Preparation of Ag@AgCl modified Bi4Ti3O12 and its visible light catalytic performance
-
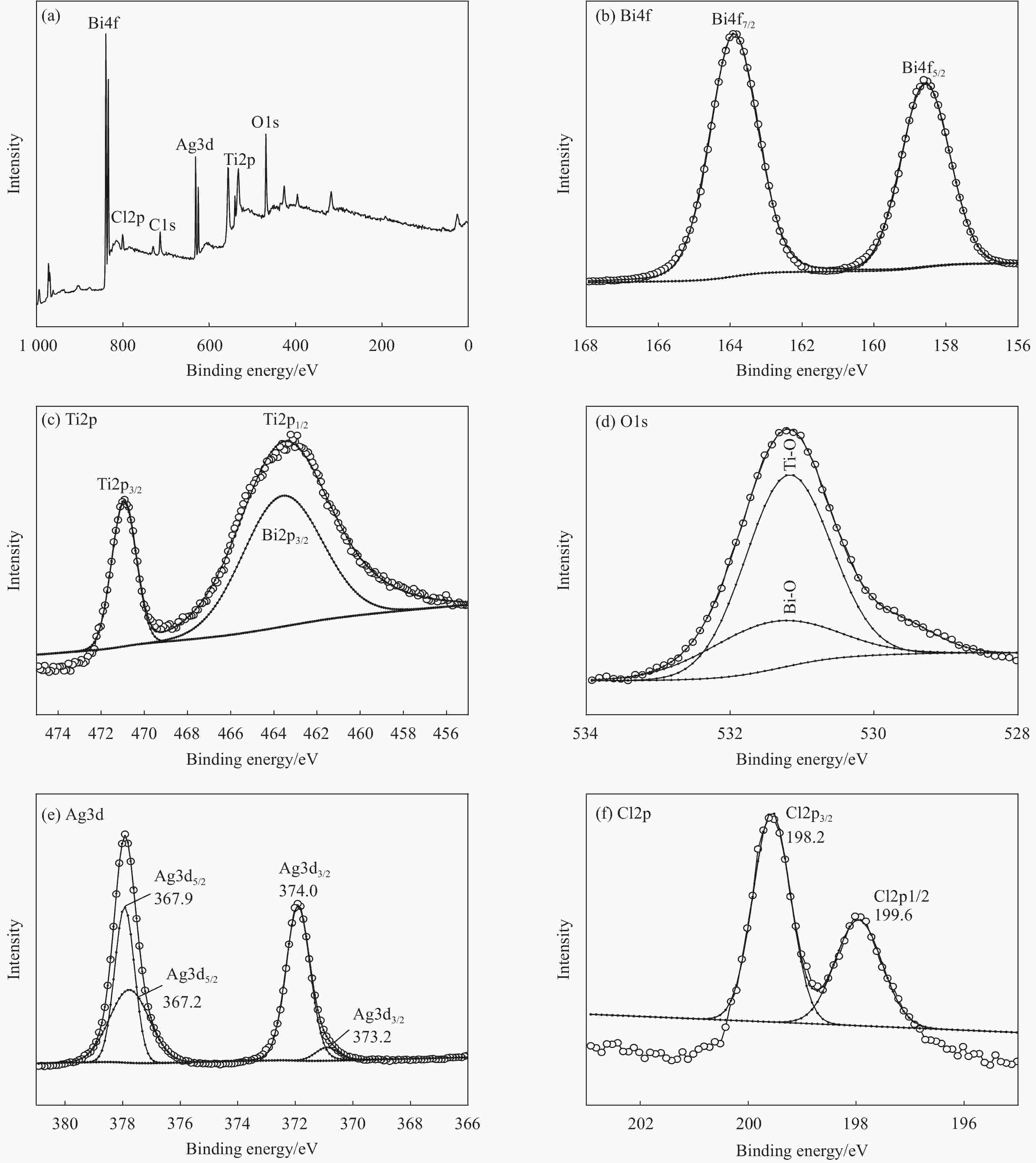
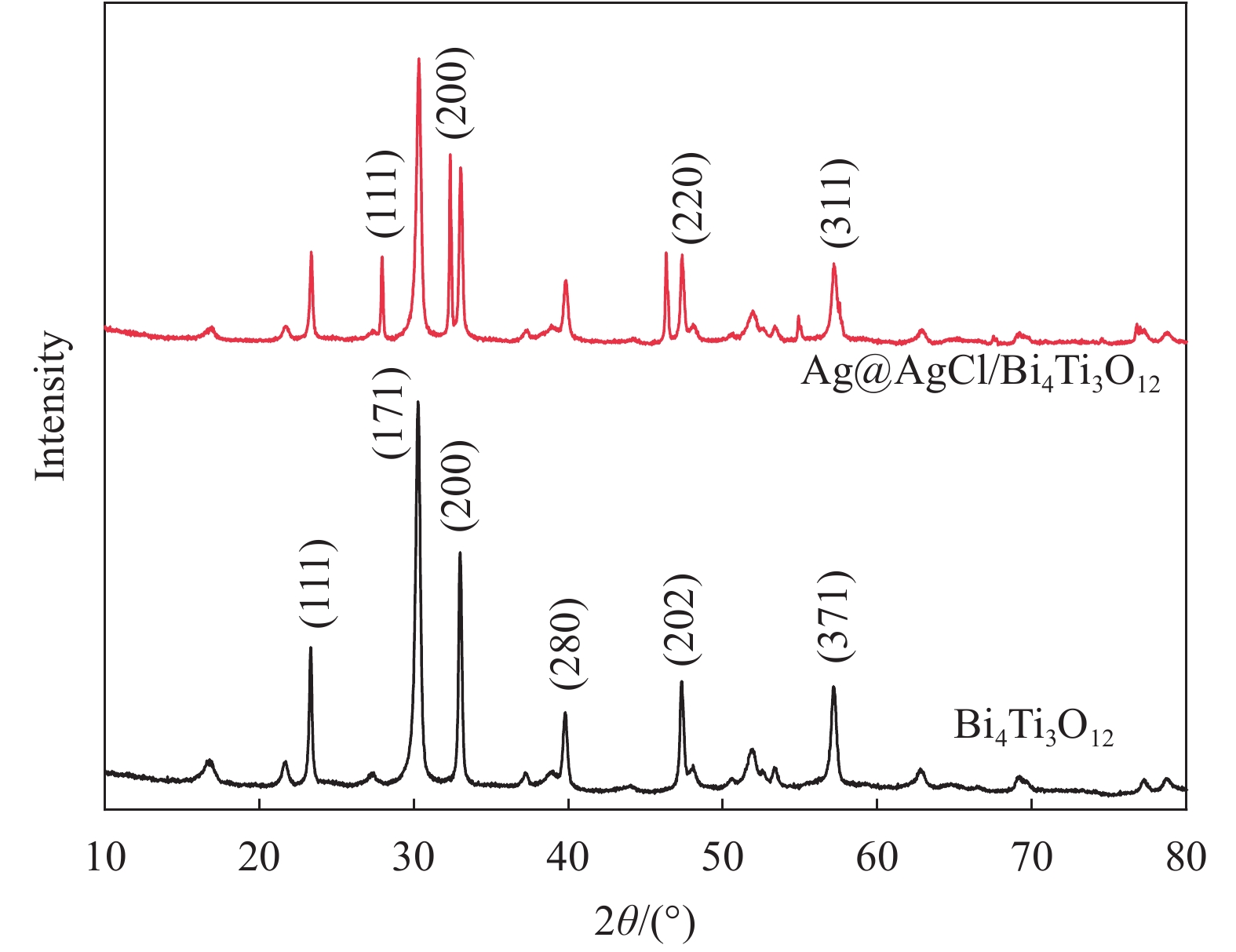
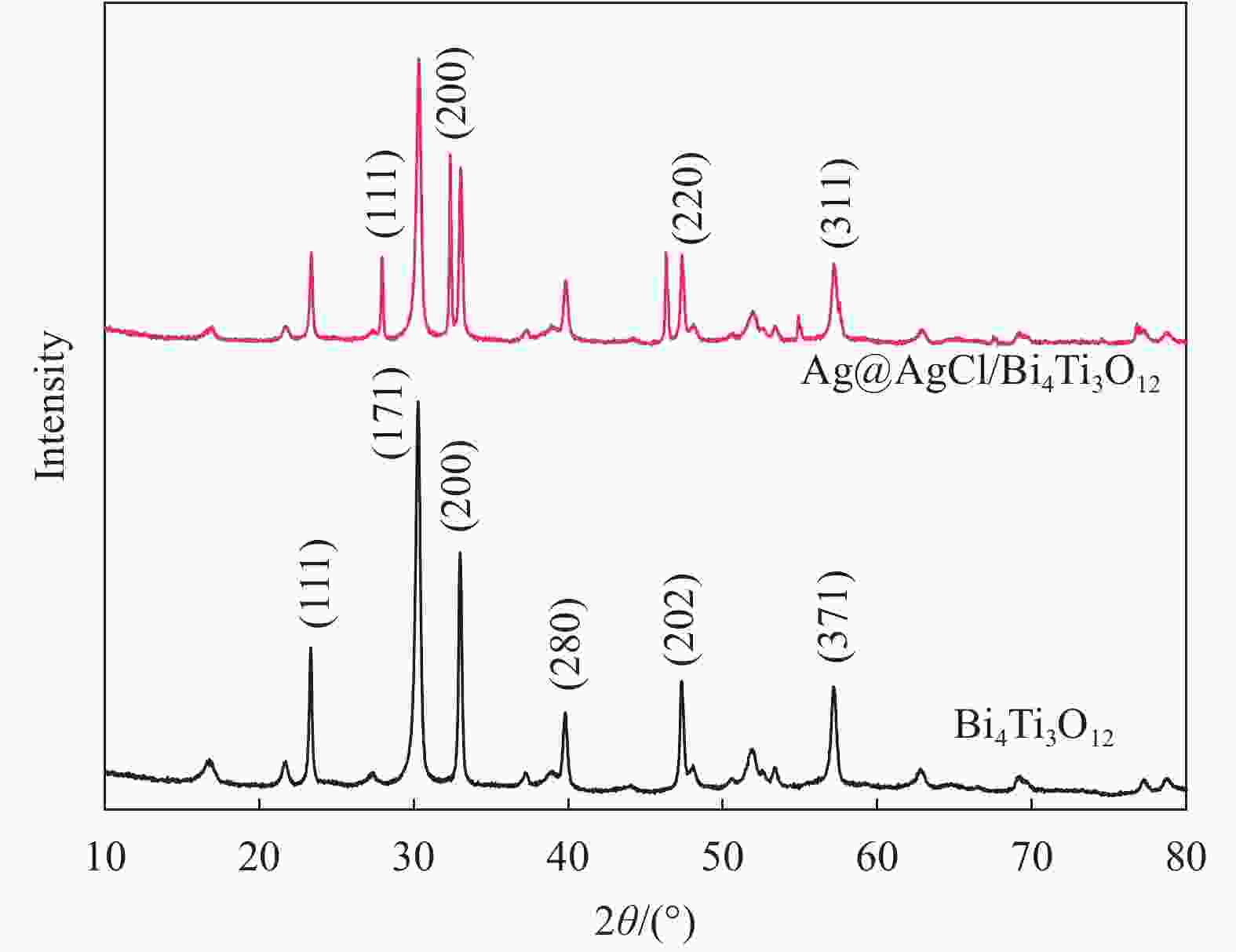
摘要: 以五水硝酸铋为铋源、钛酸四丁酯为钛源,通过水热法制备了Bi4Ti3O12,再以硝酸银为银源、盐酸为氯源,采用光照还原Bi4Ti3O12得到Ag@AgCl/Bi4Ti3O12复合材料。利用XRD、UV-Vis DRS、SEM、TEM、BET和XPS等方法对所制备材料的组成和结构进行表征和分析,并以甲基橙(MO)溶液的脱色率为评价标准,考察了所制备材料的可见光催化性能和活性物质。结果表明:制备的Bi4Ti3O12呈堆积花球状纳米片结构,Ag@AgCl颗粒沉积在Bi4Ti3O12片间,比表面积增大到14.30 m2/g,对可见光的吸收增强。当催化剂用量为0.5 g/L、在300 W氙灯照射80 mL 10 mg/L MO溶液30 min时,Ag@AgCl/Bi4Ti3O12的脱色率为96.71%,较Bi4Ti3O12提高38.28%,降解实验循环稳定性强,超氧自由基(•O2−)在降解过程中起决定性作用。Abstract: The Bi4Ti3O12was prepared by hydrothermal method using bismuth nitrate as the bismuth source and tetrabutyl titanate as the titanium source, and then the Bi4Ti3O12 was reduced by light to obtain Ag@AgCl/Bi4Ti3O12 nanocomposites using silver nitrate as the silver source and hydrochloric acid as the chlorine source. The compo-sition and structure of the as-prepared nanocomposites were characterized by XRD, UV-Vis DRS, SEM, TEM, BET and XPS, etc. Taking the decolorization rate of methyl orange (MO) as an evaluation standard, the visible light catalytic performance and active substances of the as-prepared catalyst were investigated. The results show that the prepared Bi4Ti3O12 has a stacked curd-shaped nanosheets structure and Ag@AgCl particles deposite between the sheets, the specific surface area of Ag@AgCl/Bi4Ti3O12 increases to 14.30 m2/g, and the absorption of visible light is enhanced. The decolorization rate can attain 96.71% when the 80 mL 10 mg/L MO solutions were irradiated under the 300 W xenon lamp for 30 min using 0.5 g/L Ag@AgCl/Bi4Ti3O12, which is 38.28% higher than that of pure Bi4Ti3O12. The degradation experiment has strong cycle stability and superoxide radicals (•O2−) plays a decisive role in the degradation process.
-
Key words:
- visible light catalysis /
- bismuth titanate /
- silver /
- silver chloride /
- modification /
- degradation
-
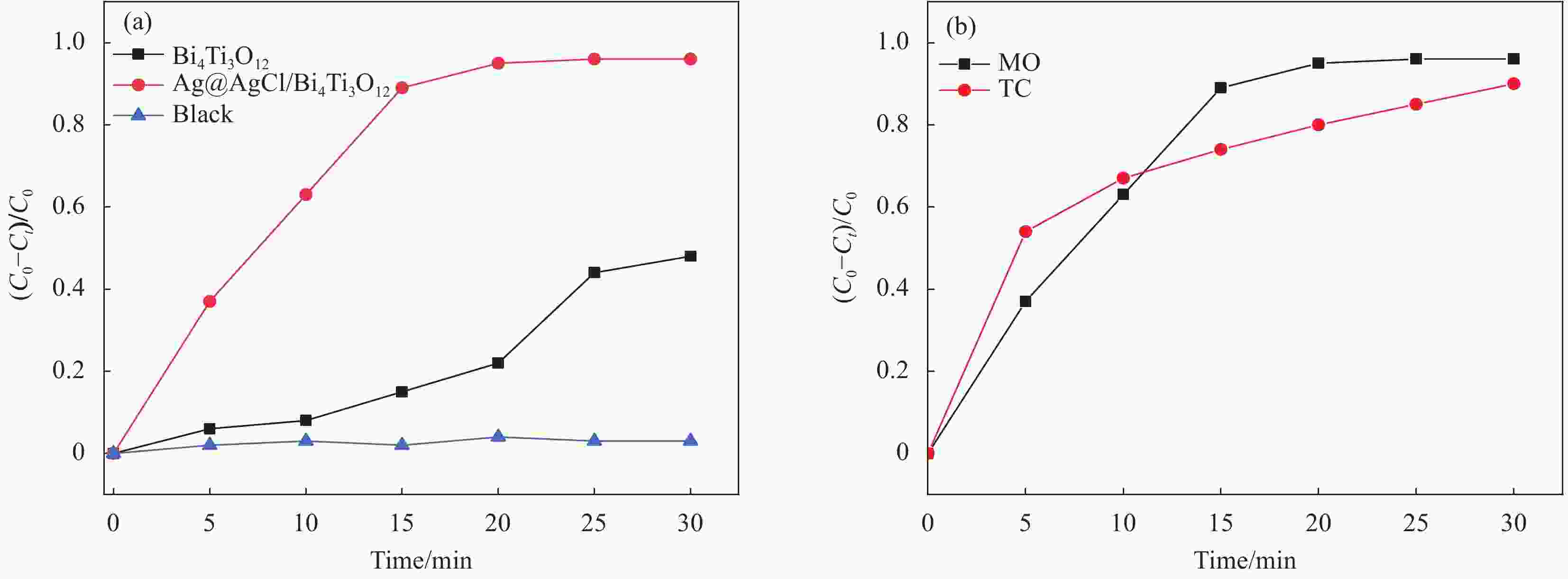
图 7 Bi4Ti3O12和Ag@AgCl/Bi4Ti3O12在甲基橙(MO)溶液降解效果 (a)、Ag@AgCl/Bi4Ti3O12对不同目标污染物降解效果 (b)
Figure 7. Degradation effect of Bi4Ti3O12 and Ag@AgCl/Bi4Ti3O12 in methyl orange (MO) solution (a) and degradation effect of Ag@AgCl/Bi4Ti3O12 on different target pollutants (b)
C0—Initial absorbance; Ct—Absorbance at light time t; TC—Tetracycline hydrochloride
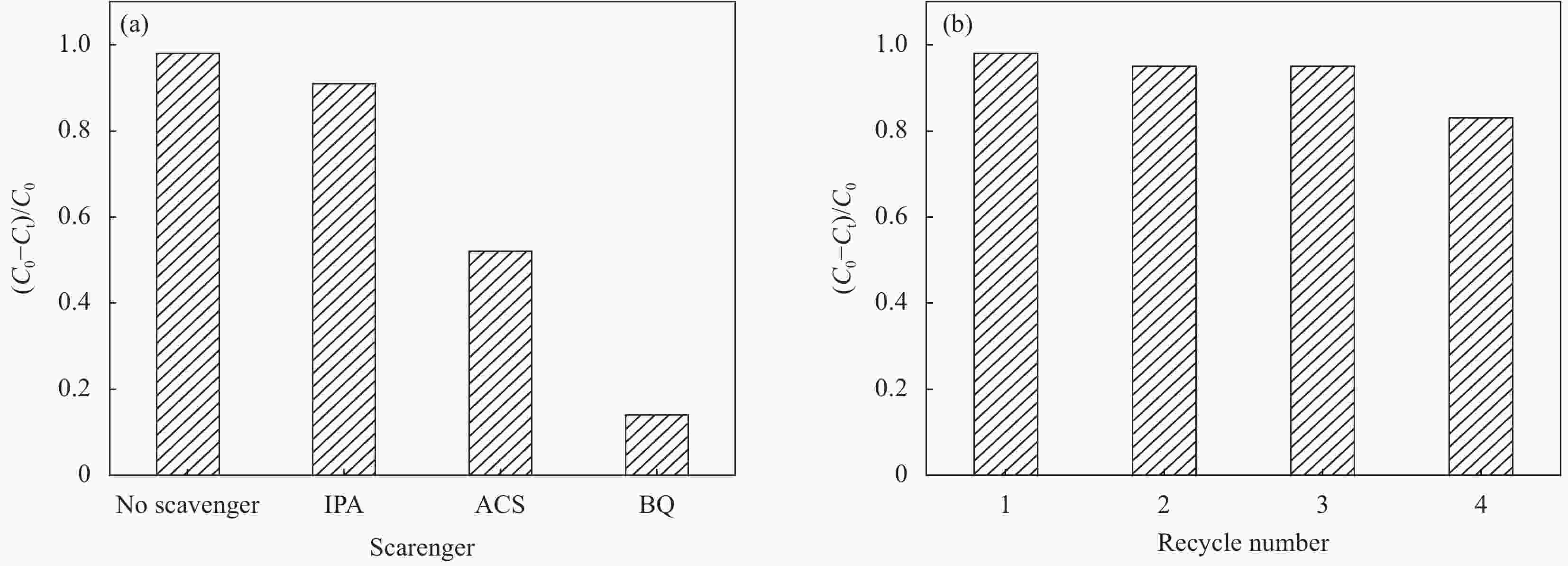
图 8 Ag@AgCl/Bi4Ti3O12在MO溶液对自由基抑制剂光催化效果 (a)和Ag@AgCl/Bi4Ti3O12在MO溶液循环反应效果 (b)
Figure 8. Photocatalytic effect of Ag@AgCl/Bi4Ti3O12 on the free radical inhibitor in MO solution (a), and the cyclic reaction effect of Ag@AgCl/Bi4Ti3O12 in MO solution (b)
IPA—Isopropanol; BQ—Benzoquinone; ACS—Ammonium oxalate
-
[1] HE R, XU D, BEI C, et al. Review on nanoscale Bi-based photocatalysts[J]. Nanoscale Horizons,2018,3:464. doi: 10.1039/C8NH00062J [2] DING Y B, ZHANG G L, WANG X, et al. Chemical and photocatalytic oxidative degradation of carbamazepine by using metastable Bi3+ self-doped NaBiO3 nanosheets as a bifunctional material[J]. Applied Catalysis B Environmental,2017:202528-202538. [3] ZHAO K, ZHANG L, WANG J, et al. Surface structure-dependent molecular oxygen activation of BiOCl single-crystalline nanosheets[J]. Journal of the American Chemical Society,2013,135(42):15750-15753. doi: 10.1021/ja4092903 [4] LIU L Z, HUANG H W, CHEN F, et al. Cooperation of oxygen vacancies and 2D ultrathin structure promoting CO2 photoreduction performance of Bi4Ti3O12[J]. Science Bulletin,2020,65(11):934-943. doi: 10.1016/j.scib.2020.02.019 [5] NIU S Y, ZHANG R Y, XIANG J C, et al. Morphology-dependent photocatalytic performance of Bi4Ti3O12[J]. Ceramics International,2020,46(5):6782-6786. doi: 10.1016/j.ceramint.2019.11.169 [6] GANG X, YANG Y, BAI H, et al. Hydrothermal synthesis and formation mechanism of the single-crystalline Bi4Ti3O12 nanosheets with dominant (010) facets[J]. Crystengcomm,2016,18(13):2268-2274. [7] ZHANG Y G, ZHENG H W, ZHANG J X, et al. Photovoltaic effects in Bi4Ti3O12 thin film prepared by a sol-gel method[J]. Materials Letters,2014,125:25-27. doi: 10.1016/j.matlet.2014.03.146 [8] ZHANG C S, GUO C J. Synthesis of plated-like template Bi4Ti3O12 particles and properties[J]. Advanced Materials Research,2011,1268(481):2170-2173. [9] ZHANG M, LIU Y, LI L, et al. BiOCl nanosheet/Bi4Ti3O12 nanofiber heterostructures with enhanced photocatalytic activity[J]. Catalysis Communications,2015,58:122-126. doi: 10.1016/j.catcom.2014.09.021 [10] NAVARRO-ROJERO M G, ROMERO J J, RUBIO-MARCOS F, et al. Intermediate phases formation during the synthesis of Bi4Ti3O12 by solid state reaction[J]. Ceramics International,2010,36(4):1319-1325. doi: 10.1016/j.ceramint.2009.12.023 [11] ZHANG M, LIU Y, LI L, et al. BiOCl nanosheet/Bi4Ti3O12 nanofiber heterostructures with enhanced photocatalytic activity[J]. Catalysis Communications, 2015, 58: 122-126. [12] HE Z, ZHAO Y, YU P, et al. Constructing Bi4Ti3O12/CdS heterostructure by atomic layer deposition for increased photocatalytic performance through suppressed electron/hole recombination[J]. Journal of Alloys and Compounds,2021,866:158978. doi: 10.1016/j.jallcom.2021.158978 [13] 张根, 黄自力, 葛胜涛, 等. Bi4Ti3O12/g-C3N4复合光催化剂的制备及其光催化性能研究[J]. 现代化工, 2020, 40(8):83-87.ZHANG G, HUANG Z L, GE S T, et al. Preparation of Bi4Ti3O12/g-C3N4 composite photocatalyst and its photocatalytic performance[J]. Modern Engineering,2020,40(8):83-87(in Chinese). [14] 李跃军, 曹铁平, 孙大伟, 等. Bi@Bi4Ti3O12/TiO2等离子体复合纤维的制备及增强可见光催化性能[J]. 复合材料学报, 2020, 37(3):609-617.LI Y J, CAO T P, SUN D W, et al. Preparation of Bi@Bi4Ti3O12/TiO2 plasma composite fiber and enhanced visible light catalytic performance[J]. Journal of Compo-site Materials,2020,37(3):609-617(in Chinese). [15] 汪玲, 李雪. Ag/AgBr/Bi4Ti3O12催化剂的制备及可见光催化降解卡马西平[J]. 信阳师范学院学报(自然科学版), 2019, 32(2):293-297.WANG L, LI X. Preparation of Ag/AgBr/Bi4Ti3O12 catalyst and visible light catalytic degradation of carbamazepine[J]. Journal of Xinyang Normal University (Natural Science Edition),2019,32(2):293-297(in Chinese). [16] JIANG C J, QIU S Q, XU D F, et al. Direct evidence and enhancement of surface plasmon resonance effect on Ag-loaded TiO2 nanotube arrays for photocatalytic CO2 reduction[J]. Applied Surface Science,2018,434:423-432. [17] WANG P, HUANG B B, QIN X Y, et al. Ag@AgCl: A highly efficient and stable photocatalyst active under visible light[J]. Angewandte Chemie International Edition,2010,47(41):7931-7933. [18] ZHAO M, YUAN Q, ZHANG H, et al. Synergy of adsorption and photocatalysis on removal of high-concentration dye by Ag/AgCl/Bi6O4(OH)4(NO3)6•H2O nanocomposite using Bi12O17Cl2 as bismuth source[J]. Journal of Alloys and Compounds,2019,782:1049-1057. doi: 10.1016/j.jallcom.2018.12.218 [19] XIAO J Q, LIN K S, YU Y. Novel Ag@AgCl@AgBr heterostructured nanotubes as high-performance visible-light photocatalysts for decomposition of dyes[J]. Catalysis Today,2018,314:10-19. doi: 10.1016/j.cattod.2018.04.019 [20] SUN L L, LIU C Y, LI J Z, et al. Fast electron transfer and enhanced visible light photocatalytic activity by using poly-o-phenylenediamine modified AgCl/g-C3N4 nanosheets[J]. Chinese Journal of Catalysis,2019,40(3):80-94. [21] ASADZADEH-KHANEGHAH S, HABIBI- YANGJEH A, ABEDI M. Decoration of carbon dots and AgCl over g-C3N4 nanosheets: Novel photocatalysts with substantially improved activity under visible light[J]. Separation and Purification Technology,2018,199:64-77. doi: 10.1016/j.seppur.2018.01.023 [22] KONG X G, LI L, GUO Z L, et al. Soft chemical in situ synthesis and photocatalytic performance of 1D Ag/AgCl/V2O5 hetero-nanostructures[J]. Materials Letters,2016,183(15):215-218. [23] 王学川, 刘轩, 韩庆鑫, 等. 光辅助原位清洁制备Ag/AgCl纳米颗粒[J]. 精细化工, 2020, 37(8):1608-1614.WANG X C, LIU X, HAN Q X, et al. Photo-assisted in-situ cleaning of Ag/AgCl nanoparticles[J]. Fine Chemicals,2020,37(8):1608-1614(in Chinese). [24] MA B W, GUO J F, DAI W L, et al. Ag-AgCl/WO3 hollow sphere with flower-like structure and superior visible photocatalytic activity[J]. Applied Catalysis B Environmental,2012,123-124(30):193-199. [25] YIN H Y, WANG X L, WANG L, et al. Ag/AgCl modified self-doped TiO2 hollow sphere with enhanced visible light photocatalytic activity[J]. Journal of Alloys and Compounds,2016,657:44-52. doi: 10.1016/j.jallcom.2015.10.055 [26] LIANG Y H, LIN S L, LIU L, et al. Oil-in-water self-assembled Ag@AgCl QDs sensitized Bi2WO6: Enhanced photocatalytic degradation under visible light irradiation[J]. Applied Catalysis B Environmental,2015:164192-164203. [27] LIU L Q, OUYANG S X, YE J H, et al. Gold-nanorod-photosensitized titanium dioxide with wide-range visible-light harvesting based on localized surface plasmon resonance[J]. Angewandte Chemie,2013,125(26):6821-6825. doi: 10.1002/ange.201300239 [28] WANG D, HUANG L, GUO Z N, et al. Enhanced photocatalytic hydrogen production over Au/SiC for water reduction by localized surface plasmon resonance effect[J]. Applied Surface Science,2018,456:871-875. doi: 10.1016/j.apsusc.2018.06.099 [29] SING S K. Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity (Recommendations 1984)[J]. Pure & Applied Chemistry,1985,57(4):603-619. [30] ZHANG S W, LI J X, WANG X K, et al. In situ ion exchange synthesis of strongly coupled Ag@AgCl/g-C3N4 porous nanosheets as plasmonic photocatalyst for highly efficient visible-light photocatalysis[J]. Acs Applied Materials & Interfaces,2014,6(24):22116-22125. [31] HOU D F, HU X L, PEI H, et al. Bi4Ti3O12 nanofibers-BiOI nanosheets p-n junction: Facile synthesis and enhanced visible-light photocatalytic activity[J]. Nanoscale,2013,5(20):9764-9772. doi: 10.1039/c3nr02458j -





 下载:
下载: