Adsorption behaviour and mechanism of tetracycline by sorghum straw-loaded HKUST-1
-
摘要:
四环素(TC)是一种难降解广谱抗生素,广泛存在于畜牧业排放的污废中,排放后会对水体生态环境造成严重的污染,通过吸附法可有效去除。本研究以高粱秸秆(SS)为基材,通过原位生长法在SS表面负载MOFs (HKUST-1)制备SS@HKUST-1复合材料,用于对TC的吸附去除,探究复合材料对TC的吸附行为及吸附机制。研究表明:当pH=7、温度T=25℃、HKUST-1的负载量为31wt%时,吸附容量达到95 mg/g。吸附过程符合准二级动力学模型,吸附等温线符合Freundlich模型,表明复合材料对TC吸附属于多分子层化学吸附。因此,SS@HKUST-1对水中TC的去除具有良好的应用前景。
Abstract:Tetracycline (TC) is a refractory broad-spectrum antibiotic, that is widely present in the waste discharged from animal husbandry, which will cause serious pollution to the ecological environment of water bodies after discharge, and it can be effectively removed by adsorption. In this work, sorghum straw (SS) was used as the substrate to prepare SS@HKUST-1 composites by in-situ growth of MOFs (HKUST-1) on the surface of SS for the adsorption and removal of TC, investigating the adsorption behaviour and adsorption mechanism of composites on TC. The results show that the adsorption capacity reaches 95 mg/g when pH=7, temperature T=25℃ and the load capacity of HKUST-1 is 31wt%. The adsorption process conforms to the pseudo-second-order kinetic model, and the adsorption isotherm conforms to the Freundlich model, which suggests the existence of multimolecular layer chemisorption between TC and adsorbent. Therefore, SS@HKUST-1 has a good application prospect for tetracycline removal in water.
-
Keywords:
- sorghum straw /
- HKUST-1 /
- in-situ growth /
- tetracycline /
- adsorption
-
近年来随着工业的快速发展,水污染问题受到越来越多的关注。其中的一部分污染是由抗生素引起的,如阿莫西林、青霉素、四环素等[1-5]。四环素(Tetracycline,TC)作为一种普遍使用的抗生素,因其独特的抗菌作用而被广泛应用于人类疾病的治疗。然而TC长期存在于水环境中难以自然降解,对生态环境和人类健康有很大危害[6-10]。因此,开发一种有效的解决方案来去除水环境中难降解的抗生素是极其重要的。半导体光催化技术因其具有环境友好、无污染、低能耗等优点引起了广泛的研究,成为当今处理有机污染物最有前景的方法之一[11-13]。
在各种光催化剂中,BiOX(X=Cl, Br, I)因其特殊的层状结构、适宜的禁带宽度和较高的稳定性,被广泛用于光降解有机污染物和光催化分解水[14-15]。其中BiOI具有较窄的禁带宽度(1.77~1.92 eV)和较宽的可见光响应范围,但由于光生电子空穴对的高复合率,其光催化性能并不理想。将BiOI与其他半导体材料相结合被认为是增强光催化性能最有前途的策略,2个或2个以上半导体相结合可以形成半导体/半导体异质结,通过提高光生电子空穴对的分离速率从而提高光催化活性。
Bi2O3的带隙介于2.1~2.8 eV之间,由于其具有较强的可见光区响应、无毒、电化学稳定性高、热稳定性好和低成本等特性[16-18],是一种很有前途的可见光光催化候选材料,可与其他半导体材料形成异质结结构。如CdS/BiOCl/Bi2O3[19]、GO/AgI/Bi2O3[20]、α-Bi2O3/g-C3N4[21]、Bi2O3/ZnS[22]等。
Wei等[23]采用一锅沉淀法在多孔Bi2O3纳米棒上成功沉积了BiOI纳米片。结果表明:与原始Bi2O3和BiOI相比,50% Bi2O3/BiOI复合材料具有更高的光生电子空穴对分离效率和更大的比表面积,在可见光照射下,其光催化还原Cr(VI)的活性显著增强。此外,50% Bi2O3/BiOI复合材料还具有优异的光化学稳定性和可回收性。Li等[24]采用化学刻蚀法制备了BiOI/Bi2O3异质结,在降解苯酚和4-氯苯酚(4-CP)方面表现出良好的光催化活性。其光催化性能的提高是由于BiOI/Bi2O3异质结的形成促进了电子空穴对的有效分离,并提出了光生电荷转移的过程。
目前所制备的同类光催化剂大多数用于去除水环境中的重金属离子和有机染料等,对降解抗生素类药物的研究较少。本文采用简单的溶剂热法制备了Bi2O3/BiOI复合光催化材料,在模拟太阳光照射下通过降解四环素研究了其光催化性能,探究了BiOI与Bi2O3不同摩尔比、反应温度、反应时间、pH等条件对光催化性能的影响。并通过活性物种捕捉实验提出了Bi2O3/BiOI复合光催化材料降解四环素可能的机制。
1. 实验材料和方法
1.1 原材料
五水硝酸铋(上海麦克林生化有限公司,AR)、碘化钾(天津市大茂化学试剂厂,AR)、乙二醇(天津市北辰方正试剂厂,AR)、四环素(上海麦克林生化有限公司,AR)。
1.2 实验仪器
EL104型电子天平(梅特勒-托利多有限公司)、HC-3018型高速离心机(安徽中科中佳科学仪器有限公司)、TGL-5A台式离心机(常州润华电器有限公司)、KSW-4D-I2型马弗炉(北京中兴伟业仪器有限公司)、HJ-1型磁力加热搅拌器(红杉实验设备厂)、101-1A型电热鼓风干燥箱(北京中兴伟业仪器有限公司)、721型可见分光光度计(上海仪电分析仪器有限公司)、KQ5200E型超声波清洗器(昆山市超声仪器有限公司)、250 W金卤灯(上海亚明)。
1.3 实验内容
1.3.1 BiOI光催化材料的制备
称取1 mmol五水硝酸铋置于15 mL乙二醇中,超声处理15 min以获得均匀悬浮液。在不断搅拌下向其中逐滴加入10 mL含1 mmol碘化钾的水溶液,继续搅拌2 h。将产物离心,用水和无水乙醇洗涤数次,在80℃下干燥12 h得到红色的BiOI。
1.3.2 Bi2O3光催化材料的制备
称取一定量的五水硝酸铋,在600℃的马弗炉里煅烧4 h,冷却至室温后,将产物研磨成粉末状,得到淡黄色的Bi2O3。
1.3.3 Bi2O3/BiOI复合光催化材料的制备
将1 mmol五水硝酸铋置于15 mL乙二醇中,超声处理15 min以获得均匀悬浮液。在不断搅拌下向其中逐滴加入10 mL含1 mmol碘化钾的水溶液,继续搅拌2 h。在此期间,用1 mol/L的H2SO4溶液将混合液的pH调至5。然后向上述溶液中加入0.8 mmol已制备好的Bi2O3,继续搅拌1 h。将得到的混合溶液转移至50 mL聚四氟乙烯内衬的不锈钢高压反应釜中,在180℃下反应20 h。自然冷却至室温后,将产物离心,用水和无水乙醇洗涤数次,在80℃下干燥12 h,得到Bi2O3/BiOI复合光催化材料。
1.3.4 光催化性能测试
使用250 W金卤灯模拟太阳光照射,通过降解四环素来评价所制备样品的光催化性能。取50 mg制得的光催化材料放入装有100 mL 25 mg/L TC溶液的烧杯中,黑暗搅拌30 min达到吸附-脱附平衡。然后将混合液置于光反应器中,光照开始计时,每隔20 min取3 mL样,将样品放入离心机中离心取其上层清液并测定吸光度。计算四环素的残余率:
η=C/C0×100%=A/A0×100% 式中:C和C0分别表示t时刻和初始四环素的质量浓度(mg·L−1);A和A0分别表示t时刻和初始四环素的吸光度。
2. 结果与讨论
2.1 Bi2O3/BiOI复合光催化材料的晶相结构
BiOI、Bi2O3和Bi2O3/BiOI的XRD图谱如图1所示。BiOI曲线在9.658°、29.645°、31.657°、37.392°、45.666°、51.345°、55.15°、66.344°和74.09°处出现的衍射峰分别对应BiOI(JCPDS 10-0445)的(001)、(102)、(110)、(112)、(104)、(114)、(212)、(214)和(302)晶面。Bi2O3在21.722°、25.757°、27.377°、33.241°、35.406°、37.595°、42.353°、46.305°、52.373°和58.563°处出现的衍射峰分别对应Bi2O3(JCPDS 41-1449)的(020)、(002)、(120)、(200)、(031)、(112)、(122)、(041)、(−321)和(−331)晶面。Bi2O3/BiOI同时出现了Bi2O3和BiOI的主要衍射峰,说明本实验成功制备了Bi2O3/BiOI复合光催化材料。
2.2 Bi2O3/BiOI复合光催化材料的微观形貌
通过SEM分析了所制备光催化材料的微观形貌,结果如图2所示。可以看出,所制备的BiOI是由纳米片自组装形成的花状微球;单一Bi2O3呈现出不同尺寸、不规则的块状结构。从图2(c)可以看出,当BiOI与Bi2O3复合后,块状Bi2O3均匀分散在花状微球的BiOI表面。
2.3 Bi2O3/BiOI复合光催化材料的结构
样品的FTIR图谱如图3所示。499 cm−1和760 cm−1处是BiOI的特征吸收峰,1617 cm−1处的吸收峰对应Bi2O3中Bi—O键的弯曲振动,再次表明BiOI和Bi2O3成功复合在一起。
2.4 Bi2O3/BiOI复合光催化材料的光学性能
利用UV-Vis DRS分析了所制备样品的光学吸收性能,如图4所示。可以看出,纯BiOI的吸收边缘位于681 nm处,纯Bi2O3的吸收边缘位于477 nm,Bi2O3/BiOI复合光催化材料的光吸收边缘位于617 nm。与纯BiOI相比,Bi2O3/BiOI复合光催化材料的光吸收边缘有轻微的蓝移,这是由于与Bi2O3耦合造成的,但其光吸收范围仍然很宽。
不同光催化材料的紫外漫反射(αhv)1/2-hv转换图如图5所示。根据Kubelka-Munk公式,纯BiOI、Bi2O3和Bi2O3/BiOI对应的禁带宽度Eg值分别为1.82 eV、2.60 eV和2.01 eV。
2.5 Bi2O3/BiOI复合光催化材料光生电子空穴对的分离
利用荧光强度来分析光生电子空穴对的复合速率,荧光强度越小,则光生电子复合速率越低,图6为不同光催化材料的荧光光谱。可以看出,在520 nm处,Bi2O3/BiOI的荧光强度低于单一BiOI和Bi2O3,表明复合光催化材料的光生电子空穴对复合速率最低,光催化活性最高。
通过电化学阻抗法研究了不同光催化材料的电荷转移效率,结果如图7所示。Bi2O3/BiOI的圆弧半径小于单一BiOI和Bi2O3,说明其电荷转移电阻较低,电导率增强。电化学阻抗图表明Bi2O3/BiOI能增强光生电子空穴对的电荷转移能力,提高其分离效率,这与荧光分析的结果一致。
2.6 Bi2O3/BiOI复合光催化材料的性能
2.6.1 不同制备条件对光催化性能的影响
通过探究反应物的不同摩尔比、反应温度、反应时间及pH对所制备材料的光催化性能的影响。从图8(a)~8(d)可以看出:当Bi2O3与BiOI的摩尔比为0.8∶1时,在pH=5、180℃下反应20 h得到的Bi2O3/BiOI复合光催化材料对四环素的降解效果最佳,在3 h内对四环素的降解率可达75%。
2.6.2 光催化性能
通过在模拟太阳光照射下降解四环素来评价所制备样品的光催化性能,图9(a)为模拟太阳光照射下降解四环素的曲线图。在3 h内,BiOI、Bi2O3、Bi2O3/BiOI对四环素的降解率分别为55%、57%、75%。图9(b)为模拟太阳光照射下降解四环素的动力学曲线,Bi2O3/BiOI的动力学速率常数(0.007 min−1)分别是BiOI(0.004 min−1)、Bi2O3(0.0045 min−1)的1.75倍、1.56倍。因此,所制备出的Bi2O3/BiOI具有较高的光催化活性。
2.7 Bi2O3/BiOI复合光催化材料降解四环素的机制
使用对苯醌(BQ)、乙二胺四乙酸二钠盐(EDTA-2Na)、异丙醇(IPA)作为·O2−、h+、·OH的捕捉剂,实验结果如图10所示。BiOI对四环素的降解率分别为50%、31%、53%、,由此可以得出h+是BiOI降解四环素的主要活性物质。Bi2O3对四环素的降解率分别为55%、52%、35%,由此可以得出·OH是Bi2O3降解四环素的主要活性物质。
当Bi2O3和BiOI复合后形成Z型异质结时,Bi2O3/BiOI光催化材料对四环素的降解率分别为35%、66%、66%,由此可以得出·O2−是光催化材料降解四环素的主要活性物质。
Bi2O3/BiOI异质结的形成可以提高光生电子空穴对的分离速率,从而提高光催化活性。BiOI 、Bi2O3的导带和价带可以通过下式计算:
ECB=X−Ee−0.5Eg Eg=EVB−ECB 式中:X为半导体的电负性,BiOI和Bi2O3的X值分别为5.94 eV 和6.23 eV;Ee为自由电子在氢标上的能量(约为4.50 eV);Eg为对应的带隙能量,由图3(b)可以得出BiOI和Bi2O3的Eg值分别为1.82 eV和2.60 eV。因此,BiOI和Bi2O3的导带(CB)边缘分别位于0.53 eV和0.43 eV,BiOI和Bi2O3的价带(VB)边缘分别位于2.35 eV和3.03 eV。在可见光照射下所制备的Bi2O3/BiOI异质结被激发并生成光生载流子且BiOI比Bi2O3的CB更正。事实上,在Bi2O3/BiOI异质结中,光子能量会激发BiOI CB上的电子到更高的电位位置(−0.68 eV),因此BiOI CB上的光生电子会转移到Bi2O3的CB上。同时,Bi2O3 VB上的空穴将转移到BiOI的VB上。而Bi2O3 CB上的电子不能与O2反应生成·O2−(O2/·O2−=−0.33 eV),·O2−是降解四环素的主要活性物质,这与捕获实验的结果不一致。结合以上结果,提出了一种更可能的光催化机制,如图11所示。BiOI和Bi2O3在可见光照射下都能产生光生电子空穴对,Bi2O3 CB上的光生电子和BiOI VB上的空穴在库仑力的作用下会重新组合。此外,BiOI的CB上的光生电子可以与O2反应生成·O2−,然后·O2−与TC反应,有效地实现了Bi2O3/BiOI异质结的光催化降解过程。综上所述,Bi2O3/BiOI异质结能够有效提高光生电子空穴对的分离效率,从而显著提高光催化性能。
3. 结 论
(1) 以五水硝酸铋为原料,采用溶剂热法制备了Bi2O3/BiOI复合光催化材料,在制备过程中加入Bi2O3可以提高单一BiOI的光催化性能,在3 h内对四环素的降解率为75%,是单一BiOI降解速率的1.75倍。
(2) BiOI、Bi2O3成功复合在一起并形成了异质结结构,Bi2O3/BiOI复合光催化材料通过提高光生电子空穴对的分离速率从而提高光催化活性。
(3) 降解机制研究表明,·O2−在降解四环素中起主要作用,且所制得的复合材料可应用于对四环素的降解,并有望进一步用于对其他抗生素的降解处理以解决实际问题。
-
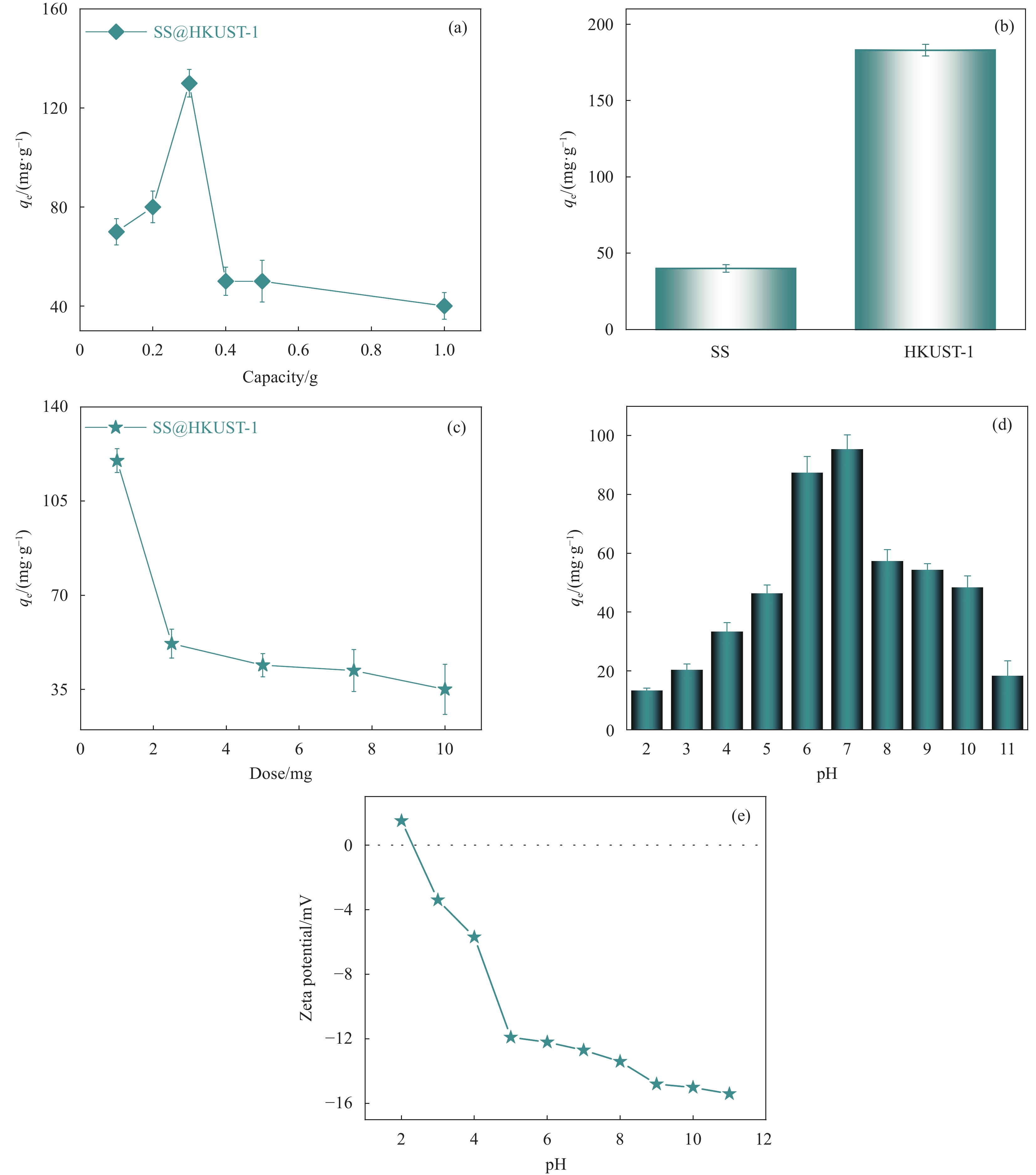
图 6 不同条件下TC吸附效果:(a)不同Cu(NO3)2质量;(b) SS和HKUST-1;(c)吸附剂用量;(d)不同溶液pH值; (e)不同pH值下SS@HKUST-1的Zeta电位
Figure 6. Effects of different condition on TC adsorption: (a) Different masses of Cu(NO3)2; (b) SS and HKUST-1; (c) Adsorbent dosages; (d) Different pH values; (e) Zeta potential of SS@HKUST-1 under different pH values
qe—Equilibrium adsorption capacity
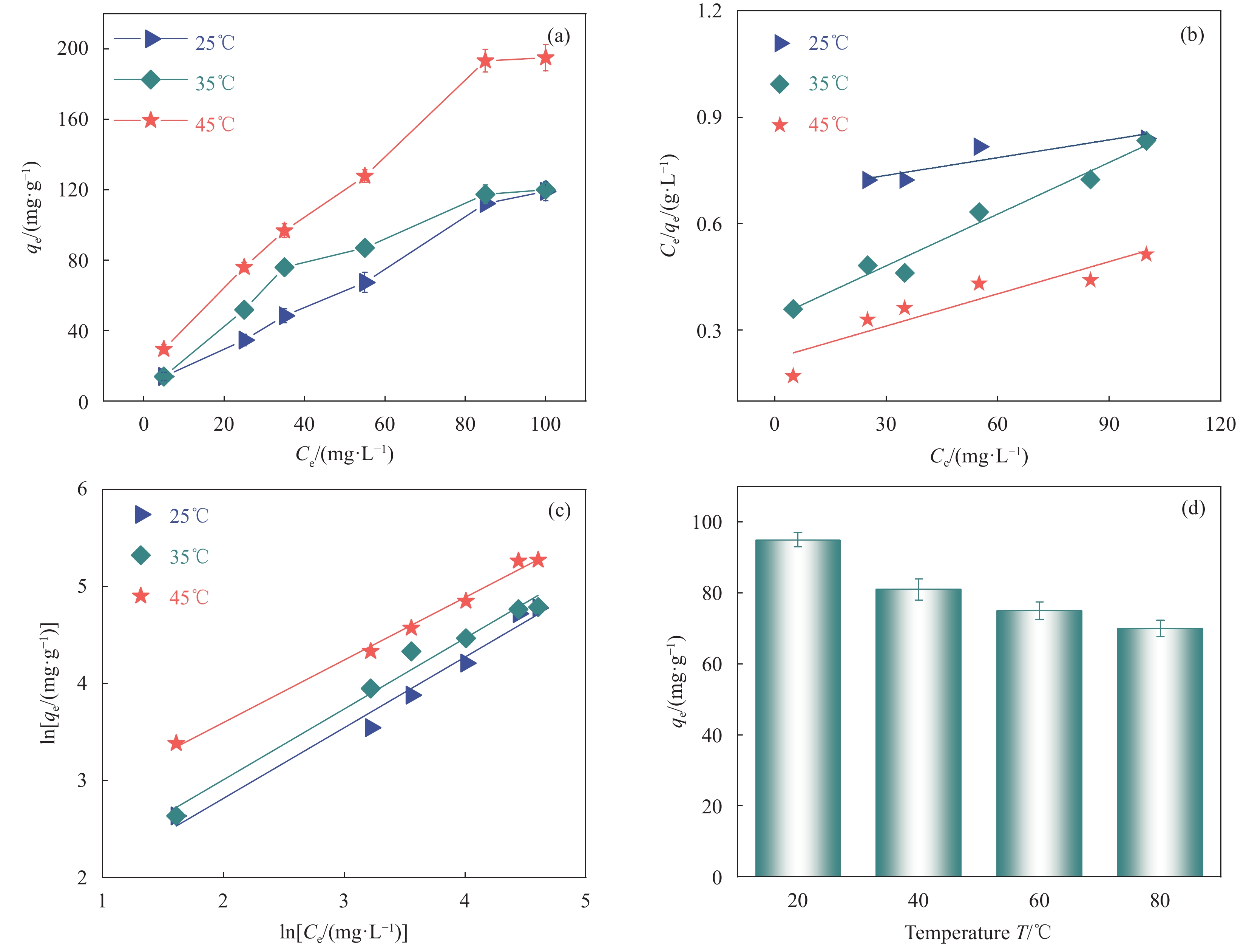
图 8 (a) TC在SS@HKUST-1表面的吸附等温线;(b) Langmuir吸附等温线;(c) Freundlich吸附等温线;(d)不同温度处理SS@HKUST-1对吸附TC的影响
Figure 8. (a) Adsorption isotherms of TC onto the surfaces of SS@HKUST-1; (b) Langmuir adsorption isotherm; (c) Freundlich adsorption isotherm; (d) Effect of different temperatures treatment SS@HKUST-1 on adsorbed TC
Ce—Concentration at adsorption equilibrium; T—Temperature
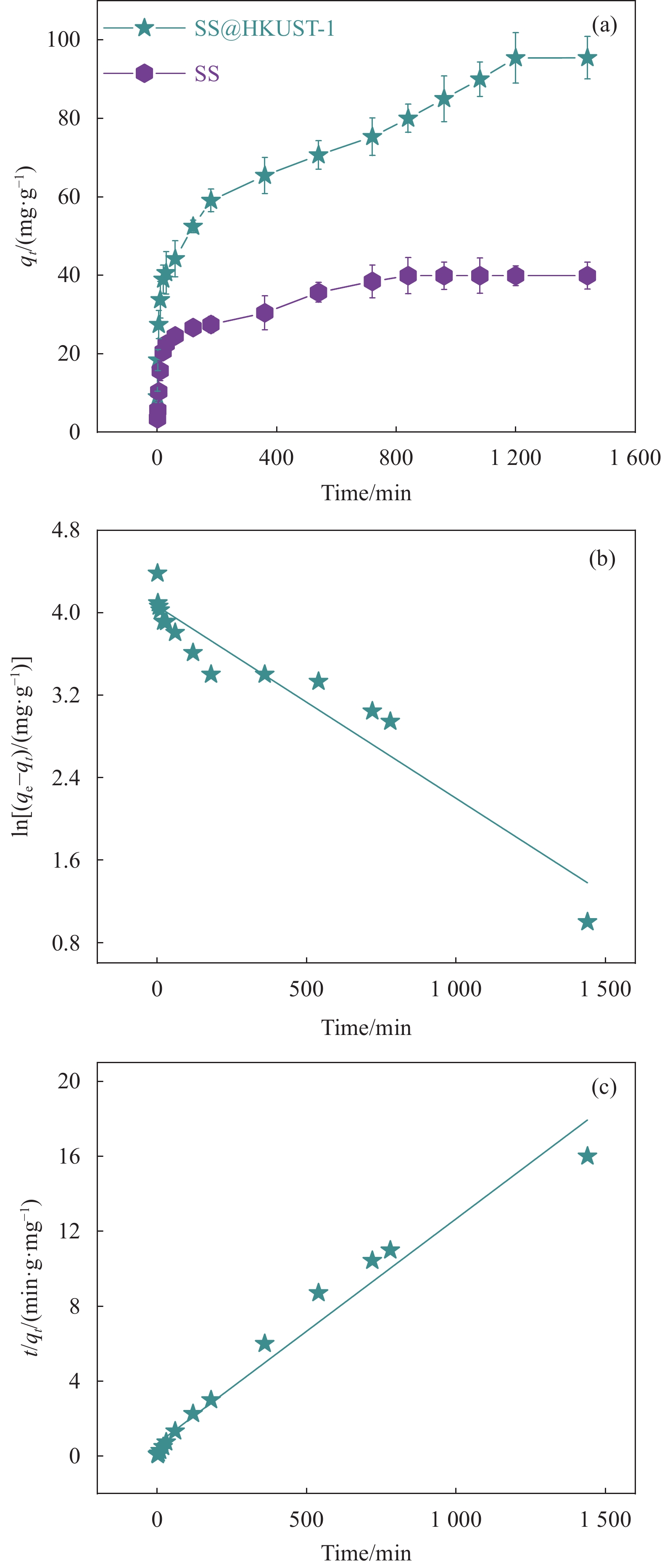
表 1 SS@HKUST-1吸附TC的动力学模型拟合参数
Table 1 Kinetic model fitting parameters for SS@HKUST-1 adsorbed TC
qe,exp/
(mg·g−1)Pseudo-first-order
kineticPseudo-second-order
kinetick1/
min−1qe,cal/
(mg·g−1)R2 k2/
(g·mg−1·min−1)qe,cal/
(mg·g −1)R2 95.44 0.002 65.06 0.947 0.010 1.87 0.994 Notes: qe,exp—Actual adsorption capacity at adsorption equilibrium; qe,cal—Calculated adsorption capacity at adsorption equilibrium; k1—Pseudo-first-order adsorption rate constant; k2—Pseudo-second-order adsorption rate constant; R2—Correlation coefficient of Langmuir and Freundlich models. 表 2 SS@HKUST-1的吸附等温线的拟合参数
Table 2 Fitting parameters of adsorption isotherms to SS@HKUST-1
Temperature/℃ Langmuir model Freundlich model qm/
(mg·g−1)b/
(L·mg−1)R2 kF/
(mg·g−1)1/n R2 25 119.10 0.686 0.901 3.88 0.7291 0.989 35 120.00 0.335 0.987 4.69 0.7302 0.991 45 194.97 0.220 0.926 10.03 0.6453 0.997 Notes: qm—Maximum adsorption capacity; b—1/qmkL, kL—Adsorption coefficient of Langmuir; kF—Adsorption coefficient of Freundlich; 1/n—Empirical parameter varied with the degree of heterogeneity of adsorbing sites. 表 3 不同吸附剂对TC的吸附去除效果对比
Table 3 Comparison of adsorption and removal effects of different adsorbents for TC
Sample qe/(mg·g−1) Ref. SS@HKUST-1 95 This work ZIF-8/CMC 78.75 [36] CS biochar 53.191 [20] WS biochar 66.67 [20] MIL-100(Fe)/PEO 85.02 [37] Fe3O4@RGO@C18 77.56 [38] AG(Mn)-88B-C 53.07 [39] Notes: ZIF-8/CMC—Metal-organic skeleton hybrid foam ZIF-8 (zeolitic imidazolate framework-8)/CMC (carboxymethyl cellulose) with cellulose; CS biochar—Corn straw biochar; WS biochar—Wheat straw biochar; MIL-100(Fe)/PEO—Polyethylene oxide modified MIL-100(Fe); Fe3O4@RGO@C18—Fe3O4 magnetic particles were coated with a layer of RGO (reduced graphene oxide), and C18 was further modified on the surface of Fe3O4@RGO material; AG(Mn)-88B-C—MIL-88B(Fe)/sodium alginate composite aerogel. -
[1] 高艳林, 景红霞, 李龙祥, 等. 溶剂热法制备Bi2O3/BiOI复合光催化材料及对四环素的降解应用[J]. 复合材料学报, 2022, 39(2): 677-684. GAO Yanlin, JING Hongxia, LI Longxiang, et al. Preparation of Bi2O3/BiOI composite photocatalytic materials by solvothermal method and application to tetracycline degradation[J]. Acta Materiae Compositae Sinica, 2022, 39(2): 677-684(in Chinese).
[2] WANG D, JIA F, WANG H, et al. Simultaneously efficient adsorption and photocatalytic degradation of tetracycline by Fe-based MOFs[J]. Journal of Colloid and Interface Science, 2018, 519: 273-284. DOI: 10.1016/j.jcis.2018.02.067
[3] ZHANG X, LIN X, HE Y, et al. Study on adsorption of tetracycline by Cu-immobilized alginate adsorbent from water environment[J]. International Journal of Biological Macromolecules, 2019, 124: 418-428. DOI: 10.1016/j.ijbiomac.2018.11.218
[4] 于晓雯, 索全义. 畜禽粪便中四环素类抗生素的残留及危害[J]. 北方农业学报, 2018, 46(3): 83-88. YU Xiaowen, SUO Quanyi. Residues and hazards of tetracycline antibiotics in livestock manure[J]. Northern Agricultural Journal, 2018, 46(3): 83-88(in Chinese).
[5] ZHU Y, LI B, WANG Y, et al. Preparation of porous Ti/RuO2-IrO2@Pt, Ti/RuO2-TiO2@Pt and Ti/Y2O3-RuO2-TiO2@Pt anodes for efficient electrocatalytic decomposition of tetracycline[J]. Molecules, 2023, 28(5): 2189. DOI: 10.3390/molecules28052189
[6] GOPAL G, ALEX S A, CHANDRASEKARAN N, et al. A review on tetracycline removal from aqueous systems by advanced treatment techniques[J]. RSC Advances, 2020, 10(45): 27081-27095.
[7] YIN F, LIN S, ZHOU X, et al. Fate of antibiotics during membrane separation followed by physical-chemical treatment processes[J]. Science of the Total Environment, 2021, 759: 143520. DOI: 10.1016/j.scitotenv.2020.143520
[8] HU Z, WANG Y, WANG L, et al. Synthesis of S-type heterostructure π-COF for photocatalytic tetracycline degradation[J]. Chemical Engineering Journal, 2023, 479: 147534.
[9] BHATT P, JEON C H, KIM W. Tetracycline bioremediation using the novel Serratia marcescens strain WW1 isolated from a wastewater treatment plant[J]. Chemosphere, 2022, 298: 134344. DOI: 10.1016/j.chemosphere.2022.134344
[10] 张甜, 姜博, 邢奕, 等. 吸附法去除水中抗生素研究进展[J]. 环境工程, 2021, 39(3): 29-39. ZHANG Tian, JIANG Bo, XING Yi, et al. Research progress of antibiotic removal from water by adsorption[J]. Environmental Engineering, 2021, 39(3): 29-39(in Chinese).
[11] CHEN W, ZHAO B, GUO Y, et al. Effect of hydrothermal pretreatment on pyrolyzed sludge biochars for tetracycline adsorption[J]. Journal of Environmental Chemical Engineering, 2021, 9(6): 106557. DOI: 10.1016/j.jece.2021.106557
[12] LUO Q, REN T, LEI Z, et al. Non-toxic chitosan-based hydrogel with strong adsorption and sensitive detection abilities for tetracycline[J]. Chemical Engineering Journal, 2022, 427: 131738. DOI: 10.1016/j.cej.2021.131738
[13] QIAO D, QU X, CHEN X, et al. Rational structural design of graphene oxide/W18O49 nanocomposites realizes highly efficient removal of tetracycline in water[J]. Applied Surface Science, 2023, 619: 156630. DOI: 10.1016/j.apsusc.2023.156630
[14] 陈刚. 功能化金属有机框架对抗生素的吸附研究[D]. 南昌: 华东交通大学, 2021. CHEN Gang. Adsorption study of antibiotics by functionalized metal-organic frameworks[D]. Nanchang: East China Jiaotong University, 2021(in Chinese).
[15] 王艳萍, 房得珍, 路淼, 等. MOFs基材料对金属离子吸附分离的研究进展[J]. 化工新型材料, 2023, 51(7): 68-73. WANG Yanping, FANG Dezhen, LU Miao, et al. Advances in the adsorption and separation of metal ions by MOFs-based materials[J]. New Materials for Chemical Industry, 2023, 51(7): 68-73(in Chinese).
[16] YANG Y, XUE Y, LI J, et al. Efficient removal of organic dyestuff in water contamination over a MOF-derived Co-based adsorbent[J]. Reaction Chemistry & Engineering, 2023, 8(9): 2195-2210.
[17] ZHANG M, LI Y, ZHOU X, et al. Preparation of ZIF-67/C3N4 composite material and adsorption of tetracycline hydrochloride[J]. Environmental Science and Pollution Research, 2023, 30(41): 94112-94125. DOI: 10.1007/s11356-023-28919-6
[18] MA X, XIONG Y, LIU Y, et al. When MOFs meet wood: From opportunities toward applications[J]. Chem, 2022, 8(9): 2342-2361. DOI: 10.1016/j.chempr.2022.06.016
[19] 李钲, 王国栋, 陈日耀, 等. 椰壳炭吸附去除四环素性能及机理研究[J]. 福建师范大学学报(自然科学版), 2023, 39(5): 117-123. LI Zheng, WANG Guodong, CHEN Riyao, et al. Study on the performance and mechanism of tetracycline removal by adsorption on coconut shell carbon[J]. Journal of Fujian Normal University (Natural Science Edition), 2023, 39(5): 117-123(in Chinese).
[20] 王诗尧, 王少华, 孙强, 等. 玉米和小麦秸秆生物炭对四环素吸附效果研究[J]. 环境科学与管理, 2023, 48(8): 84-87. WANG Shiyao, WANG Shaohua, SUN Qiang, et al. Study on the adsorption effect of tetracycline on corn and wheat straw biochar[J]. Environmental Science and Management, 2023, 48(8): 84-87(in Chinese).
[21] SHI Q, WANG W, ZHANG H, et al. Porous biochar derived from walnut shell as an efficient adsorbent for tetracycline removal[J]. Bioresource Technology, 2023, 383: 129213. DOI: 10.1016/j.biortech.2023.129213
[22] ZHUO S N, DAI T C, REN H Y, et al. Simultaneous adsorption of phosphate and tetracycline by calcium modified corn stover biochar: Performance and mechanism[J]. Bioresource Technology, 2022, 359: 127477. DOI: 10.1016/j.biortech.2022.127477
[23] WANG K, HAN T, CHEN X, et al. Insights into behavior and mechanism of tetracycline adsorption on virgin and soil-exposed microplastics[J]. Journal of Hazardous Materials, 2022, 440: 129770. DOI: 10.1016/j.jhazmat.2022.129770
[24] KANGRA B, JATRANA A, MAAN S, et al. Effective adsorption of chlorpyrifos pesticides by HKUST-1 metal-organic framework[J]. Journal of Chemical Sciences, 2022, 134(4): 104. DOI: 10.1007/s12039-022-02099-1
[25] OLIVEIRA H, SCACCHETTI F, BEZERRA F, et al. Comprehensive evaluation of HKUST-1 as an efficient adsorbent for textile dyes[J]. Environmental Science and Pollution Research, 2023, 30(37): 87242-87259. DOI: 10.1007/s11356-023-28455-3
[26] ZHAO L, AZHAR M R, LI X, et al. Adsorption of cerium (III) by HKUST-1 metal-organic framework from aqueous solution[J]. Journal of Colloid and Interface Science, 2019, 542: 421-428. DOI: 10.1016/j.jcis.2019.01.117
[27] BHORIA N, BASINA G, POKHREL J, et al. Functionalization effects on HKUST-1 and HKUST-1/graphene oxide hybrid adsorbents for hydrogen sulfide removal[J]. Journal of Hazardous Materials, 2020, 394: 122565. DOI: 10.1016/j.jhazmat.2020.122565
[28] PAN J, BAI X, LI Y, et al. HKUST-1 derived carbon adsorbents for tetracycline removal with excellent adsorption performance[J]. Environmental Research, 2022, 205: 112425. DOI: 10.1016/j.envres.2021.112425
[29] LUO J, FAN C, XIAO Z, et al. Novel graphene oxide/carboxymethyl chitosan aerogels via vacuum-assisted self-assembly for heavy metal adsorption capacity[J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2019, 578: 123584. DOI: 10.1016/j.colsurfa.2019.123584
[30] JAGÓDKA P, MATUS K, ŁAMACZ A. On the HKUST-1/GO and HKUST-1/rGO composites: The impact of synthesis method on physicochemical properties[J]. Molecules, 2022, 27(20): 7082. DOI: 10.3390/molecules27207082
[31] ZHANG S, HAN X, CAI H, et al. Aramid nanofibers/WS2 nanosheets co-assembled aerogels for efficient and stable Pb (II) adsorption in harsh environments[J]. Chemical Engineering Journal, 2022, 450: 138268. DOI: 10.1016/j.cej.2022.138268
[32] 崔芳溪, 张蓬, 曹胜凯, 等. 不同pH溶液中四环素类抗生素与金属离子配合前后的光化学性质及毒性差异[J]. 环境科学学报, 2024, 44(2): 136-144. CUI Fangxi, ZHANG Peng, CAO Shengkai, et al. Differences in photochemical properties and toxicity of tetracycline antibiotics before and after coordination with metal ions in different pH solutions[J]. Journal of Environmental Science, 2024, 44(2): 136-144(in Chinese).
[33] AL-JABARI M. Kinetic models for adsorption on mineral particles comparison between Langmuir kinetics and mass transfer[J]. Environmental Technology & Innovation, 2016, 6: 27-37.
[34] CHEN X, HOSSAIN M F, DUAN C, et al. Isotherm models for adsorption of heavy metals from water—A review[J]. Chemosphere, 2022, 307: 135545.
[35] 李微, 郭孟雅, 刘宁. ZIF-8/CMC杂化泡沫吸附TC和Cu2+研究[J/OL]. 环境工程, 1-16[2024-12-26]. LI Wei, GUO Mengya, LIU Ning. Adsorption of TC and Cu2+ by ZIF-8/CMC hybrid foam[J/OL]. Environmental Engineering, 1-16[2024-12-26]. (in Chinese).
[36] DHYANI V, KUMAR J, BHASKAR T. Thermal decomposition kinetics of sorghum straw via thermogravimetric analysis[J]. Bioresource Technology, 2017, 245: 1122-1129. DOI: 10.1016/j.biortech.2017.08.189
[37] 李微, 宁雨阳, 刘宁, 等. PEO基MOFs杂化泡沫材料对四环素和Cu2+吸附性能[J]. 环境工程, 2023, 41(7): 76-85. LI Wei, NING Yuyang, LIU Ning, et al. Adsorption properties of PEO-based MOFs hybrid foams for tetracycline and Cu2+[J]. Environmental Engineering, 2023, 41(7): 76-85(in Chinese).
[38] 李胜, 唐祝兴, 王英嘉, 等. 纳米复合材料Fe3O4@RGO@C18对四环素的吸附研究[J]. 辽宁化工, 2023, 52(4): 469-473. DOI: 10.3969/j.issn.1004-0935.2023.04.003 LI Sheng, TANG Zhuxing, WANG Yingjia, et al. Adsorption study of tetracycline on nanocomposite Fe3O4@RGO@C18[J]. Liaoning Chemical Industry, 2023, 52(4): 469-473(in Chinese). DOI: 10.3969/j.issn.1004-0935.2023.04.003
[39] 张晓东, 赵胜昊, 姜舜桐, 等. MIL-88B(Fe)/海藻酸钠气凝胶的制备及其吸附四环素性能研究[J]. 广州化学, 2023, 48(1): 10-18. ZHANG Xiaodong, ZHAO Shenghao, JIANG Shuntong, et al. Preparation of MIL-88B(Fe)/sodium alginate aerogel and its adsorption performance of tetracycline[J]. Guangzhou Chemistry, 2023, 48(1): 10-18(in Chinese).
[40] 缪家鑫. Al掺杂UiO-66-NH2的制备及其对水体中四环素和Cu(II)的吸附去除研究[D]. 雅安: 四川农业大学, 2023. MIAO Jiaxin. Preparation of Al-doped UiO-66-NH2 and its adsorption and removal of tetracycline and Cu(II) from water[D]. Ya'an: Sichuan Agricultural University, 2023(in Chinese).
[41] 符佳佳. MOFs改性石墨烯气凝胶的制备及其对污水中四环素的去除研究[D]. 雅安: 四川农业大学, 2022. FU Jiajia. Preparation of MOFs modified graphene aerogel and its removal of tetracycline from wastewater[D]. Ya'an: Sichuan Agricultural University, 2022(in Chinese).
[42] ABBASNIA A, ZAREI A, YEGANEH M, et al. Removal of tetracycline antibiotics by adsorption and photocatalytic-degradation processes in aqueous solutions using metal organic frameworks (MOFs): A systematic review[J]. Inorganic Chemistry Communications, 2022, 145: 109959. DOI: 10.1016/j.inoche.2022.109959
[43] LI W, CAO J, XIONG W, et al. In-situ growing of metal-organic frameworks on three-dimensional iron network as an efficient adsorbent for antibiotics removal[J]. Chemical Engineering Journal, 2020, 392: 124844.
-
期刊类型引用(8)
1. 谭浩,张文彬,卢文玉,祁志强,蔡红珍,杨科研. 高粱秸秆负载HKUST-1对四环素的吸附行为与机制. 复合材料学报. 2025(01): 514-526 .  本站查看
本站查看
2. 李碧秋,李希成,熊俊夫,李金韩,贾博雅,汪长征. 铋系光电催化剂降解水中有机污染物的研究进展. 材料工程. 2024(06): 92-108 .  百度学术
百度学术
3. 黄鹏伟,李静,林博,王宜民,陈仪,谢楠耿. AgI/BiOI异质结光电催化甲醇氧化. 山西化工. 2024(07): 4-6+10 .  百度学术
百度学术
4. 杜书雅,王旭东,董永浩,吕嘉辰,李洁. MOF-808/AgBr的制备及光催化降解盐酸四环素性能研究. 功能材料. 2024(11): 11137-11146 .  百度学术
百度学术
5. 严惠儒,林水源,钟祥康,黄学帅,杨玉如,冯梓盈,朱淼,谢伟. Au/BiOI花状微米球的制备及其对抗生素的降解特性. 广州化工. 2023(05): 43-46 .  百度学术
百度学术
6. 王振宇,刘燕才,陈琨,乔江浩,李晓伟. 等离子喷涂-化学气相沉积制备α/β-Bi_2O_3薄膜的相结构调控和光催化降解性能. 硅酸盐学报. 2023(07): 1800-1810 .  百度学术
百度学术
7. 刘松林,王仲民,钱熹,王童,冉兆晋,黄志民,吴晨曦,李桂银. 磁性氮掺杂石墨烯改性柿单宁复合材料对四环素的吸附行为. 复合材料学报. 2023(07): 4048-4059 .  本站查看
本站查看
8. 袁亦雷,谢水波,刘岳林,史艳丹,刘迎九. g-C_3N_4-Bi_2O_3/Al_2O_3复合材料光催化还原水中U(Ⅵ). 化工环保. 2022(05): 609-615 .  百度学术
百度学术
其他类型引用(8)
-
其他相关附件
-
目的
四环素(TC)是一种难降解广谱抗生素,广泛存在于畜牧业排放的污废中,排放后会对水体生态环境造成严重的污染。MOFs材料因具有孔隙发达、孔径可调、比表面积大、结构多样、活性位点丰富等优点被广泛应用于吸附领域。本文通过制备SS@HKUST-1复合材料来研究对TC的去除效果以及吸附机制。
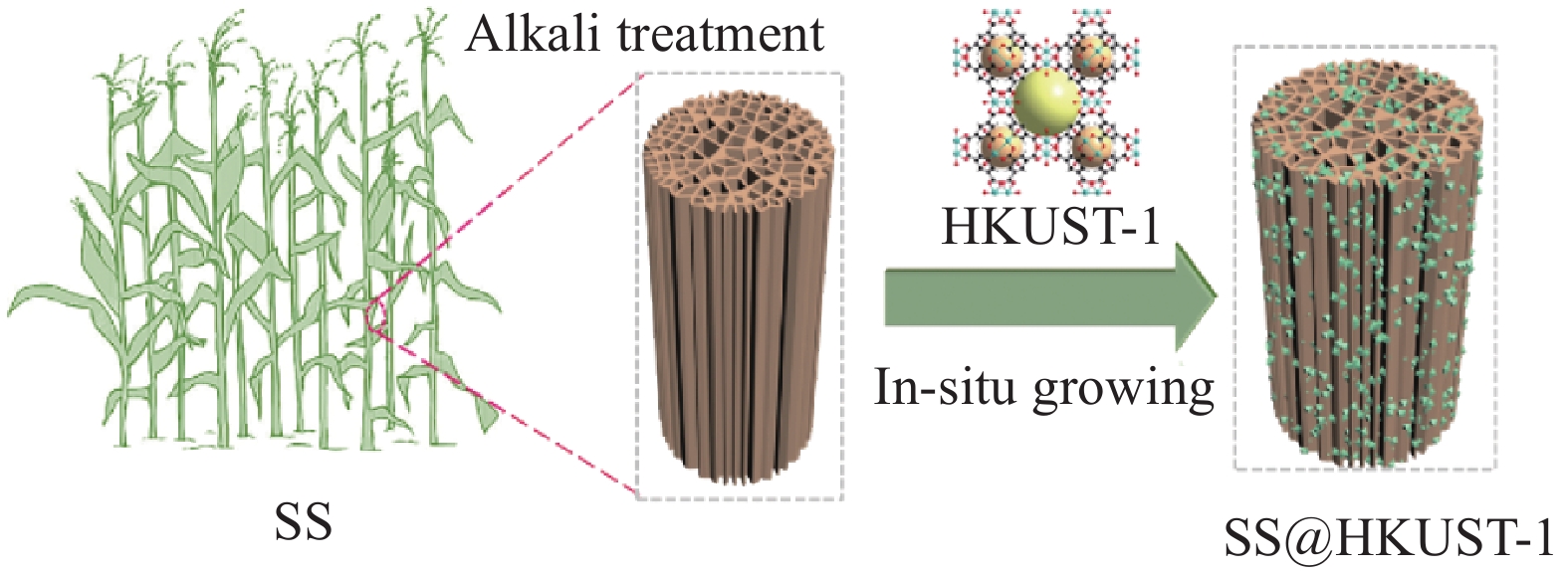
方法吸附法因成本低、操作简单而被认为是最有前途的方法之一,本研究以高粱秸秆(SS)为基材,用NaOH溶液对SS进行处理,将处理的SS与Cu(NO)溶液混合,在真空条件下诱导SS中的钠离子与铜离子交换,最终加入有机配体(HBTC)溶液在其表面原位生长HKUST-1,制备SS@HKUST-1复合材料,用于对TC的吸附去除。通过SEM、FT-IR、XRD、XPS、TG等方法来表征复合材料;通过调节TC溶液的浓度(5 mg·L、25 mg·L、35 mg·L、55 mg·L、85 mg·L、105 mg·L)、温度(25℃、35℃、45℃)、pH值(2-11)、SS@HKUST-1的接触时间(1 min、5 min、10 min、20 min、30 min、1 h、2 h、3 h、6 h、9 h、12 h、18 h、21 h、24 h)以及HKUST-1的负载量来探究最佳吸附条件。根据吸附动力学以及吸附等温线拟合出准一级动力学模型、准二级动力学模型和Langmuir模型、Freundlich模型来判断吸附类型,并通过表征吸附前后的FT-IR、XPS来确定吸附机制。
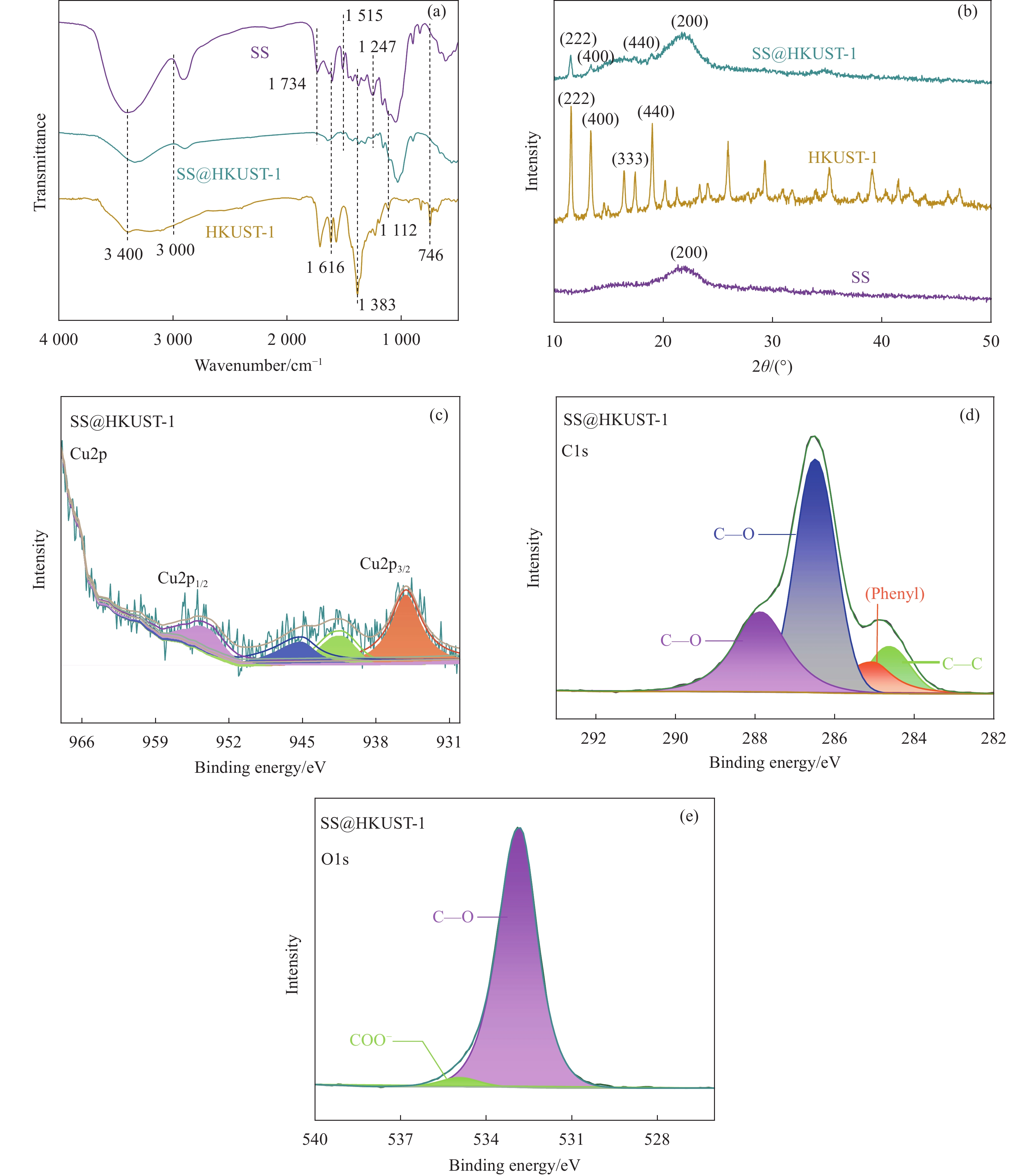
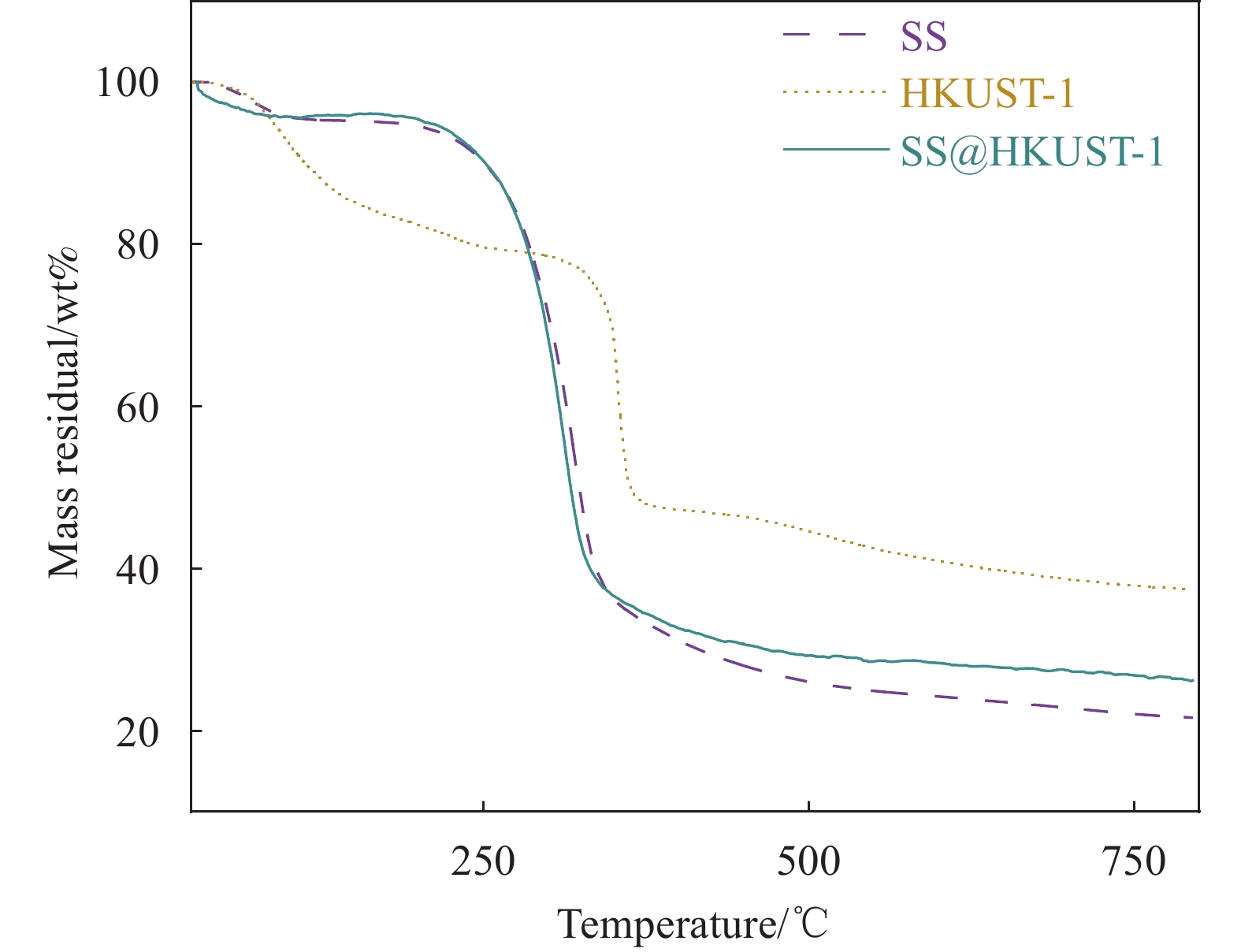
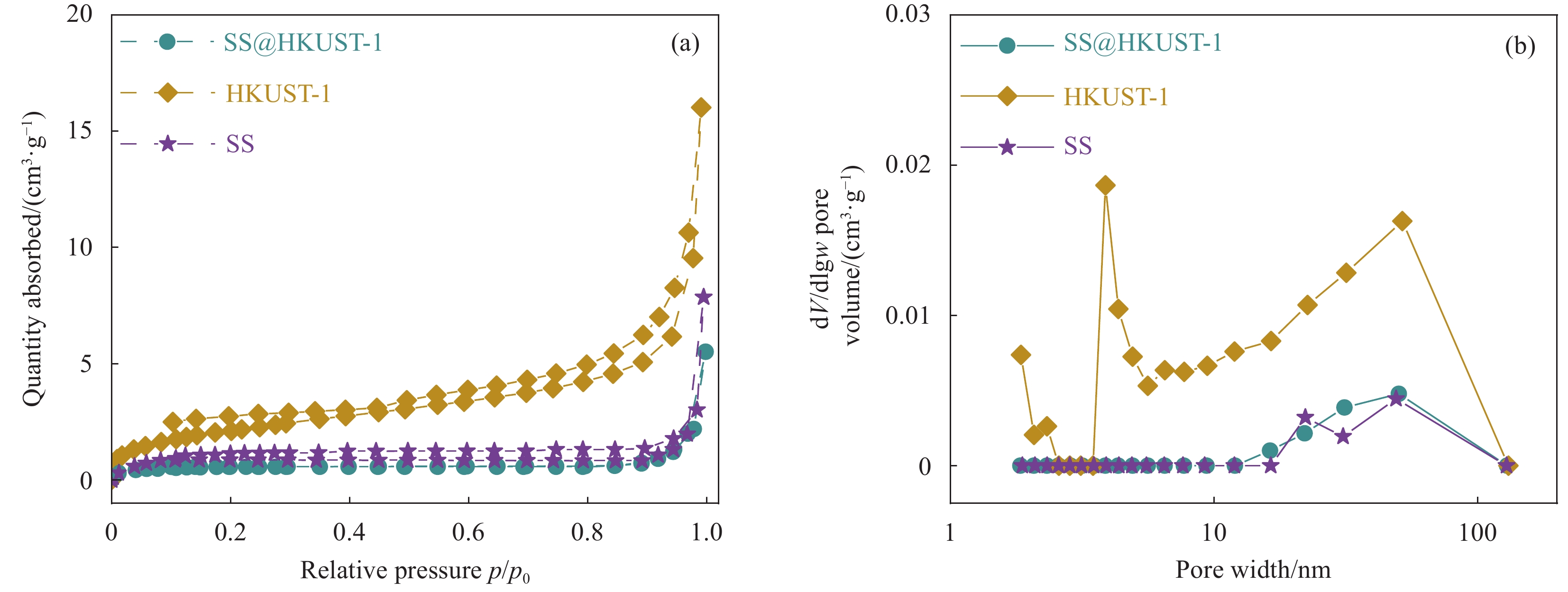
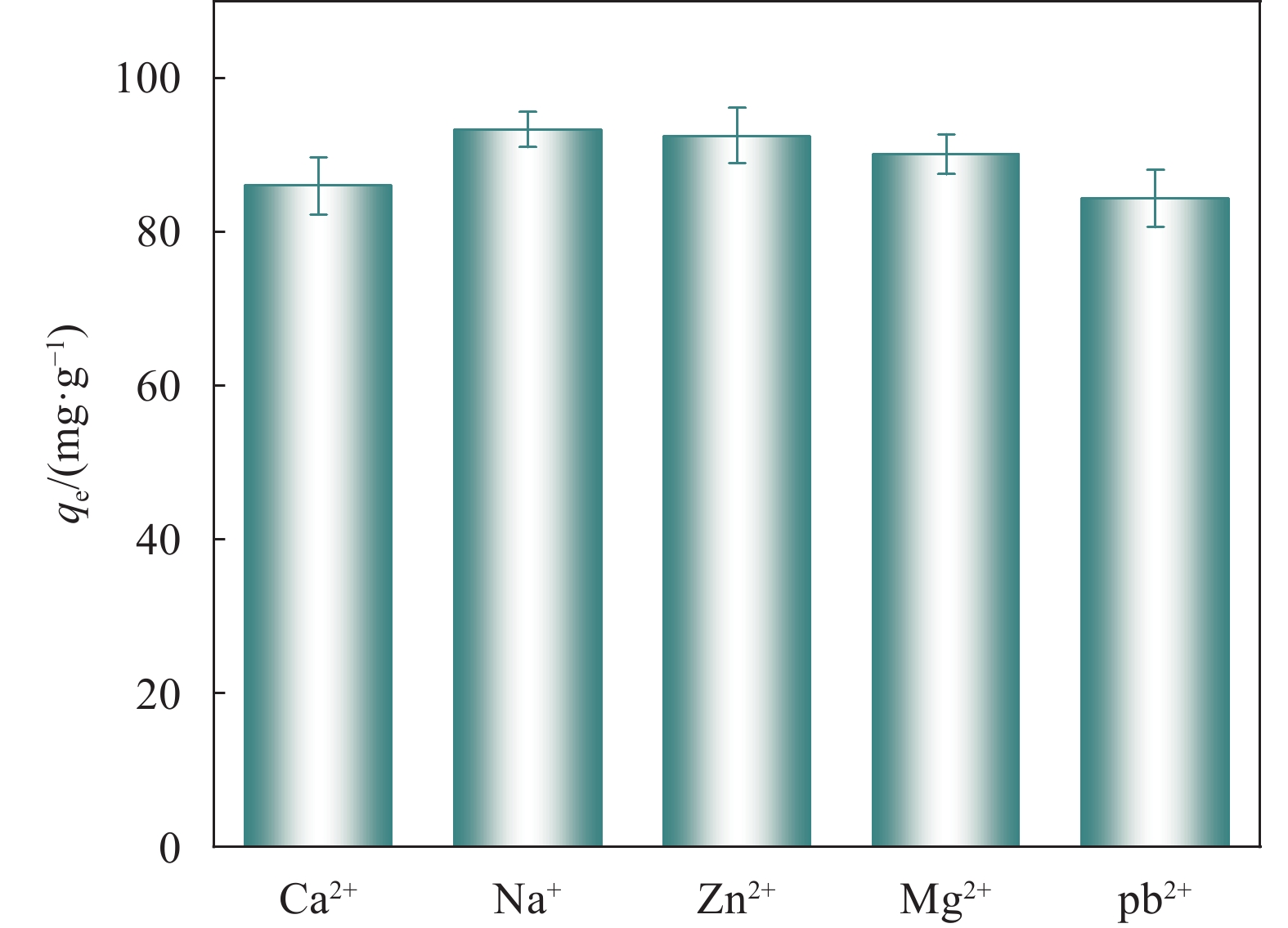
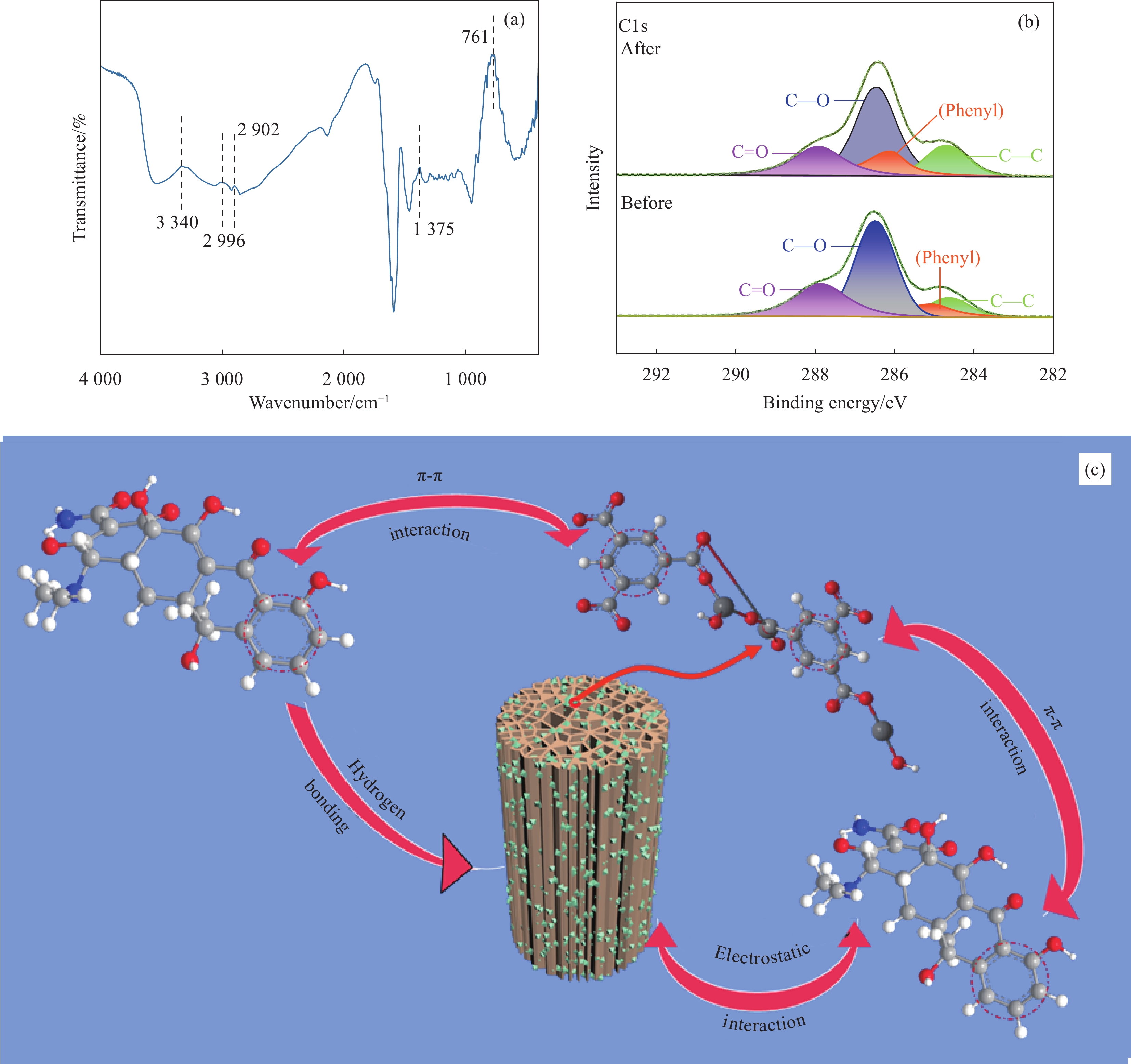
结果从SEM图像中可以看出HKUST-1晶体呈现八面体结构,晶体尺寸约2 μm;在SS@HKUST-1复合材料中观察到HKUST-1晶体在SS表面负载,并通过EDS能谱观察到Cu元素与C、O元素均匀分布;通过分析SS@HKUST-1中XPS、FT-IR、XRD的化学结构以及衍射峰得知复合材料成功制备;TG表明所制备的SS@HKUST-1具有良好的稳定性。在吸附实验阶段,实验结果表明:1通过热重图谱可以计算出HKUST-1的负载量为31%。2当pH=7、T=25°C、HKUST-1的负载量为31%时,吸附容量达到95 mg/g。3吸附动力学模型表明,20 h达到吸附平衡,吸附过程以化学吸附为主。4吸附等温线模型表明,吸附过程为多层吸附。5SS@HKUST-1经过5次吸脱附循环后,吸附容量还可以达到67 mg/g。6在共存离子的存在下,SS@HKUST-1对TC具有选择性吸附。综上所述,静电作用、氢键作用和π-π相互作用是吸附剂去除水溶液中TC过程中的主要作用机制。
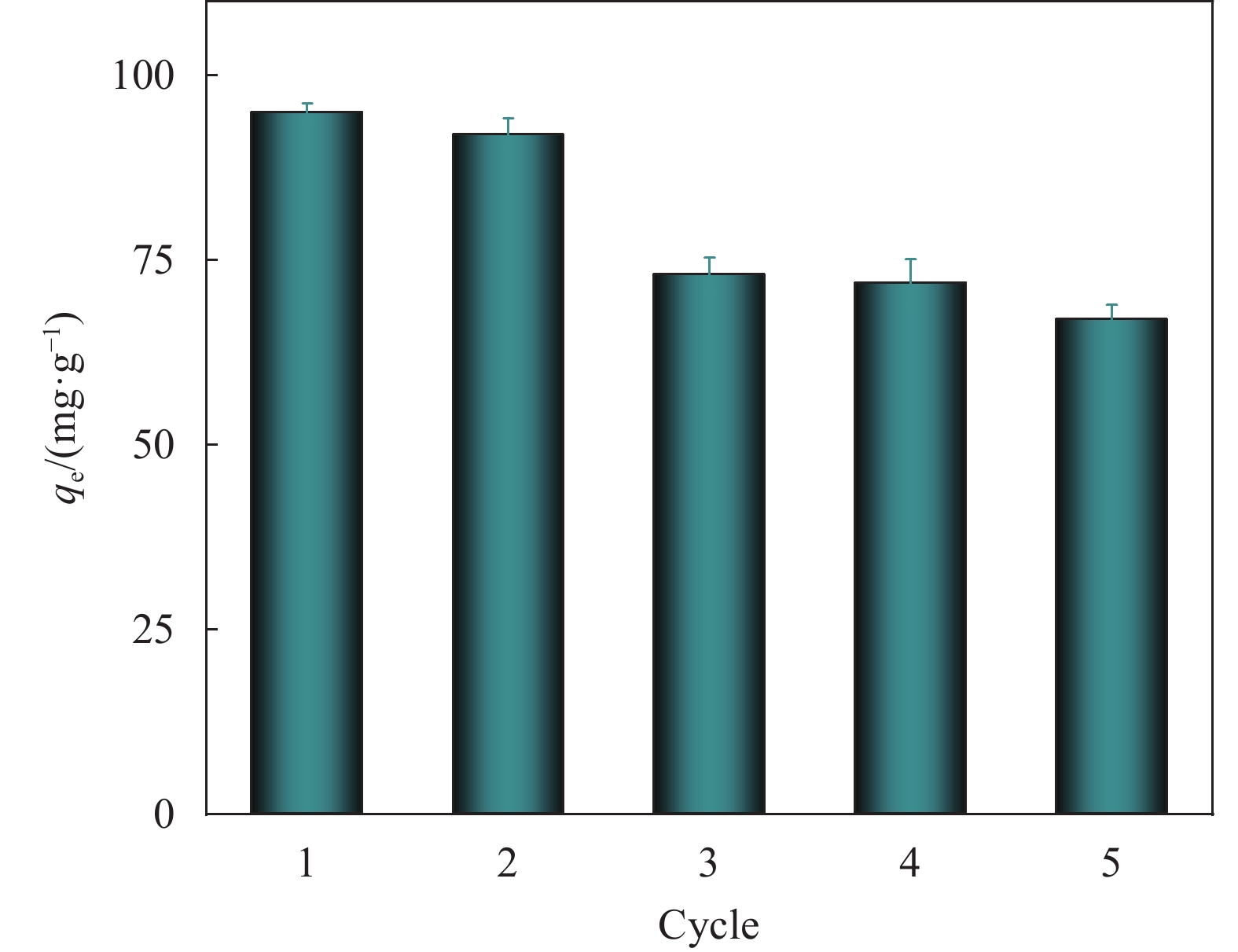
结论以高粱秸秆(SS)为基体,利用简单的原位生长方式在其表面负载HKUST-1,制备得到具有高吸附率且利于回收的SS@HKUST-1复合材料。结果表明HKUST-1均匀分布在高粱秸秆上,增加了吸附剂活性位点,二者的协同作用有利于TC的吸附,当pH=7、负载量为31%和吸附剂的添加量为1 mg吸附量达到了95 mg/g。经过5次循环实验,SS@HKUST-1仍能保持67 mg/g的吸附效果。本研究提供了一个可持续的生物质基吸附剂的制备方案。而目前生物基复合材料对污水处理仍处于实验室研究阶段,实验成分及因素单一,与工业污水和日常用水成分相差甚远。
-
四环素(TC)因抗菌效率高、价格低廉在畜牧业中得到广泛应用,然而大部分TC并未被牲畜完全吸收,会随着粪便与尿液排泄进入水体环境中,对生态环境和人体健康造成严重的威胁。其中,吸附法因成本低、操作简单而被认为是最有前途的方法之一,而金属有机框架(MOFs)材料因具有孔隙发达、孔径可调、比表面积大、结构多样、活性位点丰富等优点被广泛应用于吸附领域,但MOFs存在难以回收、成本高的问题。
本文以含有丰富极性基团(羟基、羧基)的高粱秸秆(SS)为基体,用NaOH溶液对SS进行处理,将处理的SS与Cu(NO3)2溶液混合,在真空条件下诱导SS中的钠离子与铜离子交换[18],最终加入有机配体(H3BTC)溶液在其表面原位生长HKUST-1,制备SS@HKUST-1复合材料,它不仅可以提高对四环素(TC)的吸附效果,而且也克服了MOFs的循环再利用问题。因此,所制备的SS@HKUST-1复合材料在静电作用、氢键作用和π-π相互作用下对TC的吸附容量达到95 mg·g-1,且经过5次吸脱附循环后,吸附容量仍可以达到67 mg·g-1。
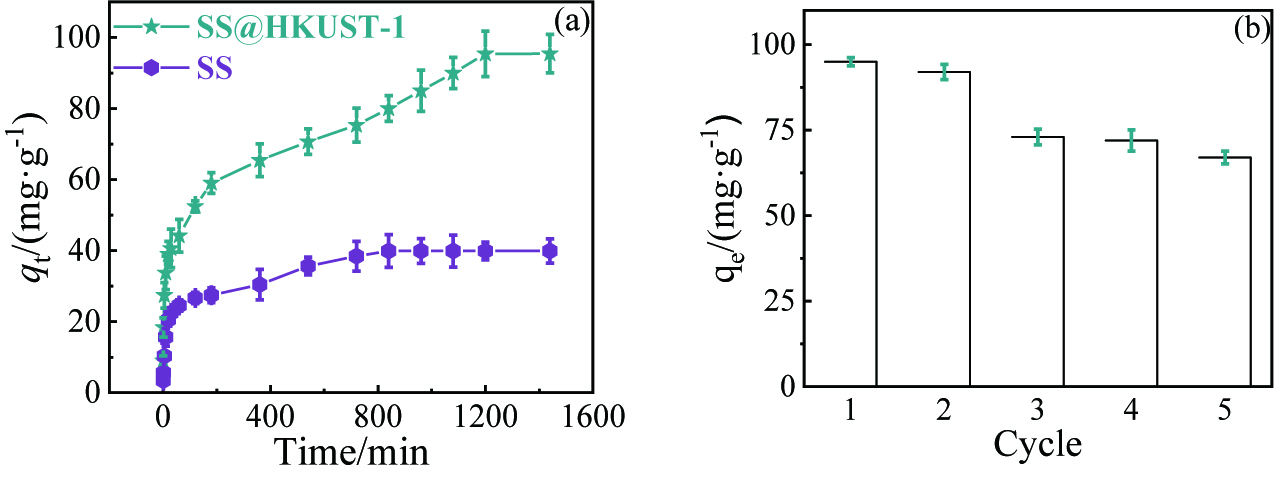
(a)吸附时间对SS@HKUST-1去除TC的影响;(b) SS@HKUST-1循环次数对去除TC的影响






 下载:
下载: