Degradation of bisphenol F by activated of peroxymonosulfate using sludge biochar loaded with cobalt iron bimetallic catalyst
-
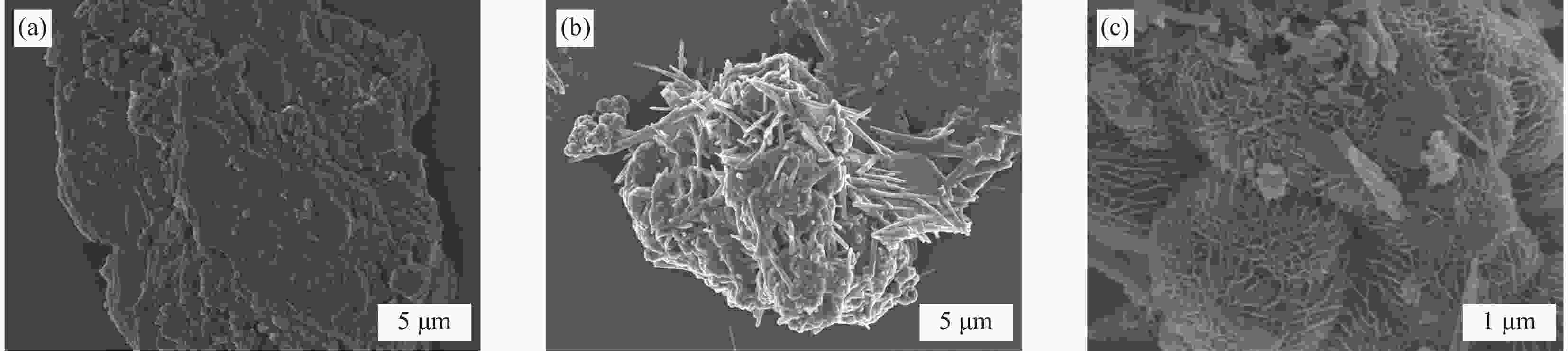
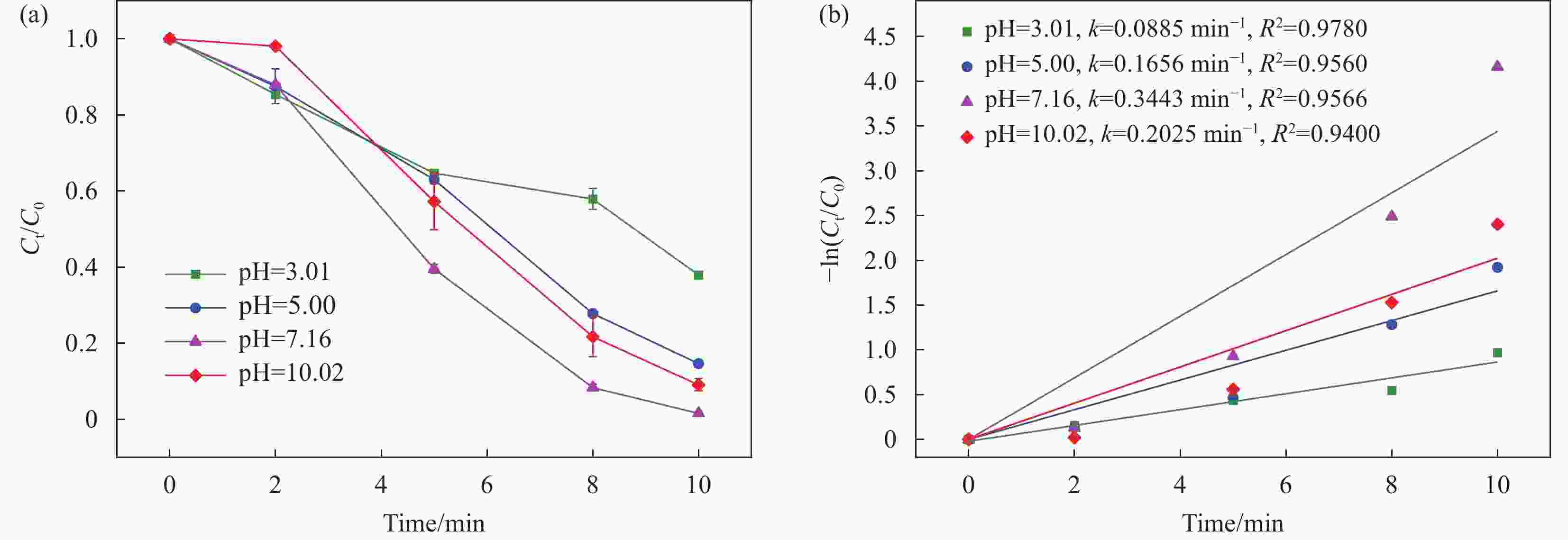
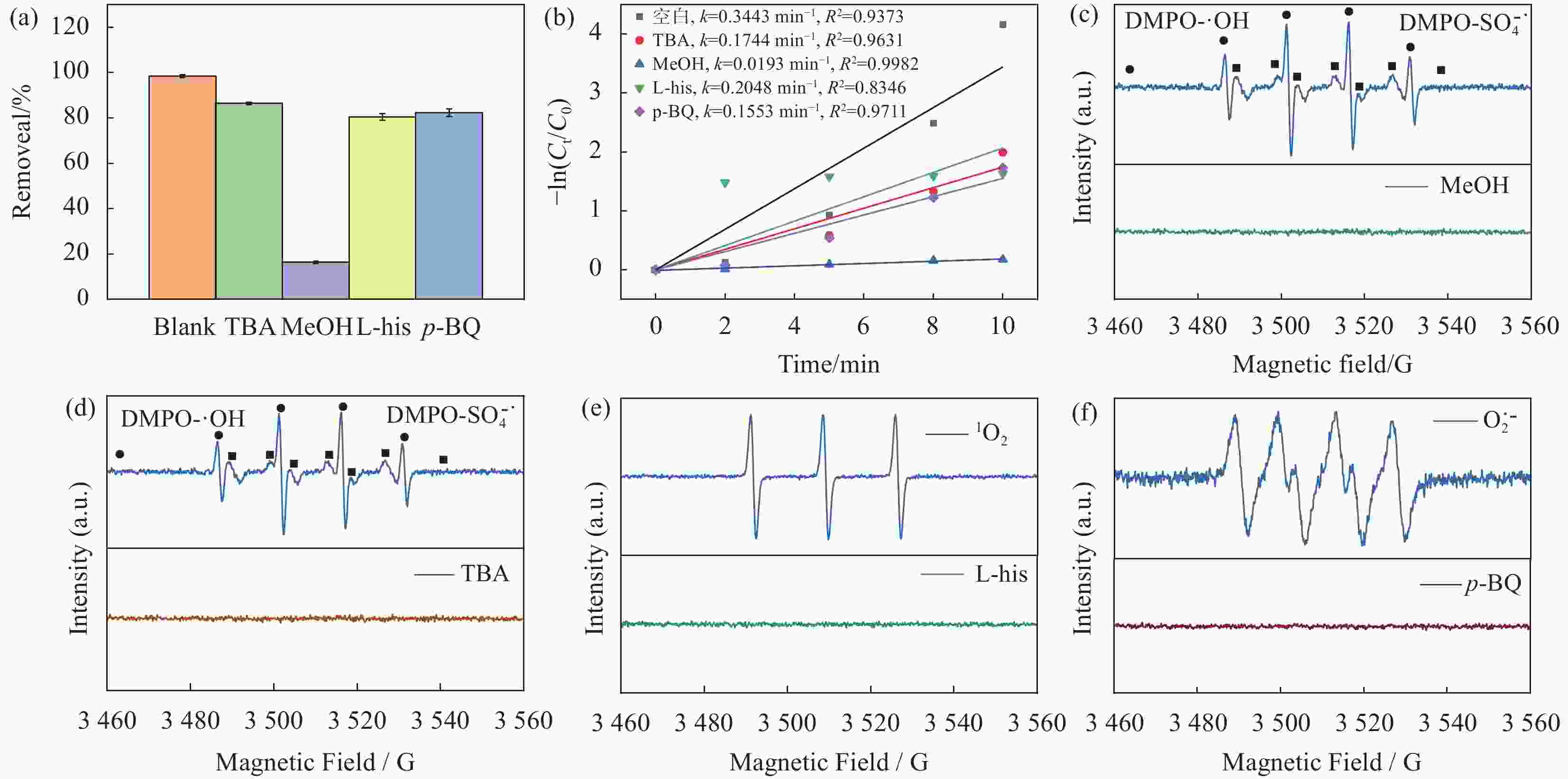
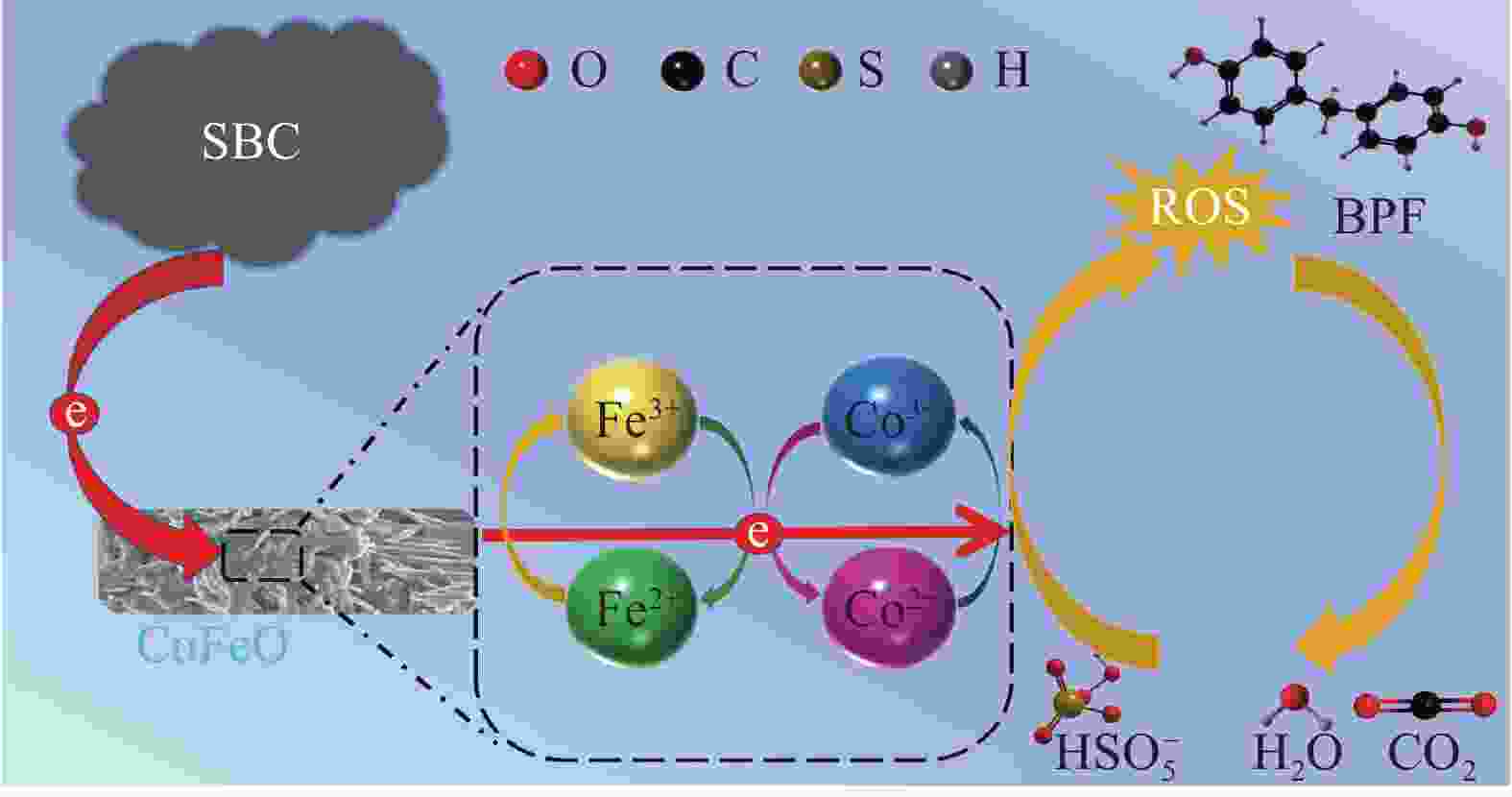
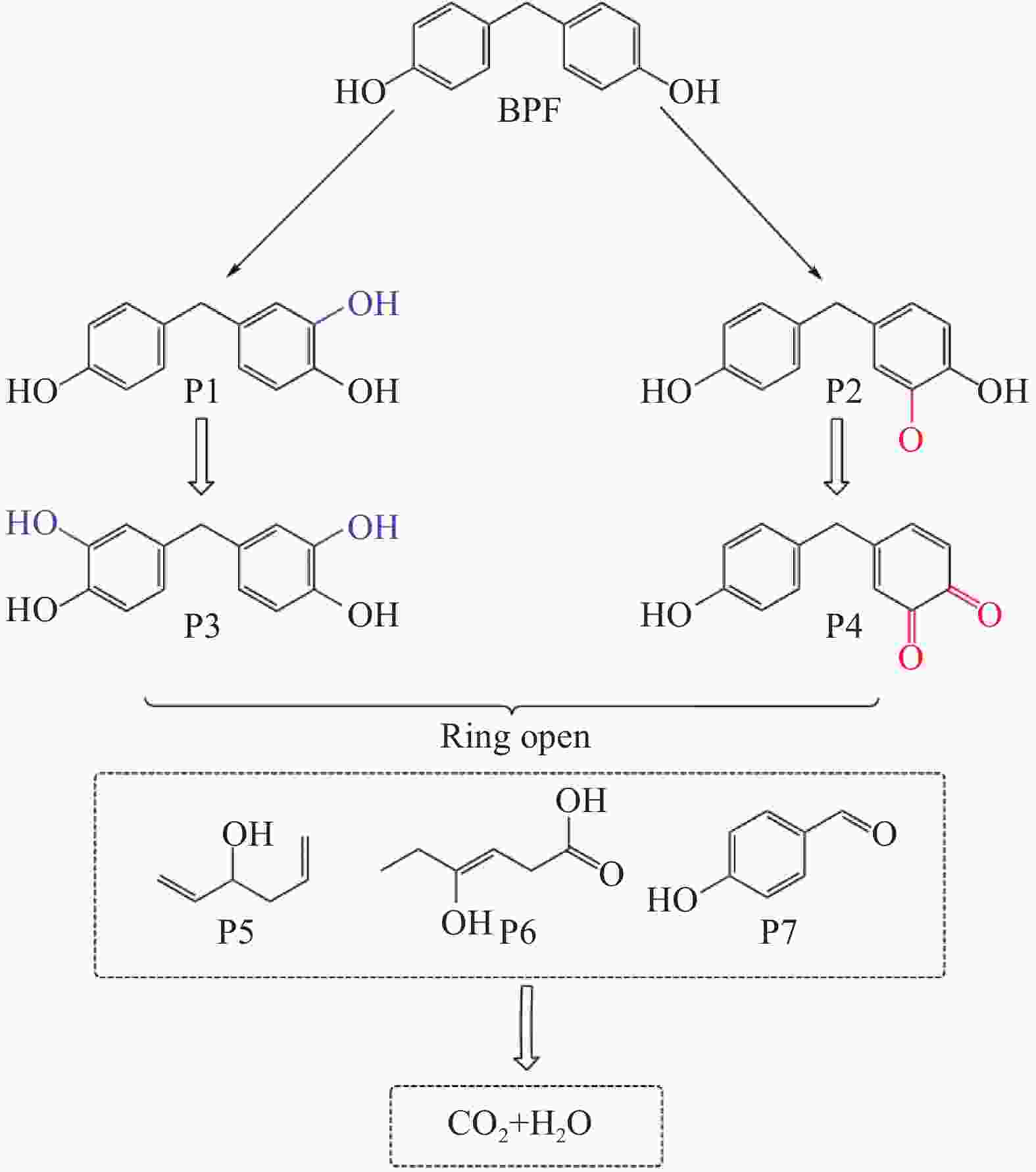
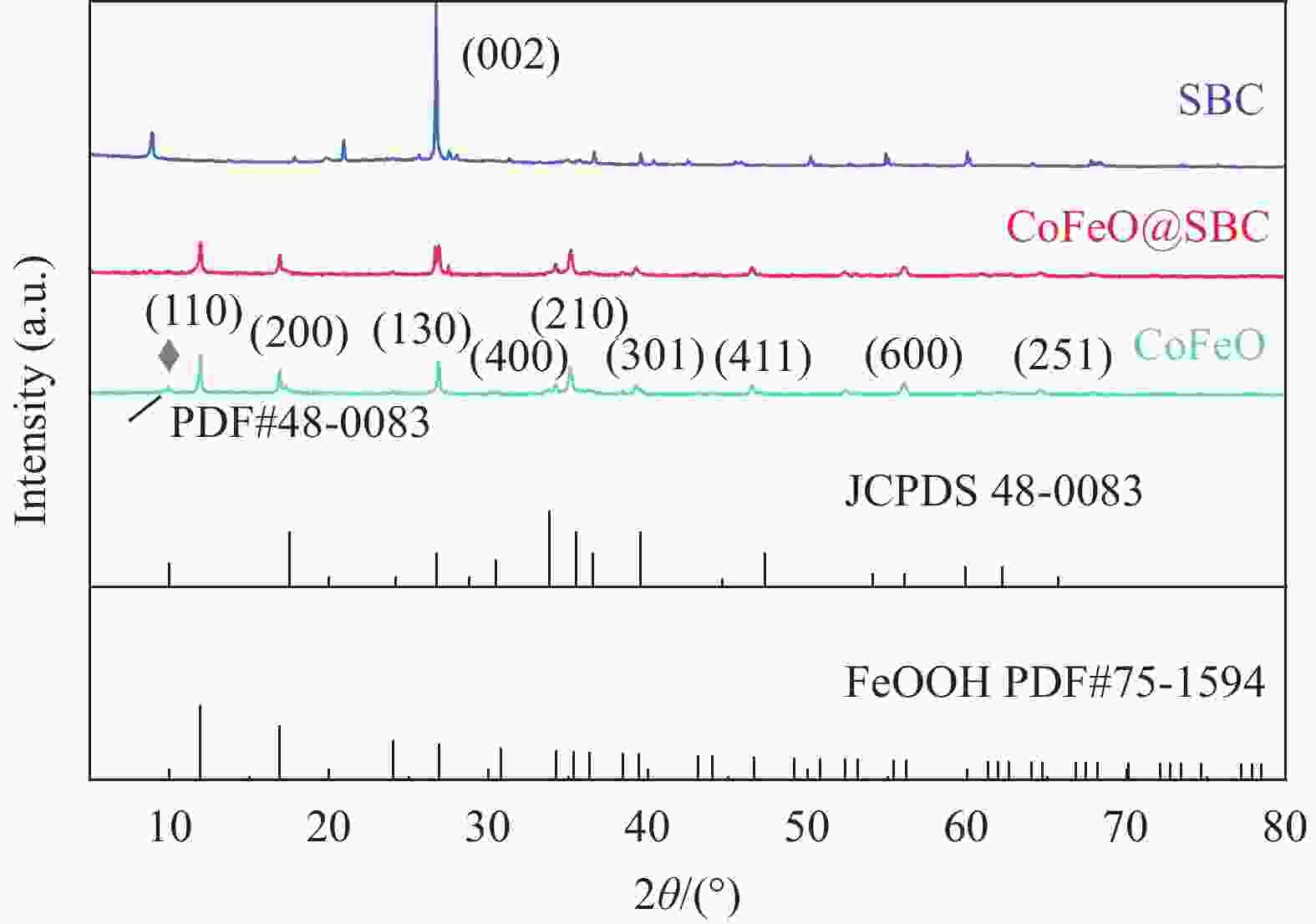
摘要: 近年来,污水处理厂的大规模建设导致污泥产量逐年增加,污泥的处理面临严峻挑战,双酚F (BPF)被广泛应用于工业中化学添加剂,在地表水,土壤和污泥中被频繁检出。本文利用市政污泥负载钴铁双金属制备了钴铁双金属@生物炭复合材料(CoFeO@SBC),通过活化过一硫酸盐(PMS)降解BPF来探究其催化性能。采用扫描电子显微镜(SEM)、比表面积测定(BET)、红外光谱(IR)、X射线衍射(XRD)和X射线光电子能谱(XPS)等表征分析所制备材料的理化性质;并考察了材料投加量、PMS投加量、初始pH和无机阴离子对CoFeO@SBC/PMS体系降解BPF效果的影响。结果表明,CoFeO与SBC复合后孔隙结构显著优化,比表面积增加了6.0倍,且具备更丰富的氧空位和还原性—OH官能团,产生了更多的Fe(Ⅱ)和Co(Ⅱ)。因此,CoFeO@SBC具有优异的催化活性,投加量为0.04 g/L时可以在10 min内几乎完全降解BPF(5 mg/L),降解速率与CoFeO相比提高了62%;Cl−和$\mathrm{NO}_3 ^{-} $对体系降解效果影响较小,而$\mathrm{HCO}_3 ^{-} $具有显著的抑制作用;通过EPR分析表明CoFeO@SBC/PMS体系存在羟基(·OH)和硫酸根($\mathrm{SO}_4 ^{\cdot-} $)自由基以及单线态氧(1O2)和超氧(${\mathrm{O}}_2^{ \cdot-} $)自由基,同时自由基淬灭实验证明,$\mathrm{SO}_4 ^{\cdot-} $是体系降解BPF的关键活性氧物种;最后通过液相色谱-质谱联用(LC-MS)对BPF的降解产物进行分析,揭示BPF在体系中的主要降解途径和机制。Abstract: In recent years, the large-scale construction of wastewater treatment plants has led to an increase in the production of sludge year after year, and the treatment of sludge is facing a serious challenge. Bisphenol F (BPF), which is widely used as a chemical additive in the industry, has been frequently detected in surface water, soil and sludge. By using municipal sludge loaded with cobalt-iron bimetal, cobalt-iron bimetallic@biochar composites (CoFeO@SBC) were prepared, and the catalytic performance was assessed by activating peroxymonosulfate (PMS) to degrade BPF. Scanning electron microscopy (SEM), specific surface area determination (BET), infrared spectroscopy (IR), X-ray diffraction (XRD), and X-ray photoelectron spectroscopy (XPS) were used to characterise and analyse the physicochemical properties of the prepared materials. The effects of the dosage of the materials, the dosage of the PMS, the initial pH, and the inorganic anions on the degradation of the BPF by the CoFeO@SBC/PMS system were investigated. The results showed that the pore structure of CoFeO was significantly improved after compounding with SBC, and the specific surface area was increased by 6.0 times. Moreover, CoFeO@SBC composites showed richer oxygen vacancies and —OH functional groups, which led to higher production of Fe(II) and Co(II). Therefore, CoFeO@SBC composites had excellent catalytic activity, which could almost completely degrade BPF (5 mg/L) within 10 min at the dosage of 0.04 g/L, and the degradation rate was 62% higher than that of CoFeO; Cl− and NO3− showed less influence on the degradation effect of the system, while HCO3− had a significant inhibitory effect; EPR analysis shows that there are hydroxyl (·OH) and sulfate ($\mathrm{SO}_4 ^{\cdot-} $) free radicals as well as singlet oxygen (1O2) and superoxide ($\mathrm{O}_2 ^{\cdot-} $) free radicals in the CoFeO@SBC/PMS system. At the same time, the free radical quenching experiment, it is proved that $\mathrm{SO}_4 ^{\cdot-} $ is the key reactive oxygen species for the degradation of BPF in the system. Finally, the degradation products of BPF were identified by liquid chromatography-mass spectrometry (LC-MS), which could elucidate the primary degradation pathways and mechanisms in CoFeO@SBC/PMS system.
-
Key words:
- bisphenol F /
- degradation /
- cobalt-iron bimetal /
- sludge biochar /
- peroxymonosulfate /
- radicals
-
图 4 (a) CoFeO和CoFeO@SBC的IR表征;(b) XPS全谱图; (c) C 1 s;(d) O 1 s;(e) Fe 2 p和(f) Co 2 p的XPS高分辨图
Figure 4. (a) Characterization of CoFeO and CoFeO@SBC by IR; (b) The XPS survey spectra of CoFeO and CoFeO@SBC; XPS high-resolution spectra of C ls spectra (c); 0 ls spectra(d); Fe 2 p spectra (e); Co 2 p spectra (f)
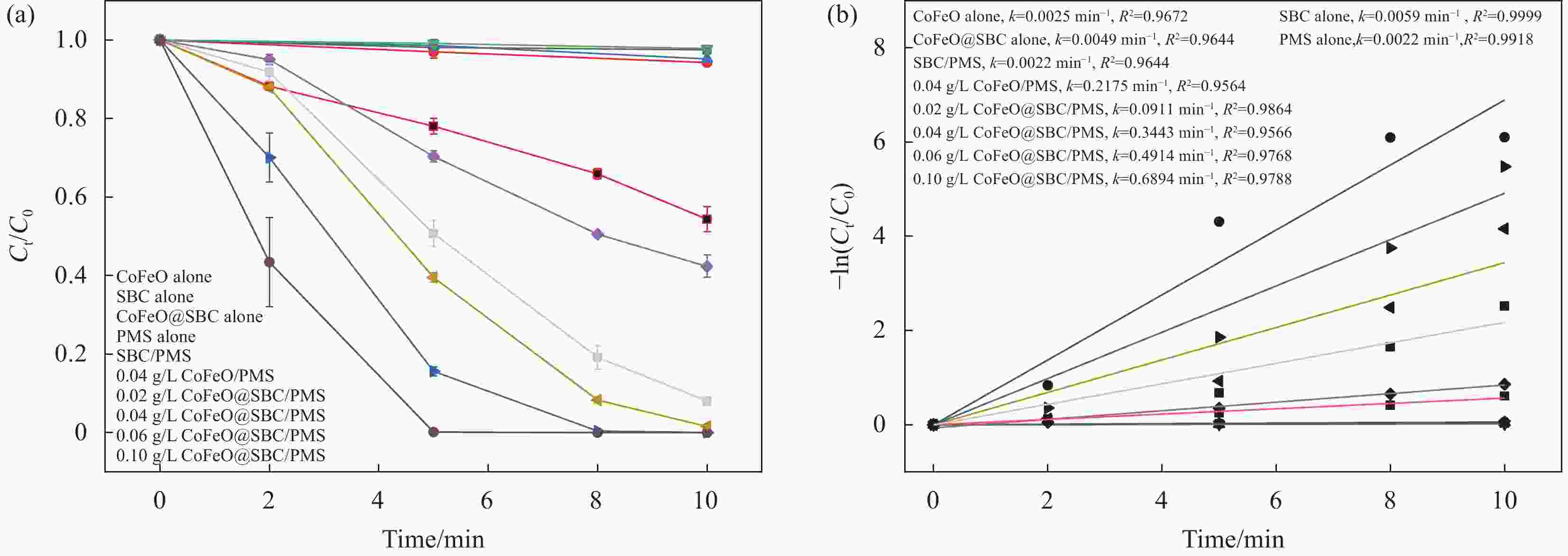
图 5 (a) 不同体系及CoFeO@SBC投加量对双酚F (BPF)去除效果的影响; (b) 不同体系及材料投加量的速率常数图
Figure 5. (a) Effect of different systems and CoFeO@SBC dosage on the removal of bisphenol F (BPF); (b)Rate constant diagram for different systems and material dosages
Ct/C0:The concentration of BPF after t min / The concentration of BPF before the reaction
表 1 不同材料的表面结构特征
Table 1. The surface structure characterization of different material
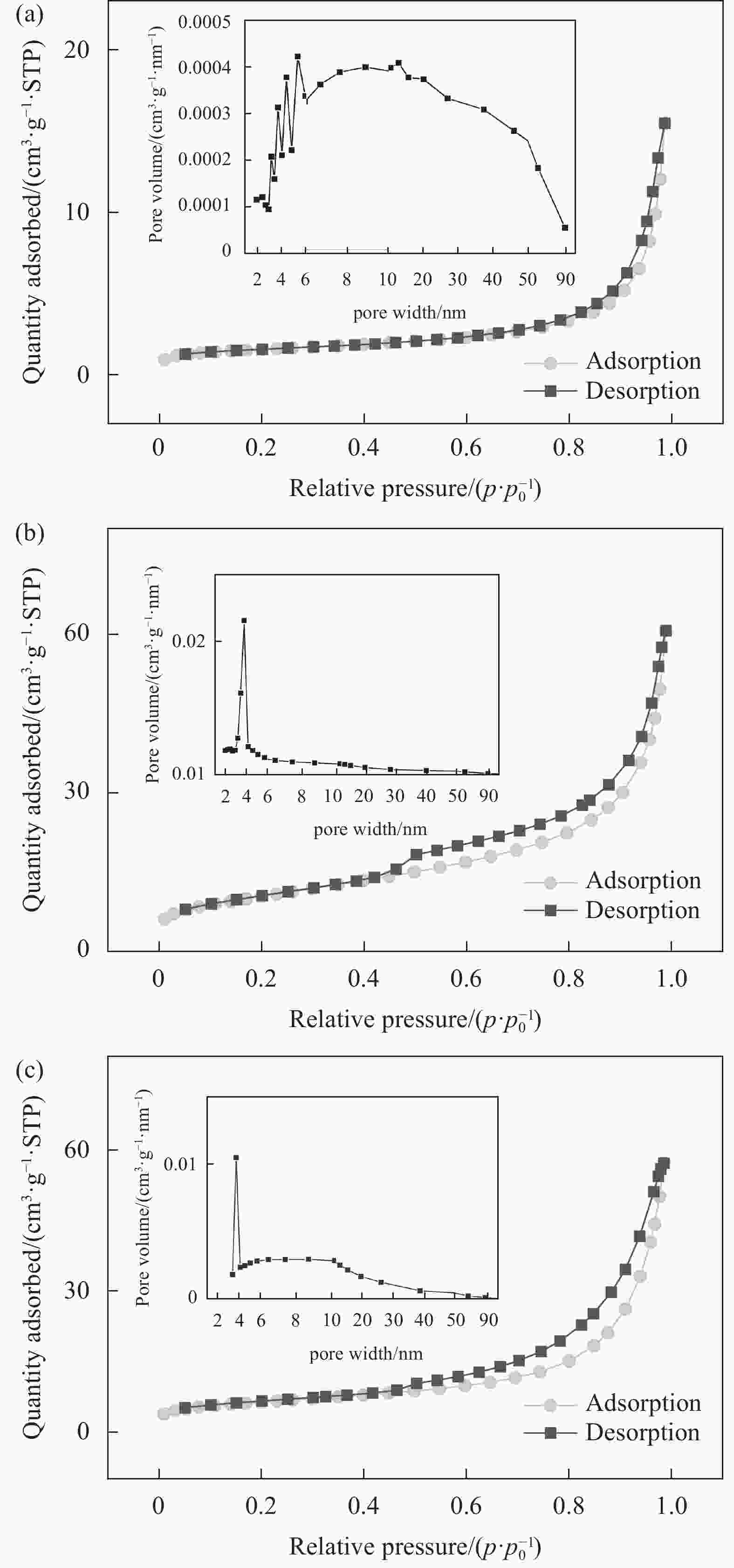
Sample Surface area/(m²·g−1) Average pore diameter/nm Pore volume/(cm³·g−1) SBC 22.0203 15.88640 0.088944 CoFeO 5.2699 17.45430 0.023892 CoFeO@SBC 37.1189 9.48554 0.093656 表 2 CoFeO和CoFeO@SBC的XPS分析数据
Table 2. XPS analysis results of CoFeO and CoFeO@SBC
Sample Fe2+/Fe3+ Co2+/Co3+ C—O O2 CoFeO 1.3 0.7 22.59 % 32.90% CoFeO@SBC 1.5 0.98 30.99 % 36.31% -
[1] 王倩倩, 王永花, 汪贝贝, 等. 双酚F和双酚S联合暴露下的斑马鱼富集及神经毒性[J]. 中国环境科学, 2020, 40(2): 865-873. doi: 10.3969/j.issn.1000-6923.2020.02.048WANG Qianqian, WANG Yonghua, WANG Beibei, et al. Accumulation and neurotoxicity of bisphenol F and bisphenol S in zebrafish under combined exposure[J]. China Environmental Science, 2020, 40(2): 865-873(in Chinese). doi: 10.3969/j.issn.1000-6923.2020.02.048 [2] GOMEZ-LASERNA O, LANZAFAME P, PAPANIKOLAOU G, et al. Analytical assessment to develop innovative nanostructured BPA-free epoxy-silica resins as multifunctional stone conservation materials[J]. Science of the Total Environment, 2018, 645: 817-826. doi: 10.1016/j.scitotenv.2018.07.188 [3] MORALES M, DE La FUENTE M, MARTIN-FOLGAR R. BPA and its analogues (BPS and BPF) modify the expression of genes involved in the endocrine pathway and apoptosis and a multi drug resistance gene of the aquatic midge Chironomus riparius (Diptera)[J]. Environmental Pollution, 2020, 265: 1148. [4] ZENG D, LI P R, HU J W, et al. Fulvic acid enhanced peroxymonosulfate activation over Co-Fe binary metals for efficient degradation of emerging bisphenols[J]. Environmental Research, 2023, 231: 116041. doi: 10.1016/j.envres.2023.116041 [5] HUANG Z, ZHAO J L, YANG Y Y, et al. Occurrence, mass loads and risks of bisphenol analogues in the Pearl River Delta region, South China: Urban rainfall runoff as a potential source for receiving rivers[J]. Environmental Pollution, 2020, 263: 114361. doi: 10.1016/j.envpol.2020.114361 [6] ČESEN M, LENARCIC K, MISLEJ V, et al. The occurrence and source identification of bisphenol compounds in wastewaters[J]. Science of the Total Environment, 2018, 616-617: 744-752. doi: 10.1016/j.scitotenv.2017.10.252 [7] STAOLES C, VAN DER Hoeven , CLARK K, et al. Distributions of concentrations of bisphenol A in North American and European surface waters and sediments determined from 19 years of monitoring data[J]. Chemosphere, 2018, 201: 448-458. [8] PANG L, YANG H Q, LV L N, et al. Occurrence and estrogenic potency of bisphenol analogs in sewage sludge from wastewater treatment plants in central China[J]. Archives of Environmental Contamination and Toxicology, 2019, 77(3): 461-470. doi: 10.1007/s00244-019-00663-4 [9] YE Q Y, WU J Y, WU P X, et al. Enhancing peroxymonosulfate activation of Fe-Al layered double hydroxide by dissolved organic matter: Performance and mechanism[J]. Water Research, 2020, 185: 116246. doi: 10.1016/j.watres.2020.116246 [10] LUO X W, YOU Y J, ZHONG M J, et al. Green synthesis of manganese–cobalt–tungsten composite oxides for degradation of doxycycline via efficient activation of peroxymonosulfate[J]. Journal of Hazardous Materials, 2022, 426: 127803. doi: 10.1016/j.jhazmat.2021.127803 [11] GIANNAKIS S, LIN K A, GHANBARI F. A review of the recent advances on the treatment of industrial wastewaters by Sulfate Radical-based Advanced Oxidation Processes (SR-AOPs)[J]. Chemical Engineering Journal, 2021, 406: 127083. doi: 10.1016/j.cej.2020.127083 [12] 王俊辉, 陆彩妹, 李泽华, 等. 磁性氮掺杂杉木屑生物炭活化过一硫酸盐降解左氧氟沙星[J]. 复合材料学报, 2023, 40(11): 6383-6394. doi: 10.13801/j.cnki.fhclxb.20230222.011 Chemical Engineering Journal, 2015, 276: 193-204.WANG Junhui, LU Caimei, LI Zehua, et al. Preparation of magnetic nitrogen-doped fir sawdust biochar to activate peroxymonosulfate for Levofloxacin degradation[J]. Acta Materiae Compositae Sinica, 2023, 40(11): 6383-6394. doi: 10.13801/j.cnki.fhclxb.20230222.011.(in Chinese). [13] FERNANDES A, MAKOS P, KHAN J A et al. Pilot scale degradation study of 16 selected volatile organic compounds by hydroxyl and sulfate radical based advanced oxidation processes[J]. Journal of Cleaner Production, 2019, 208: 54-64. doi: 10.1016/j.jclepro.2018.10.081 [14] NFODZO P, CHOI H. Sulfate radicals destroy pharmaceuticals and personal care products[J]. Environmental Engineering Science, 2011, 28(8): 605-609. doi: 10.1089/ees.2011.0045 [15] SHEN M X, HUANG Z J, LUO X W, et al. Activation of persulfate for tetracycline degradation using the catalyst regenerated from Fenton sludge containing heavy metal: Synergistic effect of Cu for catalysis[J]. Chemical Engineering Journal, 2020, 396: 125238. doi: 10.1016/j.cej.2020.125238 [16] FANG Y, LIU Q W, SONG Y, et al. Highly efficient in-situ purification of Fe(II)-rich high-arsenic groundwater under anoxic conditions: Promotion mechanisms of PMS on oxidation and adsorption[J]. Chemical Engineering Journal, 2023, 453: 139915. doi: 10.1016/j.cej.2022.139915 [17] SUN C H, JIA Z R, XU S, et al. Synergistic regulation of dielectric-magnetic dual-loss and triple heterointerface polarization via magnetic MXene for high-performance electromagnetic wave absorption[J]. Journal of Materials Science & Technology, 2022, 113: 128-137. [18] YUAN C, DAI Y D, CHEN Y C. Analysis of electric field efficacy and remediation performance of triclosan contaminated soil by Co–Fe/al oxidation electrodes coupled with peroxymonosulfate (PMS) in an ECGO system with diversified electrode configurations[J]. Chemosphere, 2022, 307: 135841. doi: 10.1016/j.chemosphere.2022.135841 [19] 张吉琛, 刘婷然, 董志强, 等. 污泥生物炭的催化机制及应用研究进展[J]. 环境化学, 2023, 42(6): 1-14.ZHANG Jichen, LIU Tingran, DONG Zhiqiang, et al. Mechanism and application for removal of contaminants by sludge derived biochar catalyst: A review[J]. Environmental Chemistry, 2023, 42(6): 1-14(in Chinese). [20] GAO Y, CONG S B, YU H Y, et al. Investigation on microwave absorbing properties of 3D C@ZnCo2O4 as a highly active heterogenous catalyst and the degradation of ciprofloxacin by activated persulfate process[J]. Separation and Purification Technology, 2021, 262: 118330. doi: 10.1016/j.seppur.2021.118330 [21] 吕鹏飞, 陈悫, 王佳程, 等. 层状双氢氧化物-生物炭复合材料在废水处理中的应用[J]. 复合材料学报, 2023, 42: 1-12.LV Pengfei, CHEN Que, WANG Jiacheng, et al. Application of layered double hydroxide-biochar composite in wastewater treatment[J]. Acta Materiae Compositae Sinica, 2023, 42: 1-12(in Chinese). [22] JI, J Q, YUAN X Z, ZHAO Y L, et al. Mechanistic insights of removing pollutant in adsorption and advanced oxidation processes by sludge biochar[J]. Journal of Hazardous Materials, 2022, 430: 128375. doi: 10.1016/j.jhazmat.2022.128375 [23] 贺德春, 胡嘉梧, 梁紫薇, 等. 同位素内标-高效液相色谱-串联质谱法测定畜禽粪便中6种雌激素[J]. 生态环境学报, 2021, 30(2): 383-390.HE Dechun, HU Jiawu, LIANG Ziwei, et al. Determination of six estrogens in livestock manures by isotope internal standard-high performance liquid chromatography coupled with tandem mass spectrometry[J]. Ecology and Environmental Science, 2021, 30(2): 383-390(in Chinese). [24] LI Y, MA S L, XU S J, et al. Novel magnetic biochar as an activator for peroxymonosulfate to degrade bisphenol A: Emphasizing the synergistic effect between graphitized structure and CoFe2O4[J]. Chemical Engineering Journal, 2020, 387: 12094. [25] WU J Y, XU Y J, WU P X, et al. Effects of different dissolved organic matter on peroxymonosulfate activation over Co-Fe binary metal: Experiments and density functional theory[J]. Chemical Engineering Journal, 2022, 450: 137770. doi: 10.1016/j.cej.2022.137770 [26] GUAN Q, CHENG J, WANG B, et al. Needle-like Co3O4 anchored on the graphene with enhanced electrochemical performance for aqueous supercapacitors[J]. ACS Appl Mater Interfaces, 2014, 6(10): 7626-7632. doi: 10.1021/am5009369 [27] WU J Y, WANG Y H, WU Z X, et al. Adsorption properties and mechanism of sepiolite modified by anionic and cationic surfactants on oxytetracycline from aqueous solutions[J]. Science of the Total Environment, 2020, 708: 134409. doi: 10.1016/j.scitotenv.2019.134409 [28] JU L T, WU P X, YANG Q L, et al. Synthesis of ZnAlTi-LDO supported C60@AgCl nanoparticles and their photocatalytic activity for photo-degradation of Bisphenol A[J]. Applied Catalysis B:Environmental, 2018, 224: 159-174. doi: 10.1016/j.apcatb.2017.10.056 [29] CHEN L Y, WU P X, CHEN M Q, et al. Preparation and characterization of the eco-friendly chitosan/vermiculite biocomposite with excellent removal capacity for cadmium and lead[J]. Applied Clay Science, 2018, 159: 74-82. doi: 10.1016/j.clay.2017.12.050 [30] LI Z, SUN Y Q, YANG Y, et al. Biochar-supported nanoscale zero-valent iron as an efficient catalyst for organic degradation in groundwater[J]. Journal of Hazardous Materials, 2020, 383: 121240. doi: 10.1016/j.jhazmat.2019.121240 [31] HAN S, XIAO P F. Catalytic degradation of tetracycline using peroxymonosulfate activated by cobalt and iron co-loaded pomelo peel biochar nanocomposite: Characterization, performance and reaction mechanism[J]. Separation and Purification Technology, 2022, 287: 120533. doi: 10.1016/j.seppur.2022.120533 [32] JIA Q Q, ZHANG X J, LI Y Q, et al. Reductive dehalogenation in groundwater by Si-Fe(II) co-precipitates enhanced by internal electric field and low vacancy concentrations[J]. Water Research, 2023, 228: 119386. doi: 10.1016/j.watres.2022.119386 [33] BAO C S, WANG H, WANG C Y, et al. Cooperation of oxygen vacancy and FeIII/FeII sites in H2-reduced Fe-MIL-101 for enhanced Fenton-like degradation of organic pollutants[J]. Journal of Hazardous Materials, 2023, 441: 129922. doi: 10.1016/j.jhazmat.2022.129922 [34] LIU J W, DU Y F, SUN W Y, et al. A granular adsorbent-supported Fe/Ni nanoparticles activating persulfate system for simultaneous adsorption and degradation of ciprofloxacin[J]. Chinese Journal of Chemical Engineering, 2020, 28(4): 1077-1084. doi: 10.1016/j.cjche.2019.12.019 [35] XIE H, XU W. Enhanced activation of persulfate by Meso-CoFe2O4/SiO2 with ultrasonic treatment for degradation of chlorpyrifos[J]. ACS Omega, 2019, 4(17): 17177-17185. doi: 10.1021/acsomega.9b01626 [36] HUANG Y M, LI G, LI M Z, et al. Kelp-derived N-doped biochar activated peroxymonosulfate for ofloxacin degradation[J]. Science of the Total Environment, 2021, 754: 141999. doi: 10.1016/j.scitotenv.2020.141999 [37] HE J, XIAO Y, TANG J C, et al. Persulfate activation with sawdust biochar in aqueous solution by enhanced electron donor-transfer effect[J]. Science of the Total Environment, 2019, 690: 768-777. doi: 10.1016/j.scitotenv.2019.07.043 [38] WANG X L, DING Y Z, DIONYSIOU D D, et al. Efficient activation of peroxymonosulfate by copper sulfide for diethyl phthalate degradation: Performance, radical generation and mechanism[J]. Science of the Total Environment, 2020, 749: 142387. doi: 10.1016/j.scitotenv.2020.142387 [39] WANG S Z, LIU Y, WANG J L. Iron and sulfur co-doped graphite carbon nitride (FeOy/S-g-C3N4) for activating peroxymonosulfate to enhance sulfamethoxazole degradation[J]. Chemical Engineering Journal, 2020, 382: 122836. doi: 10.1016/j.cej.2019.122836 [40] HU Y, CHEN D Z, ZHANG R, et al. Singlet oxygen-dominated activation of peroxymonosulfate by passion fruit shell derived biochar for catalytic degradation of tetracycline through a non-radical oxidation pathway[J]. Journal of Hazardous Materials, 2021, 419: 126495. doi: 10.1016/j.jhazmat.2021.126495 [41] LOU X Y, WW L X, GUO Y G, et al. Peroxymonosulfate activation by phosphate anion for organics degradation in water[J]. Chemosphere, 2014, 117: 582-585. doi: 10.1016/j.chemosphere.2014.09.046 [42] LI X N, WANG Z H, ZHANG B, et al. FexCo3−xO4 nanocages derived from nanoscale metal–organic frameworks for removal of bisphenol A by activation of peroxymonosulfate[J]. Applied Catalysis B:Environmental, 2016, 181: 788-799. doi: 10.1016/j.apcatb.2015.08.050 [43] WANG Z W, LI Q, SU R D, et al. Enhanced degradation of bisphenol F in a porphyrin-MOF based visible-light system under high salinity conditions[J]. Chemical Engineering Journal, 2022, 428: 132106. doi: 10.1016/j.cej.2021.132106 -

 点击查看大图
点击查看大图
计量
- 文章访问数: 52
- HTML全文浏览量: 26
- 被引次数: 0




 下载:
下载: