Preparation and properties of bifunctional magnetic fluorescent core-shell nanoparticles
-
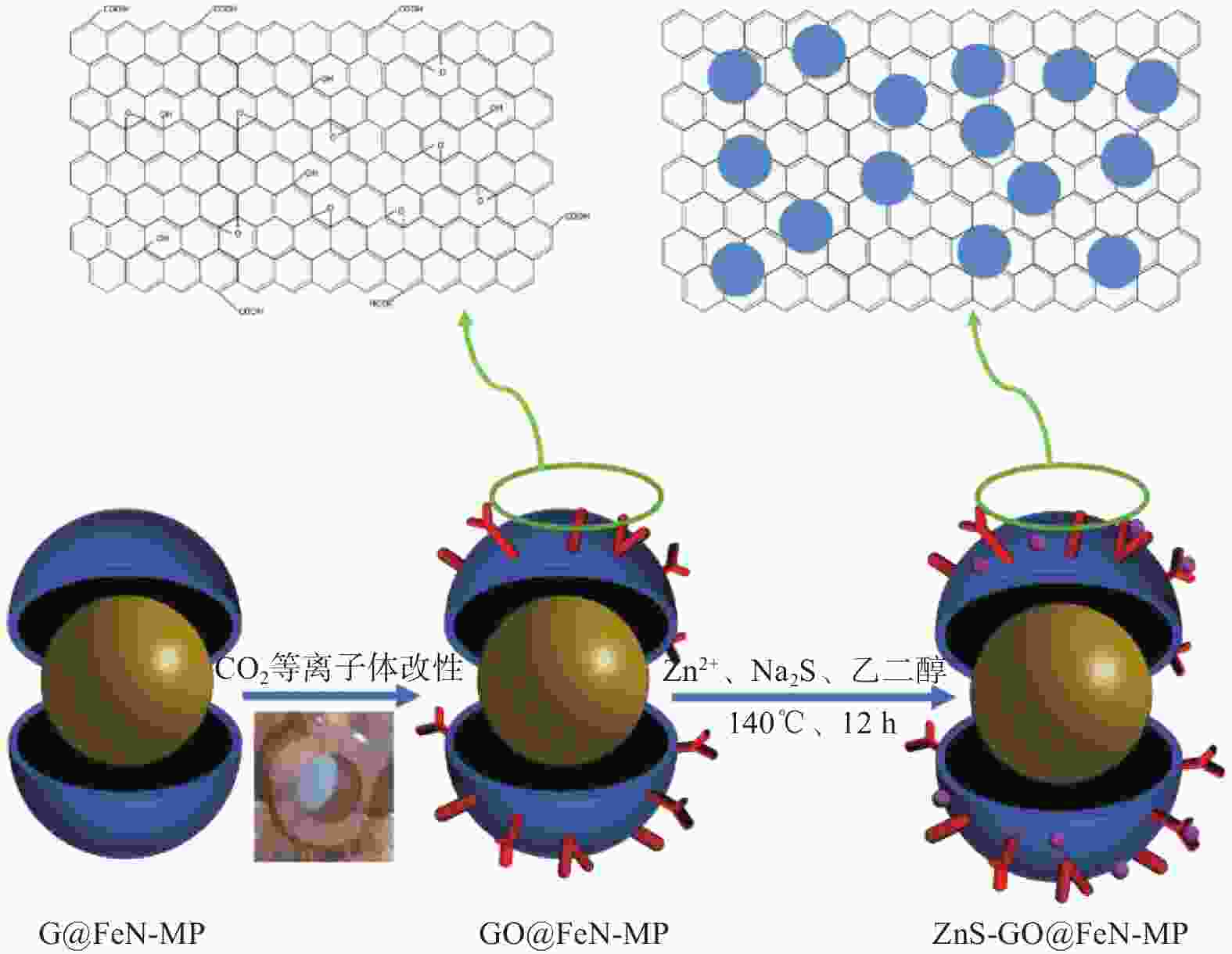
摘要: 采用等离子体表面改性技术对石墨烯包覆氮化铁磁性纳米颗粒 (G@FeN-MP)进行CO2等离子体改性得到氧化石墨烯包覆氮化铁磁性纳米颗粒 (GO@FeN-MP)。通过溶剂热法进一步制备了硫化锌修饰的氧化石墨烯包覆氮化铁磁性纳米颗粒 (ZnS-GO@FeN-MP),利用X射线衍射 (XRD)、X射线光电子能谱 (XPS)、透射电子显微镜 (TEM)、傅立叶变换红外光谱 (FTIR)、紫外可见光谱 (UV)、光致发光光谱 (PL)和拉曼光谱 (Raman)对复合材料进行表征,同时进行A549细胞毒性评估与ZnS-GO@FeN-MP偶联二抗的联合应用揭示了其在实验生物学功能中的潜在应用,研究结果确认了该纳米复合材料的优良生物相容性。Abstract: Graphene coated ferronitride magnetic nanoparticles (G@FeN-MP) were modified by CO2 plasma surface modification technology to obtain graphene oxide coated ferronitride magnetic nanoparticles (GO@FeN-MP). Zinc sulfide modified graphene oxide coated ferronitride magnetic nanoparticles (ZnS-GO@FeN-MP) were prepared by solvothermal method, and the composites were characterized by X-ray diffraction (XRD), X-ray photoelectron spectroscopy (XPS), transmission electron microscopy (TEM), Fourier transform infrared spectroscopy (FTIR), ultraviolet visible spectroscopy (UV), photoluminescence spectroscopy (PL) and Raman spectroscopy (Raman), and concurrent cytotoxicity assessment of A549 cells and the coupled application of ZnS-GO@FeN-MP secondary antibodies have unveiled their potential in experimental biological functions, with study results affirming the excellent biocompatibility of this nanocomposite material.
-
Key words:
- Core-shell magnetic nanoparticles /
- plasma /
- fluorescence /
- ZnS /
- biocompatibility
-
图 4 G@FeN-MP、GO@FeN-MP、ZnS-GO@FeN-MP-1和ZnS-GO@FeN-MP-2的(a)全谱图 和(b) C 1s高分辨XPS谱图; ZnS-GO@FeN-MP-1和ZnS-GO@FeN-MP-2的(c) Zn 2p的高分辨XPS谱图和(d) S 2p的高分辨XPS谱图
Figure 4. (a) The full spectrum and (b) C 1s of G@FeN-MP, GO@FeN-MP, ZnS-GO@FeN-MP-1and ZnS-GO@FeN-MP-2; (c) high-resolution Zn 2p and (d) S 2p spectra of ZnS-GO@FeN-MP-1and ZnS-GO@FeN-MP-2
表 1 实验合成材料统计表
Table 1. Statistical table of experimental synthetic materials:
Material Name Synthetic Raw Material Synthetic Method G@FeN-MP Fe、Fe3O4、Fe3N、C Microwave plasma
GO@FeN-MPG@FeN-MP CO2 plasma modification
ZnS-GO@FeN-MP-1GO@FeN-MP(20-50 mg)、ZnSO4·7H2O(1 mmoL,0.28756 g)、Na2S·9H2O
(1 mmoL,0.24018 g)、ethanediol (15 mL)
Solvothermal method
ZnS-GO@FeN-MP-2GO@FeN-MP(20-50 mg)、Na2S·9H2O(1 mmoL,0.24018 g)、Zn(CH3COO)2·2H2O(1 mmoL,0.21951 g)、ethanediol (15 mL)
Solvothermal method表 2 1 mg ZnS-GO@FeN-MP-1在450 nm下的吸光度值
Table 2. Absorbance value of 1 mg ZnS-GO@FeN-MP-1 at 450 nm
ZnS-GO@FeN-MP-1 Second Antibody -ZnS-GO@FeN-MP-1 Second Antibody 0.24 0.285 0.385 0.164 0.321 0.371 0.205 0.248 0.364 Average value 0.203 0.285 0.373 表 3 1 mg ZnS-GO@FeN-MP-2在450 nm下的吸光度值
Table 3. Absorbance value of 1 mg ZnS-GO@FeN-MP-2 at 450 nm
ZnS-GO@FeN-MP-2 Second Antibody-ZnS-GO@FeN-MP-2 Second Antibody 0.600 0.821 0.385 0.581 0.766 0.371 0.585 0.877 0.364 Average value 0.589 0.821 0.373 -
[1] REN Z Q, ZHU X Q, LV H H, et al. A fluorescent method based on magnetic nanoparticles for detection of CGG trinucleotide repeat genes[J]. New Journal of Chemistry, 2019, 43: 1322-1327. doi: 10.1039/C8NJ04686G [2] AKSHAY P, VIJAY M, SOURAV T, et al. Nanotechnology Derived Nanotools in Biomedical Perspectives: An Update[J]. Current Nanoscience, 2019, 15(2): 137-146. doi: 10.2174/1573413714666180426112851 [3] JIANG L, LIU S, ZHANG D, et al. Catechol-formaldehyde resin microspheres@UiO-66-NH2@CdS core-shell nanohybrids as robust visible-light photocatalyst for multifunctional applications[J]. Materials Today Chemistry, 2024, 35: 101875. doi: 10.1016/j.mtchem.2023.101875 [4] HOSSAIN M D, MAYANOVIC R A, SAKIDJA R, et al. Magnetic properties of core-shell nanoparticles possessing a novel Fe(II)-chromia phase: an experimental and theoretical approach[J]. Nanoscale, 2018, 10: 2138-2147. doi: 10.1039/C7NR04770C [5] CHEN F, LI Y, LIN X, et al. Polymeric Systems Containing Supramolecular Coordination Complexes for Drug Delivery[J]. Polymers, 2021, 13(3): 370. doi: 10.3390/polym13030370 [6] NONG J, GLASSMAN P M, MUZYKANTOV V R. Targeting vascular inflammation through emerging methods and drug carriers[J]. Advanced Drug Delivery Reviews, 2022, 184: 114180. doi: 10.1016/j.addr.2022.114180 [7] BEKOVOC M, BAN I, DROFENIK M, et al. Magnetic nanoparticles as mediators for magnetic hyperthermia therapy applications: a status review[J]. Applied Sciences, 2023, 13(17): 9548. doi: 10.3390/app13179548 [8] LIU X L, ZHENG J J, SUN W, et al. Ferrimagnetic Vortex Nanoring-Mediated Mild Magnetic Hyperthermia Imparts Potent Immunological Effect for Treating Cancer Metastasis[J]. ACS Nano, 2019, 13(8): 8811-8825. doi: 10.1021/acsnano.9b01979 [9] RAMACHANDRAN A, JANA M, KUMAR A, et al. Evaluation of Infantile Brachial Plexopathy Using 3T MRI and High-Resolution Ultrasound: Experience From a Tertiary Care Centre[J]. Neurology India, 2024, 72(2): 326-333. doi: 10.4103/ni.ni_818_21 [10] MUI T S M, SILVA L L G, PRYSIAZHNYI V, et al. Surface modification of aluminium alloys by atmospheric pressure plasma treatments for enhancement of their adhesion properties[J]. Surface and Coatings Technology, 2017, 312: 32-36. doi: 10.1016/j.surfcoat.2016.08.024 [11] MOOSBURGER-WILL J, LACHNER E, LOEFFLER M, et al. Adhesion of carbon fibers to amine hardened epoxy resin: Influence of ammonia plasma functionalization of carbon fibers[J]. Applied Surface Science, 2018, 453: 141-152. doi: 10.1016/j.apsusc.2018.05.057 [12] CROISSANT J G, FATIEIEV Y, ALMALIK A, et al. Mesoporous Silica and Organosilica Nanoparticles: Physical Chemistry, Biosafety, Delivery Strategies, and Biomedical Applications[J]. Advanced Healthcare Materials, 2018, 7(4): 1700831. doi: 10.1002/adhm.201700831 [13] SOBHANAN J, ANAS A, BIJU V. Nanomaterials for Fluorescence and Multimodal Bioimaging[J]. Chemical record, 2023, 23(3): 1527. [14] MCHUGH K J, JING L, BEHRENS A M, et al. Biocompatible Semiconductor Quantum Dots as Cancer Imaging Agents[J]. Advanced Materials, 2018, 30(18): 1706356. doi: 10.1002/adma.201706356 [15] ZHANG J, WANG L, CHEN F, et al. Optical properties of multinary copper chalcogenide semiconductor nanocrystals and their applications in electroluminescent devices[J]. Chinese Science Bulletin, 2021, 66(17): 2162-2178. doi: 10.1360/TB-2020-1633 [16] ORTAKAYA S, DUQUE C A. Infrared relative dielectric permittivity and refractive index on the spherical CdSe/ZnS heteronanocrystals with self-consistent field[J]. Physica Scripta, 2024, 99(4): 045968. doi: 10.1088/1402-4896/ad328a [17] FONTENOT R S, ALLISON S W, LYNCH K J, et al. Mechanical, spectral, and luminescence properties of ZnS: Mn doped PDMS[J]. Journal of Luminescence, 2016, 170: 194-199. doi: 10.1016/j.jlumin.2015.10.047 [18] AKHGARI F, FATTAHI H, OSKOEI Y M, et al. Recent advances in nanomaterial-based sensors for detection of trace nitroaromatic explosives[J]. Sensors & Actuators B Chemical, 2015, 221: 867-878. [19] DEVADOSS A, SUDHAGAR P, TERASHIMA C, et al. Photoelectrochemical biosensors: New insights into promising photoelectrodes and signal amplification strategies[J]. Journal of Photochemistry and Photobiology C: Photochemistry Reviews, 2015, 24: 43-63. doi: 10.1016/j.jphotochemrev.2015.06.002 [20] LI X Q, WANG Q, YU Y F. Surface modification by CO2 plasma boosting core shells structural Fe/Fe3C/FeN@graphite carbon nanoparticles toward high performance microwave absorber[J]. Materials Today Nano, 2024, 25: 100465. doi: 10.1016/j.mtnano.2024.100465 [21] FAN X L, ZHU Y J, LUO C, et al. In situ lithiated FeF3/C nanocomposite as high energy conversion-reaction cathode for lithium-ion batteries[J]. Journal of Power Sources, 2016, 307: 435-442. doi: 10.1016/j.jpowsour.2016.01.004 [22] MAO C M, XU X X, WANG S W, et al. Mass production of porous Fe3O4 nanoparticle networks wrapped with ultrathin nitrogen-doped carbon for superior lithium ion batteries[J]. Applied Surface Science, 2020, 505: 144525. doi: 10.1016/j.apsusc.2019.144525 [23] REN M M, YANG M Z, LIU W L, et al. Ultra-small Fe3O4 nanocrystals decorated on 2D graphene nanosheets with excellent cycling stability as anode materials for lithium ion batteries[J]. Electrochimica Acta, 2016, 194: 219-227. doi: 10.1016/j.electacta.2016.02.091 [24] SHAO M W, WANG D B, YU G H, et al. The synthesis of carbon nanotubes at low temperature via carbon suboxide disproportionation[J]. Carbon, 2004, 42(1): 183-185. doi: 10.1016/j.carbon.2003.10.010 [25] PENG C C, YANG C, CHEN P, et al. Mesoporous carbons and Fe collectively boost the capacity increases upon Long-term cycling of Ni/Fe/NiFe2O4@C anode for Lithium-ion batteries[J]. Applied Surface Science, 2023, 623: 156994. doi: 10.1016/j.apsusc.2023.156994 [26] YUAN K F, NI Y H, ZHANG L. Facile hydrothermal synthesis of polyhedral Fe3O4 nanocrystals, influencing factors and application in the electrochemical detection of H2O2[J]. Journal of Alloys & Compounds, 2012, 532: 10-15. [27] RAO T P, KUMAR M C S, ANGAYARKANNI S A, et al. Effect of stress on optical band gap of ZnO thin films with substrate temperature by spray pyrolysis[J]. Journal of Alloys and Compounds, 2009, 485(1-2): 413-417. doi: 10.1016/j.jallcom.2009.05.116 [28] HUANG N B, DONG W J, FENG Y, et al. Using dopamine interlayers to construct Fe/Fe3C@FeNC microspheres of high N-content for bifunctional oxygen electrocatalysts of Zn–air batteries[J]. Dalton Transactions, 2023, 52(8): 2373-2383. doi: 10.1039/D2DT03522G [29] WANG H, FENG X Y, LIU N, et al. Engineering Fe-Fe3C@Fe-N-C Active Sites and Hybrid Structures from Dual Metal-Organic Frameworks for Oxygen Reduction Reaction in H2-O2 Fuel Cell and Li-O2 Battery[J]. Advanced Functional Materials, 2019, 29(23): 1901531. doi: 10.1002/adfm.201901531 [30] FELTEN A, BITTENCOURT C, PIREAUX J J, et al. Radio-frequency plasma functionalization of carbon nanotubes surface O2, NH3, and CF4 treatments[J]. Journal of Applied Physics, 2005, 98(7): 074308. doi: 10.1063/1.2071455 [31] WANG Y L, ZHENG J X, WANG J L, et al. Rapid microwave-assisted synthesis of highly luminescent nitrogen-doped carbon dots for white light-emitting diodes[J]. Optical Materials, 2017, 73: 319-329. doi: 10.1016/j.optmat.2017.08.032 [32] LIU S, LIU L W, CHEN X W, et al. On an Easy Way to Prepare Fe, S, N Tri-Doped Mesoporous Carbon Materials as Efficient Electrocatalysts for Oxygen Reduction Reaction[J]. Electrocatalysis, 2019, 10: 72-81. doi: 10.1007/s12678-018-0496-9 [33] CAO Q C, DING X B, LI F, et al. Zinc, sulfur and nitrogen co-doped carbon from sodium chloride/zinc chloride-assisted pyrolysis of thiourea/sucrose for highly efficient oxygen reduction reaction in both acidic and alkaline media[J]. Journal of Colloid and Interface Science, 2020, 576: 139-146. doi: 10.1016/j.jcis.2020.05.020 [34] LI Y F, LIU Y Z, SHEN W Z, et al. Graphene-ZnS quantum dot nanocomposites produced by solvothermal route[J]. Materials Letters, 2011, 65(15): 2518-2521. [35] ZHAO L P, GAO L. Coating multi-walled carbon nanotubes with zinc sulfide[J]. Journal of Materials Chemistry, 2004, 14(6): 1001-1004. doi: 10.1039/b315450e [36] LI Y F, LIU Y Z, SHEN W Z, et al. Graphene-ZnS quantum dot nanocomposites produced by solvothermal route[J]. Materials Letters, 2011, 65(15): 2518-2521. -

 点击查看大图
点击查看大图
计量
- 文章访问数: 60
- HTML全文浏览量: 25
- 被引次数: 0




 下载:
下载:















