Gel polymer electrolytes for lithium sulfur batteries: Synthesis and advanced characterization techniques
-
摘要:
凝胶聚合物电解质(GPE)的应用为改善锂硫电池的安全性和抑制穿梭效应提供一种有希望的方案。凝胶聚合物电解质能够改善全固态电解质与双电极之间存在的高界面阻抗所带来的电荷转移受阻、锂沉积不均匀等问题,有效解决容量衰减快、循环稳定性差等缺陷。本文针对锂硫电池中制备凝胶聚合物电解质所采用的原位聚合和非原位聚合两种不同的工艺手段进行介绍,通过阐述不同合成工艺改进凝胶聚合物电解质基底的方法,重点分析不同工艺所带来的“收益”,并介绍了具有实时性和精准性的原位表征仪器在锂硫电池中的应用,指出原位先进表征技术对锂硫电池电极材料设计的指导作用,并供科研工作者开发研究更适宜产业化的凝胶聚合物电解质的合成工艺,展望未来锂硫电池凝胶聚合物电解质合成设计的发展方向。
Abstract:The application of gel-polymer electrolyte (GPE) provides a promising scheme for improving the safety of lithium-sulfur batteries and inhibiting the shuttle effect. The GPE can improve the charge transfer obstruction and uneven lithium deposition caused by the high interface impedance between the all-solid electrolyte and the double electrode, and effectively solve the defects such as fast capacity decay and poor cycle stability. In this paper, two different technological means, in-situ polymerization and non-in-situ polymerization, are introduced for the preparation of GPE in lithium-sulfur batteries. The methods of improving the GPE base by different synthetic processes are described, and the "benefits" brought by different processes are analyzed, and the application of real-time and accurate in-situ characterization instruments in lithium-sulfur batteries is introduced. It is pointed out that in situ advanced characterization technology can guide the electrode material design of lithium-sulfur battery and provide researchers with more suitable synthesis technology of GPE for industrialization.
-
锂离子电池自20世纪90年代实现商业化应用以来,广泛应用于便携式摄像机、手机、笔记本电脑以及新能源汽车等领域[1-5]。随着全球能源需求的快速增长推动高能量密度二次电池的发展,现有传统锂离子电池的能量密度难以满足人们未来对于储能的应用需求[6],开发下一代高能量密度绿色能源储能器件已引起人们的高度重视[7]。锂硫电池因其超高理论能量密度(
2600 W·h·kg−1)、硫在自然界储量丰富、环境友好等优点,已成为有前景的下一代储能装置[8]。然而,由于长链多硫化锂溶解在醚类电解液中导致穿梭效应、充放电过程中正极活性物质材料硫及其放电产物(Li2S/Li2S2)电导率低等因素,所导致的锂硫电池面临活性物质硫利用率低、锂金属负极腐蚀、界面稳定性差等问题的限制,使锂硫电池市场化应用仍然面临众多挑战[9]。为克服上述挑战,研究者们在正极材料改性[10]、电解液优化、隔膜修饰[11]、金属锂负极保护[12]等方面进行了诸多努力。电解液作为锂硫电池的重要组成部分,起到输送离子传递电子的作用,电池的循环效率、使用寿命、安全性能都与电解液息息相关[13-14]。
现阶段商品化的锂硫电池醚类电解液的应用中,仍存在多硫化锂穿梭效应、锂枝晶生长等问题。
为了解决上述问题,研究者们在电解液中引入少量添加剂。2008年,Mikhaylik等[15]首次提出硝酸锂作为锂硫电池电解液添加剂,此项研究颠覆人们以往对于锂硫电池电解液配方的认知,以硝酸锂作为电解液添加剂后,锂硫电池电化学性能显著提高,该项研究对此后锂硫电池电解液添加剂的探索具有引领作用。近年来,Shi等[16]采用三聚硫氰酸(TMT)作为添加剂,放电时TMT和多硫化锂反应,改变多硫化锂的转化过程,有效降低电解液中多硫化锂的浓度。充电时TMT和多硫化物的反应产物进一步和Li2S反应生成Li和在锂负极表面生成稳定的固体电解质界面(SEI)有效保护金属锂不被腐蚀。Qiu等[17]选用六氟磷酸铵(AFP)作为双功能添加剂,经测试后发现,AFP有效促进不导电性Li2S在电解液中的分解,AFP分解产生PF6−,在锂负极生成含F的SEI,保护锂负极不被腐蚀。以上研究在电解液中加入少量添加剂,在锂负极表面生成稳定SEI,阻止多硫化锂沉积到锂负极上,保护金属锂不被多硫化锂腐蚀并改变多硫化物的转化过程。以上工作能够减轻多硫化锂的穿梭效应,生成的SEI保护金属锂不被腐蚀,提升锂硫电池电化学性能。但这类研究策略未能完全解决多硫化锂在电解液中的溶解穿梭问题,且商品化的醚类电解液沸点较低,在充放电过程中易产生可燃性气体,安全性有待提高。
近些年来,固态电解质因其能够避免多硫化锂在电解质中的穿梭效应和提高锂硫电池的安全性被广泛关注[18]。锂硫电池固态电解质分为聚合物电解质和无机固态电解质,聚合物电解质具有良好的柔韧性和机械稳定性,电极之间的界面阻抗低,和聚合物电解质相比,无机固态电解质在室温下电导率较高[19],但目前固态电解质在锂硫电池的应用仍然受到离子电导率低于传统醚类电解液和界面阻抗高等问题的制约。将液态电解液和聚合物电解质进行复合得到凝胶聚合物电解质(GPE)有望克服以上技术阻碍,成为未来发展柔性电池技术的有利候选者[20]。GPE结合了固态聚合物电解质卓越的力学性能和传统液态电解液的良好电化学行为和优异界面稳定性。因此,GPE很好的平衡了电池的整体性能,包括安全性、寿命、稳定性、润湿性、离子电导率和放电比容量。
本文聚焦锂硫电池GPE的合成工艺设计和先进表征手段进行系统阐述(图1),分析和阐明锂硫电池GPE不同合成工艺所展现的优势及其对锂硫电池体系的调控机制,提出利用先进表征手段对揭示锂硫电池反应机制的重要性,同时展望了锂硫电池GPE的设计与应用前景。
1. GPE中存在问题及其解决过程
GPE主要是由聚合物基底、增塑剂和锂盐组成,锂盐和增塑剂均匀分布在聚合物基底中[21]。聚合物基底通过非晶区链段运动促使锂盐解离,增塑剂的主要作用是降低聚合物基底的结晶率,提高离子电导率。锂硫电池中GPE主要采用聚环氧乙烷(PEO)、聚偏二氟乙烯(PVDF)、聚甲基丙烯酸甲酯(PMMA)、聚丙烯腈(PAN)等材料为基底。由于聚合物基底的分子结构不同,应用在GPE中基底材料特性有所差异,需要克服缺陷不同。早在1973年,Wright课题组[22]最早发现PEO结构中的醚键与电解液有良好亲和力,浸泡在电解液中容易溶胀,有利于锂离子传导,但PEO容易结晶导致基底中非晶区链段运动受阻,使Li+电导率降低,该缺陷阻碍了PEO基GPE锂硫电池的发展;2000年,Michot等[23]研究发现PVDF具有强吸电子能力的—CF2基团,有助于加速锂盐溶解,使电解质具有高离子电导率,现已成为锂硫电池GPE中最常用的聚合物基底,但PVDF基GPE与锂负极的界面稳定性差导致锂硫电池循环性能差、容量保持率不好[24];PMMA的链段结构中含有丰富的羰基,与醚类电解液中的氧原子有较强的相互作用,因此它与传统醚类电解液有良好的相容性,吸液性强更容易形成凝胶态,但PMMA的低机械强度和低离子电导率使由PMMA基组装的锂硫电池阻抗高放电比容量低、电化学性能差[25];Qian等[26]指出PAN由于离子电导率高经常作为锂硫电池GPE基底,但PAN中的—CN易与锂负极反应,在锂负极表面生成一层“钝化层”导致GPE/负极界面稳定性差影响电池循环性能。
GPE基底材料的选择会影响锂硫电池GPE性能,GPE生产工艺不同也会使其表现出不同特性。目前,GPE的制备方法主要包括原位聚合和非原位聚合两种方法,两种合成工艺的共同特点是都要经历从液态前驱体到凝胶电解质的转变过程。传统非原位聚合工艺是将前驱体溶液固化成电解质薄膜,再浸泡在电解液中使其溶胀形成GPE;运用原位聚合工艺合成GPE,扬弃了传统GPE合成过程中的聚合物材料溶解、干燥涂膜等复杂工序,使GPE的合成及电池组装一步完成,有效地降低成本。为进一步探究原位/非原位合成工艺制备出的GPE不同特性,研究者们运用多种原位表征手段,通过分析锂硫电池中电化学反应现象,进而深入分析锂硫电池的反应原理,以便更合理设计出性能优异锂硫电池材料。为了解决基底材料的缺陷,研究者们运用不同合成工艺对锂硫电池GPE进行各种功能改性。
2. 锂硫电池凝胶聚合物电解质的合成工艺及其应用
固态电解质因其能够避免多硫化锂在电解质中的穿梭效应和提高锂硫电池的安全性近些年来被广泛关注。Hu等[27]运用硫化物固态电解质(Li6PS5Cl)组装了锂硫电池,表现出良好循环稳定性和高机械强度,有效提高电池安全性。但全固态锂硫电池中电解质和硫正极界面接触较差,在常温下电解质中离子迁移速率低等问题都阻碍着全固态锂硫电池的发展。而GPE因其半固态结构,具有和电极接触更加紧密、化学和电化学稳定性好、电解质中离子迁移速率高、安全性能高和能够在分子水平上进行设计等优点。因此GPE被认为是解决上述问题的有效途径。
近些年,国内外诸多学者以PVDF、PEO、PMMA等材料为基体制备了不同的GPE[28-30]。在过去的研究中,锂硫电池凝胶电解质的相关综述通常按基体分类,对合成工艺的描述相对较少。本文按合成工艺进行下述分类(表1)。GPE合成工艺主要分为原位聚合和非原位聚合,已广泛应用于锂硫电池体系中,原位聚合是通过将引发剂加入电解液中,在电池组装完毕静置过程中自发形成凝胶态电解质;非原位聚合则是通过预先合成凝胶态薄膜后再对电池进行组装。合成工艺的差异,导致形成GPE状态不同,功能性也有所差异。
2.1 非原位聚合凝胶聚合物电解质
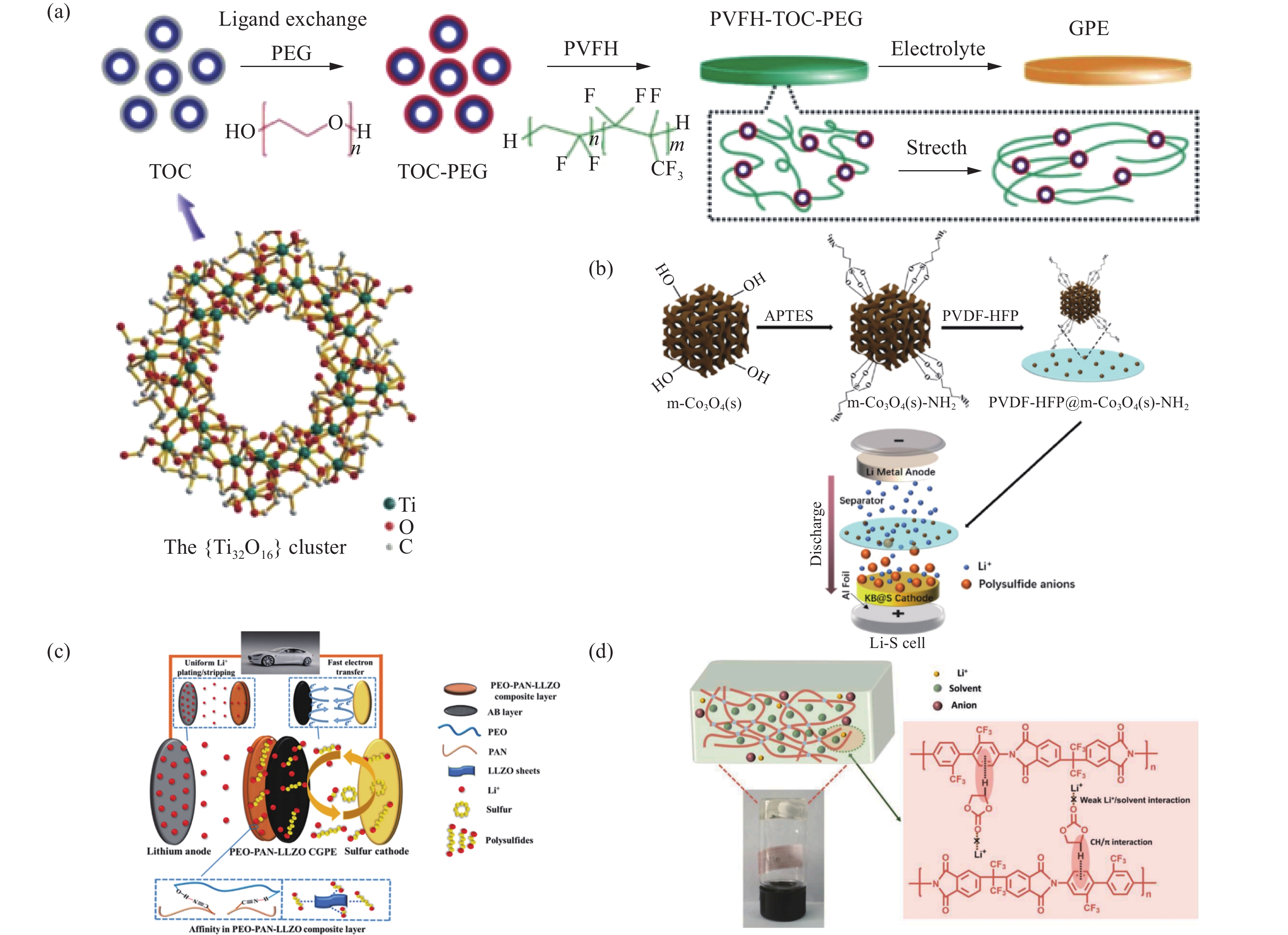
近年来在锂硫电池体系中,非原位聚合手段因其合成凝胶流程简便,更容易在分子水平进行修饰,兼容性更好,易于整合多种材料的优点[31]。非原位聚合方法被广泛应用于制备多功能凝胶聚合物电解质。Pei等[32]首次发掘金属-氧簇化合物在锂硫电池GPE中的潜力,选用聚偏氟乙烯-共六氟丙烯(PVFH)作为聚合物基体,以TOC (Ti32O16(OCH2CH2O)32(RCOO)16(EGH)16(R:t-CH3-CH2CH2CH2—;EGH:—OCH2CH2OH))作为可进行基团改性的填料。将TOC的EGH配体基团在室温下被聚乙二醇(PEG)取代,PEG的取代有利于增强TOC对PVFH的亲和作用,形成PVFH-TOC-PEG凝胶聚合物电解质(图2(a))。改性后的TOC能够促进聚合物基体成为凝胶薄膜,制成的GPE薄膜能够抑制锂硫电池中多硫化锂的穿梭效应,引导锂离子的均匀沉积。经TOC改性后的GPE,在提高锂硫电池能量密度和电池循环稳定性方面具有很大潜力。通过非原位聚合工艺合成的GPE,可以通过引入纳米氧化物类无机填料构成复合GPE,该类填料分子表面具有路易斯酸性位点可以与聚合物链段上具有路易斯碱性位点的氧化性基团产生作用,降低聚合物基底结晶度,进而使非晶区链段运动速率更快,提升锂离子迁移速率,为改进锂硫电池凝胶电解质的功能提供全新的思路。
![]() 图 2 (a) TOC晶体结构及聚偏氟乙烯-共六氟丙烯(PVFH)-TOC-聚乙二醇(PEG)电解质合成示意图[32];(b) PVDF-六氟丙烯(HFP)@氨化介孔Co3O4 (m-Co3O4(s)-NH2)的合成路线示意图[33];(c)聚环氧乙烷(PEO)-聚丙烯腈(PAN)-LLZO GPE在Li-S电池中的性能提升机制示意图[35];(d)聚合物凝胶形成机制示意图[36]Figure 2. (a) Crystal structure of TOC and schematic illustration for the synthesis of the polyvinylidene fluoride-hexafluoropropylene (PVFH)-TOC-polyethylene glycol (PEG) electrolyte[32]; (b) Schematic illustration of the synthesis route of poly(vinylidene fluoride)-hexafluoropropylene (PVDF-HFP)@ammoniated mesoporous Co3O4 (m-Co3O4(s)-NH2)[33]; (c) Schematic illustration for the performance improvement mechanisms of polyethylene oxide (PEO)-polyacrylonitrile (PAN)-LLZO GPE applied in the Li-S battery[35]; (d) Schematic diagram of formation mechanism for the polymer gel polymer[36]TOC—Ti32O16(OCH2CH2O)32(RCOO)16(EGH)16 (R: t-CH3-CH2CH2CH2—; EGH: —OCH2CH2OH); APTES—3-aminopropyltriethoxysilane; LLZO—Li7La3Zr2O2
图 2 (a) TOC晶体结构及聚偏氟乙烯-共六氟丙烯(PVFH)-TOC-聚乙二醇(PEG)电解质合成示意图[32];(b) PVDF-六氟丙烯(HFP)@氨化介孔Co3O4 (m-Co3O4(s)-NH2)的合成路线示意图[33];(c)聚环氧乙烷(PEO)-聚丙烯腈(PAN)-LLZO GPE在Li-S电池中的性能提升机制示意图[35];(d)聚合物凝胶形成机制示意图[36]Figure 2. (a) Crystal structure of TOC and schematic illustration for the synthesis of the polyvinylidene fluoride-hexafluoropropylene (PVFH)-TOC-polyethylene glycol (PEG) electrolyte[32]; (b) Schematic illustration of the synthesis route of poly(vinylidene fluoride)-hexafluoropropylene (PVDF-HFP)@ammoniated mesoporous Co3O4 (m-Co3O4(s)-NH2)[33]; (c) Schematic illustration for the performance improvement mechanisms of polyethylene oxide (PEO)-polyacrylonitrile (PAN)-LLZO GPE applied in the Li-S battery[35]; (d) Schematic diagram of formation mechanism for the polymer gel polymer[36]TOC—Ti32O16(OCH2CH2O)32(RCOO)16(EGH)16 (R: t-CH3-CH2CH2CH2—; EGH: —OCH2CH2OH); APTES—3-aminopropyltriethoxysilane; LLZO—Li7La3Zr2O2其他研究者们也在相同基体中加入无机填料进行GPE功能改性。Li等[33]合成了氨化介孔Co3O4 (m-Co3O4-NH2)作为功能性无机填料并将其填充到PVDF-六氟丙烯(HFP)中(图2(b)),制备高性能杂化凝胶聚合物电解质(PVDF-HFP@m-Co3O4-NH2),介孔填料具有更大的孔隙,有利于锂离子转移、避免孔隙被多硫化锂堵塞等优点。在PVDF-HFP基体中加入m-Co3O4后,在室温下凝胶电解质的离子电导率由1.37×10−3 S·cm−1 提升到3.23×10−3 S·cm−1,组装锂硫电池测试后与纯PVDF-HFP基相比,混合无机填料的凝胶聚合物电解质的电池表现出更优异的电化学性能。该研究拓展了应用在锂硫电池GPE研究中无机填料的种类,促进GPE在锂硫电池中的应用。同样Wang等[34]也在相同基体中加入三维γ-Al2O3无机填料进行GPE功能改性,制备出复合材料Al2O3-PVDF-HFP GPE。γ-Al2O3纳米纤维可以通过Lewis酸碱相互作用有效锚定多硫化锂,抑制穿梭效应。与此同时TFSI−与γ-Al2O3纳米纤维之间的Lewis酸碱相互作用可以促进锂盐解离,提高锂离子电导率和锂离子转移数,使锂负极均匀沉积。此外由于γ-Al2O3不可燃,经过γ-Al2O3填料进行改性的GPE组装的锂硫电池安全性显著提升。
在非原位聚合凝胶电解质的过程中,合理引入无机填料可以获得多功能凝胶聚合物电解质,提高锂硫电池电化学性能,对基体进行基体链段基团改性也是提升电化学性能的有效途径之一。Xie等[35]利用纤维素无纺布作为骨架,设计出双面涂层(乙炔黑、PEO-PAN-Li7La3Zr2O2)的复合凝胶聚合物电解质(图2(c))。纤维素无纺布作为凝胶聚合物电解质的骨架能有效提高PEO-PAN-Li7La3Zr2O2复合材料的力学性能,同时PEO中的羟基和PAN中的氰基之间形成化学键,减少氰基与锂负极之间的不良反应,PEO和PAN复合可以有效继承PEO良好的力学性能和PAN优异的离子电导率,增加锂负极与凝胶聚合物电解质之间的界面稳定性,促进锂离子扩散。加入的Li7La3Zr2O2无机填料具有较高的离子电导率和对可溶性多硫化锂较强的化学亲和力,抑制多硫化锂对金属锂的副反应。这样设计的新型多功能凝胶聚合物电解质可以抑制多硫化锂的穿梭效应和稳定锂负极,这种在非原位聚合工艺中对基体进行基团改性获得多功能凝胶聚合物电解质的方法受到大家的认可。Zhang等[36]合成一种三氟甲基基团的聚酰亚胺(PI)作为聚合物凝胶电解质,可以将碳酸盐电解质凝胶化来制备阻燃凝胶聚合物电解质,PI链上的三氟甲基提供一个负电环境有利于锂离子转移,芳香苯环和溶剂烷基形成了CH/π相互作用,促进锂离子和溶剂之间相互作用使锂离子均匀沉积(图2(d))。该研究还采用了硫化聚丙烯腈(SPAN)作为正极活性材料,与传统的C@S正极依靠从固相到液相硫的分解沉积机制不同,SPAN与酯类电解质匹配,采用固-固转化机制支撑,消除了多硫化锂在传统电解液中的溶解抑制了穿梭效应。由此构建的阻燃型凝胶聚合物电解质不仅可以使锂硫电池的安全性显著提升,还具有抑制穿梭效应促进锂离子均匀沉积等多种功能,推动锂硫电池商业化进程。
上述非原位聚合凝胶电解质在锂硫电池外部聚合而成,通过筛选,在相应的基体中添加有效提高锂离子电导率的无机填料或通过嵌段共聚能吸附多硫化锂的官能团进行基团改性。非原位聚合工艺手段更易获得多功能凝胶聚合物电解质,进而改善锂硫电池的电化学性能。
2.2 原位聚合凝胶聚合物电解质
在锂硫电池的聚合物凝胶电解质体系中,与非原位聚合凝胶电解质相比,原位聚合GPE是在电池内部形成电解质,电极和电解质之间界面稳定性更强,界面阻抗更小,离子迁移速率更高。此外还具有制取便捷,操作安全,更易于实现产业化等优点。近年来,在锂硫电池体系中,原位聚合GPE的合成工艺受到科研工作者的青睐,不同原位聚合策略获得的凝胶聚合物电解质在界面稳定性等方面表现出明显差异。
2.2.1 原位界面聚合凝胶聚合物电解质
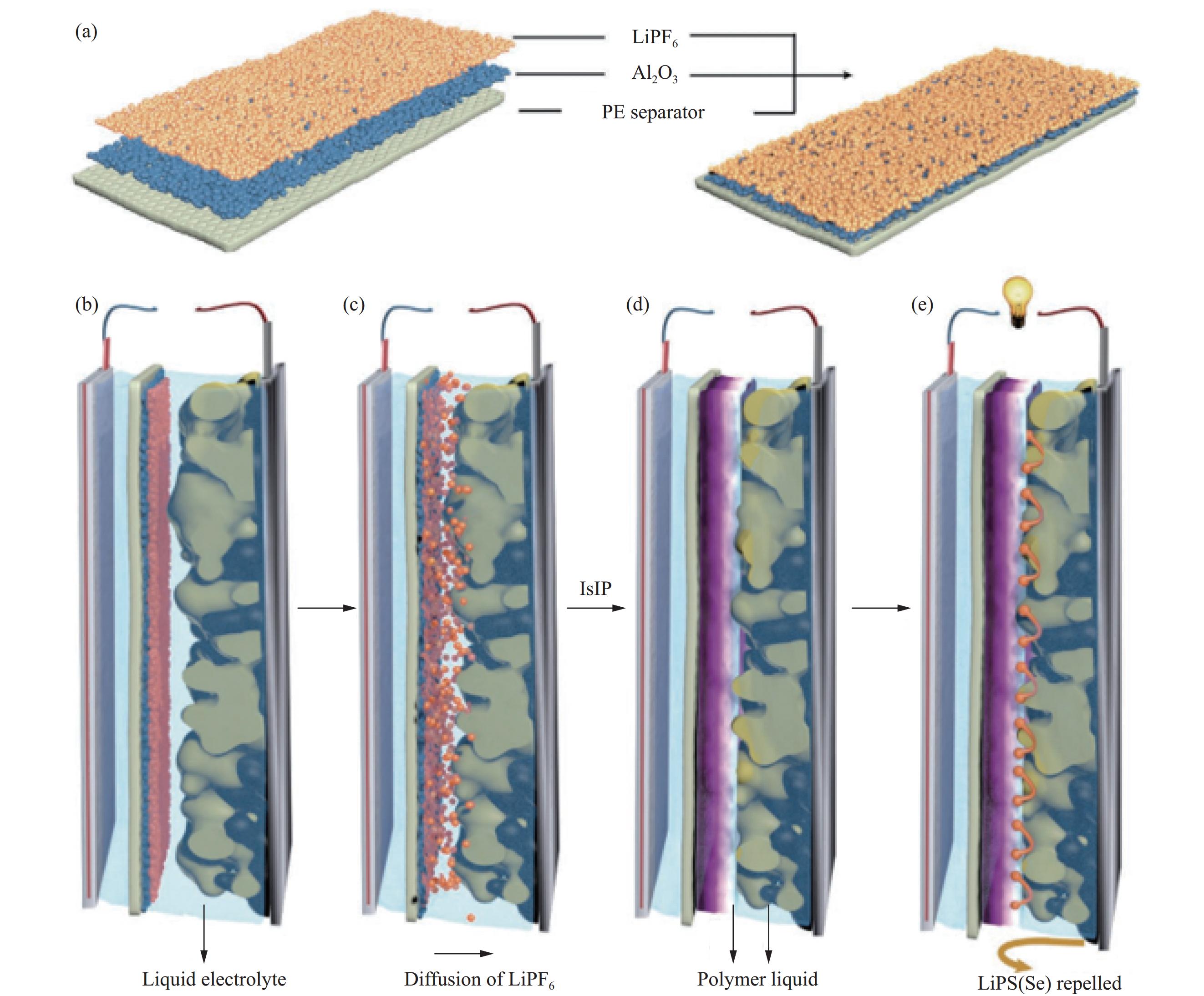
通过原位界面聚合这一工艺手段,能够实现在硫正极到锂负极间形成固化程度不同的GPE,在正极侧所形成未固化的液态电解质使Li2S适当分解,解决正极/GPE界面稳定性较差的问题,负极侧形成固化程度较高的GPE阻碍多硫化锂的穿梭效应。Wang等[37]经过原位界面聚合提出一种新的凝胶聚合物电解质,融合传统电解液和凝胶聚合物电解质的优点。制备一种用于锂硫电池的功能复合隔膜,用商用聚丙烯隔膜作为底层,顶层由PEO溶解的六氟磷酸锂组成,二者之间采用Al2O3纳米颗粒作为无机填料为顶层与底层之间提供亲和力,PEO层与硫正极相向,六氟磷酸锂由隔膜进入电解液引发1,3-二氧戊环(DOL)原位聚合(图3)。电解质由两种凝固程度不同的连续相组成,在正极和隔膜之间形成的原位聚合物凝胶相有助于抑制可溶性多硫化锂的穿梭,而正极内部则保留了电解液。混合电解质中电解液使正极和电解质之间界面稳定性良好,不易析出导电性差的Li2S,同时凝胶态电解质抑制多硫化锂的穿梭效应。这样“一箭双雕”的原位合成策略,为锂硫电池的原位聚合凝胶聚合物电解质提供了全新思路。
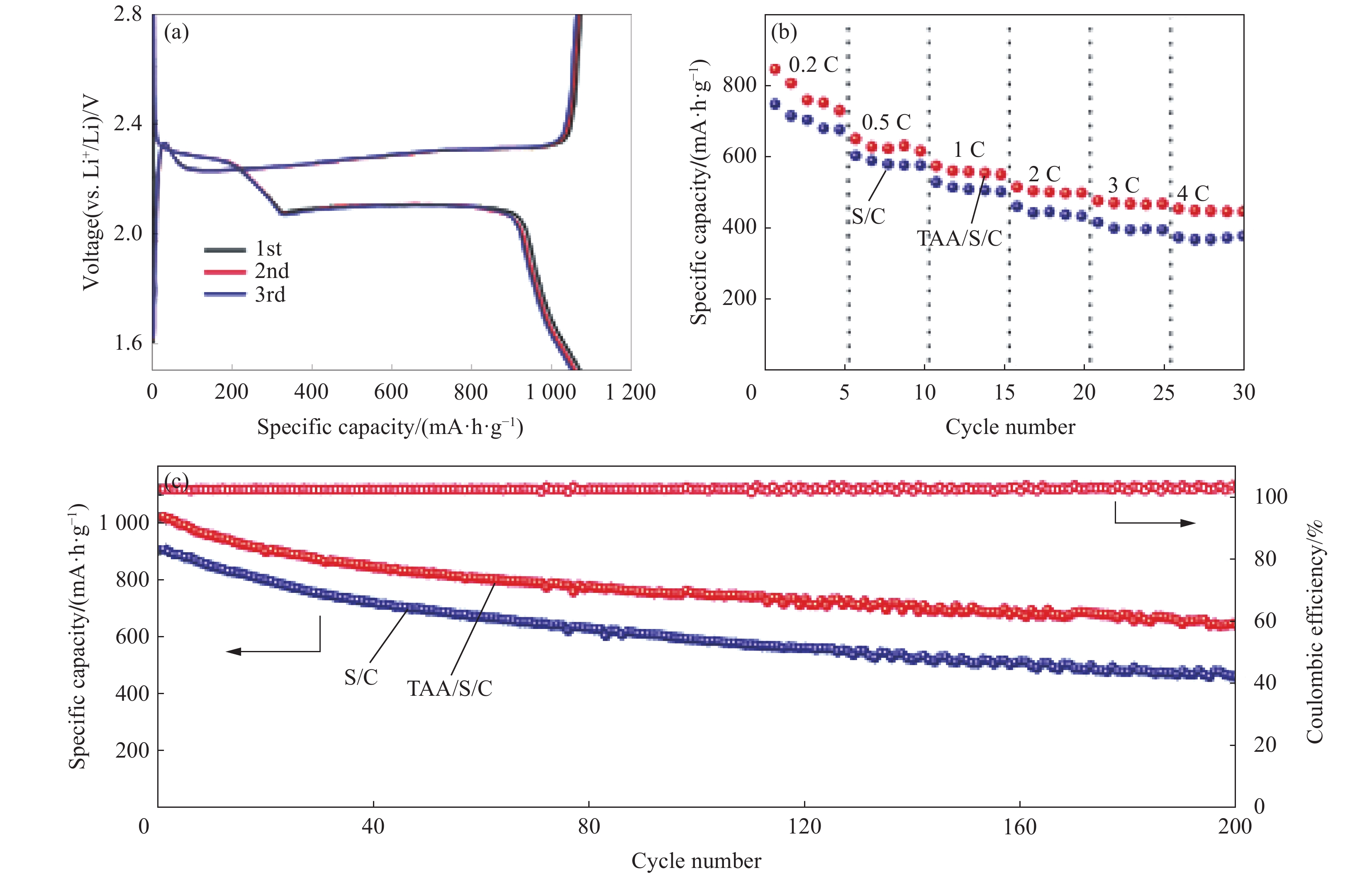
Li等[38]在硫正极中加入硫乙酰胺(TAA)作为添加剂,利用DOL进行原位聚合形成凝胶聚合物电解质,同样达到“双赢”的目的。TAA的加入可以有效抑制单质硫附近的DOL聚合而远离单质硫的DOL聚合则不受影响,使正极和GPE之间形成液态中间层。TAA进入中间层后,有助于Li2S适当的分解于凝胶聚合物液态中间层中,防止Li2S在正极堆积。Li2S分解进入中间层一方面可以防止导电性差的Li2S在正极堆积,另一方面多硫化锂的溶解会导致穿梭效应。所以此项研究应用固化程度更高的GPE抑制多硫化锂的穿梭效应。改善凝胶聚合物电解质锂硫电池中正极和GPE界面稳定性差的问题。TAA作为正极添加剂的锂硫电池在4 C倍率下具有450 mA·h·g−1的高放电比容量。0.1 C倍率下经200次充放电循环后保持645 mA·h·g−1的比容量,库仑效率接近100% (图4)。该项研究采用了通过修饰硫正极来引发电解液原位界面聚合,在合成聚合物凝胶电解质的过程中操作简便,组装电池后进行静置合成凝胶聚合物电解质,简化凝胶聚合物电解质的合成工艺。
![]() 图 4 (a)倍率为0.1 C时Li-硫乙酰胺(TAA)/S/C电池的充放电循环曲线;(b) Li-TAA/S/C和Li-S/C电池的倍率性能;(c) Li-TAA/S/C和Li-S/C电池的循环性能(倍率为0.1 C)[38]Figure 4. (a) Charge and discharge cycle curves of Li-thioacetamide (TAA)/S/C battery at 0.1 C rate; (b) Rate performance of Li-TAA/S/C and Li-S/C cells; (c) Cycling performance of Li-TAA/S/C and Li-S/C cells (Rate: 0.1 C)[38]
图 4 (a)倍率为0.1 C时Li-硫乙酰胺(TAA)/S/C电池的充放电循环曲线;(b) Li-TAA/S/C和Li-S/C电池的倍率性能;(c) Li-TAA/S/C和Li-S/C电池的循环性能(倍率为0.1 C)[38]Figure 4. (a) Charge and discharge cycle curves of Li-thioacetamide (TAA)/S/C battery at 0.1 C rate; (b) Rate performance of Li-TAA/S/C and Li-S/C cells; (c) Cycling performance of Li-TAA/S/C and Li-S/C cells (Rate: 0.1 C)[38]原位界面聚合策略制备出的凝胶聚合物电解质不仅可有效简化GPE制备工艺,并仍能保证正极和电解质之间的界面稳定性,制备出的混合电解质结合电解液和凝胶态电解质的优点,这种新型的聚合工艺已成为制备锂硫电池凝胶聚合物电解质的热点之一。
2.2.2 原位溶液聚合凝胶聚合物电解质
在锂硫电池凝胶聚合物电解质的发展中,原位溶液聚合是制备凝胶聚合物电解质的常用工艺。原位溶液聚合而成的凝胶聚合物电解质基体选择性少,为了制备出功能更加优越的GPE,进一步提升锂硫电池的性能,研究人员提出了多种可行策略。
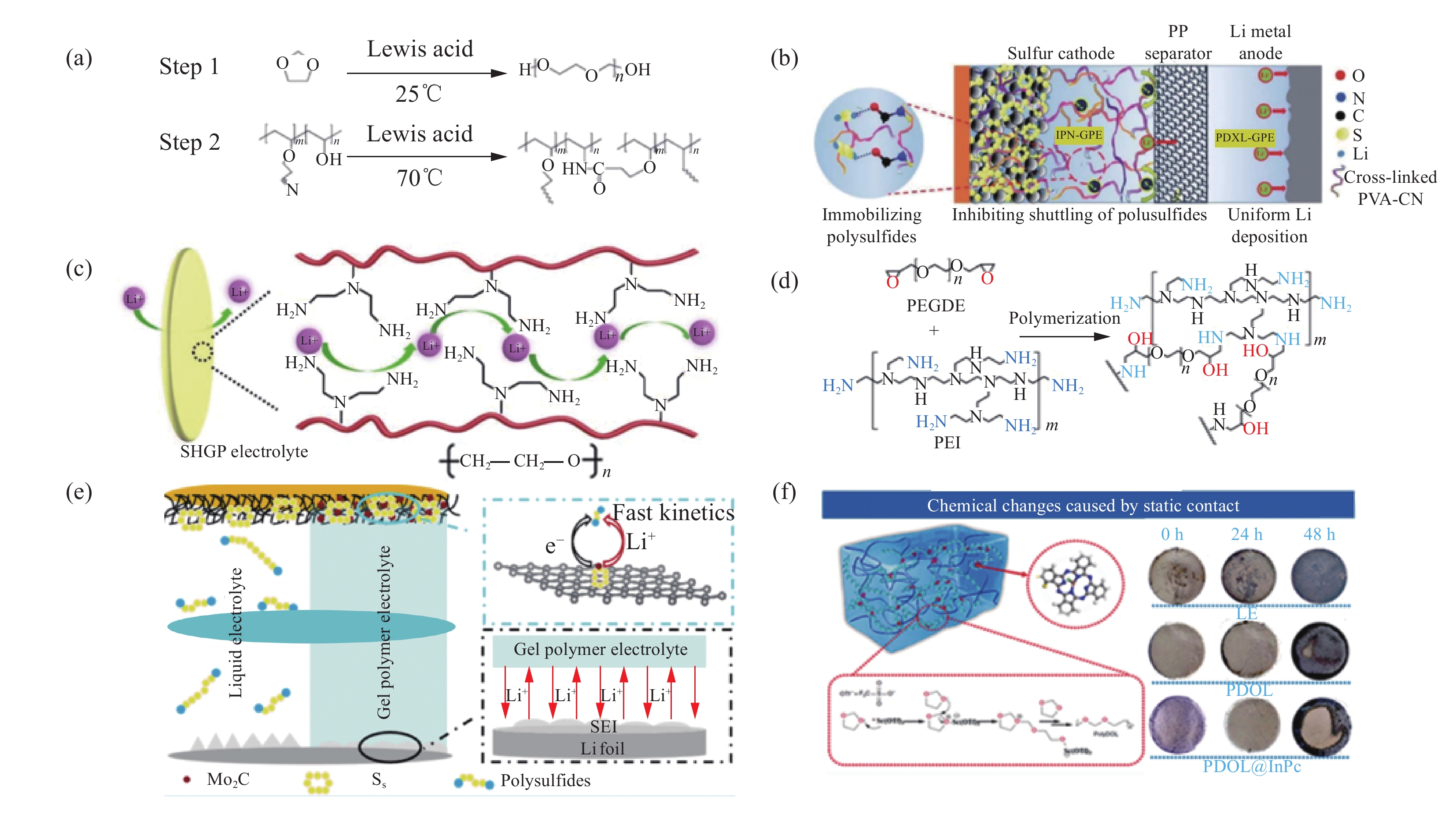
Yang等[39]利用能够原位聚合的物质引入基体中,进行交联形成互穿网络结构的策略,通过原位阳离子开环聚合1,3-二氧戊环(DOL)和氰基聚乙烯醇(PVA-CN)交联制备具有互穿网络结构的不对称双功能凝胶聚合物电解质。将PVA-CN引入基底的GPE在正极侧,羰基结构对于可溶性多硫化锂具有较高的结合能,可以固定多硫化锂,而负极侧未交联PVA-CN的PDOL基GPE拥有聚醚结构,对锂负极有很好的保护作用,可以促进锂离子均匀沉积(图5(a)、图5(b))。由此形成的不对称双功能凝胶聚合物电解质锂硫电池具有良好的循环稳定性(0.5 C倍率下进行充放电循环500次后比容量为807 mA·h·g−1)和高库仑效率(~99.6%)。Zhou等[40]通过聚乙二醇二缩水甘油酯(PEGDE)和支化聚乙烯亚胺的开环反应,发现一种新型的超高离子导电凝胶聚合物(SHGP)电解质(图5(c)、图5(d))。凝胶聚合物电解质中醚链与锂离子有很强的相互作用,能够提高锂离子的离子电导率。聚乙烯亚胺中的含氮基团中的氮原子含有孤对电子,可以通过与锂离子形成配位键来促进锂离子传导。与此同时凝胶聚合物电解质中含有的极性基团与多硫化锂发生化学作用,可以抑制多硫化锂的穿梭效应。电化学性能表明组装锂硫电池后在0.2 C倍率下进行充放电循环时获得950 mA·h·g−1的高比容量,在0.5 C倍率下充放电循环100次后的容量保持率高达98%,表现出优异的循环稳定性。
![]() 图 5 (a) 1,3-二氧戊环(DOL)与氰基聚乙烯醇(PVA-CN)合成互传网络(IPN)-GPE的两步反应机制;(b)具有非对称GPE网络的Li|IPN-GPE|S电池示意图[39];(c)超高离子导电凝胶聚合物(SHGP)电解质的原理图和工作原理;(d)聚乙二醇二缩水甘油酯(PEGDE)和支化聚乙烯亚胺(PEI)聚合合成方案[40];(e) 采用碳纳米管(CNT)/Mo2C/S正极和GPE的电池中的性能提升机制示意图[41];(f)当与锂箔静态接触时,电解液(LE)、PDOL和PDOL@酞菁铟(InPc)的原理图和光学图像[42]Figure 5. (a) The two-step reaction mechanisms of 1,3-dioxolane (DOL) and cyanoethyl polyvinyl alcohol (PVA-CN) to form the interpenetrating network (IPN)-GPE; (b) Schematic diagram of the Li|IPN-GPE|S battery with the asymmetric GPE network[39]; (c) Schematic illustration and the working principle of super-high ionic conductive gel polymer (SHGP) electrolyte; (d) Synthesis scheme of the polar polymer by polymerization of poly(ethylene glycol) diglycidyl ether (PEGDE) and branched polyethylenimine (PEI)[40]; (e) Schematic illustration for the performance improvement mechanisms of the cells with carbon nanotubes (CNT)/Mo2C/S cathodes and GPE[41]; (f) Schematic and optical images of electrolyte (LE), PDOL, and PDOL@indium phthalocyanine (InPc) when in static contact with lithium foil[42]
图 5 (a) 1,3-二氧戊环(DOL)与氰基聚乙烯醇(PVA-CN)合成互传网络(IPN)-GPE的两步反应机制;(b)具有非对称GPE网络的Li|IPN-GPE|S电池示意图[39];(c)超高离子导电凝胶聚合物(SHGP)电解质的原理图和工作原理;(d)聚乙二醇二缩水甘油酯(PEGDE)和支化聚乙烯亚胺(PEI)聚合合成方案[40];(e) 采用碳纳米管(CNT)/Mo2C/S正极和GPE的电池中的性能提升机制示意图[41];(f)当与锂箔静态接触时,电解液(LE)、PDOL和PDOL@酞菁铟(InPc)的原理图和光学图像[42]Figure 5. (a) The two-step reaction mechanisms of 1,3-dioxolane (DOL) and cyanoethyl polyvinyl alcohol (PVA-CN) to form the interpenetrating network (IPN)-GPE; (b) Schematic diagram of the Li|IPN-GPE|S battery with the asymmetric GPE network[39]; (c) Schematic illustration and the working principle of super-high ionic conductive gel polymer (SHGP) electrolyte; (d) Synthesis scheme of the polar polymer by polymerization of poly(ethylene glycol) diglycidyl ether (PEGDE) and branched polyethylenimine (PEI)[40]; (e) Schematic illustration for the performance improvement mechanisms of the cells with carbon nanotubes (CNT)/Mo2C/S cathodes and GPE[41]; (f) Schematic and optical images of electrolyte (LE), PDOL, and PDOL@indium phthalocyanine (InPc) when in static contact with lithium foil[42]在原位溶液聚合工艺中对硫正极进行修饰来改善硫正极的氧化还原反应动力学,在提升原位聚合GPE锂硫电池性能也有显著作用。Zhang等[41]通过在碳纳米管上修饰Mo2C纳米颗粒作为S活性物质的宿主来改善硫的氧化还原反应动力学,用六氟磷酸锂引发DOL进行原位开环聚合来合成凝胶聚合物电解质(图5(e))。由此形成的GPE与硫正极和锂负极均具有良好的界面稳定性和较高的锂离子电导率,有助于抑制穿梭效应和锂枝晶的生长。从密度泛函理论结果表明,Mo2C通过提高硫的氧化还原反应动力学,抑制循环过程中多硫化锂的穿梭效应。与传统的原位聚合凝胶聚合物电解质相比,该体系具有良好的电化学性能,由该GPE组装的锂硫电池在0.05 C倍率下进行充放电循环,放电比容量可达到
1024 mA·h·g−1。在0.5 C倍率下充放电循环100次后,放电比容量仍保持521 mA·h·g−1。在聚合反应过程中加入添加剂对凝胶聚合物电解质的功能进行合理设计,对于原位聚合GPE锂硫电池电化学性能的提升同为重要。Guo等[42]在原位聚合反应过程中加入可溶性酞菁铟(InPc),在引发剂三氟磺酸钪(Sc(OTf)3)的引发下发生开环反应制备一种新型凝胶聚合物电解质。通过物理及化学表征技术及理论计算相结合,结果表明引入的酞菁铟通过重建电场诱导了锂离子均匀沉积,同时共轭酞菁铟基团有效控制凝胶聚合物电解质局部配位环境,抑制可溶性多硫化锂的穿梭效应,此外含有InPc的凝胶聚合物电解质可以与锂负极相互作用形成类似锂铟合金结构的保护层(图5(f))。用PDOL@InPc组装的Li-S袋状电池在8.0 mg·cm−2的硫负载,0.2 C倍率下进行充放电循环具有
1194.7 mA·h·g−1的放电比容量和325 W·h·kg−1的高能量密度,表明该凝胶聚合物Li-S电池在新能源汽车实际应用中具有巨大的潜力。表 1 运用不同合成工艺GPE的优点及其组装锂硫电池的性能参数Table 1. Advantages of using different synthetic processes GPE and the performance parameters of assembling lithium-sulfur batteries合成工艺 优点 GPE 离子电导率/(S·cm−1) 面载量/(mg·cm−2) 放电比容量/(mA·h·g−1) 循环性能 非原位聚合法 合成凝胶流程简便,容易在分子水平进行修饰,兼容性更好,易于整合多种材料的优点 PVFH-TOC-PEG 8×10−3(25℃) 5 1103 (2 mA·cm−1)8 mA·cm−2、650次循环后放电比容量680 mA·h·g−1 PVDF-HFP-m-Co3O4-NH3 3.23×10−3(室温) 2 — 0.5 C、150次循环后放电比容量620 mA·h·g−1 PVDF-HFP-Al2O3 1.85×10−3(室温) 1 1233 (0.1 C)0.1 C、150次循环后放电比容量841.5 mA·h·g−1 PEO-PAN-Li7La3Zr2O2 2.1×10−3(30℃) 2±0.3 1459 (0.1 C)1 C、300次循环后放电比容量575 mA·h·g−1 PI10 6.22×10−3(室温) 1 — 0.2 C、450次循环后放电比容量 1154.3 mA·h·g−1原位界面聚合法 在硫正极到锂负极间形成固化程度不同的GPE,解决正极/GPE界面稳定性较差的问题 PDOL 5.56×10−3(室温) 2 1102 (0.1 C)0.1 C、200次循环后放电比容量805 mA·h·g−1 PDOL — 1.5 760(0.5 C) 0.1 C、200次循环后放电比容量645 mA·h·g−1 原位溶液聚合法 扬弃了传统GPE合成过程中的聚合物材料溶解、干燥涂膜等复杂工序,制取便捷、操作安全 PVA-CN/PDOL 3.23×10−3(25℃) 1.5 — 0.5 C、500次循环后放电比容量807 mA·h·g−1 SHGP 0.75×10−3(30℃) 2.5 950(0.2 C) 0.5 C、100次循环后放电比容量715 mA·h·g−1 PDOL 2.5×10−2(室温) 1 1024 (0.05 C)0.5 C、100次循环后放电比容量521 mA·h·g−1 PDOL@InPc 3.7×10−3(室温) 1.5±0.02 1194.7 (0.2 C)0.2 C、260次循环后放电比容量673.5 mA·h·g−1 Notes: PI10—Polyimide10; PDOL—Poly (1, 3-dioxopentylene); SHGP—Novel gel polymer synthesized from polyethylene glycol diglycidyl ester and branched polyethylene imide; PDOL@InPc—Indium phthalocyanine added to PDOL. 3. 原位表征技术在锂硫电池GPE中的应用
在锂硫电池凝胶聚合物电解质的发展过程中,为了合理设计凝胶聚合物电解质的成分和结构[43],研究者还需对锂硫电池凝胶聚合物电解质的反应机制及其对电化学性能的影响进行全面而深入的了解。近年来出现了众多原位表征方法包括:原位X射线吸收光谱法(In-situ XAS)、原位透射电子显微镜(In-situ TEM)、原位原子力显微镜(In-situ AFM)、原位拉曼光谱(In-situ Raman)、原位紫外可见吸收光谱(In-situ UV-Vis)等[44-45],这些技术已被用于探究锂硫电池电化学反应过程的内部结构变化,通过联用其他设备也可以获取不同发现,为进一步合理设计GPE锂硫电池提供助力,根据仪器的功能属性,本文进行了下述分类(表2)。
表 2 不同原位表征方法及其用途Table 2. Different in situ characterization methods and their uses原位表征方法 特点 测试对象 在锂硫电池中的用途 In-situ TEM 提供电化学反应过程中电极在高空间分辨率下的实时综合信息,探究锂硫电池中的微观结构演变和化学组成变化 锂硫电池循环过程中的固态产物 观测锂硫电池中Li2S的结晶状态及其演变 In-situ AFM 结合模拟电池环境条件,并跟踪这种环境下观测物的形貌演变 固体材料表面结构(包括绝缘体) 观测正极侧不溶性Li2S及Li2S2演变过程和负极侧SEI形成及演变途径 In-situ Raman及
In-situ UV-Vis根据拉曼散射光谱和紫外吸收光谱的不同来确定不同分子的组成结构 锂硫电池循环过程中产生的多硫化物 观测锂硫电池体系中,正极/电解质界面处多硫化锂的分布情况 In-situ XAS 不依赖长程有序结构而对目标原子近邻结构敏感,能够得到材料局部几何结构和电子结构信息 催化剂及多硫化物 定量测定硫中几种含硫物种的含量,研究材料与多硫化物相互作用 3.1 形貌及结构表征
在锂硫电池反应体系中,为了探究锂硫电池内部多硫化锂的穿梭效应,锂枝晶的生长以及凝胶聚合物的形貌状态,在过去的几十年里,各种形貌及结构表征方法,如SEM、TEM、AFM等来进行锂硫电池工作状态内部形貌演变的观测中[46]。
3.1.1 原位透射电子显微镜
原位透射电镜技术能够提供电化学反应过程中电极在高空间分辨率下的实时综合信息,可以探究锂硫电池中的微观结构演变和化学组成变化。Wang等[47]采用原位透射电镜技术和原位池装置研究了在固态锂硫电池中Li2S的沉淀和分解并通过电子衍射实时表征了Li2S的结构变化。此外,通过联用微机电系统(MEMS)加热装置,以研究温度对该过程的影响。结果表明,随着电化学反应的进行,Li2S由非晶/纳米晶向多晶态转变,温度越高,Li2S的析出更彻底,随着Li+在高温下传导Li2S的分解变得容易,Li+在高温下可增强Li2S可逆性(图6)。这一研究表明In-situ TEM可以为锂硫电池中Li2S的结晶状态及其演变提供清晰的解释,并提出了一种通过控制反应温度来操纵其可逆性的方法,为锂硫电池凝胶聚合物电解质功能设计指明道路。
3.1.2 原位原子力显微镜
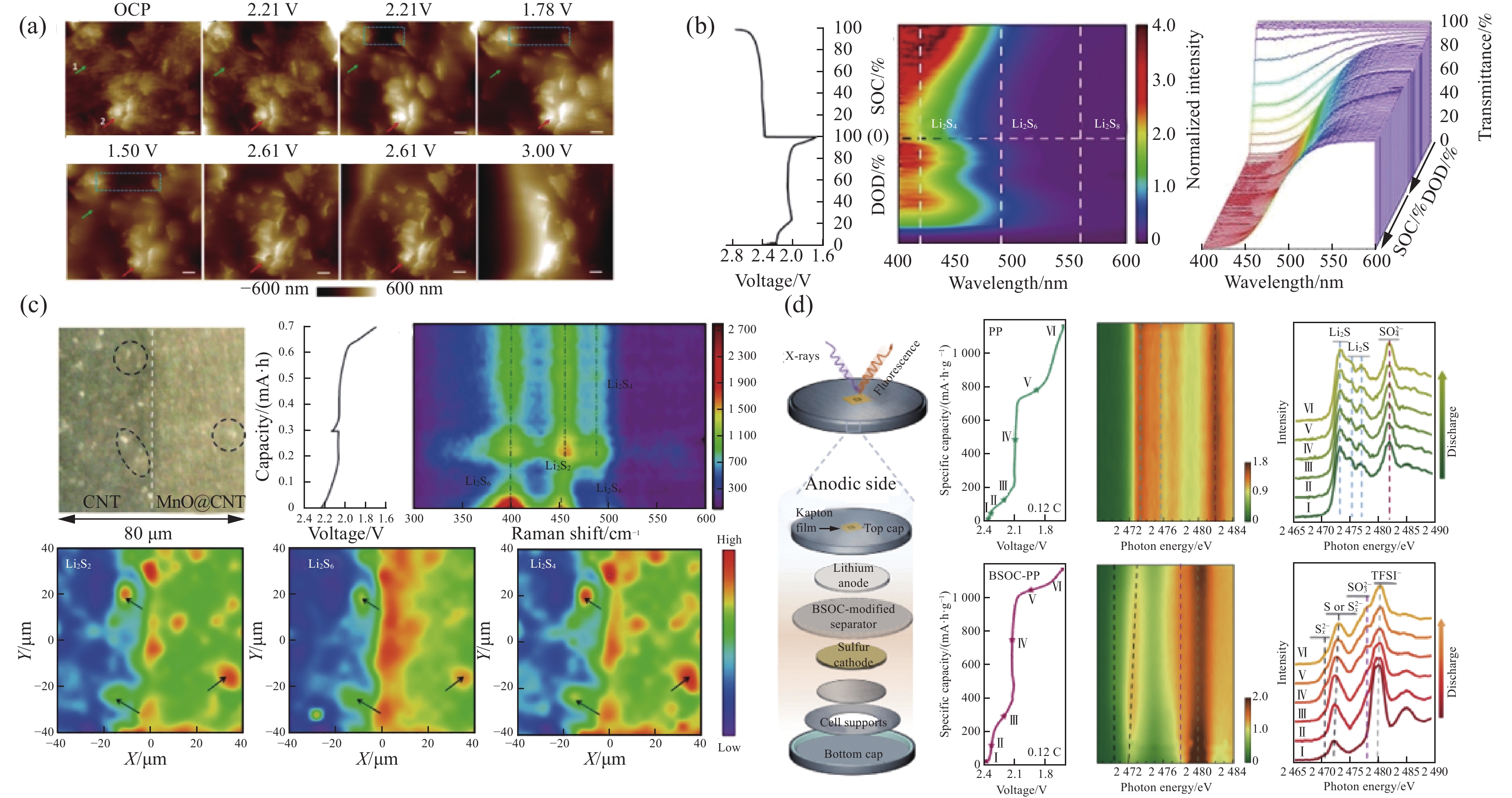
与In-situ TEM主要强调对观测物形貌演变的直接观察不同,In-situ AFM可以结合模拟电池环境条件,并跟踪这种环境下观测物的形貌演变[48]。Song等[49]通过原位光学显微镜(In-situ OM)及In-situ AFM探究了硫正极/电解质界面处不溶性的Li2S2和Li2S的成核、生长、沉积、溶解等动态过程(图7(a))[50],通过In-situ OM监测,实现了硫正极与电解质界面反应的直接可视化。
![]() 图 7 (a) Li2S2和Li2S的成核、生长、沉积、溶解等动态过程[50];(b)可溶性多硫化锂的In-situ UV-Vis光谱;(c)硫正极上Li2Sx的动态分布情况[53];(d)有/无碳基体中的硫化铋/氧化铋纳米团簇(BSOC)的Li/S电池负极侧的原位S K-edge XAS光谱[54]Figure 7. (a) Dynamic processes of nucleation, growth, deposition, and dissolution of Li2S2 and Li2S[50]; (b) In-situ UV-Vis spectra of soluble lithium polysulfide; (c) Dynamic distribution of Li2Sx on the positive sulfur electrode[53]; (d) In situ/operando S K-edge XAS observations at the anodic side of Li/S cells with/without bismuth sulfide/bismuth oxide nanoclusters in a carbon matrix (BSOC) layer[54]OCP—Open circuit potential; DOD—Depth of discharge; SOC—State of charge; PP—Polypropylene; TFSI—Thium bistrifluoromethane sulfonimide
图 7 (a) Li2S2和Li2S的成核、生长、沉积、溶解等动态过程[50];(b)可溶性多硫化锂的In-situ UV-Vis光谱;(c)硫正极上Li2Sx的动态分布情况[53];(d)有/无碳基体中的硫化铋/氧化铋纳米团簇(BSOC)的Li/S电池负极侧的原位S K-edge XAS光谱[54]Figure 7. (a) Dynamic processes of nucleation, growth, deposition, and dissolution of Li2S2 and Li2S[50]; (b) In-situ UV-Vis spectra of soluble lithium polysulfide; (c) Dynamic distribution of Li2Sx on the positive sulfur electrode[53]; (d) In situ/operando S K-edge XAS observations at the anodic side of Li/S cells with/without bismuth sulfide/bismuth oxide nanoclusters in a carbon matrix (BSOC) layer[54]OCP—Open circuit potential; DOD—Depth of discharge; SOC—State of charge; PP—Polypropylene; TFSI—Thium bistrifluoromethane sulfonimide通过对锂硫电池的内部形貌进行表征能够对电极/电解质界面物质的结构演变情况有一个直观的判断,为更有针对性地提出锂硫电池改进策略,为GPE的结构设计提供参考。
3.2 元素表征
在锂硫电池体系中,形貌表征仅适用于固相,如硫、Li2S2和Li2S等。全固态电解质可以避免锂硫电池体系中多硫化锂的穿梭效应,因此在全固态锂硫电池中结构及形貌表征多用于探究固态电解质的结构及元素组成。凝胶聚合物电解质中含有醚类电解液,在锂硫电池电化学反应过程中会产生长链多硫化锂,由于长链多硫化锂可溶于传统醚类电解液,探究不同链长多硫化锂在电解液中的化学转变及其与电极之间的反应对于全面了解锂硫电池化学反应体系中的整个电化学反应过程至关重要,为了探究凝胶电解质中多硫化锂的转化及其分布情况,锂硫电池电解质的结构及元素表征同样重要。
3.2.1 原位紫外可见吸收光谱及原位拉曼光谱
UV-Vis是利用某些物质的分子吸收10~100 nm光谱区的辐射来进行分析测定的方法。因此被广泛应用于锂硫电池中多硫化锂的定性及定量检测。UV-Vis通常与Raman进行联用来观察固态锂硫电池中多硫化锂的分布情况。Raman是一种散射光谱,是根据入射光频率不同的散射光谱进行分析以得到分子的振动、转动方面的信息并应用于分子结构研究的一种分析方法。根据拉曼光谱中的拉曼信号响应,可以探究锂硫电池充放电循环前后正极/电解质界面上多硫化锂的沉积情况[51]。将UV-Vis、Raman和自制原位池联用可以探究锂硫电池体系中,正极/电解质界面处多硫化锂的分布情况。
Luo等[52]运用原位紫外可见光谱及拉曼光谱观测出在固态锂硫电池体系充放电循环前后,很大一部分多硫化锂不参与锂硫电池氧化还原反应,被禁锢在固态电解质之中。在电解液锂硫电池体系观测到,多硫化锂一旦在正极界面上生成就会迅速扩散到电解液当中。表明固态电解质和电解液相比有助于抑制多硫化锂的穿梭效应。在最近的研究中,Meng等[53]用原位紫外可见吸收光谱和原位拉曼光谱等原位表征观测到Li2S2在锂硫电池体系中的演化。通过In-situ UV-Vis实时观测锂硫电池体系中可溶性多硫化锂浓度(图7(b)),In-situ Raman观测硫正极上Li2Sx的动态分布情况(图7(c)),结果表明,在放电过程中,Li2S2同时经历电化学还原和化学歧化,使多硫化锂进一步溶解和Li2S生成,但Li2S2的化学反应对放电比容量没有贡献。与完全氧化的Li2S相比,Li2S2在充电末期仍有残余,Li2S2电化学转化和副化学反应是限制硫利用率的决定性因素。原位可见光谱技术的应用能够对锂硫电池体系中多硫化锂的组分进行动态的监测,对于了解整个电化学反应过程至关重要,研究者们与其他仪器进行联用对锂硫电池内部体系进行更深层次的探究,对合成高性能锂硫电池GPE提供启发。
3.2.2 原位X射线吸收光谱法
X射线吸收光谱法(XAS)是研究锂硫电池体系的重要方法之一。X射线吸收光谱可以揭示被探测原子的化学状态和提供材料与物质之间的相互作用等信息。X射线吸收光谱法主要包括:前缘吸收谱、X射线吸收近边结构谱(XANES)和广延X射线吸收精细结构谱(EXAFS)。其中,XANES对样品是否为晶态没有要求,主要与内层电子从低能级到高能级的跃迁有关,反映原子中电子的状态[52]。将XANES与电化学工作站联用在凝胶聚合物电解质锂硫电池中是表征催化剂重要手段,Meng等[53]利用XANES证明了其研发的催化剂SA-Ni@HCS中的Ni价态和镍酞菁中的Ni相似。X射线吸收精细结构谱定量拟合证明Ni是由N四配位的,配位数为4.2。表明已将单原子Ni-N4位点锚定在导电中空纳米球,成功设计出一种模型催化剂(SA-Ni@HCS)。此研究表明X射线吸收光谱法对于表征固态锂硫电池中催化剂的价态、配位数、配位原子、轨道杂化方式等结构信息十分有力,为探索GPE锂硫电池中的物质信息奠定基础。
研究者们为了追踪多硫化锂在锂硫电池体系中的迁移情况,Jia等[54]利用隔膜修饰方法,嵌入碳基(BSOC)的混合硫化铋和氧化铋纳米团簇作为Li/S电池的催化层,联用X射线吸收光谱和原位池进行原位表征(图7(d)),全面分析经修饰的隔膜和未经修饰的隔膜组装的Li-S电池中穿梭效应的动态过程。上述In-situ XAS技术促进对锂硫电池体系多硫化锂穿梭效应的认识,为设计出合理锂硫电池GPE提供基础。
综上所述,原位表征技术能够实时检测锂硫电池充放电反应过程中内部的复杂转化机制,对研究者们理解锂硫电池的电化学反应和反应机制有着至关重要的作用,为凝胶聚合物电解质、电极材料和电池结构的合理设计奠定了基础,进而加速锂硫电池整体性能的提升[55]。
4. 总结与展望
因其适用于低成本高能量密度储能器件,锂硫电池以其高容量正极和高能量密度而被视为下一代储能系统。然而,电极与电解质之间以及电极内部不稳定的多界面仍然限制了其商业应用。因此开发功能化凝胶聚合物电解质(GPE)锂硫电池是抑制多硫化锂穿梭效应、稳定电极界面和提升电池安全性的必要手段,但GPE基底缺陷仍然阻碍着GPE锂硫电池的商业化进程,合理设计功能化GPE可以解决上述问题。近年来,科研人员提出多种聚合工艺,合理采用不同聚合工艺实现锂硫电池GPE功能化。综上所述,针对锂硫电池GPE合成工艺及功能化改进进行深入探究,利用先进表征技术阐明锂硫电池电化学作用机制,为推动锂硫电池的商业化提供助力。基于上述研究进展,经过功能化的GPE锂硫电池在一定程度上能够改善锂硫电池体系中的关键问题,但凝胶聚合物锂硫电池商业化进程仍然面临诸多挑战。未来锂硫电池GPE设计应集中于以下几点:GPE中液体泄露仍然使GPE锂硫电池具有安全隐患,在GPE中加入阻燃增塑剂是改进此类缺陷的绝妙选择;结合理论计算预测加入不同填料时GPE的离子电导率以优化填料选择[56];应用更先进的原位表征技术对锂硫电池转化动力学实时检测,从而为锂硫电池GPE的合理设计提供依据。
-
图 2 (a) TOC晶体结构及聚偏氟乙烯-共六氟丙烯(PVFH)-TOC-聚乙二醇(PEG)电解质合成示意图[32];(b) PVDF-六氟丙烯(HFP)@氨化介孔Co3O4 (m-Co3O4(s)-NH2)的合成路线示意图[33];(c)聚环氧乙烷(PEO)-聚丙烯腈(PAN)-LLZO GPE在Li-S电池中的性能提升机制示意图[35];(d)聚合物凝胶形成机制示意图[36]
Figure 2. (a) Crystal structure of TOC and schematic illustration for the synthesis of the polyvinylidene fluoride-hexafluoropropylene (PVFH)-TOC-polyethylene glycol (PEG) electrolyte[32]; (b) Schematic illustration of the synthesis route of poly(vinylidene fluoride)-hexafluoropropylene (PVDF-HFP)@ammoniated mesoporous Co3O4 (m-Co3O4(s)-NH2)[33]; (c) Schematic illustration for the performance improvement mechanisms of polyethylene oxide (PEO)-polyacrylonitrile (PAN)-LLZO GPE applied in the Li-S battery[35]; (d) Schematic diagram of formation mechanism for the polymer gel polymer[36]
TOC—Ti32O16(OCH2CH2O)32(RCOO)16(EGH)16 (R: t-CH3-CH2CH2CH2—; EGH: —OCH2CH2OH); APTES—3-aminopropyltriethoxysilane; LLZO—Li7La3Zr2O2
图 4 (a)倍率为0.1 C时Li-硫乙酰胺(TAA)/S/C电池的充放电循环曲线;(b) Li-TAA/S/C和Li-S/C电池的倍率性能;(c) Li-TAA/S/C和Li-S/C电池的循环性能(倍率为0.1 C)[38]
Figure 4. (a) Charge and discharge cycle curves of Li-thioacetamide (TAA)/S/C battery at 0.1 C rate; (b) Rate performance of Li-TAA/S/C and Li-S/C cells; (c) Cycling performance of Li-TAA/S/C and Li-S/C cells (Rate: 0.1 C)[38]
图 5 (a) 1,3-二氧戊环(DOL)与氰基聚乙烯醇(PVA-CN)合成互传网络(IPN)-GPE的两步反应机制;(b)具有非对称GPE网络的Li|IPN-GPE|S电池示意图[39];(c)超高离子导电凝胶聚合物(SHGP)电解质的原理图和工作原理;(d)聚乙二醇二缩水甘油酯(PEGDE)和支化聚乙烯亚胺(PEI)聚合合成方案[40];(e) 采用碳纳米管(CNT)/Mo2C/S正极和GPE的电池中的性能提升机制示意图[41];(f)当与锂箔静态接触时,电解液(LE)、PDOL和PDOL@酞菁铟(InPc)的原理图和光学图像[42]
Figure 5. (a) The two-step reaction mechanisms of 1,3-dioxolane (DOL) and cyanoethyl polyvinyl alcohol (PVA-CN) to form the interpenetrating network (IPN)-GPE; (b) Schematic diagram of the Li|IPN-GPE|S battery with the asymmetric GPE network[39]; (c) Schematic illustration and the working principle of super-high ionic conductive gel polymer (SHGP) electrolyte; (d) Synthesis scheme of the polar polymer by polymerization of poly(ethylene glycol) diglycidyl ether (PEGDE) and branched polyethylenimine (PEI)[40]; (e) Schematic illustration for the performance improvement mechanisms of the cells with carbon nanotubes (CNT)/Mo2C/S cathodes and GPE[41]; (f) Schematic and optical images of electrolyte (LE), PDOL, and PDOL@indium phthalocyanine (InPc) when in static contact with lithium foil[42]
图 7 (a) Li2S2和Li2S的成核、生长、沉积、溶解等动态过程[50];(b)可溶性多硫化锂的In-situ UV-Vis光谱;(c)硫正极上Li2Sx的动态分布情况[53];(d)有/无碳基体中的硫化铋/氧化铋纳米团簇(BSOC)的Li/S电池负极侧的原位S K-edge XAS光谱[54]
Figure 7. (a) Dynamic processes of nucleation, growth, deposition, and dissolution of Li2S2 and Li2S[50]; (b) In-situ UV-Vis spectra of soluble lithium polysulfide; (c) Dynamic distribution of Li2Sx on the positive sulfur electrode[53]; (d) In situ/operando S K-edge XAS observations at the anodic side of Li/S cells with/without bismuth sulfide/bismuth oxide nanoclusters in a carbon matrix (BSOC) layer[54]
OCP—Open circuit potential; DOD—Depth of discharge; SOC—State of charge; PP—Polypropylene; TFSI—Thium bistrifluoromethane sulfonimide
表 1 运用不同合成工艺GPE的优点及其组装锂硫电池的性能参数
Table 1 Advantages of using different synthetic processes GPE and the performance parameters of assembling lithium-sulfur batteries
合成工艺 优点 GPE 离子电导率/(S·cm−1) 面载量/(mg·cm−2) 放电比容量/(mA·h·g−1) 循环性能 非原位聚合法 合成凝胶流程简便,容易在分子水平进行修饰,兼容性更好,易于整合多种材料的优点 PVFH-TOC-PEG 8×10−3(25℃) 5 1103 (2 mA·cm−1)8 mA·cm−2、650次循环后放电比容量680 mA·h·g−1 PVDF-HFP-m-Co3O4-NH3 3.23×10−3(室温) 2 — 0.5 C、150次循环后放电比容量620 mA·h·g−1 PVDF-HFP-Al2O3 1.85×10−3(室温) 1 1233 (0.1 C)0.1 C、150次循环后放电比容量841.5 mA·h·g−1 PEO-PAN-Li7La3Zr2O2 2.1×10−3(30℃) 2±0.3 1459 (0.1 C)1 C、300次循环后放电比容量575 mA·h·g−1 PI10 6.22×10−3(室温) 1 — 0.2 C、450次循环后放电比容量 1154.3 mA·h·g−1原位界面聚合法 在硫正极到锂负极间形成固化程度不同的GPE,解决正极/GPE界面稳定性较差的问题 PDOL 5.56×10−3(室温) 2 1102 (0.1 C)0.1 C、200次循环后放电比容量805 mA·h·g−1 PDOL — 1.5 760(0.5 C) 0.1 C、200次循环后放电比容量645 mA·h·g−1 原位溶液聚合法 扬弃了传统GPE合成过程中的聚合物材料溶解、干燥涂膜等复杂工序,制取便捷、操作安全 PVA-CN/PDOL 3.23×10−3(25℃) 1.5 — 0.5 C、500次循环后放电比容量807 mA·h·g−1 SHGP 0.75×10−3(30℃) 2.5 950(0.2 C) 0.5 C、100次循环后放电比容量715 mA·h·g−1 PDOL 2.5×10−2(室温) 1 1024 (0.05 C)0.5 C、100次循环后放电比容量521 mA·h·g−1 PDOL@InPc 3.7×10−3(室温) 1.5±0.02 1194.7 (0.2 C)0.2 C、260次循环后放电比容量673.5 mA·h·g−1 Notes: PI10—Polyimide10; PDOL—Poly (1, 3-dioxopentylene); SHGP—Novel gel polymer synthesized from polyethylene glycol diglycidyl ester and branched polyethylene imide; PDOL@InPc—Indium phthalocyanine added to PDOL. 表 2 不同原位表征方法及其用途
Table 2 Different in situ characterization methods and their uses
原位表征方法 特点 测试对象 在锂硫电池中的用途 In-situ TEM 提供电化学反应过程中电极在高空间分辨率下的实时综合信息,探究锂硫电池中的微观结构演变和化学组成变化 锂硫电池循环过程中的固态产物 观测锂硫电池中Li2S的结晶状态及其演变 In-situ AFM 结合模拟电池环境条件,并跟踪这种环境下观测物的形貌演变 固体材料表面结构(包括绝缘体) 观测正极侧不溶性Li2S及Li2S2演变过程和负极侧SEI形成及演变途径 In-situ Raman及
In-situ UV-Vis根据拉曼散射光谱和紫外吸收光谱的不同来确定不同分子的组成结构 锂硫电池循环过程中产生的多硫化物 观测锂硫电池体系中,正极/电解质界面处多硫化锂的分布情况 In-situ XAS 不依赖长程有序结构而对目标原子近邻结构敏感,能够得到材料局部几何结构和电子结构信息 催化剂及多硫化物 定量测定硫中几种含硫物种的含量,研究材料与多硫化物相互作用 -
[1] EVARTS E. Lithium batteries: To the limits of lithium[J]. Nature, 2015, 526: S93-S95. DOI: 10.1038/526S93a
[2] LI S, HUANG J, CUI Y, et al. A robust all-organic protective layer towards ultrahigh-rate and large-capacity Li metal anodes[J]. Nature Nanotechnology, 2022, 17: 613-621. DOI: 10.1038/s41565-022-01107-2
[3] YUE L, WANG X, CHEN L, et al. In situ interface engineering of highly nitrogen-rich triazine-based covalent organic frameworks for an ultra-stable, dendrite-free lithium-metal anode[J]. Energy & Environmental Science, 2024, 17(3): 1117-1131.
[4] SU C C, AMINE K. Dicarbonyl electrolyte for high-voltage lithium metal batteries: Importance of the entropy of solvation in bidentate solvent[J]. ACS Energy Letters, 2023, 9(1): 118-125.
[5] LIU X, WANG G, LYU Z, et al. A perspective on uniform plating behavior of Mg metal anode: Diffusion limited theory versus nucleation theory[J]. Advanced Materials, 2024, 36(9): 2306395. DOI: 10.1002/adma.202306395
[6] JI L, JIA Y, WANG X, et al. Strong adsorption, catalysis and lithiophilic modulation of carbon nitride for lithium/sulfur battery[J]. Nanotechnology, 2021, 32(19): 192002. DOI: 10.1088/1361-6528/abe002
[7] LIU S, LI J, YAN X, et al. Superhierarchical cobalt-embedded nitrogen-doped porous carbon nanosheets as two-in-one hosts for high-performance lithium-sulfur batteries[J]. Advanced Materials, 2018, 30(12): 1706895. DOI: 10.1002/adma.201706895
[8] MANTHIRAM A, FU Y, CHUNG S H, et al. Rechargeable lithium-sulfur batteries[J]. Chemical Reviews, 2014, 114(23): 11751-11787. DOI: 10.1021/cr500062v
[9] JI L, WANG X, JIA Y, et al. Flexible electrocatalytic nanofiber membrane reactor for lithium/sulfur conversion chemistry[J]. Advanced Functional Materials, 2020, 30(28): 1910533. DOI: 10.1002/adfm.201910533
[10] WANG Z, FAN Q, SI Y, et al. A self-regulatory organosulfur copolymer cathode towards high performance lithium-sulfur batteries[J]. Energy Storage Materials, 2023, 58: 222-231. DOI: 10.1016/j.ensm.2023.03.020
[11] HUANG Y, WANG Y, FU Y. A thermoregulating separator based on black phosphorus/MOFs heterostructure for thermo-stable lithium-sulfur batteries[J]. Chemical Engineering Journal, 2023, 454: 140250. DOI: 10.1016/j.cej.2022.140250
[12] HAN Z, REN H R, HUANG Z, et al. A permselective coating protects lithium anode toward a practical lithium-sulfur battery[J]. ACS Nano, 2023, 17(5): 4453-4462. DOI: 10.1021/acsnano.2c10047
[13] WU T, YE J, LI T, et al. Tetrathiafulvalene as a multifunctional electrolyte additive for simultaneous interface amelioration, electron conduction, and polysulfide redox regulation in lithium-sulfur batteries[J]. Journal of Power Sources, 2022, 536: 231482. DOI: 10.1016/j.jpowsour.2022.231482
[14] WU T, SUN G, LU W, et al. A polypyrrole/black-TiO2/S double-shelled composite fixing polysulfides for lithium-sulfur batteries[J]. Electrochimica Acta, 2020, 353: 136529. DOI: 10.1016/j.electacta.2020.136529
[15] MIKHAYLIK Y V. Electrolytes for lithium sulfur cells: US patent, 7358012 B2[P]. 2008-04-15.
[16] SHI C, SHAO S, ZONG C, et al. Organothiols for dual-interface modification of high performance lithium-sulfur batteries[J]. Chemical Engineering Journal, 2022, 448: 137552. DOI: 10.1016/j.cej.2022.137552
[17] QIU H, SONG Y, GU J, et al. A bifunctional electrolyte additive ammonium hexafluorophosphate for long cycle life lithium-sulfur batteries[J]. Materials Letters, 2023, 351: 134986. DOI: 10.1016/j.matlet.2023.134986
[18] XU R, ZHANG S, WANG X, et al. Recent developments of all-solid-state lithium secondary batteries with sulfide inorganic electrolytes[J]. Chemistry—A European Journal, 2018, 24(23): 6007-6018. DOI: 10.1002/chem.201704568
[19] FU K K, GONG Y, HITZ G T, et al. Three-dimensional bilayer garnet solid electrolyte based high energy density lithium metal-sulfur batteries[J]. Energy & Environmental Science, 2017, 10(7): 1568-1575.
[20] YIN Y X, XIN S, GUO Y G, et al. Lithium-sulfur batteries: Electrochemistry, materials, and prospects[J]. Angewandte Chemie International Edition, 2013, 52(50): 13186-13200. DOI: 10.1002/anie.201304762
[21] CUI Y, LI J, YUAN X, et al. Emerging strategies for gel polymer electrolytes with improved dual-electrode side regulation mechanisms for lithium-sulfur batteries[J]. Chemistry—An Asian Journal, 2022, 17(21): e202200746. DOI: 10.1002/asia.202200746
[22] FENTON D E, PARKER J M, WRIGHT P V. Complex of alkali metal ions with poly(ethylene oxide)[J]. Polymer, 1973, 14: 589.
[23] MICHOT T, NISHIMOTO A, WATANABE M. Electrochemical properties of polymer gel electrolytes based on poly (vinylidene fluoride) copolymer and homopolymer[J]. Electrochimica Acta, 2000, 45(8-9): 1347-1360. DOI: 10.1016/S0013-4686(99)00343-6
[24] JIA M, LI T, YANG D, et al. Polymer electrolytes for lithium-sulfur batteries: Progress and challenges[J]. Batteries, 2023, 9(10): 488. DOI: 10.3390/batteries9100488
[25] NAIR J R, BELLA F, ANGULAKSHMI N, et al. Nanocellulose-laden composite polymer electrolytes for high performing lithium-sulphur batteries[J]. Energy Storage Materials, 2016, 3: 69-76. DOI: 10.1016/j.ensm.2016.01.008
[26] QIAN J, JIN B, LI Y, et al. Research progress on gel polymer electrolytes for lithium-sulfur batteries[J]. Journal of Energy Chemistry, 2021, 56: 420-437. DOI: 10.1016/j.jechem.2020.08.026
[27] HU J K, YUAN H, YANG S J, et al. Dry electrode technology for scalable and flexible high-energy sulfur cathodes in all-solid-state lithium-sulfur batteries[J]. Journal of Energy Chemistry, 2022, 71: 612-618. DOI: 10.1016/j.jechem.2022.04.048
[28] SONG J, NOH H, LEE H, et al. Polysulfide rejection layer from alpha-lipoic acid for high performance lithium–sulfur battery[J]. Journal of Materials Chemistry A, 2015, 3(1): 323-330. DOI: 10.1039/C4TA03625E
[29] LI W, PANG Y, ZHU T, et al. A gel polymer electrolyte based lithium-sulfur battery with low self-discharge[J]. Solid State Ionics, 2018, 318: 82-87. DOI: 10.1016/j.ssi.2017.08.018
[30] CHIU L L, CHUNG S H. Composite gel-polymer electrolyte for high-loading polysulfide cathodes[J]. Journal of Materials Chemistry A, 2022, 10(26): 13719-13726. DOI: 10.1039/D2TA01867E
[31] ZHANG H, LU H, CHEN J, et al. A novel filler for gel polymer electrolyte with a high lithium-ion transference number toward stable cycling for lithium-metal anodes in lithium-sulfur batteries[J]. ACS Applied Materials & Interfaces, 2021, 13(41): 48622-48633.
[32] PEI F, DAI S, GUO B, et al. Titanium-oxo cluster reinforced gel polymer electrolyte enabling lithium-sulfur batteries with high gravimetric energy densities[J]. Energy & Environmental Science, 2021, 14(2): 975-985.
[33] LI J, CHEN X, YUE W. Mesoporous Co3O4-modified gel polymer electrolyte applied in lithium-sulfur batteries[J]. ACS Applied Energy Materials, 2022, 5(12): 15548-15558. DOI: 10.1021/acsaem.2c03130
[34] WANG H M, WANG Z Y, ZHOU C, et al. A gel polymer electrolyte with Al2O3 nanofibers skeleton for lithium-sulfur batteries[J]. Science China Materials, 2023, 66(3): 913-922. DOI: 10.1007/s40843-022-2252-1
[35] XIE P, YANG R, ZHOU Y, et al. Rationally designing composite gel polymer electrolyte enables high sulfur utilization and stable lithium anode[J]. Chemical Engineering Journal, 2022, 450: 138195. DOI: 10.1016/j.cej.2022.138195
[36] ZHANG H, CHEN J, LIU J, et al. Gel electrolyte with flame retardant polymer stabilizing lithium metal towards lithium-sulfur battery[J]. Energy Storage Materials, 2023, 61: 102885. DOI: 10.1016/j.ensm.2023.102885
[37] WANG W P, ZHANG J, YIN Y X, et al. A rational reconfiguration of electrolyte for high-energy and long-life lithium-chalcogen batteries[J]. Advanced Materials, 2020, 32(23): 2000302. DOI: 10.1002/adma.202000302
[38] LI C C, WANG W P, FENG X X, et al. High-performance quasi-solid-state lithium-sulfur battery with a controllably solidified cathode-electrolyte interface[J]. ACS Applied Materials & Interfaces, 2023, 15(15): 19066-19074.
[39] YANG Y J, WANG R, XUE J X, et al. In situ forming asymmetric bi-functional gel polymer electrolyte in lithium–sulfur batteries[J]. Journal of Materials Chemistry A, 2021, 9(48): 27390-27397. DOI: 10.1039/D1TA06007D
[40] ZHOU J, JI H, LIU J, et al. A new high ionic conductive gel polymer electrolyte enables highly stable quasi-solid-state lithium sulfur battery[J]. Energy Storage Materials, 2019, 22: 256-264. DOI: 10.1016/j.ensm.2019.01.024
[41] ZHANG Y J, XING Z Y, WANG W P, et al. Mo2C electrocatalysts for kinetically boosting polysulfide conversion in quasi-solid-state lithium-sulfur batteries[J]. ACS Applied Materials & Interfaces, 2021, 13(38): 45651-45660.
[42] GUO Y, LU J, JIN Z, et al. InPc-modified gel electrolyte based on in situ polymerization in practical high-loading lithium-sulfur batteries[J]. Chemical Engineering Journal, 2023, 469: 143714. DOI: 10.1016/j.cej.2023.143714
[43] HAN D D, LIU S, LIU Y T, et al. Lithiophilic gel polymer electrolyte to stabilize the lithium anode for a quasi-solid-state lithium-sulfur battery[J]. Journal of Materials Chemistry A, 2018, 6(38): 18627-18634. DOI: 10.1039/C8TA07685E
[44] ZHAO E, NIE K, YU X, et al. Advanced characterization techniques in promoting mechanism understanding for lithium-sulfur batteries[J]. Advanced Functional Materials, 2018, 28(38): 1707543. DOI: 10.1002/adfm.201707543
[45] ZHANG L, QIAN T, ZHU X, et al. In situ optical spectroscopy characterization for optimal design of lithium-sulfur batteries[J]. Chemical Society Reviews, 2019, 48(22): 5432-5453. DOI: 10.1039/C9CS00381A
[46] MAHANKALI K, THANGAVEL N K, REDDY A L M. In situ electrochemical mapping of lithium-sulfur battery interfaces using AFM-SECM[J]. Nano Letters, 2019, 19(8): 5229-5236. DOI: 10.1021/acs.nanolett.9b01636
[47] WANG Z, TANG Y, ZHANG L, et al. In situ TEM observations of discharging/charging of solid-state lithium-sulfur batteries at high temperatures[J]. Small, 2020, 16(28): 2001899. DOI: 10.1002/smll.202001899
[48] LANG S Y, SHI Y, GUO Y G, et al. Insight into the interfacial process and mechanism in lithium-sulfur batteries: An in situ AFM study[J]. Angewandte Chemie International Edition, 2016, 55(51): 15835-15839. DOI: 10.1002/anie.201608730
[49] SONG Y X, SHI Y, WAN J, et al. Dynamic visualization of cathode/electrolyte evolution in quasi-solid-state lithium batteries[J]. Advanced Energy Materials, 2020, 10(25): 2000465. DOI: 10.1002/aenm.202000465
[50] TAN J, LIU D, XU X, et al. In situ/operando characterization techniques for rechargeable lithium-sulfur batteries: A review[J]. Nanoscale, 2017, 9(48): 19001-19016. DOI: 10.1039/C7NR06819K
[51] WU H L, HUFF L A, GEWIRTH A A. In situ Raman spectroscopy of sulfur speciation in lithium-sulfur batteries[J]. ACS Applied Materials & Interfaces, 2015, 7(3): 1709-1719.
[52] LUO Y, FANG Z, DUAN S, et al. Direct monitoring of Li2S2 evolution and its influence on the reversible capacities of lithium-sulfur batteries[J]. Angewandte Chemie International Edition, 2023, 62(11): e202215802. DOI: 10.1002/anie.202215802
[53] MENG X, LIU Y, MA Y, et al. Diagnosing and correcting the failure of the solid-state polymer electrolyte for enhancing solid-state lithium-sulfur batteries[J]. Advanced Materials, 2023, 35(22): 2212039. DOI: 10.1002/adma.202212039
[54] JIA L, WANG J, REN S, et al. Unraveling shuttle effect and suppression strategy in lithium/sulfur cells by in situ/operando X-ray absorption spectroscopic characterization[J]. Energy & Environmental Materials, 2021, 4(2): 222-228.
[55] PREHAL C, VON MENTLEN J M, DRVARIČ T S, et al. On the nanoscale structural evolution of solid discharge products in lithium-sulfur batteries using operando scattering[J]. Nature Communications, 2022, 13(1): 6326. DOI: 10.1038/s41467-022-33931-4
[56] ZHANG X, LI X, ZHANG Y, et al. Accelerated Li+ desolvation for diffusion booster enabling low-temperature sulfur redox kinetics via electrocatalytic carbon-grazfted-CoP porous nanosheets[J]. Advanced Functional Materials, 2023, 33(36): 2302624. DOI: 10.1002/adfm.202302624
-
期刊类型引用(2)
1. 雷东,刘锟,杨富康,裴坤坤,胡国新,高寒阳. 石墨烯/聚氨酯复合材料的制备及其力学性能分析. 功能材料. 2022(01): 1181-1184+1189 .  百度学术
百度学术
2. 彭旭涛,熊巍,侯世维,刘濮源,何文俊,李贤娟,李晓燕. 后处理对连续玻璃纤维增强聚氨酯复合材料的影响. 塑料工业. 2022(07): 97-105 .  百度学术
百度学术
其他类型引用(3)
-
目的
凝胶聚合物电解质能够改善全固态电解质与双电极之间存在的高界面阻抗所带来的电荷转移受阻、锂沉积不均匀等问题,有效解决容量衰减快、循环稳定性差等缺陷。本文针对锂硫电池中制备凝胶聚合物电解质所采用的原位聚合和非原位聚合两种不同的工艺手段进行介绍,并讨论供科研工作者开发研究更适宜产业化的凝胶聚合物电解质的合成工艺,展望未来锂硫电池凝胶聚合物电解质合成设计的发展方向。
方法通过对近些年来国内外凝胶聚合物在锂硫电池中应用的相关文献整理,分析了制备凝胶类聚合物的主要方法,并分别对制备方法的原理、优缺点进行了总结;按照聚合凝胶聚合物的类型,将其分为原位聚合凝胶聚合物和非原位聚合凝胶聚合物,并分析了不同凝胶聚合物基底的特点;基于对凝胶聚合物电解质的研究,总结了不同原位表征手段在锂硫电池凝胶聚合物中的相关应用。
结果凝胶聚合物的制备方法主要分为:(1)非原位聚合:因其合成凝胶流程简便,更容易在分子水平进行修饰,兼容性更好,易于整合多种材料的优点。非原位聚合方法被广泛应用于制备多功能凝胶聚合物电解质。(2)原位聚合:与非原位聚合凝胶电解质相比,原位聚合凝胶聚合物电解质是在电池内部形成电解质,电极和电解质之间界面稳定性更强,界面阻抗更小,离子迁移速率更高。此外还具有制取便捷,操作安全,更易于实现产业化等优点。为了合理设计凝胶聚合物电解质的成分和结构,研究者还需对锂硫电池凝胶聚合物电解质的反应机理及其对电化学性能的影响进行全面而深入的了解。近年来出现了众多原位表征方法包括:原位X射线吸收光谱法(in-situ XAS)、原位透射电子显微镜(in-situ TEM)、原位原子力显微镜(in-situ AFM)、原位拉曼光谱(in-situ Raman)、原位紫外可见吸收光谱(in-situ UV-Vis)等,这些技术已被用于探究锂硫电池电化学反应过程的内部结构变化,通过联用其他设备也可以获取不同发现,为进一步合理设计凝胶聚合物电解质锂硫电池提供助力。
结论开发功能化凝胶聚合物电解质锂硫电池是抑制多硫化锂穿梭效应、稳定电极界面和提升电池安全性的必要手段,但凝胶聚合物电解质基底缺陷仍然阻碍着凝胶聚合物电解质锂硫电池的商业化进程,合理设计功能化凝胶聚合物电解质可以解决上述问题。近年来,科研人员提出多种聚合工艺,合理采用不同聚合工艺实现锂硫电池凝胶聚合物电解质功能化。综上所述,针对锂硫电池凝胶聚合物电解质合成工艺及功能化改进进行深入探究,利用先进表征技术阐明锂硫电池电化学作用机制,为推动锂硫电池的商业化提供助力。




 下载:
下载: