersion of composite Janus cage (bottom), oil soluble dye dil-C18 is added in toluene and the pH value of aqueous dispersion is 5; (e) pH value remains to be 5 and magnetic Janus cages are collected by magnets after vibration while dyed oil remains unchanged进而在荧光显微镜下观察复合Janus笼对油相pH响应性的吸释过程,如图8所示。将甲苯加入香豆素-6荧光染料标记后再用十二烷基硫酸钠(SDS)为乳化剂和水乳化得到水包油乳液,乳液液滴在荧光显微镜下呈现蓝色。将pH响应性复合Janus笼先用FITC进行荧光标记,染料可吸附在Janus笼壳层便于后续观察。再将标记后的Janus笼加入上述荧光标记的水和甲苯的乳液中,分散均匀,调节pH为9左右,避光搅拌后再置于荧光显微镜下观察,发现蓝色的甲苯液滴(圆圈内)被吸入到Janus笼腔体内(图8(a)),磁分离Janus笼后取水相置于荧光显微镜下观察,蓝色液滴消失(图8(b)),证明在pH=9的条件下,Janus笼将甲苯完全吸入腔内;把pH调至5,壳层内聚合物由疏水转变为亲水,甲苯被挤出,此时在荧光显微镜下仅仅看到Janus笼呈现绿色的光圈,腔内蓝色液滴消失(图8(c)),磁分离后对水相取样观察,出现蓝色液滴(球形),证明甲苯被Janus笼响应性释放(图8(d)),说明复合Janus笼可通过改变环境pH值可以实现可控的油水分离。
![]() 图 8 pH=9时复合Janus笼吸附荧光染色甲苯的荧光显微镜图像(a)和用磁铁分离吸附甲苯的Janus笼后,上层水样的荧光显微镜图像(b); pH=5时Janus笼释放出甲苯后的荧光显微镜图像(c)和磁铁分离Janus笼后上层水样的荧光显微镜图像(d)Figure 8. Fluorescence microscopy images of the Janus composite cage after absorption of toluene with pH of the circumstance of 9 (a) and the supernatant water after magnetic collection of the Janus cage (b); Fluorescence microscopy images of the Janus composite cage after release of toluene with pH of the circumstance of 5 (c) and the supernatant water after magnetic collection of the Janus cage (d)
图 8 pH=9时复合Janus笼吸附荧光染色甲苯的荧光显微镜图像(a)和用磁铁分离吸附甲苯的Janus笼后,上层水样的荧光显微镜图像(b); pH=5时Janus笼释放出甲苯后的荧光显微镜图像(c)和磁铁分离Janus笼后上层水样的荧光显微镜图像(d)Figure 8. Fluorescence microscopy images of the Janus composite cage after absorption of toluene with pH of the circumstance of 9 (a) and the supernatant water after magnetic collection of the Janus cage (b); Fluorescence microscopy images of the Janus composite cage after release of toluene with pH of the circumstance of 5 (c) and the supernatant water after magnetic collection of the Janus cage (d)3. 结 论
(1) 通过乳液界面溶胶凝胶和两种表面活性剂在界面微相分离先制备磁性无机Janus笼,内部修饰的烷基基团使无机笼壳层具有良好支撑性,同时可通过调节吐温80 (Tween 80)含量实现孔径在40 nm~1 μm可调,进一步利用同样修饰在内表面的卤素基团,通过“Cu媒介”的活性自由基聚合法在温和的反应条件下成功在内表面接枝pH响应性聚合物得到功能无机/有机复合Janus笼。
(2) 将复合Janus笼用于油水分离,证明了其可通过调控环境pH来实现对油相的响应性吸收和释放。这种pH敏感性的微容器且表面带有增强传质的孔道,有望用于药物的装载和体内靶向释放等领域。
-
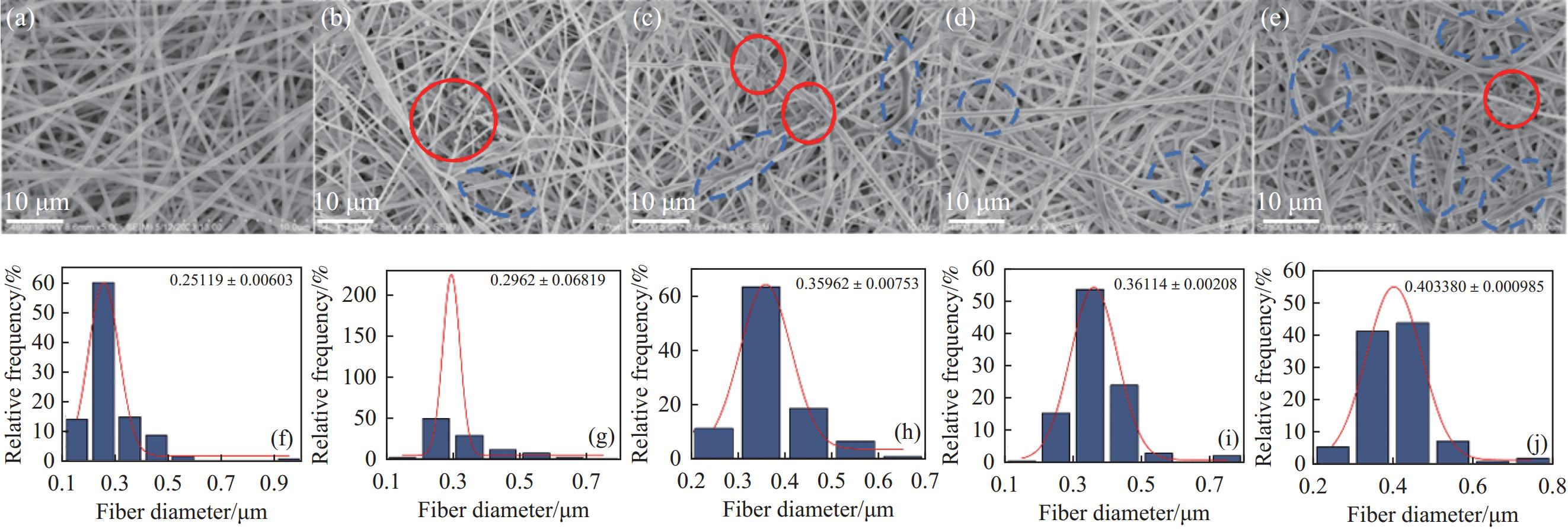
图 6 复合纳米纤维膜的SEM图像和直径分布:((a), (f)) PVA;((b), (g)) PVA/O-CN-0.6;((c), (h)) PVA/O-CN-4;((d), (i)) PVA/O-CN-8;((e), (j)) PVA/O-CN-16
Figure 6. SEM images and diameter distributions of composite nanofiber films: ((a), (f)) PVA; ((b), (g)) PVA/O-CN-0.6; ((c), (h)) PVA/O-CN-4; ((d), (i)) PVA/O-CN-8; ((e), (j)) PVA/O-CN-16
图 9 光照下CN ((a), (f))、O-CN ((b)~(e), (g)~(j))、PVA/CN ((a'), (g'))、PVA/O-CN ((b')~(e'), (h')~(k'))和PVA ((f'), (l'))对大肠杆菌和金黄色葡萄球菌的抑菌圈结果
Figure 9. Inhibitory zone results of CN ((a), (f)), O-CN ((b)-(e), (g)-(j)), PVA/CN ((a'), (g')), PVA/O-CN ((b')-(e'), (h')-(k')) and PVA ((f'), (l')) under illumination against E. coli and S. aureus
图 10 光照下CN ((a), (f))、O-CN ((b)~(e), (g)~(j))、PVA/CN ((a'), (g'))、PVA/O-CN ((b')~(e'), (h')~(k'))和PVA ((f'), (l'))对大肠杆菌和金黄色葡萄球菌的平板计数结果
Figure 10. Plate count results of CN ((a), (f)), O-CN ((b)-(e), (g)-(j)), PVA/CN ((a'), (g')), PVA/O-CN ((b')-(e'), (h')-(k')) and PVA ((f'), (l')) under illumination against E. coli and S. aureus
图 12 在黑暗和可见光(波长λ≥420 nm)照射下O-CN-0.6和PVA/O-CN-0.6中5,5-二甲基-1-吡咯烷氧基-氮氧自由基(DMPO)捕获·OH (a)、2,2,6,6-四甲基哌啶氮氧化物(TEMP)捕获1O2 (b)和DMPO捕获·O−2 (c)的ESR光谱
Figure 12. ESR spectra of ·OH trapping by dimethyl pyridine N-oxide (DMPO) (a), 1O2 trapping by 2,2,6,6-tetramethylpiperidine-1-oxyl (TEMP) (b) and ·O−2 trapping by DMPO (c) for O-CN-0.6 and PVA/O-CN-0.6 in the dark and under visible-light (Wavelength λ≥420 nm) irradiation
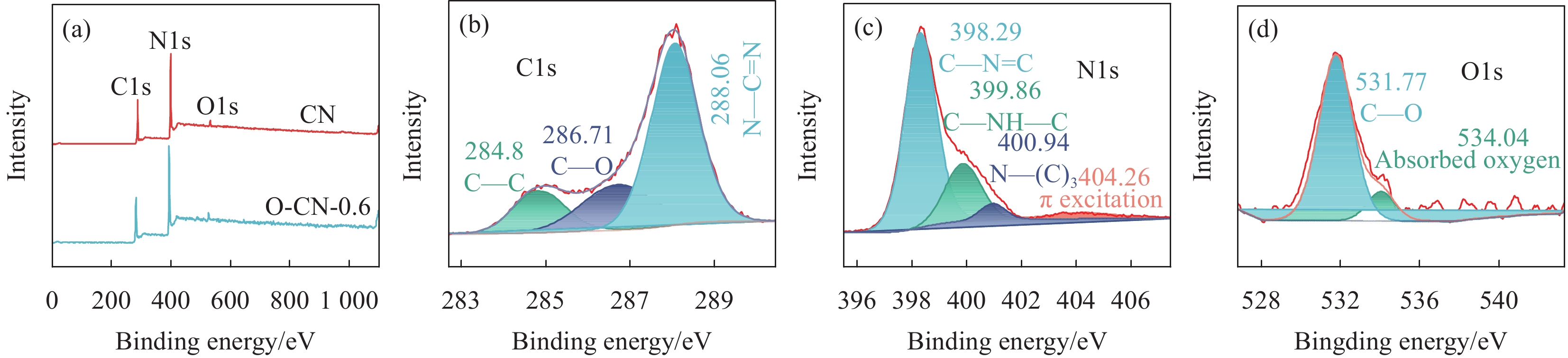
表 1 O-CN-X纳米片的组成
Table 1 Composition of O-CN-X nanosheet
Sample Mass of urea/g Volume of formic acid/mL O-CN-0.6 20 0.6 O-CN-4 20 4 O-CN-8 20 8 O-CN-16 20 16 表 2 CN和O-CN-0.6的原子百分比
Table 2 Atomic percentage of CN and O-CN-0.6
Element CN O-CN-0.6 C1s/at% 42.13 42.77 N1s/at% 55.13 53.55 O1s/at% 2.74 3.69 -
[1] ZHANG J, WANG J, XU H H, et al. The effective photocatalysis and antibacterial properties of AgBr/AgVO3 composites under visible-light[J]. RSC Advances, 2019, 9(63): 37109-37118. DOI: 10.1039/C9RA06810D
[2] ZHAO H B, XING Z P, SU S, et al. Recent advances in metal organic frame photocatalysts for environment and energy applications[J]. Applied Materials Today, 2020, 21: 100821. DOI: 10.1016/j.apmt.2020.100821
[3] HASIJA V, RAIZADA P, SUDHAIK A, et al. Recent advances in noble metal free doped graphitic carbon nitride based nanohybrids for photocatalysis of organic contaminants in water: A review[J]. Applied Materials Today, 2019, 15: 494-524. DOI: 10.1016/j.apmt.2019.04.003
[4] AMELIA S R, ROHMATULLOH Y, LISTIANI P, et al. One pot synthesis and performance of N- and (Mg, B, N)-doped ZnO for photocatalytic and antibacterial applications: Experimental and theoretical investigations[J]. Ceramics International, 2024, 50(7): 11216-11235.
[5] REGMI A, BASNET Y, BHATTARAI S, et al. Cadmium sulfide nanoparticles: Synthesis, characterization, and antimicrobial study[J]. Journal of Nanomaterials, 2023, 2023: 8187000.
[6] REN F, LI Y, ZHANG M, et al. Photocatalytic inactivation mechanism of nano-BiPO4 against Vibrio parahaemolyticus and its application in abalone[J]. Food Research International, 2024, 177: 113806. DOI: 10.1016/j.foodres.2023.113806
[7] HUY B T, NHI P T, VY N T T, et al. Design of novel p-n heterojunction ZnBi2O4-ZnS photocatalysts with impressive photocatalytic and antibacterial activities under visible light[J]. Environmental Science and Pollution Research, 2022, 29(56): 84471-84486. DOI: 10.1007/s11356-022-21810-w
[8] DAS M, SETHY C, KUNDU C N, et al. Synergetic reinforcing effect of graphene oxide and nanosilver on carboxymethyl cellulose/sodium alginate nanocomposite films: Assessment of physicochemical and antibacterial properties[J]. International Journal of Biological Macromolecules, 2023, 239: 124185. DOI: 10.1016/j.ijbiomac.2023.124185
[9] INDUJA M, SIVAPRAKASH K, GOMATHI P P. et al. Facile green synthesis and antimicrobial performance of Cu2O nanospheres decorated g-C3N4 nanocomposite[J]. Materials Research Bulletin, 2019, 112: 331-335. DOI: 10.1016/j.materresbull.2018.12.030
[10] THURSTON J H, HUNTER N M, WAYMENT L J. et al. Urea-derived graphitic carbon nitride (u-g-C3N4) films with highly enhanced antimicrobial and sporicidal activity[J]. Journal of Colloid and Interface Science, 2017, 505: 910-918. DOI: 10.1016/j.jcis.2017.06.089
[11] ORCUTT E K, VARAPRAGASAM S J, PETERSON Z C, et al. Ultrafast charge injection in silver-modified graphitic carbon nitride[J]. ACS Applied Materials & Interfaces, 2023, 15(12): 15478-15485.
[12] LYU C Y, LI W, LIN Q W, et al. Efficient photocatalytic hydrogen evolution: A novel multi-modified carbon nitride based on physical adsorption[J]. Journal of Materials Chemistry A, 2023, 11(38): 20701-20711. DOI: 10.1039/D3TA04906J
[13] LI F, ZHANG D N, XIANG Q J. Nanosheet-assembled hierarchical flower-like g-C3N4 for enhanced photocatalytic CO2 reduction activity[J]. Chemical Communications, 2020, 56(16): 2443-2446. DOI: 10.1039/C9CC08793A
[14] ZHANG R Y, ZHANG A L, YANG Y, et al. Surface modification to control the secondary pollution of photocatalytic nitric oxide removal over monolithic protonated g-C3N4/graphene oxide aerogel[J]. Journal of Hazardous Materials, 2020, 397: 122822. DOI: 10.1016/j.jhazmat.2020.122822
[15] LU L L, XU X X, AN K L, et al. Coordination polymer derived NiS@g-C3N4 composite photocatalyst for sulfur vacancy and photothermal effect synergistic enhanced H2 production[J]. ACS Sustainable Chemistry & Engineering, 2018, 6(9): 11869-11876.
[16] WANG Y, WANG X C, ANTONIETTI M. Polymeric graphitic carbon nitride as a heterogeneous organocatalyst: From photochemistry to multipurpose catalysis to sustainable chemistry[J]. Angewandte Chemie International Edition, 2012, 51(1): 68-89. DOI: 10.1002/anie.201101182
[17] STOLBOV S, ZULUAGA S. Sulfur doping effects on the electronic and geometric structures of graphitic carbon nitride photocatalyst: Insights from first principles[J]. Journal of Physics: Condensed Matter, 2013, 25(8): 085507. DOI: 10.1088/0953-8984/25/8/085507
[18] ZHANG Z Z, SUN Q M, JI R, et al. Boosted photogenerated charge carrier separation by synergy of oxygen and phosphorus co-doping of graphitic carbon nitride for efficient 2-chlorophenol photocatalytic degradation[J]. Chemical Engineering Journal, 2023, 471: 144388. DOI: 10.1016/j.cej.2023.144388
[19] ZHAO H B, ZHANG Y. Sulfur-doped carbon nitride with carbon vacancies: Enhanced photocatalytic activity for degradation of tetracycline hydrochloride[J]. Diamond and Related Materials, 2023, 139: 110239. DOI: 10.1016/j.diamond.2023.110239
[20] CHEN L, NING S B, LIANG L, et al. Potassium doped and nitrogen defect modified graphitic carbon nitride for boosted photocatalytic hydrogen production[J]. International Journal of Hydrogen Energy, 2022, 47(30): 14044-14052. DOI: 10.1016/j.ijhydene.2022.02.147
[21] ZHAO D M, WANG Y Q, DONG C L, et al. Boron-doped nitrogen-deficient carbon nitride-based Z-scheme heterostructures for photocatalytic overall water splitting[J]. Nature Energy, 2021, 6(4): 388-397. DOI: 10.1038/s41560-021-00795-9
[22] LEI L, FAN H Q, JIA Y X, et al. Cyanuric acid-assisted synthesis of hierarchical amorphous carbon nitride assembled by ultrathin oxygen-doped nanosheets for excellent photocatalytic hydrogen generation[J]. ACS Applied Materials & Interfaces, 2024, 16(12): 14809-14821.
[23] HU S Z, WANG K Y, LI P, et al. The effect of hydroxyl group grafting on the photocatalytic phenolic compounds oxidation ability of g-C3N4 prepared by a novel H2O2-alkali hydrothermal method[J]. Applied Surface Science, 2020, 513: 145783. DOI: https://doi.org/10.1016/j.apsusc.2020.145783
[24] ZENG Y X, LIU X, LIU C B, et al. Scalable one-step production of porous oxygen-doped g-C3N4 nanorods with effective electron separation for excellent visible-light photocatalytic activity[J]. Applied Catalysis B: Environmental, 2018, 224: 1-9. DOI: 10.1016/j.apcatb.2017.10.042
[25] 温家奇. 氧掺杂多孔氮化碳的制备及其光催化制氢性能研究[D]. 郑州: 郑州大学, 2022. WEN Jiaqi. Preparation of oxygen-doped porous carbon nitride and its photocatalytic performance for hydrogen production[D]. Zhengzhou: Zhengzhou University, 2022(in Chinese).
[26] XU X Q, WANG S, HU T, et al. Fabrication of Mn/O co-doped g-C3N4: Excellent charge separation and transfer for enhancing photocatalytic activity under visible light irradiation[J]. Dyes and Pigments, 2020, 175: 108107. DOI: 10.1016/j.dyepig.2019.108107
[27] LI F, ZHU P, WANG S M, et al. One-pot construction of Cu and O co-doped porous g-C3N4 with enhanced photocatalytic performance towards the degradation of levofloxacin[J]. RSC Advances, 2019, 9(36): 20633-20642. DOI: 10.1039/C9RA02411E
[28] IQBAL S, BAHADUR A, ANWER S, et al. Designing novel morphologies of l-cysteine surface capped 2D covellite (CuS) nanoplates to study the effect of CuS morphologies on dye degradation rate under visible light[J]. CrystEngComm, 2020, 22(24): 4162-4173. DOI: 10.1039/D0CE00421A
[29] XING Q F, ZANG M, XU X Q, et al. Rapid photocatalytic inactivation of E. coli by polyethyleneimine grafted O-doped g-C3N4: Synergetic effects of the boosted reactive oxygen species production and adhesion performance[J]. Applied Surface Science, 2022, 573: 151496. DOI: 10.1016/j.apsusc.2021.151496
[30] YANG Y Y, GUO W T, ZHAI Y P, et al. Oxygen-doped and nitrogen vacancy co-modified carbon nitride for the efficient visible light photocatalytic hydrogen evolution[J]. New Journal of Chemistry, 2020, 44(38): 16320-16328. DOI: 10.1039/D0NJ03695A
[31] HU B, GUO F S, LI S R, et al. Facile fabrication of oxygen-doped carbon nitride with enhanced visible-light photocatalytic degradation of methyl mercaptan[J]. Research on Chemical Intermediates, 2022, 48(6): 2295-2311. DOI: 10.1007/s11164-022-04712-x
[32] JINGUJI K, WATANABE M, MORITA R, et al. Visible light driven hydrogen peroxide production by oxygen and phosphorus co-doped CoP-C3N4 photocatalyst[J]. Catalysis Today, 2024, 426: 114400. DOI: 10.1016/j.cattod.2023.114400
[33] SONG X H, ZHANG X Y, LI X, et al. Enhanced light utilization efficiency and fast charge transfer for excellent CO2 photoreduction activity by constructing defect structures in carbon nitride[J]. Journal of Colloid and Interface Science, 2020, 578: 574-583. DOI: 10.1016/j.jcis.2020.06.035
[34] WANG L Q, LI R Y, ZHANG Y M, et al. Tetracycline degradation mechanism of peroxymonosulfate activated by oxygen-doped carbon nitride[J]. RSC Advances, 2023, 13(10): 6368-6377. DOI: 10.1039/D3RA00345K
[35] MAJIDI M, GIVIANRAD M H, SABER-TEHRANI M. et al. A novel method to synthesis of oxygen doped graphitic carbon nitride with outstanding photocatalytic efficiency for the degradation organic pollutants[J]. Diamond and Related Materials, 2023, 139: 110431. DOI: 10.1016/j.diamond.2023.110431
[36] FENG T, ZHANG J, YU F H, et al. Broad-bandgap porous graphitic carbon nitride with nitrogen vacancies and oxygen doping for efficient visible-light photocatalytic degradation of antibiotics[J]. Environmental Pollution, 2023, 335: 122268. DOI: 10.1016/j.envpol.2023.122268
[37] WEN D, SU Y R, FANG J Y, et al. Synergistically boosted photocatalytic production of hydrogen peroxide via protonation and oxygen doping on graphitic carbon nitride[J]. Nano Energy, 2023, 117: 108917. DOI: 10.1016/j.nanoen.2023.108917
[38] FENG C Y, TANG L, DENG Y C, et al. Synthesis of leaf-vein-like g-C3N4 with tunable band structures and charge transfer properties for selective photocatalytic H2O2 evolution[J]. Advanced Functional Materials, 2020, 30(39): 2001922. DOI: 10.1002/adfm.202001922
[39] YU P, ZHOU X Q, YAN Y C, et al. Enhanced visible-light-driven photocatalytic disinfection using AgBr-modified g-C3N4 composite and its mechanism[J]. Colloids and Surfaces B: Biointerfaces, 2019, 179: 170-179. DOI: 10.1016/j.colsurfb.2019.03.074
[40] ZHANG H H, HAN X X, YU H J, et al. Enhanced photocatalytic performance of boron and phosphorous co-doped graphitic carbon nitride nanosheets for removal of organic pollutants[J]. Separation and Purification Technology, 2019, 226: 128-137. DOI: 10.1016/j.seppur.2019.05.066
[41] ABD EL-AZIZ A M, EL-MAGHRABY A, TAHA N A. Comparison between polyvinyl alcohol (PVA) nanofiber and polyvinyl alcohol (PVA) nanofiber/hydroxyapatite (HA) for removal of Zn2+ ions from wastewater[J]. Arabian Journal of Chemistry, 2017, 10(8): 1052-1060. DOI: 10.1016/j.arabjc.2016.09.025
[42] TARI E, UGRASKAN V, YAZICI O. Enhanced mechanical, thermal and optical properties of poly (vinyl alcohol)/functionalized-graphitic carbon nitride composites[J]. Fullerenes, Nanotubes and Carbon Nanostructures, 2023, 32(5): 464-470.
[43] WANG Y M, CAI H Y, QIAN F F, et al. Facile one-step synthesis of onion-like carbon modified ultrathin g-C3N4 2D nanosheets with enhanced visible-light photocatalytic performance[J]. Journal of Colloid and Interface Science, 2019, 533: 47-58. DOI: 10.1016/j.jcis.2018.08.039
[44] LIU D, TIAN R, WANG J, et al. Photoelectrocatalytic degradation of methylene blue using F doped TiO2 photoelectrode under visible light irradiation[J]. Chemosphere, 2017, 185: 574-581.
[45] NAMBIAR A P, PILLAI R, VADIKKEETTIL Y, et al. Glutaraldehyde-crosslinked poly(vinyl alcohol)/halloysite composite films as adsorbent for methylene blue in water[J]. Materials Chemistry and Physics, 2022, 291: 126752. DOI: 10.1016/j.matchemphys.2022.126752
[46] ZIDAN H M, EL-GHAMAZ N A, ABDELGHANY A M. et al. Photodegradation of methylene blue with PVA/PVP blend under UV light irradiation[J]. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 2018, 199: 220-227. DOI: 10.1016/j.saa.2018.03.057
[47] JASEELA P K, GARVASIS J, JOSEPH A. Selective adsorption of methylene blue (MB) dye from aqueous mixture of MB and methyl orange (MO) using mesoporous titania (TiO2)-poly vinyl alcohol (PVA) nanocomposite[J]. Journal of Molecular Liquids, 2019, 286: 110908. DOI: 10.1016/j.molliq.2019.110908
[48] ALGHASHAM H A. Development of wound healing scaffolds based on polymeric blends of polyvinyl alcohol and hyaluronic acid doped with super antibacterials of silver phosphate with magnesium vanadate[J]. New Journal of Chemistry, 2024, 48: 4529-4538. DOI: 10.1039/D3NJ03842D
[49] KONG X Y, LIU X M, ZHENG Y F, et al. Graphitic carbon nitride-based materials for photocatalytic antibacterial application[J]. Materials Science and Engineering: R: Reports, 2021, 145: 100610. DOI: 10.1016/j.mser.2021.100610
[50] ALGHASHAM H A. Development of wound healing scaffolds based on polymeric blends of polyvinyl alcohol and hyaluronic acid doped with super antibacterials of silver phosphate with magnesium vanadate[J]. New Journal of Chemistry, 2024, 48(10): 4529-4538. DOI: 10.1039/D3NJ03842D
[51] ZHANG L L, ZHANG L F, XU J G. Chemical composition, antibacterial activity and action mechanism of different extracts from hawthorn (Crataegus pinnatifida Bge. )[J]. Scientific Reports, 2020, 10(1): 8876. DOI: 10.1038/s41598-020-65802-7
[52] HAO W, WANG W, LIU C, et al. Synthesis of Ag@CeO2@Ti3C2 heterojunction and its photocatalytic bacteriostatic properties[J]. Materials Letters, 2022, 308: 131202. DOI: 10.1016/j.matlet.2021.131202
[53] NEMATI D, ASHJARI M, RASHEDI H, et al. PVA based nanofiber containing cellulose modified with graphitic carbon nitride/nettles/trachyspermum accelerates wound healing[J]. Biotechnology Progress, 2021, 37(6): e3200. DOI: 10.1002/btpr.3200
[54] SHE P, LI J, BAO H G, et al. Green synthesis of Ag nanoparticles decorated phosphorus doped g-C3N4 with enhanced visible-light-driven bactericidal activity[J]. Journal of Photochemistry and Photobiology A: Chemistry, 2019, 384: 112028. DOI: 10.1016/j.jphotochem.2019.112028
[55] IQBAL S, BAHADUR A, ALI S, et al. Critical role of the heterojunction interface of silver decorated ZnO nanocomposite with sulfurized graphitic carbon nitride heterostructure materials for photocatalytic applications[J]. Journal of Alloys and Compounds, 2021, 858: 158338. DOI: 10.1016/j.jallcom.2020.158338
[56] ZHANG D, LIU Y, LIU Z G, et al. Advances in antibacterial functionalized coatings on Mg and its alloys for medical use—A review[J]. Coatings, 2020, 10(9): 828.
[57] LIU X, ZHAO Y X, YANG X F, et al. Porous Ni5P4 as a promising cocatalyst for boosting the photocatalytic hydrogen evolution reaction performance[J]. Applied Catalysis B: Environmental, 2020, 275: 119144. DOI: 10.1016/j.apcatb.2020.119144
-
期刊类型引用(10)
1. 翟兆阳,李欣欣,张延超,刘忠明,杜春华,张华明. 连续光纤激光切割金属薄壁材料工艺研究. 红外与激光工程. 2024(02): 33-43 .  百度学术
百度学术
2. 陶洋,李存静,逄增媛,张典堂. 展宽布/网胎针刺C/C复合材料制备及力学性能. 复合材料学报. 2024(04): 1934-1944 .  本站查看
本站查看
3. 董志刚,王中旺,冉乙川,鲍岩,康仁科. 碳纤维增强陶瓷基复合材料超声振动辅助铣削加工技术的研究进展. 机械工程学报. 2024(09): 26-56 .  百度学术
百度学术
4. 席翔,李海龙,陈友元,裴景奇,廖城坤,薛琳,储洪强,冉千平. 碳纤维增强碳基复合材料的介电性能对应力的自感知. 高分子材料科学与工程. 2024(05): 115-124 .  百度学术
百度学术
5. 钱奇伟,张昕,杨贞军,沈镇,校金友. 基于CT图像深度学习的三维编织C/C复合材料微观组分与缺陷智能识别. 复合材料学报. 2024(07): 3536-3543 .  本站查看
本站查看
6. 翟兆阳,李欣欣,张延超,刘忠明,杜春华,张华明. 基于正交试验的金属薄壁材料激光切割工艺优化. 中国机械工程. 2024(07): 1279-1289 .  百度学术
百度学术
7. 何金玲. 纤维复材浆料流变性能分析及矿混匀质量应用研究. 粘接. 2024(09): 87-90 .  百度学术
百度学术
8. 姚先龙. 碳基复合材料的应用及相关制备方法. 信息记录材料. 2023(01): 36-38 .  百度学术
百度学术
9. 石磊,罗浩,罗瑞盈. 胶层厚度对C/C复合材料剪切粘接性能的影响. 炭素技术. 2023(04): 22-26 .  百度学术
百度学术
10. 刘科众,陈舟,王泽鹏,韩保恒. C/C复合材料增密过程孔隙结构及演化研究. 机械设计与制造工程. 2022(10): 33-36 .  百度学术
百度学术
其他类型引用(10)
-
其他相关附件
-
目的
大量有机污染物对水循环造成严重污染,同时过度使用抗生素产生的耐药细菌对人类健康也构成严重威胁。光催化纳米材料由于广谱和低能耗的特点已成为抗菌领域的研究热点。本文选择石墨相氮化碳(CN)作为光催化材料,通过氧掺杂和静电纺丝制备出柔性纳米纤维催化剂,探索其在光催化去除有机污染物和抑菌等领域的应用。
方法通过热聚合法,利用甲酸与尿素共热,制备一系列氧掺杂氮化碳(O-CN),再将其与聚乙烯醇(PVA)溶液复合,通过静电纺丝方法制备了聚乙烯醇/氧掺杂氮化碳(PVA/O-CN)复合纳米纤维膜,利用SEM和TEM观察O-CN和PVA/O-CN的形貌特征,利用XRD、FT-IR和XPS研究了O-CN的结构特征,利用PL和UV-vis探究了O-CN的光学性能,利用应力-应变测试探究了PVA/O-CN的力学性能。并且以MB为模型污染物,分析讨论了可见光照射下复合纳米纤维膜的光催化去除有机污染物的能力和循环使用情况。通过抑菌圈法和平板计数法分别测试了O-CN和PVA/O-CN对大肠杆菌()和金黄葡萄球菌()的抑菌能力。并通过电子自旋共振自由基捕获法研究了O-CN和PVA/O-CN中ROS的种类和相对强度。推导了PVA/O-CN光催化去除有机物和抑菌的原理,可能是在可见光下,O-CN表面吸收光能,激发电子跃迁,产生电子-空穴对并分离。光生载流子能够与催化剂表面的O和HO分别发生氧化还原反应,生成ROS(、、),通过纳米纤维表面作用于MB分子和细菌。
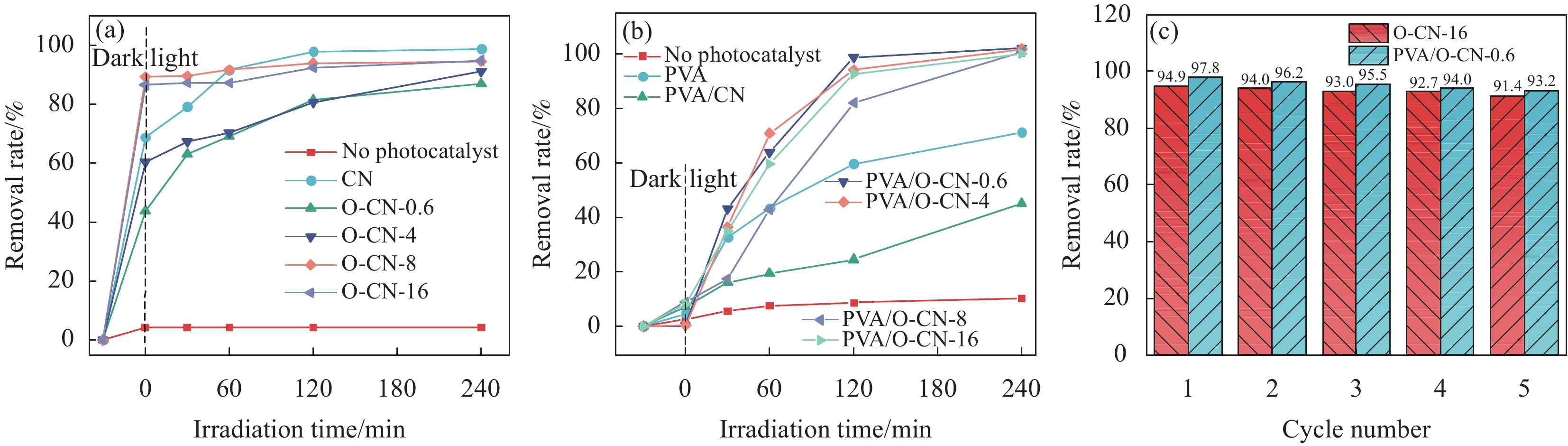
结果通过以上方法的测试结果并进行数据分析得到,O-CN与CN相比,其形貌中多孔结构以及片层状结构增多,随着氧含量增加,O-CN的颜色变深;通过分析O-CN和CN的结构特征,O元素可能取代CN中三-三嗪环结构中N的位置,并且导致材料缺陷增多;O-CN的可见光吸收能力和电子-空穴对分离率较CN有明显提高,PVA/O-CN纳米纤维中随着氧含量的增加,纤维直径变大。 PVA/O-CN-0.6复合纳米纤维膜对MB的去除率均在95%以上,连续实验5次之后,没有观察到明显的失活现象。PVA/O-CN复合纳米纤维膜对E.coli和S.aureus的抑菌率最大分别达到96%和93.7%。
结论聚乙烯醇/氧掺杂氮化碳(PVA/O-CN)纳米纤维膜光催化去除亚甲基蓝(MB)性能和光催化抑菌能力提升明显。尤其是PVA/O-CN-0.6复合膜,在实验范围内,在吸附和光催化作用下,去除MB达到97.7%。PVA/O-CN-0.6复合膜对和有最好的抑制效果,最大抑制直径分别为24.36±0.15 mm和20.48±0.35 mm,并且对两种菌的抑制率达到96%和93.7%。用ESR测试证实了在光激发下材料产生ROS,ROS是提高抑菌性能和光催化去除效率的关键因素;并提供了一种可能的抑菌机理。可以预测PVA/O-CN纳米纤维膜可用于包括但不限于医疗保健、食品包装和处理、家居用品、医美等多种领域。
-
g-C3N4具有合适的能带结构、良好的稳定性和光吸收能力,而且来源丰富,廉价易得。然而,它也存在量子效率低、可见光吸收范围窄、比表面积较小、电子-空穴对复合率高等缺点,纳米催化剂在应用过程中也存在难回收以及产生二次污染的问题。
本文通过用甲酸处理尿素获得前驱体,以热聚合法制备了氧掺杂氮化碳(O-CN)光催化剂,再利用静电纺丝技术制备负载O-CN的聚乙烯醇(PVA)纳米纤维膜,不仅改善g-C3N4本身存在的可见光捕获率不足,电子-空穴对复合率高等缺点,并且PVA纳米纤维膜的高比表面积和均匀的孔径为光催化反应产生更多的活性位点,实现光催化去除有机污染物和抑菌的效果。制备的PVA/O-CN-0.6纳米纤维膜光催化抑菌能力显著提高,对大肠杆菌(E.coli)和金黄色葡萄球菌(S.aureus)的抑菌圈直径达到24.36±0.15 mm和20.48±0.35 mm,抑制率为96%和93.7%;而纯石墨相氮化碳(CN)对两种菌的抑菌圈直径为4.40±0.52 mm和3.58±0.47 mm,抑制率只有30.2%和30%;同时对有机污染物有高效的降解作用(对亚甲基蓝(MB)的去除效率为97.7%),为抑菌和处理水污染提供新的设计方案。
PVA/O-CN复合纳米纤维膜的抑菌结果对比(a)和光催化去除MB效率(b)






 下载:
下载: