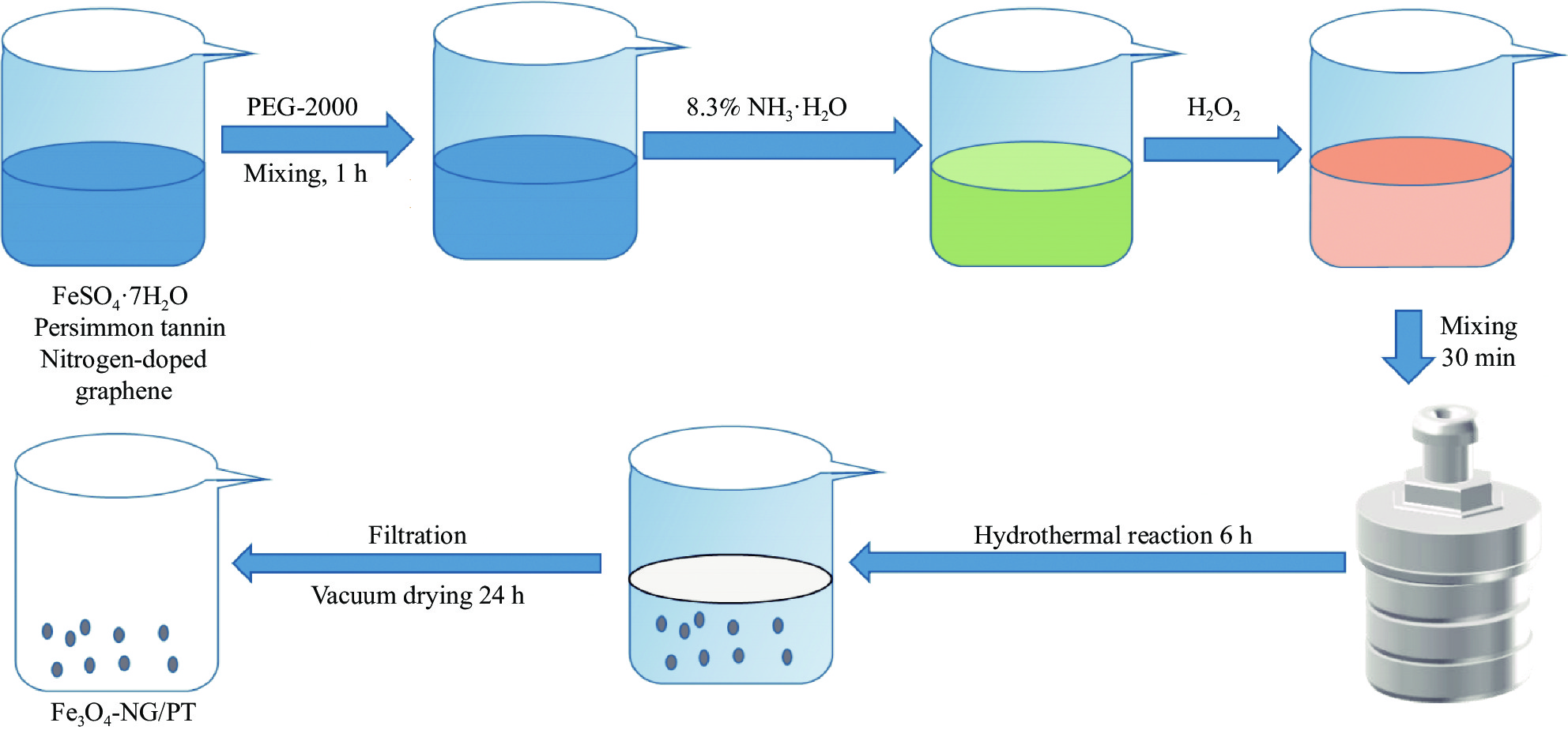
Adsorption behaviors of magnetic nitrogen-doped graphene-modified persimmon tannins for tetracycline
-
摘要: 四环素(TC)作为广谱类抗生素药物被大量应用于养殖业,因其不易自然降解的特性,造成了严重的水污染。本文以柿单宁(PT)为基体,通过氮掺杂石墨烯(NG)对其改性,并以Fe3O4作为磁性功能,采用水热法制得磁性复合材料Fe3O4-NG/PT。并针对此复合材料在吸附TC方面的应用进行研究。NG以其疏水性和比表面积大的优势,弥补了PT在吸附上的不足,Fe3O4解决了吸附剂难以回收的问题。通过SEM、EDS、FTIR、XPS、BET、XRD等表明其机制主要是静电作用、孔填充、氢键作用和π-π相互作用;通过XRD与磁强计表明Fe3O4的成功引入及其高效回收性能。并对吸附条件优化,在Fe3O4∶PT质量比4∶1、pH为7、180 min、投加量为0.6 g/L时,吸附效果最佳。吸附动力学符合拟二级动力学模型,化学吸附为主;吸附等温符合Freundlich模型,表明其为多层吸附。318 K时吸附的最大容量达到315.65 mg/g,吸附效率达到94.5%。Fe3O4-NG/PT复合材料因其较大比表面积、多孔、回收方便、大量酚羟基结构的优点,在吸附领域有一定的应用前景。
-
关键词:
- 柿单宁(PT) /
- 氮掺杂石墨烯(NG) /
- 吸附 /
- 四环素(TC) /
- 吸附模型
Abstract: Tetracycline (TC), as a broad-spectrum class of antibiotics, is used in large quantities in the farming industry and has caused serious water pollution due to its characteristics of not being easily degraded naturally. In this paper, a magnetic composite Fe3O4-NG/PT was produced by hydrothermal method, persimmon tannin (PT) was as the matrix, and modified by nitrogen-doped graphene (NG), Fe3O4 was as a magnetic filler. The application of the composite in TC adsorption was also investigated. NG with its hydrophobicity and the large specific surface area made up for the lack of persistent tannin in adsorption, and Fe3O4 solved the problem of difficult recovery of adsorbent. The mechanism was shown by SEM, chemisorption was dominant. The adsorption isotherm is consistent with the Freundlich model, indicating that it is multilayer adsorption. The maximum capacity of adsorption at 318 K reaches 315.65 mg/g, and the adsorption efficiency reaches 94.5%. The Fe3O4-NG/PT composites have the advantages of large specific surface area, high porosity, and phenolic hydroxyl groups, which will broaden their application fields. -
近年来随着工业的快速发展,水污染问题受到越来越多的关注。其中的一部分污染是由抗生素引起的,如阿莫西林、青霉素、四环素等[1-5]。四环素(Tetracycline,TC)作为一种普遍使用的抗生素,因其独特的抗菌作用而被广泛应用于人类疾病的治疗。然而TC长期存在于水环境中难以自然降解,对生态环境和人类健康有很大危害[6-10]。因此,开发一种有效的解决方案来去除水环境中难降解的抗生素是极其重要的。半导体光催化技术因其具有环境友好、无污染、低能耗等优点引起了广泛的研究,成为当今处理有机污染物最有前景的方法之一[11-13]。
在各种光催化剂中,BiOX(X=Cl, Br, I)因其特殊的层状结构、适宜的禁带宽度和较高的稳定性,被广泛用于光降解有机污染物和光催化分解水[14-15]。其中BiOI具有较窄的禁带宽度(1.77~1.92 eV)和较宽的可见光响应范围,但由于光生电子空穴对的高复合率,其光催化性能并不理想。将BiOI与其他半导体材料相结合被认为是增强光催化性能最有前途的策略,2个或2个以上半导体相结合可以形成半导体/半导体异质结,通过提高光生电子空穴对的分离速率从而提高光催化活性。
Bi2O3的带隙介于2.1~2.8 eV之间,由于其具有较强的可见光区响应、无毒、电化学稳定性高、热稳定性好和低成本等特性[16-18],是一种很有前途的可见光光催化候选材料,可与其他半导体材料形成异质结结构。如CdS/BiOCl/Bi2O3[19]、GO/AgI/Bi2O3[20]、α-Bi2O3/g-C3N4[21]、Bi2O3/ZnS[22]等。
Wei等[23]采用一锅沉淀法在多孔Bi2O3纳米棒上成功沉积了BiOI纳米片。结果表明:与原始Bi2O3和BiOI相比,50% Bi2O3/BiOI复合材料具有更高的光生电子空穴对分离效率和更大的比表面积,在可见光照射下,其光催化还原Cr(VI)的活性显著增强。此外,50% Bi2O3/BiOI复合材料还具有优异的光化学稳定性和可回收性。Li等[24]采用化学刻蚀法制备了BiOI/Bi2O3异质结,在降解苯酚和4-氯苯酚(4-CP)方面表现出良好的光催化活性。其光催化性能的提高是由于BiOI/Bi2O3异质结的形成促进了电子空穴对的有效分离,并提出了光生电荷转移的过程。
目前所制备的同类光催化剂大多数用于去除水环境中的重金属离子和有机染料等,对降解抗生素类药物的研究较少。本文采用简单的溶剂热法制备了Bi2O3/BiOI复合光催化材料,在模拟太阳光照射下通过降解四环素研究了其光催化性能,探究了BiOI与Bi2O3不同摩尔比、反应温度、反应时间、pH等条件对光催化性能的影响。并通过活性物种捕捉实验提出了Bi2O3/BiOI复合光催化材料降解四环素可能的机制。
1. 实验材料和方法
1.1 原材料
五水硝酸铋(上海麦克林生化有限公司,AR)、碘化钾(天津市大茂化学试剂厂,AR)、乙二醇(天津市北辰方正试剂厂,AR)、四环素(上海麦克林生化有限公司,AR)。
1.2 实验仪器
EL104型电子天平(梅特勒-托利多有限公司)、HC-3018型高速离心机(安徽中科中佳科学仪器有限公司)、TGL-5A台式离心机(常州润华电器有限公司)、KSW-4D-I2型马弗炉(北京中兴伟业仪器有限公司)、HJ-1型磁力加热搅拌器(红杉实验设备厂)、101-1A型电热鼓风干燥箱(北京中兴伟业仪器有限公司)、721型可见分光光度计(上海仪电分析仪器有限公司)、KQ5200E型超声波清洗器(昆山市超声仪器有限公司)、250 W金卤灯(上海亚明)。
1.3 实验内容
1.3.1 BiOI光催化材料的制备
称取1 mmol五水硝酸铋置于15 mL乙二醇中,超声处理15 min以获得均匀悬浮液。在不断搅拌下向其中逐滴加入10 mL含1 mmol碘化钾的水溶液,继续搅拌2 h。将产物离心,用水和无水乙醇洗涤数次,在80℃下干燥12 h得到红色的BiOI。
1.3.2 Bi2O3光催化材料的制备
称取一定量的五水硝酸铋,在600℃的马弗炉里煅烧4 h,冷却至室温后,将产物研磨成粉末状,得到淡黄色的Bi2O3。
1.3.3 Bi2O3/BiOI复合光催化材料的制备
将1 mmol五水硝酸铋置于15 mL乙二醇中,超声处理15 min以获得均匀悬浮液。在不断搅拌下向其中逐滴加入10 mL含1 mmol碘化钾的水溶液,继续搅拌2 h。在此期间,用1 mol/L的H2SO4溶液将混合液的pH调至5。然后向上述溶液中加入0.8 mmol已制备好的Bi2O3,继续搅拌1 h。将得到的混合溶液转移至50 mL聚四氟乙烯内衬的不锈钢高压反应釜中,在180℃下反应20 h。自然冷却至室温后,将产物离心,用水和无水乙醇洗涤数次,在80℃下干燥12 h,得到Bi2O3/BiOI复合光催化材料。
1.3.4 光催化性能测试
使用250 W金卤灯模拟太阳光照射,通过降解四环素来评价所制备样品的光催化性能。取50 mg制得的光催化材料放入装有100 mL 25 mg/L TC溶液的烧杯中,黑暗搅拌30 min达到吸附-脱附平衡。然后将混合液置于光反应器中,光照开始计时,每隔20 min取3 mL样,将样品放入离心机中离心取其上层清液并测定吸光度。计算四环素的残余率:
η=C/C0×100%=A/A0×100% 式中:C和C0分别表示t时刻和初始四环素的质量浓度(mg·L−1);A和A0分别表示t时刻和初始四环素的吸光度。
2. 结果与讨论
2.1 Bi2O3/BiOI复合光催化材料的晶相结构
BiOI、Bi2O3和Bi2O3/BiOI的XRD图谱如图1所示。BiOI曲线在9.658°、29.645°、31.657°、37.392°、45.666°、51.345°、55.15°、66.344°和74.09°处出现的衍射峰分别对应BiOI(JCPDS 10-0445)的(001)、(102)、(110)、(112)、(104)、(114)、(212)、(214)和(302)晶面。Bi2O3在21.722°、25.757°、27.377°、33.241°、35.406°、37.595°、42.353°、46.305°、52.373°和58.563°处出现的衍射峰分别对应Bi2O3(JCPDS 41-1449)的(020)、(002)、(120)、(200)、(031)、(112)、(122)、(041)、(−321)和(−331)晶面。Bi2O3/BiOI同时出现了Bi2O3和BiOI的主要衍射峰,说明本实验成功制备了Bi2O3/BiOI复合光催化材料。
2.2 Bi2O3/BiOI复合光催化材料的微观形貌
通过SEM分析了所制备光催化材料的微观形貌,结果如图2所示。可以看出,所制备的BiOI是由纳米片自组装形成的花状微球;单一Bi2O3呈现出不同尺寸、不规则的块状结构。从图2(c)可以看出,当BiOI与Bi2O3复合后,块状Bi2O3均匀分散在花状微球的BiOI表面。
2.3 Bi2O3/BiOI复合光催化材料的结构
样品的FTIR图谱如图3所示。499 cm−1和760 cm−1处是BiOI的特征吸收峰,1617 cm−1处的吸收峰对应Bi2O3中Bi—O键的弯曲振动,再次表明BiOI和Bi2O3成功复合在一起。
2.4 Bi2O3/BiOI复合光催化材料的光学性能
利用UV-Vis DRS分析了所制备样品的光学吸收性能,如图4所示。可以看出,纯BiOI的吸收边缘位于681 nm处,纯Bi2O3的吸收边缘位于477 nm,Bi2O3/BiOI复合光催化材料的光吸收边缘位于617 nm。与纯BiOI相比,Bi2O3/BiOI复合光催化材料的光吸收边缘有轻微的蓝移,这是由于与Bi2O3耦合造成的,但其光吸收范围仍然很宽。
不同光催化材料的紫外漫反射(αhv)1/2-hv转换图如图5所示。根据Kubelka-Munk公式,纯BiOI、Bi2O3和Bi2O3/BiOI对应的禁带宽度Eg值分别为1.82 eV、2.60 eV和2.01 eV。
2.5 Bi2O3/BiOI复合光催化材料光生电子空穴对的分离
利用荧光强度来分析光生电子空穴对的复合速率,荧光强度越小,则光生电子复合速率越低,图6为不同光催化材料的荧光光谱。可以看出,在520 nm处,Bi2O3/BiOI的荧光强度低于单一BiOI和Bi2O3,表明复合光催化材料的光生电子空穴对复合速率最低,光催化活性最高。
通过电化学阻抗法研究了不同光催化材料的电荷转移效率,结果如图7所示。Bi2O3/BiOI的圆弧半径小于单一BiOI和Bi2O3,说明其电荷转移电阻较低,电导率增强。电化学阻抗图表明Bi2O3/BiOI能增强光生电子空穴对的电荷转移能力,提高其分离效率,这与荧光分析的结果一致。
2.6 Bi2O3/BiOI复合光催化材料的性能
2.6.1 不同制备条件对光催化性能的影响
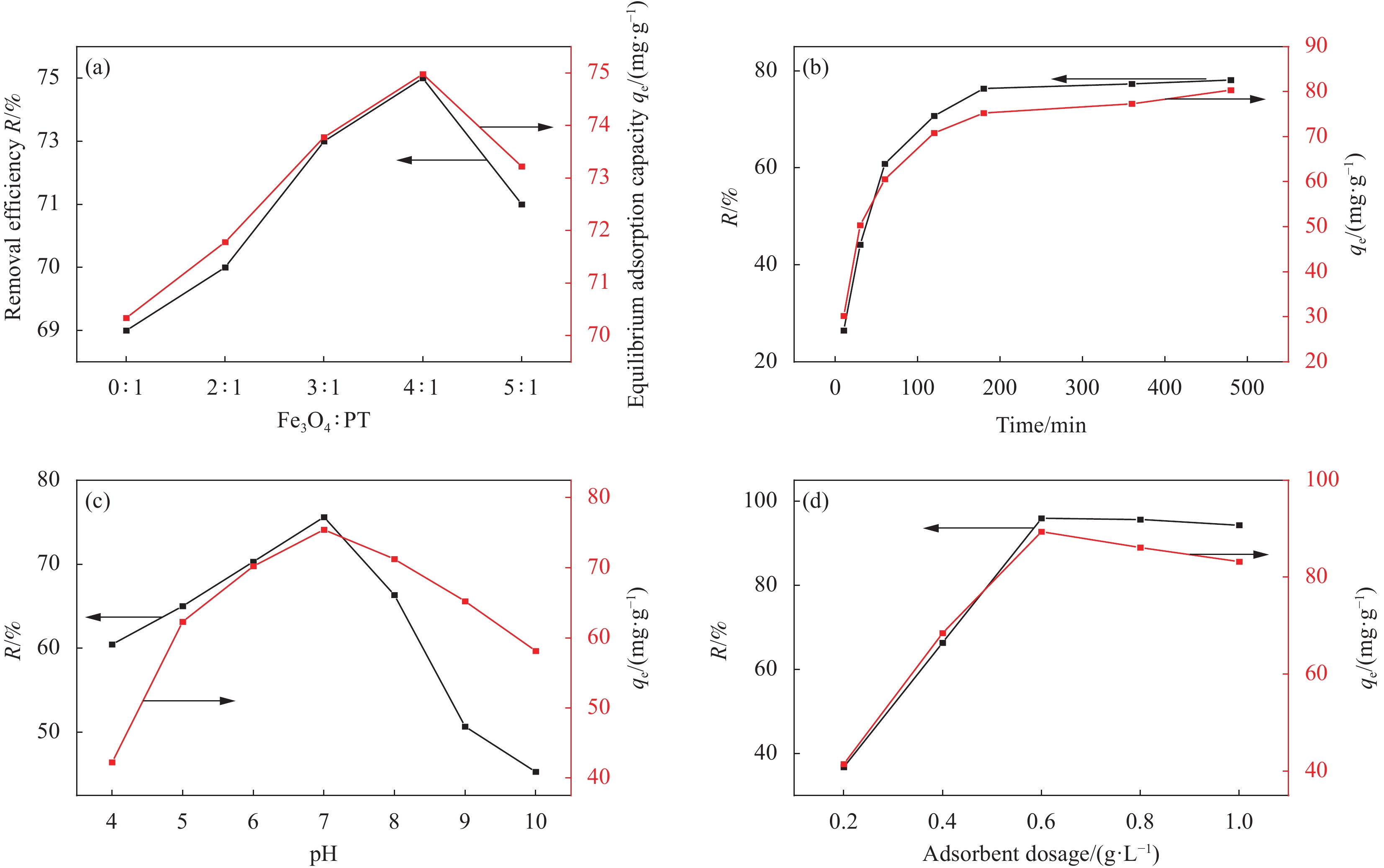
通过探究反应物的不同摩尔比、反应温度、反应时间及pH对所制备材料的光催化性能的影响。从图8(a)~8(d)可以看出:当Bi2O3与BiOI的摩尔比为0.8∶1时,在pH=5、180℃下反应20 h得到的Bi2O3/BiOI复合光催化材料对四环素的降解效果最佳,在3 h内对四环素的降解率可达75%。
2.6.2 光催化性能
通过在模拟太阳光照射下降解四环素来评价所制备样品的光催化性能,图9(a)为模拟太阳光照射下降解四环素的曲线图。在3 h内,BiOI、Bi2O3、Bi2O3/BiOI对四环素的降解率分别为55%、57%、75%。图9(b)为模拟太阳光照射下降解四环素的动力学曲线,Bi2O3/BiOI的动力学速率常数(0.007 min−1)分别是BiOI(0.004 min−1)、Bi2O3(0.0045 min−1)的1.75倍、1.56倍。因此,所制备出的Bi2O3/BiOI具有较高的光催化活性。
2.7 Bi2O3/BiOI复合光催化材料降解四环素的机制
使用对苯醌(BQ)、乙二胺四乙酸二钠盐(EDTA-2Na)、异丙醇(IPA)作为·O2−、h+、·OH的捕捉剂,实验结果如图10所示。BiOI对四环素的降解率分别为50%、31%、53%、,由此可以得出h+是BiOI降解四环素的主要活性物质。Bi2O3对四环素的降解率分别为55%、52%、35%,由此可以得出·OH是Bi2O3降解四环素的主要活性物质。
当Bi2O3和BiOI复合后形成Z型异质结时,Bi2O3/BiOI光催化材料对四环素的降解率分别为35%、66%、66%,由此可以得出·O2−是光催化材料降解四环素的主要活性物质。
Bi2O3/BiOI异质结的形成可以提高光生电子空穴对的分离速率,从而提高光催化活性。BiOI 、Bi2O3的导带和价带可以通过下式计算:
ECB=X−Ee−0.5Eg Eg=EVB−ECB 式中:X为半导体的电负性,BiOI和Bi2O3的X值分别为5.94 eV 和6.23 eV;Ee为自由电子在氢标上的能量(约为4.50 eV);Eg为对应的带隙能量,由图3(b)可以得出BiOI和Bi2O3的Eg值分别为1.82 eV和2.60 eV。因此,BiOI和Bi2O3的导带(CB)边缘分别位于0.53 eV和0.43 eV,BiOI和Bi2O3的价带(VB)边缘分别位于2.35 eV和3.03 eV。在可见光照射下所制备的Bi2O3/BiOI异质结被激发并生成光生载流子且BiOI比Bi2O3的CB更正。事实上,在Bi2O3/BiOI异质结中,光子能量会激发BiOI CB上的电子到更高的电位位置(−0.68 eV),因此BiOI CB上的光生电子会转移到Bi2O3的CB上。同时,Bi2O3 VB上的空穴将转移到BiOI的VB上。而Bi2O3 CB上的电子不能与O2反应生成·O2−(O2/·O2−=−0.33 eV),·O2−是降解四环素的主要活性物质,这与捕获实验的结果不一致。结合以上结果,提出了一种更可能的光催化机制,如图11所示。BiOI和Bi2O3在可见光照射下都能产生光生电子空穴对,Bi2O3 CB上的光生电子和BiOI VB上的空穴在库仑力的作用下会重新组合。此外,BiOI的CB上的光生电子可以与O2反应生成·O2−,然后·O2−与TC反应,有效地实现了Bi2O3/BiOI异质结的光催化降解过程。综上所述,Bi2O3/BiOI异质结能够有效提高光生电子空穴对的分离效率,从而显著提高光催化性能。
3. 结 论
(1) 以五水硝酸铋为原料,采用溶剂热法制备了Bi2O3/BiOI复合光催化材料,在制备过程中加入Bi2O3可以提高单一BiOI的光催化性能,在3 h内对四环素的降解率为75%,是单一BiOI降解速率的1.75倍。
(2) BiOI、Bi2O3成功复合在一起并形成了异质结结构,Bi2O3/BiOI复合光催化材料通过提高光生电子空穴对的分离速率从而提高光催化活性。
(3) 降解机制研究表明,·O2−在降解四环素中起主要作用,且所制得的复合材料可应用于对四环素的降解,并有望进一步用于对其他抗生素的降解处理以解决实际问题。
-
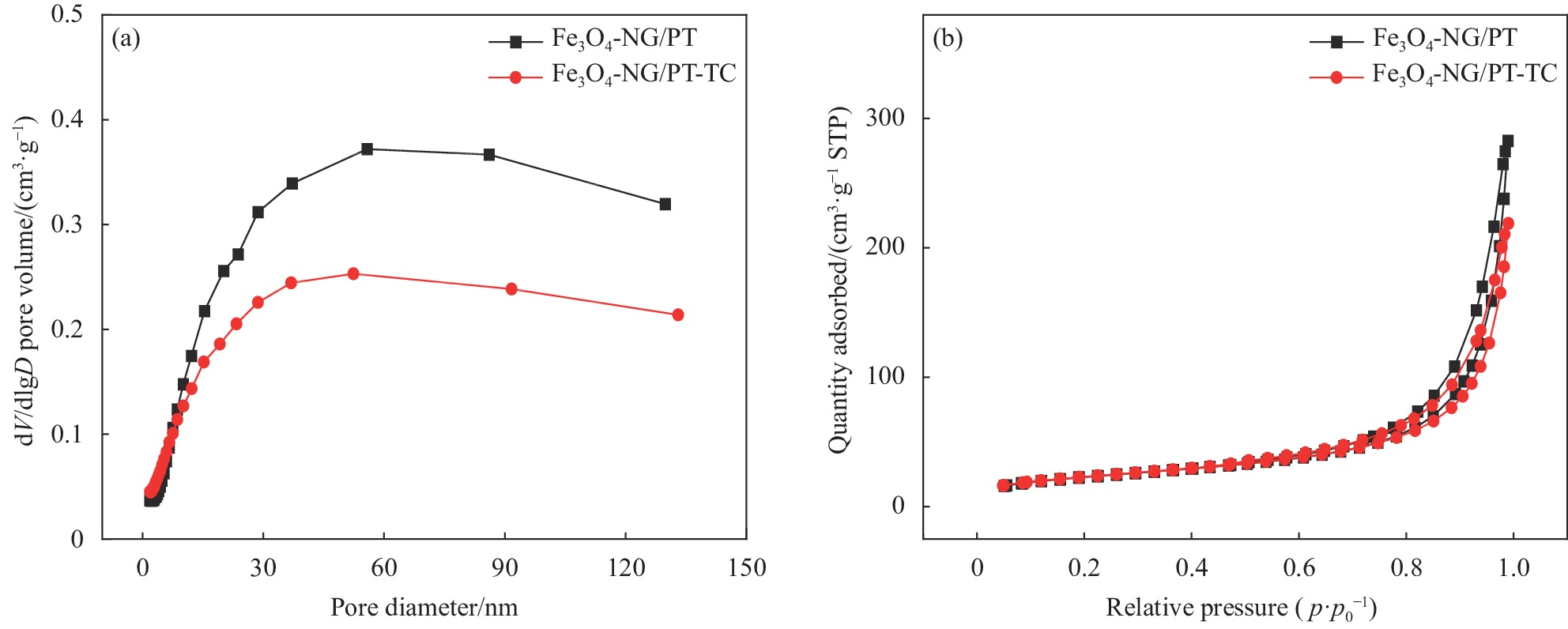
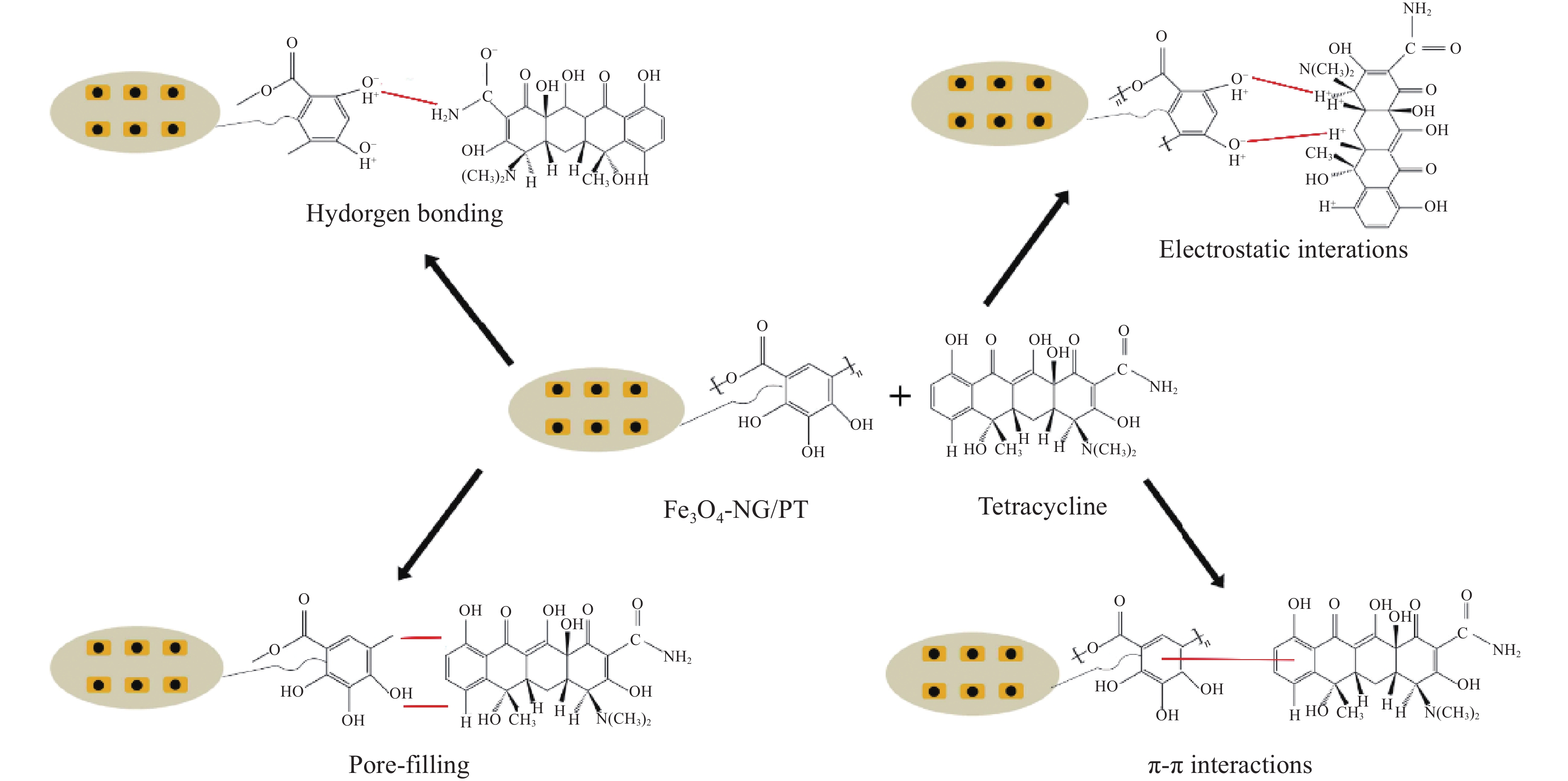
图 3 ((a), (b)) NG/PT、Fe3O4-NG/PT的SEM图像;((c), (d)) Fe3O4-NG/PT吸附TC前后的SEM图像;((e), (i)) C元素吸附前后元素分布图;((f), (j)) O元素吸附前后元素分布图;((g), (k)) N元素吸附前后元素分布图;((h), (l)) Fe元素吸附前后元素分布图
Figure 3. ((a), (b)) SEM images of NG/PT and Fe3O4-NG/PT; ((c), (d)) SEM images of Fe3O4-NG/PT before and after TC adsorption; ((e), (i)) Elemental distribution before and after adsorption of element C; ((f), (j)) Elemental distribution before and after adsorption of element O; ((g), (k)) Elemental distribution before and after adsorption of element N; ((h), (l)) Elemental distribution before and after adsorption of element Fe
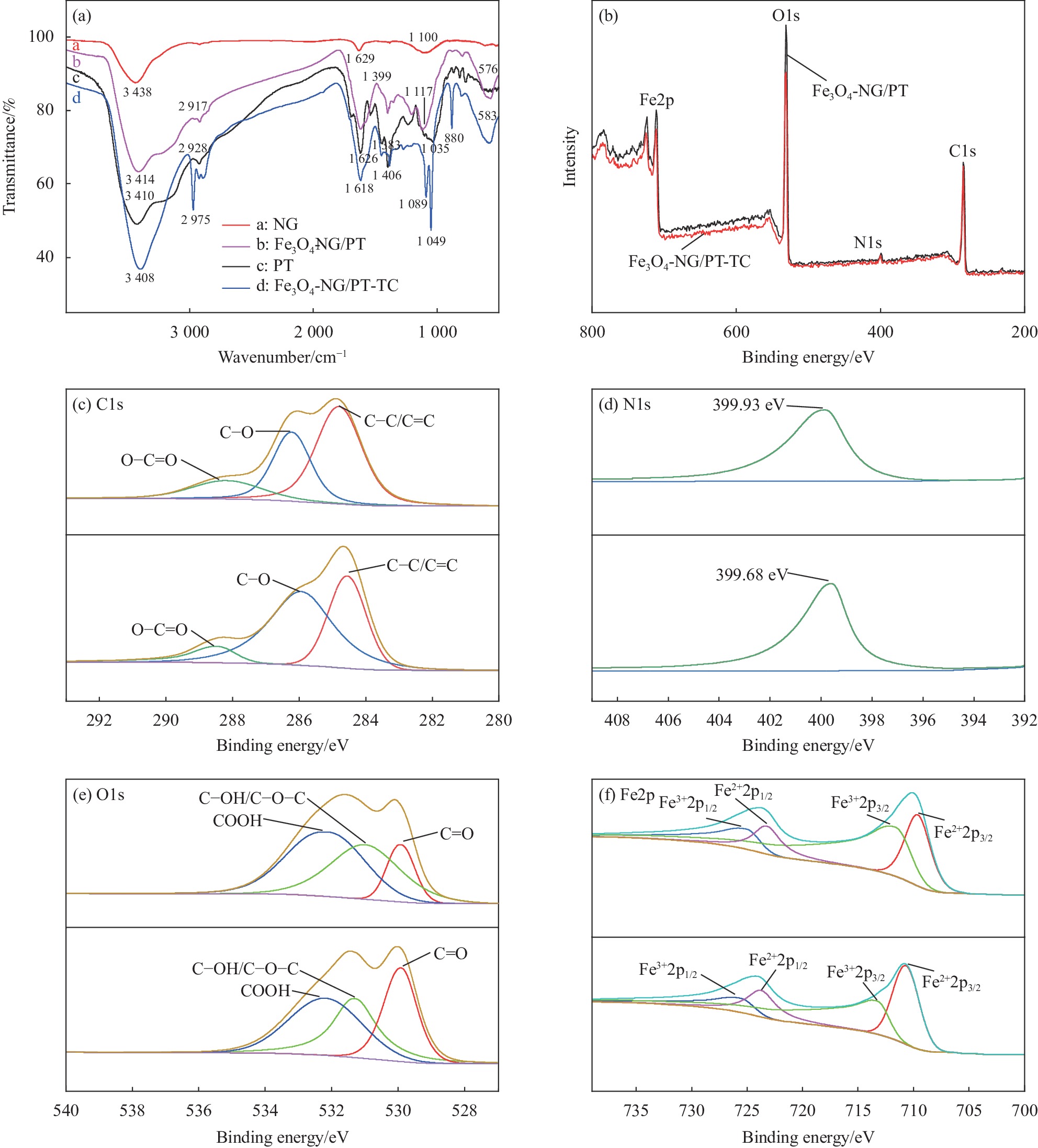
图 4 (a) NG、PT、Fe3O4-NG/PT、Fe3O4-NG/PT吸附TC后的FTIR图谱;(b) Fe3O4-NG/PT和Fe3O4-NG/PT吸附TC后的XPS图谱;(c) C1s峰;(d) N1s峰;(e) O1s峰;(f) Fe2p峰
Figure 4. (a) FTIR spectra of NG, PT, Fe3O4-NG/PT and Fe3O4-NG/PT after TC adsorption; (b) XPS spectra of Fe3O4-NG/PT and Fe3O4-NG/PT after TC adsorption; (c) C1s peak; (d) N1s peak; (e) O1s peak; (f) Fe2p peak
图 8 Fe3O4-NG/PT吸附TC的动力学拟合:(a) 不同TC浓度相同时间下对吸附量的影响;(b) Fe3O4-NG/PT吸附TC的拟一级动力学拟合图;(c) Fe3O4-NG/PT吸附TC的拟二级动力学拟合图;(d) Fe3O4-NG/PT吸附TC的内扩散拟合图
Figure 8. Kinetic fitting of TC adsorption by Fe3O4-NG/PT: (a) Effect of different TC concentrations for the same time on the amount of adsorption; (b) Proposed primary kinetic fitting plot of TC adsorption by Fe3O4-NG/PT; (c) Proposed secondary kinetic fitting plot of TC adsorption by Fe3O4-NG/PT; (d) Fitting of internal diffusion of TC adsorbed by Fe3O4-NG/PT
qt—Adsorption capacity of time t
-
[1] 高艳林, 景红霞, 李龙祥, 等. 溶剂热法制备Bi2O3/BiOI复合光催化材料及对四环素的降解应用[J]. 复合材料学报, 2022, 39(2):677-684. GAO Yanlin, JING Hongxia, LI Longxiang, et al. Preparation of Bi2O3/BiOI composite photocatalytic material by solvothermal method and its application to the degradation of tetracycline[J]. Acta Materiae Compositae Sinica,2022,39(2):677-684(in Chinese).
[2] LI X, LIU C, CHEN Y, et al. Antibiotic residues in liquid manure from swine feedlot and their effects on nearby groundwater in regions of North China[J]. Environmental Science and Pollution Research,2018,25(12):11565-11575. DOI: 10.1007/s11356-018-1339-1
[3] EKAMBARAM S P, PERUMALPE S S, BALAKRISHNAN A. Scope of hydrolysable tannins as possible antimicrobial agent[J]. Phytotherapy Research,2016,30(7):1035-1045. DOI: 10.1002/ptr.5616
[4] XU Y, GUO C, LUO Y, et al. Occurrence and distribution of antibiotics, antibiotic resistance genes in the urban rivers in Beijing, China[J]. Environmental Pollution,2016,213:833-840. DOI: 10.1016/j.envpol.2016.03.054
[5] GRENDA K, AMOLD J, GAMELAS A F, et al. Up-scaling of tannin-based coagulants for wastewater treatment: Performance in a water treatment plant[J]. Environmental Science and Pollution Research,2020,27(2):1202-1213. DOI: 10.1007/s11356-018-2570-5
[6] ARBENZ A, AV'EROUS L. Chemical modification of tannins to elaborate aromatic biobased macromolecular architectures[J]. Green Chemistry,2015,17(5):2626-2646. DOI: 10.1039/C5GC00282F
[7] GEORGIEVA V G, GONSALVESH L, TAVLIEVA M P. Thermodynamics and kinetics of the removal of nickel (II) ions from aqueous solutions by biochar adsorbent made from agro-waste walnut shells[J]. Journal of Molecular Liquids,2020,312:112788. DOI: 10.1016/j.molliq.2020.112788
[8] 袁帅, 赵雪松, 王丽, 等. 磁性松塔/松针基活性炭的制备及对Pb~(2+)吸附性能研究[J]. 应用化工, 2022, 51(6):1675-1680. DOI: 10.3969/j.issn.1671-3206.2022.06.028 YUAN Shuai, ZHAO Xuesong, WANG Li, et al. Study on preparation of activated carbon based on magnetic pine cone/pine needle and adsorption properties of Pb~(2+)[J]. Applied Chemical Industry,2022,51(6):1675-1680(in Chinese). DOI: 10.3969/j.issn.1671-3206.2022.06.028
[9] 杨奇亮, 吴平霄. 改性多孔生物炭的制备及其对水中四环素的吸附性能研究[J]. 环境科学学报, 2019, 39(12):3973-3984. DOI: 10.13671/j.hjkxxb.2019.0235 YANG Qiliang, WU Pingxiao. Preparation of modified porous biochar and its adsorption properties for tetracycline in water[J]. Acta Scientiae Circumstantiae,2019,39(12):3973-3984(in Chinese). DOI: 10.13671/j.hjkxxb.2019.0235
[10] WANG Z M, GAO M M, LI X J, et al. Efficient adsorption of methylene blue from aqueous solution by graphene oxide modified persimmon tannins[J]. Materials Science and Engineering: C,2020,108:110196. DOI: 10.1016/j.msec.2019.110196
[11] GAO L, WANG Z M, QIN C K, et al. Preparation and application of iron oxide/persimmon tannin/graphene oxide nanocomposites for efficient adsorption of erbium from aqueous solution[J]. Journal of Rare Earths,2020,38(12):1344-1353. DOI: 10.1016/j.jre.2020.02.003
[12] ZHANG Y P, ZHANG C Q, PARKER D B, et al. Occurrence of antimicrobials and antimicrobial resistance genes in beef cattle storage ponds and swine treatment lagoons[J]. Science of the Total Environment,2013,463:631-638.
[13] AZANU D, STYRISHAVE B, DARKO G, et al. Occurrence and risk assessment of antibiotics in water and lettuce in Ghana[J]. Science of the Total Environment,2018,622:293-305.
[14] BOXALL A B, FOGG L, BACKWELL P, et al. Veterinary medicines in the environment[J]. Reviews of Environmental Contamination and Toxicology,2004,199:1-91.
[15] CABELLO F C, GODFREY H P, TOMOVA A, et al. Antimicrobial use in aquaculture re-examined: Its relevance to antimicrobial resistance and to animal and human health[J]. Environmental Microbiology,2013,15(7):1917-1942. DOI: 10.1111/1462-2920.12134
[16] LI S Z, GONG Y B, YANG Y C, et al. Recyclable CNTs/Fe3O4 magnetic nanocomposites as adsorbents to remove bisphenol A from water and their regeneration[J]. Chemical Engineering Journal,2015,26:231-239.
[17] ZHOU Z, HUANG Y, LIANG J, et al. Extraction, purification and anti-radiation activity of persimmon tannin from Diospyros kaki L.f.[J]. Journal of Environmental Radioactivity,2016,162-163:182-188. DOI: 10.1016/j.jenvrad.2016.05.034
[18] TENKORANG A, YEBOAH A M, BUAMAH R, et al. Promoting sustainable sanitation through wastewater-fed aquaculture: A case study from Ghana[J]. Water International,2012,37(7):831-842. DOI: 10.1080/02508060.2012.733669
[19] 叶林静, 关卫省, 卢勋, 等. 改性纳米Fe3O4去除水溶液中四环素的研究[J]. 安全与环境学报, 2014, 14(1):202-207. YE Linjing, GUAN Weisheng, LU Xun, et al. Study on the removal of tetracycline from aqueous solution by modified Fe3O4 nanoparticles[J]. Journal of Safety and Environment,2014,14(1):202-207(in Chinese).
[20] HARNISZ M, KORZENIEWSKA E, GOLAS I. The impact of a freshwater fish farm on the community of tetracycline-resistant bacteria and the structure of tetracycline resistance genes in river water[J]. Chemosphere,2015,128:134-141. DOI: 10.1016/j.chemosphere.2015.01.035
[21] GUPTA V K, FAKHRI A, AGARWAL S, et al. Synthesis and characterization of MnO2/NiO nanocomposites for photocatalysis of tetracycline antibiotic and modification with guanidine for carriers of Caffeic acid phenethyl ester-an anticancer drug[J]. Journal of Photochemistry and Photobiology B: Biology,2017,174:235-242. DOI: 10.1016/j.jphotobiol.2017.08.006
[22] ZHANG L, ZHANG T, DONG L, et al. Assessment of halogenated POPs and PAHs in three cities in the Yangtze River Delta using high-volume samplers[J]. Science of the Total Environment,2013,454:619-626.
[23] LU Y, LI S Z. Preparation of hierarchically interconnected porous banana peel activated carbon for methylene blue adsorption[J]. Journal of Wuhan University of Technology (Materials Science), 2019, 34(2): 472-480.
[24] LUNDSTROM S V, OSTAMN M, BENGTSSON P J, et al. Minimal selective concentrations of tetracycline in complex aquatic bacterial biofilms[J]. Science of the Total Environment,2016,553:587-595. DOI: 10.1016/j.scitotenv.2016.02.103
[25] TORRES-ACOSTA M A, PEREIRA J F B, FREIRE M G, et al. Economic evaluation of the primary recovery of tetracycline with traditional and novel aqueous two-phase systems[J]. Separation and Purification Technology,2018,203:178-184. DOI: 10.1016/j.seppur.2018.04.041
[26] MIN Z, SHENG Y L, LIANG J Z, et al. Occurrence, fate and mass loadings of antibiotics in two swine wastewater treatment systems[J]. Science of the Total Environment,2018,639:142-148.
[27] DU Z J, ZHANG Y, LI Z J, et al. Facile one-pot fabrication of nano-Fe3O4/carboxyl-functionalized baker's yeast composites and their application in methylene blue dye adsorption[J]. Applied Surface Science,2017,392:312-320. DOI: 10.1016/j.apsusc.2016.09.050
[28] ZHANG X, LIN Q, ZHANG X, et al. A novel 3D conductive network-based polyaniline/graphitic mesoporous carbon composite electrode with excellent electrochemical performance[J]. Journal of Power Sources,2018,401:278-286. DOI: 10.1016/j.jpowsour.2018.08.091
[29] LI T, LI M, ZHANG M, et al. Immobilization of Fe3N nanoparticles within N-doped carbon nanosheet frameworks as a high-efficiency electrocatalyst for oxygen reduction reaction in Zn-air battery[J]. Carbon,2019,153:364-371. DOI: 10.1016/j.carbon.2019.07.044
[30] DENG W J, LI N, YING G G. Antibiotic distribution, risk assessment, and microbial diversity in river water and sediment in Hong Kong[J]. Environmental Geochemistry & Health,2018,40(5):2191-2203.
[31] XIE A, DAI J, CHEN X, et al. Ultrahigh adsorption of typical antibiotics onto novel hierarchical porous carbons derived from renewable lignin via halloysite nanotubes-template and in-situ activation[J]. Chemical Engineering Journal,2016,304:609-620. DOI: 10.1016/j.cej.2016.06.138
[32] YANG J, DAI J, WANG L, et al. Ultrahigh adsorption of tetracycline on willow branche-derived porous carbons with tunable pore structure: Isotherm, kinetics, thermodynamic and new mechanism study[J]. Journal of the Taiwan Institute of Chemical Engineers,2019,96:473-482. DOI: 10.1016/j.jtice.2018.12.017
[33] TANG J, WANG S, FAN J, et al. Predicting distribution coefficients for antibiotics in a river water sediment using quantitative models based on their spatiotemporal variations[J]. Science of the Total Environment,2019,65:1301-1310.
[34] 赵丹华, 张少丽, 陈作义, 等. CaC2O4-直接枣红B/壳聚糖复合材料的制备及对乙基紫的吸附性能[J]. 复合材料学报, 2022, 39(2):736-745. ZHAO Danhua, ZHANG Shaoli, CHEN Zuoyi, et al. Preparation of CaC2O4-direct claret B/chitosan composite materials and adsorption properties for ethyl violet[J]. Acta Materiae Compositae Sinica,2022,39(2):736-745(in Chinese).
[35] 范甲, 胡世琴, 魏柏, 等. 磁性γ-Fe2O3/玉米秸秆淀粉的制备及其对废水中U(VI)吸附性能[J]. 复合材料学报, 2022, 39(10):4898-4907. FAN Jia, HU Shiqin, WEI Bai, et al. Preparation of magnetic γ-Fe2O3/corn stalk starch and its adsorption performance for U(VI) in wastewater[J]. Acta Materiae Compositae Sinica,2022,39(10):4898-4907(in Chinese).
[36] LIN Y, XU S, LI J. Fast and highly efficient tetracyclines removal from environmental waters by graphene oxide functionalized magnetic particles[J]. Chemical Engineering Journal,2013,225:679-685. DOI: 10.1016/j.cej.2013.03.104
[37] KHAN M H, BAE H, JUNG J. Tetracycline degradation by ozonation in the aqueous phase: Proposed degradation intermediates and pathway[J]. Journal of Hazardous Materials,2010,181(1):659-665. DOI: 10.1016/j.jhazmat.2010.05.063
-
期刊类型引用(8)
1. 谭浩,张文彬,卢文玉,祁志强,蔡红珍,杨科研. 高粱秸秆负载HKUST-1对四环素的吸附行为与机制. 复合材料学报. 2025(01): 514-526 .  本站查看
本站查看
2. 李碧秋,李希成,熊俊夫,李金韩,贾博雅,汪长征. 铋系光电催化剂降解水中有机污染物的研究进展. 材料工程. 2024(06): 92-108 .  百度学术
百度学术
3. 黄鹏伟,李静,林博,王宜民,陈仪,谢楠耿. AgI/BiOI异质结光电催化甲醇氧化. 山西化工. 2024(07): 4-6+10 .  百度学术
百度学术
4. 杜书雅,王旭东,董永浩,吕嘉辰,李洁. MOF-808/AgBr的制备及光催化降解盐酸四环素性能研究. 功能材料. 2024(11): 11137-11146 .  百度学术
百度学术
5. 严惠儒,林水源,钟祥康,黄学帅,杨玉如,冯梓盈,朱淼,谢伟. Au/BiOI花状微米球的制备及其对抗生素的降解特性. 广州化工. 2023(05): 43-46 .  百度学术
百度学术
6. 王振宇,刘燕才,陈琨,乔江浩,李晓伟. 等离子喷涂-化学气相沉积制备α/β-Bi_2O_3薄膜的相结构调控和光催化降解性能. 硅酸盐学报. 2023(07): 1800-1810 .  百度学术
百度学术
7. 刘松林,王仲民,钱熹,王童,冉兆晋,黄志民,吴晨曦,李桂银. 磁性氮掺杂石墨烯改性柿单宁复合材料对四环素的吸附行为. 复合材料学报. 2023(07): 4048-4059 .  本站查看
本站查看
8. 袁亦雷,谢水波,刘岳林,史艳丹,刘迎九. g-C_3N_4-Bi_2O_3/Al_2O_3复合材料光催化还原水中U(Ⅵ). 化工环保. 2022(05): 609-615 .  百度学术
百度学术
其他类型引用(8)
-
-
磁性氮掺杂石墨烯改性柿单宁材料对四环素的吸附行为研究
柿单宁材料以其比表面积大、表面官能团丰富、成本低廉、环保等优点,在吸附领域吸引了越来越多的研究。但由于其具有亲水性,导致其在吸附过程的分离较差,回收困难,阻碍了其吸附领域的应用。
以柿单宁(PT)为基体,通过氮掺杂石墨烯(NG)对其改性,并以四氧化三铁(Fe3O4)作为磁性功能,采用水热法制得磁性复合材料(Fe3O4-NG/PT)。氮掺杂石墨烯以其疏水性和比表面积大的优势,弥补了柿单宁在吸附上的不足,四氧化三铁解决了吸附剂难以回收的问题。针对此复合材料在吸附四环素方面的应用进行研究,通过SEM、EDS、FTIR、XPS、BET表明其机制主要是静电作用、孔填充、氢键作用和π-π相互作用;通过XRD与磁强计表明Fe3O4-NG/PT 的成功引入以及其高效回收性能。并对吸附条件优化,在Fe3O4:PT为4 :1,PH为7,投加量为0.6 g/L,吸附180min吸附效果最佳。研究其磁性复合材料对四环素的吸附过程符合拟二级动力学模型和Freundlich模型,其主要为化学吸附为主的多层吸附,308K时吸附的饱和容量达到315.65 mg/g,吸附效率达到94.5%。
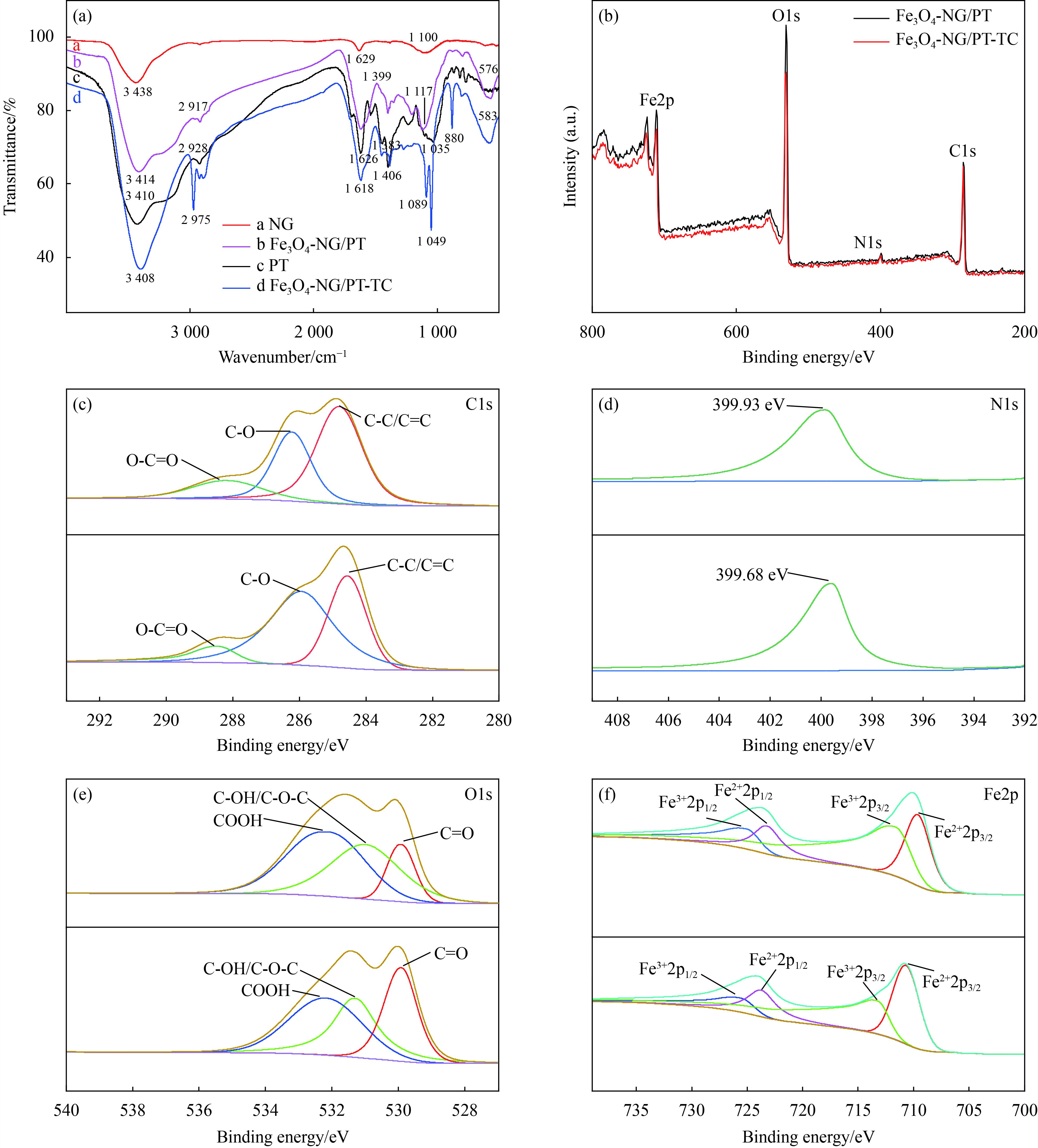
(A) FTIR profiles of NG, PT, Fe3O4-NG/PT and Fe3O4-NG/PT after TC adsorption (B) XPS profiles of Fe3O4-NG/PT and Fe3O4-NG/PT after TC adsorption (C) C1s peak (D) N1s peak (E) O1s peak (F) Fe2p






 下载:
下载: