Preparation and properties of CNSs-BA/PET composites integrated with flame retardancy and melt-drop resistance
-
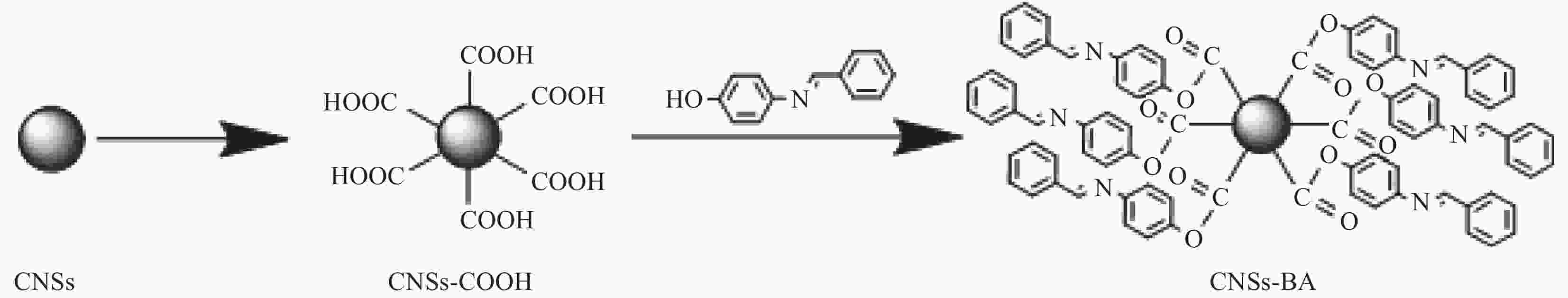
摘要: 聚对苯二甲酸乙二醇酯(PET)的易燃问题极大地威胁着人们的生命和财产安全,但通用阻燃剂无法兼顾其阻燃和抗熔滴需求。为同时改善PET的阻燃性和抗熔滴性,在碳纳米球(CNSs)表面接枝芳香席夫碱(BA)制备了一种新型的纳米碳基复合阻燃剂CNSs-BA,并将其通过熔融共混法引入PET中制备了复合材料。对CNSs-BA阻燃剂的形貌结构和热稳定性进行了表征,并研究了CNSs-BA/PET复合材料的阻燃性及其阻燃机制。结果表明:CNSs-BA为粒径50 nm左右的球状颗粒,热稳定性良好。当CNSs-BA添加量仅为2.0wt.%时,CNSs-BA/PET的LOI从PET的21.0%提高至28.1%,阻燃等级达到UL-94 V-0级,峰值热释放速率降低了46.3%。阻燃机制研究表明:CNSs-BA/PET表现出典型的凝聚相阻燃机制,CNSs-BA的引入可显著提高PET的成炭性,CNSs-BA/PET的高温残炭量较纯PET提高了55.4%,且其成炭量的实际值大于理论值。与纯PET相比,CNSs-BA阻燃剂的引入使PET熔融之后发生了高温交联,使得CNSs-BA/PET燃烧生成的炭层致密性、连续性和热稳定性均显著提高。
-
关键词:
- 碳纳米球 /
- 聚对苯二甲酸乙二醇酯 /
- 复合材料 /
- 阻燃 /
- 抗熔滴
Abstract: The inherent flammability of polyethylene terephthalate (PET) poses a considerable THReat to human lives and property, yet conventional flame retardants fail to adequately address both flame resistance and melt-drop concerns. To effectively enhance both the flame retardancy and melt-drop resistance of PET, a novel nanocarbon- method. The morphology structure and thermal stability of CNSs-BA were characterized. The flame retardancy of based composite flame retardant, CNSs-BA, was prepared by grafting aromatic Schiff bases (BA) on the surface of carbon nanospheres (CNSs), and the composite was prepared by introducing CNSs-BA into PET via melt blending CNSs-BA/PET composites and its flame retardant mechanism were investigated. The results show that the CNSs-BA with the addition of just 2.0wt.% of CNSs-BA, the limiting oxygen index (LOI) of the CNSs-BA/PET composite increased significantly from 21.0% to 28.1%, achieving a UL-94 V-0 flame retardant grade. Furthermore, the peak heat exhibit a spherical shape with a particle size approximating 50 nm and possess remarkable thermal stability. Notably, release rate was reduced by an impressive 46.3%. The investigation into the flame retardant mechanism revealed that the CNSs-BA/PET composite exhibits a typical condensed-phase flame retardant behavior. The introduction of CNSs-BA greatly enhances the char formation of PET, with the high-temperature char residual of the composite increasing by 55.4% compared to pure PET, and the actual value of char formation exceeds the theoretical value. Moreover, the addition of CNSs-BA flame retardant triggers high-temperature crosslinking in PET after melting, leading to a substantial improvement in the density, continuity and thermal stability of the char layer formed during the combustion of the CNSs-BA/PET composite.-
Key words:
- carbon nanospheres /
- PET /
- composites /
- flame retardancy /
- melt-drop resistance
-
表 1 LOI和UL 94垂直燃烧测试结果
Table 1. LOI and UL 94 vertical burning test results
Samples Flame retardant content/wt.% LOI /% UL 94 Vertical combustion test results t1 /s t2 /s t3 /s Ignite cotton? Rate PET — 21.0 Burn out — — Yes NR CNSs/PET 0.5 23.2 2.6 2.5 0 Yes V-2 1.0 25.0 2.4 2.4 0 Yes V-2 2.0 26.2 2.4 2.8 0 Yes V-2 3.0 24.6 3.1 2.2 0 Yes V-2 CNSs-BA/PET 0.5 24.0 1.5 2.3 0 Yes V-2 1.0 26.9 1.2 2.1 0 Yes V-2 2.0 28.1 0.5 2.2 0 No V-0 3.0 27.5 0.6 1.9 0 No V-0 Notes: t1-Afterglow time after the first application of flame, t2-Afterglow time after the second application of flame, t3-Afterglow time. 表 2 锥形量热仪测试数据
Table 2. Data of cone calorimeter test
Samples FR content/wt.% TTI/s Time to pk-HRR/s pk-HRR/(kW·m−2) THR/(MJ·m−2) PET 0 47 104 810.45 150.27 CNSs/PET 0.5 44 34 528.96 151.64 1 34 34 503.44 148.03 2 40 41 470.72 146.06 3 30 29 501.49 143.19 CNSs-BA/PET 0.5 35 39 485.54 146.54 1 31 55 469.98 156.04 2 34 39 433.90 146.54 3 30 39 466.05 156.69 Notes: TTI- Time to ignition, pk-HRR- Peak heat release rate, THR- Total heat release. 表 3 CNSs、CNSs-BA以及PET、CNSs/PET、CNSs-BA/PET在氮气气氛下的TG-DTG数据
Table 3. TG-DTG data of CNSs, CNSs-BA, PET, CNSs/PET and CNSs-BA/PET under nitrogen atmosphere
Samples Tonset /℃ Tmax /℃ CR500℃ /% ∆CR500℃/%c exp.a/cal.b CNSs >800℃ - 96.88/- - CNSs-BA 476.4 - 94.92/- - PET 379.1 419.1 10.09/- - CNSs/PET 380.1 421.4 13.97/11.52 2.45 CNSs-BA/PET 382.0 420.1 15.68/11.79 3.89 Note: a CR500℃, exp. is the experimental value of char residue; b CR500℃, cal. is the calculated value of char residue; c ∆CR500℃ = CR500℃, exp. - CR500℃, cal. 表 4 PET、CNSs/PET和CNSs-BA/PET在空气气氛下的TG-DTG数据
Table 4. TG-DTG data of PET, CNSs/PET and CNSs-BA/PET under air atmosphere
Samples Tonset/℃ Tmax-1/℃ Tmax-2/℃ PET 397.3 433.4 585.1 CNSs/PET 359.1 438.4 567.5 CNSs-BA/PET 391.0 439.7 563.3 Notes: Tonset - initial weightlessness temperature, Tmax-1- Maximum weightlessness temperature in the first stage, Tmax-2- Maximum weightlessness temperature of the second stage. 表 5 PET、CNSs/PET和CNSs-BA/PET裂解产物
Table 5. Pyrolysis products of PET, CNSs/PET and CNSs-BA/PET
Pyrolysis products which found only in PET Tetrahydropyran, 2,2-dimethylpropanal, 4,8,12-trimethyl-tridecanoic acid methyl ester, 2,2-dimethoxybutane, 2-methyl-1,5-hexadien-3-yne, 1,6-heptadiyne, p-xylene, decane, methyl benzoate, dodecylethyl ketone, 1-(3-methylphenyl), benzyl, (2-methyl-1-methylenepropylidene), 4-methylphenyl-1-pentyn-3 -ol phenol, dimethyl 1,3-benzenedicarboxylate, vinylmethyl terephthalate, diphenylacetylene, biphenyl-4-ylacetophenone, 1-(5,5-dimethyl-1,3-dioxocyclohexan-2-ylidene)-2-(N-ethylbenzothiazol-2-ylidene)-ethanes, phthalic acid, 4-formylphenyl ester, o-tertiaryl tricyclic [8.2.2.2(4,7)]hexadeca- 2,4,6,8,10,12,13,15-octene, 4-(diethylaminomethyl)-2,5-dimethylphenol Pyrolysis products which found only in CNSs/PET Phenol, 1,2-dihydro-indene, 1-(4-methylphenyl)-ethanone, stilbene, 1 H-cyclopropyl[l]phenanthrene, dihydro-p-terphenyl, 1-naphthol, fluorene-9-methanol, 2-ethyl-1,1'-biphenyl, 1,1-diphenylethene, 4-(2-benzoyl-5-phenyl-3-thienyl)-1,2-dihydrophenanthrene 2-phenylnaphthalenyl benzoate, 1,1-dihydro-2-phenylnaphthalenyl benzoate, 3-chlorobenzylnononyl, 1-(2,5-dimethylphenyl)ethanone, 1-(2,5-dimethylphenyl)ethanone, 1-(2,5-dimethylphenyl)ethanone, 1-(2,5-dimethylphenethyl) ethanone, 3-chlorobenzylnonyl, 1-(2,5-dimethylphenyl) acetone dimethyl-1 H-indene, diethylmalonic acid, 3-chlorobenzylnonyl ester, 1-(2,5-dimethylphenyl) ethanone Pyrolysis products which found only in CNSs-BA/PET 1,5-Hexadiyne, dimethylamine, nitrous oxide, 1,1'-(1,4-phenylene)bis-acetophenone, 2-methylindene, azobenzene, benzene, (1-methyl-2-cyclopropen-1-yl)-2-methylindene, stilbene, ethylketone, 1-(3,4-dimethylphenyl), ethylketone, 1-(4-methylphenyl), 1-ethenyl-4-methylbenzene, dibenzofuran, 2-naphthol, 4-hydroxy-1,2,3,4-tetrahydrophenanthrene, 9,10-dihydrophenanthrene, benzopropiophenone, fluorene, 4-vinylbiphenyl, 1,2,3,4-tetrahydrofil, 9,10-dihydrofil, fluorene, 4-vinylbiphenyl, 1,4-vinylbiphenyl. hydroxy-1,2,3,4-tetrahydrophenanthrene, 9,10-dihydrophenanthrene, phenylacetone, fluorene, 4-vinylbiphenyl, 1,3,5-cycloheptatriene, 2-phenylnaphthalene, 1-acrylbenzene, 2-methylnaphthalene, 4-(2-benzoyl-5-phenyl-3-thienyl)-methylbenzoic acid, 1,3-dimethyl-1 H-indene, tricyclohexen-8-ol, hexaethylcyclohexane, 9- phenyl-9-fluorenol, ethylene oxide, methoxyphenyltricyclohexadecen-5-ylmethanol, 4-benzylbiphenyl, tritylbenzene, 9-phenylanthracene, 3-(1-phenylethoxy)-3 H-isobenzofuran-1-one, 4-phenyl-3,4-dihydroisoquinoline, oxetane,-2-phenyl,-3-phenylethynyl, tetraphenyl, 1-[4-(2-phenylethenyl)phenyl]-ethanone, acenaphthene, 1,2 ,3,5-tetraisopropyl-cyclohexane, 6,9-dimethoxy-phenazine-1-carboxylic acid, [1,1'-biphenyl]-4-yl-phenylmethanone, 1,1':4',1''-3'-methyltriphenylene Pyrolysis products which found both in PET and CNSs/PET Acetophenone, benzoic acid, biphenyl, 2-methyl-1,1'-biphenyl, 1,1'-(1,4-phenylene)bisacetophenone, p-terphenyl Pyrolysis products which found both in PET and CNSs-BA/PET Styrene, acetophenone, benzoic acid, biphenyl, 2-ethyl-1,1'-biphenyl, benzophenone, 9 H-fluoren-9-one, p-terphenyl Pyrolysis products which found both in CNSs/PET and CNSs-BA/PET Benzene, biphenyl, acetophenone, naphthalene, toluene, phenanthrene, indene, 6,6-diphenylfulvene, p-terphenyl, methylstyrene, biphenylacetophenone, 4-ethylbiphenyl, diphenylmethane Pyrolysis products found in PET, CNS/PET and CNSs-BA/PET Acetophenone, benzoic acid, biphenyl, p-terphenyl -
[1] 中国化学纤维工业协会. 中国化纤行业发展规划研究(2021~2025) [M]. 北京: 中国纺织出版社, 2021.China Chemical Fiber Industry Association. Study on the Development Plan of China's Chemical Fiber Industry (2021~2025) [M]. Beijing: China Textile Press, 2021(in Chinese). [2] 刘天明, 赵东, 沈育才, 等. 热塑性聚酯弹性体复合材料的制备与阻燃性能[J]. 复合材料学报, 2024, 41(3): 1249-1258.LIU T M, ZHAO D, SHEN Y C, et al. Preparation and flame retardancy of thermoplastic polyester elastomer composites[J]. Acta Materiae Compositae Sinica, 2024, 41(3): 1249-1258(in Chinese). [3] NI Y P, WU W S, CHEN L, et al. How hydrogen bond interactions affect the flame retardancy and anti-dripping performances of PET[J]. Macromolecular Materials and Engineering, 2019, 305(1): 1900661. [4] SALMEIA K A, GOONEIE A, SIMONETTI P, et al. Comprehensive study on flame retardant polyesters from phosphorus additives[J]. Polymer Degradation and Stability, 2018, 155: 22-34. doi: 10.1016/j.polymdegradstab.2018.07.006 [5] WANG C, WU L, DAI Y, et al. Application of self-templated PHMA sub-microtubes in enhancing flame-retardance and anti-dripping of PET[J]. Polymer Degradation and Stability, 2018, 154: 239-247. doi: 10.1016/j.polymdegradstab.2018.06.005 [6] LI Z, FU T, GUO D M, et al. Trinity flame retardant with benzimidazole structure towards unsaturated polyester possessing high thermal stability, fire-safety and smoke suppression with in-depth insight into the smoke suppression mechanism[J]. Polymer, 2023, 275: 125928. doi: 10.1016/j.polymer.2023.125928 [7] ZHAO H B, WANG Y Z Design and synthesis of PET-based copolyesters with flame-retardant and antidripping performance[J]. Macromolecular Rapid Communications, 2017, 38(23): 1700451. [8] CHEN L, ZHAO H, NI Y, et al. 3D printable robust shape memory PET copolyesters with fire safety via π-stacking and synergistic crosslinking[J]. Journal of Materials Chemistry A, 2019, 7: 17037-17045. doi: 10.1039/C9TA04187G [9] WU J N, CHEN L, FU T, et al. New application for aromatic Schiff base: High efficient flame-retardant and anti-dripping action for polyesters[J]. Chemical Engineering Journal, 2018, 336: 622-632. doi: 10.1016/j.cej.2017.12.047 [10] LIU B, LIU Q, PAN Y, et al. An impact-resistant and flame-retardant CNTs/STF/Kevlar composite with conductive property for safe wearable design[J]. Composites Part A: Applied Science and Manufacturing, 2023, 168: 107489. doi: 10.1016/j.compositesa.2023.107489 [11] GUO Z, WANG Z, FANG Z. Fabrication of 9, 10-dihydro-9-oxa-10-phosphaphenanTHRene-10-oxide-decorated fullerene to improve the anti-oxidative and flame-retardant properties of polypropylene[J]. Composites Part B: Engineering, 2019, 183: 107672. [12] LIANG Q, PAN Y, LU T, et al. Study on the thermal stability and flame retardancy of nanocarbon black based layer-by-layer self-assembled coating modified flexible polyurethane foam[J]. Polymer-Plastics Technology and Materials, 2022, 5(8): 61. [13] YUAN B, SUN Y, CHEN X, et al. Poorly-/well-dispersed graphene: Abnormal influence on flammability and fire behavior of intumescent flame retardant[J]. Composites Part A: Applied Science and Manufacturing, 2018, 109: 345-354. doi: 10.1016/j.compositesa.2018.03.022 [14] 季亚明, 杨雅茹, 姚勇波等. 碳纳米球基氮-磷-硫复合阻燃剂的合成及其对环氧树脂的阻燃性能[J]. 材料研究学报, 2021, 35(12): 918-924.JI Y M, YANG Y R, YAO Y B, et al. Synthesis of carbon nanosphere-based nitrogen-phosphorus-sulfur composite flame retardants and their flame retardant properties on epoxy resin[J]. Journal of Materials Research, 2021, 35(12): 918-924 (in Chinese). [15] WANG X, KALALI E N, WAN J T, et al. Carbon-family materials for flame retardant polymeric materials[J]. Progress in Polymer Science, 2017, 69: 22-46. doi: 10.1016/j.progpolymsci.2017.02.001 [16] LEE G W, KIM J, YOON J, et al. Structural characterization of carboxylated multi-walled carbon nanotubes[J]. Thin Solid Films, 2008, 516(17): 5781-5784. doi: 10.1016/j.tsf.2007.10.071 [17] HUI Q C, YAN X L I,. TIAN W Z Combustion characteristics and flame retardant mechanism of warm-mixed flame-retardant asphalt based on cone calorimeter[J]. World of mining-surface & underground, 2022, 1: 74. [18] JIANG J, CHEN H, WANG Z, et al. Nitrogen-doped hierarchical porous carbon microsphere THRough KOH activation for supercapacitors[J]. Journal of Cold and Interface Science, 2015, 452: 54-61. doi: 10.1016/j.jcis.2015.04.012 [19] ZHI Y, WANG X, QIAN L, et al. Adsorption charring flame retardant effect of phosphaphenanTHRene derivate intercalated micro-expanded graphite composite system in rigid polyurethane foams[J]. Polymer Degradation and Stability, 2023, 216: 110493. doi: 10.1016/j.polymdegradstab.2023.110493 [20] TANG W, QIAN L, PROLONGO S G, et al. Small core of piperazine/silane aggregation initiate efficient charring flame retardant effect in polypropylene composites[J]. Polymer Degradation and Stability, 2023, 208: 110265. doi: 10.1016/j.polymdegradstab.2023.110265 [21] ZHAO H B, CHEN L, YANG J C, et al. A novel flame-retardant-free copolyester: cross-linking towards self extinguishing and non-dripping[J]. Journal of Materials Chemistry, 2012, 22(37): 19849-19857. doi: 10.1039/c2jm34376b [22] COONEY J D, DAY M, WILES D M Thermal degradation of poly(ethylene terephthalate): A kinetic analysis of thermogravimetric data[J]. Journal of Applied Polymer Science, 1983, 28(9): 2887-2902. [23] DU Y, JIANG X, LV G, et al. TG-DSC and FTIR study on pyrolysis of irradiation cross-linked polyethylene[J]. Journal of Material Cycles and Waste Management, 2017, 19(4): 1400-1404. doi: 10.1007/s10163-016-0530-z [24] GHYSELS S, RATHNAYAKE D, MAZIARKA P, et al. Biochar stability scores from analytical pyrolysis (Py-GC-MS)[J]. Journal of analytical & applied pyrolysis, 2022, 161: 105412. [25] LIU H Y, XIN F, DU J Y, et al. The behavior and mechanism of tris-(3-dopo-propyl)-triazine trione flame retardant polyethylene terephthalate[J]. Journal of Applied Polymer Science, 2023, 140(29): 54069. doi: 10.1002/app.54069 -

 点击查看大图
点击查看大图
计量
- 文章访问数: 75
- HTML全文浏览量: 87
- 被引次数: 0




 下载:
下载:










