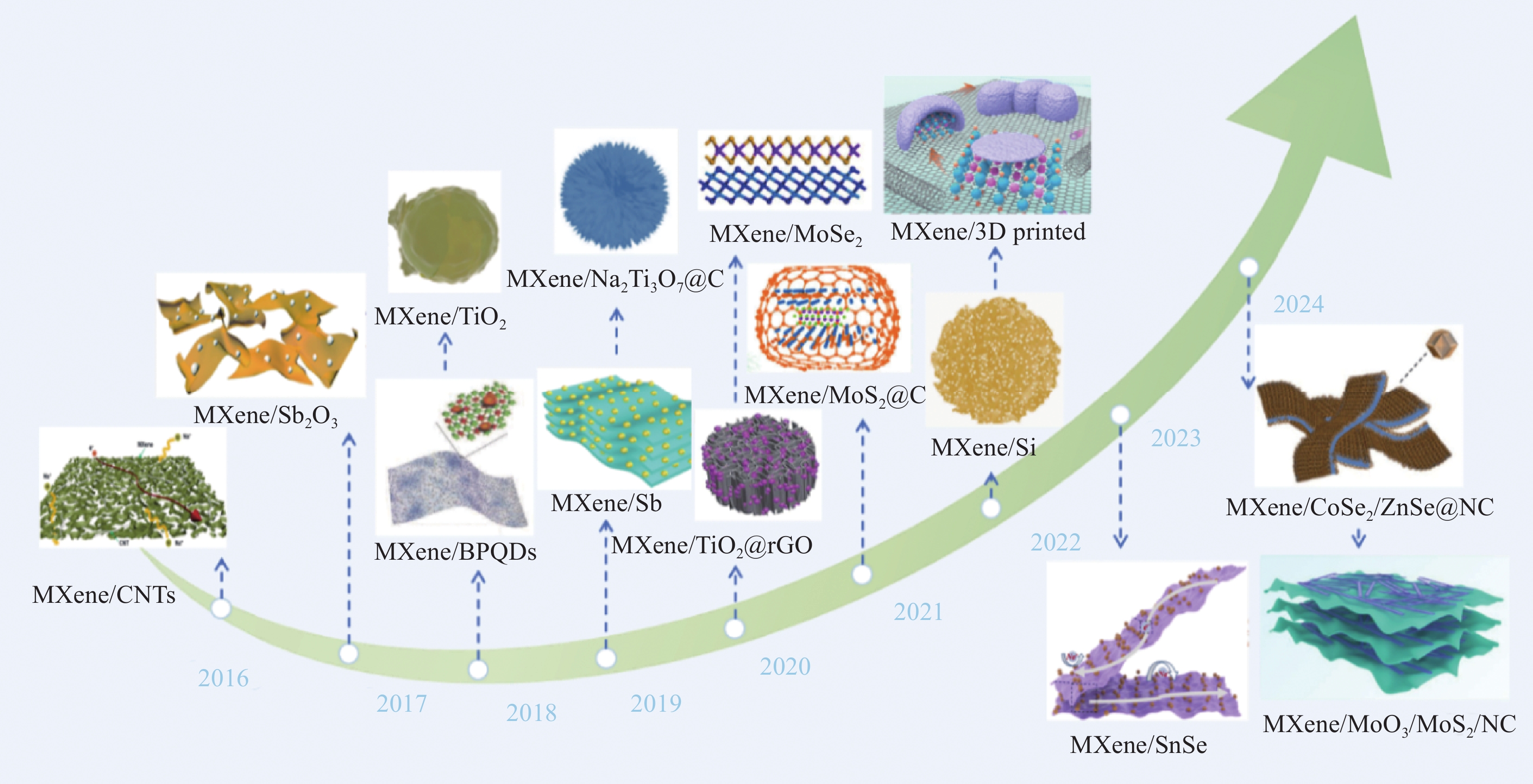
Research progress of MXene as anodes for sodium-ion batteries
-
摘要:
钠离子电池是近年来热门研究的二次离子电池之一,具有成本低、资源丰富的优点。但钠的原子质量和离子半径较大,导致钠离子电池结构形变,影响其电化学性能。MXene是一种新型的二维层状结构材料,具有高导电性、高可塑性、高耐腐蚀性和大比表面积的特性,在钠离子电池中具有广泛的应用。但MXene作为电极材料时,由于层间的范德华力使得片层易再次堆叠,从而减少了表面活性位点,加长了离子扩散路径,进而影响了钠离子电池的比容量和倍率性能。本文综述了MXene材料在钠离子电池负极中的研究进展,介绍了MXene材料的基本性质和制备方法,详细论述了MXene材料优化改性的方法和储钠机制,总结了MXene在钠离子电池负极实际应用中面临的困难和挑战,并展望了MXene材料进一步发展的方向,希望对MXene材料的实际应用有所启发。
Abstract:Sodium-ion battery is one of the most popular secondary ion batteries studied in recent years, which has the advantages of low cost and abundant resources. However, the large atomic mass and ionic radius of sodium lead to structural deformation of sodium-ion batteries, which affects their electrochemical performance. MXene is a novel two-dimensional layered structural material with high conductivity, high plasticity, high corrosion resistance, and large specific surface area, which has been widely used in sodium-ion batteries. However, when MXene is used as an electrode material, their layers are easy to re-stacked due to the van der Waals force between the layers, which reduces the surface-active sites and lengthens the ion diffusion paths, thus affecting the specific capacity and rate performance of sodium-ion batteries. This paper reviews the research progress of MXene as anodes of sodium-ion batteries. It introduces the basic properties and preparation methods of MXene, discusses the modification and optimization methods and the sodium storage mechanism of MXene in detail. It also summarizes the difficulties and challenges faced by MXene in the practical application as anodes of sodium-ion batteries and looks forward to the direction of further development of MXene. It is hoped that this will strengthen the practical application of MXene.
-
目前,全球变暖带来的负面效应极大地影响了人类的生产生活[1-2],能源的过度消耗和匮乏使得环境问题进一步增加,如何做好热管理是目前面临的主要问题[3-5]。地球表面温度为300 K,而外太空平均温度为3 K[6-7],根据热力学第二定律,地球上物体的热量由于热量差可以通过辐射的方式将热量传递到外太空。因此,辐射制冷技术是地球上的物体通过“大气窗口”波段(8~13 μm)将热量辐射到外太空[8-10],以此实现自身降温冷却的过程。
虽然辐射制冷材料在节能环保方面显示出极大的应用潜力[10],但现有材料多为白色或银白色,外观单调,利用率极低。染料会使材料的表面颜色发生改变,但染料的可见色会吸收热量,并在近红外波段内吸收额外的热量[11-12],降低了材料本身的制冷效果。到目前为止,克服这一问题的主要策略是提高在可见光区域内的反射率和在“大气窗口”波段(8~13 μm)的发射率[13-16]。为避免染料吸收热量的现象,结构色辐射制冷材料引发人们的广泛关注[17-18]。利用硅蛋白石可以制备具有结构色变化的辐射制冷材料,但这种方法不能实现亚环境冷却[19],并且生产工艺和条件复杂且苛刻,很难进行大批量生产和应用。
纤维素纳米晶体(CNC)是刚性的棒状颗粒,长度为数十至数百纳米,直径可以达到数十纳米[20],具有结晶度高、降解性好等特点。CNC可以从棉花、木材和纸浆等可再生资源中提取,具有成本低、绿色、可持续等特性[21-23]。CNC可以在水中自发地组织成手性向列结构的液晶相,这种有序结构在干燥过程中可以得到保留,直至得到具有手性向列结构的CNC薄膜[24]。双折射的CNC纳米棒在薄膜中呈现的螺旋式排列会使纳米结构的折射率产生周期性的变化,进而引起对可见光的强烈反射[25]。因此,结构色的调节与螺旋结构的周期性密切相关。
目前已有研究致力于制造色彩更为丰富的纤维素辐射制冷材料。Shanker等[26]通过将CNC自组装成结构色薄膜,与硅片基底结合,得到了一种结构色辐射制冷装置。通过控制CNC/甘油(GLU) 的质量比得到颜色由蓝紫色转变为红色的复合薄膜。结构色复合薄膜表现出在太阳光谱范围内低吸收率和“大气窗口”内高发射率,且绿色和红色样品降温效果可达9℃左右,蓝紫色样品的降温效果可达6℃左右,为制备纤维素结构色辐射制冷薄膜提供了研究基础。
本文将CNC与聚乙二醇(PEG)复合,通过自组装方法制备了高太阳光反射率和在“大气窗口”波段高发射率的结构色薄膜。通过控制CNC与PEG的质量比,调控复合薄膜的结构色,以实现不同波段的热辐射率调控。探究不同含量PEG的添加对CNC复合结构色薄膜的结构、光学性能及制冷性能的影响。并将复合结构色薄膜粘贴到醋酸纤维素滤膜上制备成双层复合膜,进一步探究双层复合膜的辐射制冷效应。
1. 实验材料及方法
1.1 原材料
醋酸纤维素:上海兴亚净化材料厂;浓硫酸(H2SO4):分析纯,阿拉丁世纪有限公司;聚乙二醇400:分析纯,天津市科密欧化学试剂有限公司。
1.2 实验过程
CNC的制备:利用酸水解法制备CNC,将200 mL 64%硫酸溶液缓慢倒入25 g硫酸盐漂白针叶浆中,在45℃的水浴加热中搅拌1 h,后加入大量蒸馏水来终止水解反应。静置2~3 d,去除上清液,加入去离子水通过高速离心机 (CT14D型,上海天美仪器有限公司) 进行多次离心洗涤。得到的CNC悬浮液,将CNC在去离子水中透析,直至pH为中性。最后将CNC悬浮液浓缩至3wt%,冷藏备用。
CNC/PEG复合薄膜的制备:称取聚乙二醇5.0 g,加入45 g超纯水中,在室温搅拌1 h,得到质量分数10wt%的聚乙二醇溶液。取一定量的质量分数为3wt%的CNC悬浮液超声5 min,取4份3.0 g CNC悬浮液分别与
0.1000 g、0.2250 g、0.3857 g和0.6000 g PEG的溶液进行混合,将上述溶液充分搅拌2 h,后将混合液体倒入圆形容器中,室温缓慢蒸发2~3 d,得到PEG浓度为10wt%、20wt%、30wt%和40wt%的CNC/PEG复合结构色薄膜。按照聚合物的种类和含量对复合薄膜进行命名,分别为CNC/PEG-10%、CNC/PEG-20%、CNC/PEG-30%、CNC/PEG-40%,具体见表1。表 1 纤维素纳米晶体/聚乙二醇(CNC/PEG)复合辐射制冷薄膜和CNC/PEG-醋酸纤维素(CA)结构色辐射制冷双层复合膜的命名Table 1. Naming of cellulose nanocrystal/polyethylene glycol (CNC/PEG) composite radiative cooling films and CNC/PEG-cellulose acetate (CA) structure-colored radiation-cooled bilayer composite filmsSample Mass fraction of
CNC/wt%Mass fraction of
PEG/wt%CA CNC/PEG-10% 3 10 — CNC/PEG-20% 3 20 — CNC/PEG-30% 3 30 — CNC/PEG-40% 3 40 — CNC/PEG-10%-CA 3 10 0.0740 g CNC/PEG-20%-CA 3 20 0.0740 g CNC/PEG-30%-CA 3 30 0.0740 g CNC/PEG-40%-CA 3 40 0.0740 g 双层复合膜的制备:将不同质量比的复合结构色薄膜与醋酸纤维素膜用双面胶粘在一起,双面胶放在醋酸纤维素膜的边缘起固定作用,不会影响双层复合结构色薄膜的结构,双层复合结构色薄膜分别命名为CNC/PEG-10%-CA、CNC/PEG-20%-CA、CNC/PEG-30%-CA,具体见表1。
1.3 测试与表征
1.3.1 CNC/PEG复合膜的性能分析
采用Malvern Zetasizer nano ZS90测试CNC的Zeta电位和粒径。利用偏光显微镜(POM,XPF-550C,上海蔡康光学仪器公司)观察复合薄膜样品的液晶特性。使用反射光谱仪(UV-vis,HR4000 CG-NIR型,海洋光学公司)对薄膜进行反射光谱测试(可见光区域)。将复合薄膜用液氮进行脆断,用双面导电胶粘到截面制样台上,喷金60 s,利用扫描电子显微镜(SEM,JSM-7500F,日本电子株式会社)观察复合薄膜的横截面微观形貌。使用带有积分球附件的紫外-可见-近红外分光光度计(UV-VIS-NIR Spectrometer,美国Perkins Elmer公司)检测样品在300~
2500 nm波长范围内的反射率变化。利用傅里叶变换红外光谱仪(FTIR,Nicolet-IS10,美国Thermo Fisher公司)检测样品在2.5~25 μm的波长范围内样品的吸收率变化。采用太阳光谱匹配良好的高功率氙灯(中教金源HXF300)来模拟太阳光照射,在聚苯乙烯(PS)泡沫箱子中裁剪一个1 cm×1 cm×1 cm的空腔,将薄膜或复合薄膜放置在空腔中固定,利用聚乙烯薄膜覆盖泡沫箱,消除环境热对流影响。将多通道温度计(JINKO,JK804)与两个k型热电偶进行连接,一个k型热电偶测试复合薄膜覆盖下空腔的内部温度,另一个k型热电偶用来测量聚乙烯膜覆盖下的装置内的环境温度。由于CNC/PEG-40%复合薄膜在表征过程中会吸收环境的水分,手性结构发生润胀,螺距在实际测量中会发生变化,不利于复合薄膜在实际环境中的应用,因此后续将不会对CNC/PEG-40%复合薄膜进行扫描电镜、光谱学和辐射制冷性能的测试与分析。1.3.2 双层复合膜的性能分析
将醋酸纤维素膜用导电胶粘贴到水平制样台上,喷金60 s,利用扫描电子显微镜观察醋酸纤维素薄膜的表面形貌。在同一氙灯光源照射下,观察不同样品在红外热成像仪(FTIR-E390,美国FLIR SYSTEMS公司)下的温度变化。利用上述自组装装置分别测量双层复合薄膜下方温度及被聚乙烯膜覆盖的整个装置内的环境温度。利用实验室自组装装置对样品进行户外降温性能测试,将室内自组装降温性能测试装置放置在用铝箔纸包裹的纸壳箱中,整体放置在泡沫箱上,用湿度计记录测试过程中的环境湿度变化。
2. 结果与讨论
2.1 纤维素纳米晶的电位粒径分析
图1为CNC的TEM图像、粒径分布和电位曲线,酸水解法制备的CNC具有棒状形态,平均粒径为144.1 nm (图1(b))。在水解过程中,CNC表面形成较多的负电荷,Zate电位高达−32.2 mV (图1(c))。CNC表面较多的负电荷会促进静电排斥作用,使CNC溶液的稳定性增强,为进一步制备结构色复合薄膜奠定基础。
2.2 纤维素纳米晶复合薄膜结构色光学特性分析
图2(a)~2(d)是PEG含量不同的复合薄膜光学照片。随着PEG含量的增加,薄膜反射颜色发生红移,逐渐由蓝绿色转变为红色。因此,复合薄膜的结构色红移现象与PEG的含量相关。通过对紫外-可见反射光谱(图2(e))分析可得,随着PEG含量的增加,4种复合薄膜分别在427 nm、487 nm、576 nm和654 nm处存在清晰的高峰,复合薄膜反射光谱中的最高峰发生红移,与薄膜结构色的红移相对应。通过图2(f)可知,复合薄膜通过反射可见光和发射热量来实现自身降温,为后续辐射制冷的研究提供理论基础。
2.3 纤维素纳米晶复合薄膜微观形貌分析
图3(a)~3(c)为PEG含量不同复合薄膜的横截断面扫描电镜图。当PEG含量为40%时,复合薄膜会吸收环境中的水分,手性结构发生润胀,螺距在实际测量中会发生变化,因此只对PEG含量为10%~30%的CNC/PEG复合薄膜进行扫描电镜的研究,不对CNC/PEG-40%复合薄膜进行扫描电镜的测试与分析。纯CNC薄膜具有周期性的层状结构,CNC通过逆时针方向旋转后形成了左旋的手性向列螺旋结构,这种左旋的手性向列结构反射特定波长的左旋圆偏振光,从而使复合薄膜表现出独特的虹彩色。聚合物的加入并不会改变CNC原有的手性向列结构,随着聚合物含量的增加,CNC手性向列结构的螺距明显增加。布拉格方程式中手性向列的螺距(P)定义为CNC棒状颗粒旋转360°产生的层间距,在电镜图(SEM)中表现为相邻两层结构的间距。
CNC/PEG复合薄膜的反射光遵循布拉格方程:
λ=nPcos(θ) (1) 其中:λ为反射波长;n为薄膜的平均折射率;θ为入射角;P为手性向列结构的层间距。因为CNC和PEG的折射率相似,分别为1.41和1.44,所以薄膜的平均折射率(n)可以认为是常量,当入射角(θ)恒定时,λ取决于手性向列结构的螺距P。图3(a)~3(c)的平均螺距分别为0.30、0.36和0.46 μm。随着平均螺距P的增加其反射波长λ也逐渐增大,主要原因是在加入PEG后,PEG高分子进入到CNC手性向列结构中,导致CNC手性向列结构的螺距P增大,复合薄膜颜色红移。
2.4 纤维素纳米晶复合薄膜折射现象
图4是PEG含量不同的复合薄膜偏光显微镜(POM)图像。通过观察图4(a)~4(d)可以得知薄膜具有明显的双折射现象,高倍POM图像(图4(e)~4(h))可以看出其具有明显的指纹结构,这说明CNC/PEG在干燥过程中CNC自组装了手性向列结构,并且在完全成膜后,仍然保留其手性向列结构。因此,适量PEG的加入并不会破坏CNC自组装所形成的手性向列结构。复合薄膜的指纹结构的纹理间隔随着PEG含量的增加逐渐变宽,分别为2.05 μm、2.33 μm、2.84 μm和3.38 μm,颜色由蓝绿色逐渐转化成蓝红色。PEG的添加占据了手性向列结构CNC之间的空间,导致螺距P增大,从而发生红移。因此,通过对POM结果分析,证明了PEG的添加不会破坏CNC的手性向列结构和双折射现象,控制PEG含量可以有效调控CNC手性向列层间距,进而调控复合薄膜结构色的变化。
2.5 纤维素纳米晶结构色复合薄膜和双层复合薄膜的光谱学分析
由基尔霍夫定律可知,样品的发射率(T)等同于吸收率(A)。通过观察图5(a)可知,在室内湿度为42%时,样品在大气窗口波段(8~13 μm)都有较高的发射率,其中当PEG的含量为30%时,复合薄膜的发射率最高可达93.0%,这样可以最大限度的向天空辐射红外热量。CNC/PEG复合薄膜具有高发射率,这是由于O—H (6.9~7.6 μm)、C—O (7.6~9.5 μm)、C—H (11.1~14.3 μm)键在大气窗口范围(8~13 μm)内产生强烈的拉伸与弯曲振动所导致的。图5(b)是PEG含量不同的复合薄膜在太阳波段(0.3~2.5 μm)范围内太阳光反射率曲线,结构色复合薄膜在近红外范围内的反射率最高可达68.5%。随着PEG的含量增加,其反射率也随之变高。
图6为不同PEG含量的双层复合制冷膜的发射率曲线和在可见光范围内的反射率曲线。通过观察图6(a)可知,在室内湿度为42%的测量环境下,双层复合膜在大气窗口的发射率高于醋酸纤维素膜的发射率,随着PEG含量的增加双层复合膜的发射率也逐步提高,当PEG含量为30%时,双层复合膜的发射率最高可达68.0%。图6(b)是PEG含量不同的双层复合膜在太阳波段(0.3~2.5 μm)范围内的太阳光反射率曲线,在近红外范围内的反射率最高可达91.8%。
2.6 纤维素纳米晶复合结构色薄膜和双层复合薄膜的辐射制冷性能分析
图7(a)为室内氙灯模拟图,利用100 mW/cm2高功率氙灯来照射,不仅可以模拟太阳光照射还可以将光均匀地分布在样品表面。图7(b)为用来测量样品温度及装置内空气温度的自组装装置,聚乙烯(PE)薄膜既可以保证氙灯的光照射到装置内部,又可以减少热对流对辐射制冷结果的影响。由图7(c)、图7(d)可知,将氙灯打开后,PE薄膜覆盖的装置内部温度迅速上升,在5 min后样品逐渐达到热稳定状态。当温度逐渐趋向平衡时,薄膜下方温度明显低于PE覆盖装置内的空气温度,不同结构色CNC/PEG复合薄膜辐射制冷性能相似,薄膜平均降温可达3.4℃。
![]() 图 7 (a)室内氙灯模拟装置图;(b)自组装温度测量装置图;CNC/PEG-10%和空气(c)、CNC/PEG-30%和空气(d) 温度对比图Figure 7. (a) Photos of indoor xenon lamp simulation device; (b) Self assembling temperature measuring device; Temperature comparison of CNC/PEG-10% and air (c), CNC/PEG-30% and air (d)PE—Polyethylene; IR—Infrared spectroscopy
图 7 (a)室内氙灯模拟装置图;(b)自组装温度测量装置图;CNC/PEG-10%和空气(c)、CNC/PEG-30%和空气(d) 温度对比图Figure 7. (a) Photos of indoor xenon lamp simulation device; (b) Self assembling temperature measuring device; Temperature comparison of CNC/PEG-10% and air (c), CNC/PEG-30% and air (d)PE—Polyethylene; IR—Infrared spectroscopy图8(a)~8(c)为不同纤维素基底在同一光源照射下的红外热成像图。在氙灯的照射下,采用红外热成像观察5 min,可以看出,醋酸纤维素薄膜的表面温度最低,滤纸的表面温度略高于醋酸纤维素薄膜,而A4纸的表面温度最高。通过观察醋酸纤维素的SEM图像(图8(d))可知,醋酸纤维素膜具有多孔结构,可以有效地反射可见光。图8(e)为醋酸纤维素膜下温度与环境温度对比曲线,膜下温度平均比环境温度低15℃左右。综上表明,醋酸纤维素薄膜具有良好的辐射降温能力,是作为双层复合薄膜的较优选择。
利用红外热成像分别观察CNC/PEG-20%、CNC/PEG-20%-CA和带有蓝色涂料的CA薄膜在相同时间和相同光照下其表面的降温能力,如图9(a)~9(c)所示。结果可知,CNC/PEG-20%-CA的表面降温能力较强,CNC/PEG-20%的降温能力次之,而带有蓝色涂料的CA薄膜的表面降温能力最差。通过分析CNC/PEG-20%和带有蓝色涂料的CA薄膜的温度曲线(图9(d)、图9(e)),进一步证实双层复合薄膜具有良好的制冷性能。由图9(f)、图9(g)可知,PE薄膜覆盖下装置内的空气温度与双层复合制冷膜下方温度的起始温度大致相同,氙灯打开后,两者温度迅速上升,5 min后双层复合制冷膜下方温度与装置内空气温度逐渐达到热稳定状态。当温度逐渐趋向平衡时,双层复合制冷膜下方温度远低于装置内空气温度,双层复合膜的辐射制冷性能几乎不受PEG含量的影响,双层复合薄膜平均降温可达14.3℃,双层复合膜的降温性能优于复合薄膜的降温性能。实验结果表明:CNC/PEG复合薄膜平均可降温3.4℃左右,醋酸纤维素膜是作为双层复合膜的理想基底,双层复合制冷膜的降温性能优于复合薄膜,平均降温可达14.3℃左右。
![]() 图 9 ((a)~(c)) CNC/PEG-20%、CNC/PEG-20%-CA与带有蓝色涂料的CA薄膜红外热成像图;带蓝色涂料的CA和空气(d)、CNC/PEG-20%-CA和空气(e)、CNC/PEG-10%-CA和空气(f)、CNC/PEG-30%-CA和空气(g)温度对比图Figure 9. ((a)-(c)) Infrared thermograms of CNC/PEG-20%, CNC/PEG-20%-CA and and CA films with blue coatings; Temperature comparison of CA with blue painting and air (d), CNC/PEG-20%-CA and air (e), CNC/PEG-10%-CA and air (f), CNC/PEG-30%-CA and air (g)
图 9 ((a)~(c)) CNC/PEG-20%、CNC/PEG-20%-CA与带有蓝色涂料的CA薄膜红外热成像图;带蓝色涂料的CA和空气(d)、CNC/PEG-20%-CA和空气(e)、CNC/PEG-10%-CA和空气(f)、CNC/PEG-30%-CA和空气(g)温度对比图Figure 9. ((a)-(c)) Infrared thermograms of CNC/PEG-20%, CNC/PEG-20%-CA and and CA films with blue coatings; Temperature comparison of CA with blue painting and air (d), CNC/PEG-20%-CA and air (e), CNC/PEG-10%-CA and air (f), CNC/PEG-30%-CA and air (g)图10(a)、图10(b)为测量CNC/PEG-30%-CA、CNC/PEG-30%及PE覆盖装置内空气温度的户外装置图,利用铝箔纸包裹整个装置以减少周围建筑物对装置热辐射的影响,在装置顶部覆盖一层PE膜来减少环境中的热对流及热传导对整个装置的影响,装置下方的泡沫箱用来隔绝地面对测量温度的热影响,利用热电偶分别记录样品覆盖空腔中的温度及PE膜覆盖下装置的空气温度。通过分析图10(c)可以看出,在平均温度为25℃,平均湿度51%的户外环境中,与PE覆盖装置中空气温度对比,复合薄膜可以实现平均2℃左右的降温效果,而双层复合薄膜可以实现平均6℃左右的降温效果。
![]() 图 10 ((a), (b))测试CNC/PEG-30%-CA、CNC/PEG-30%和空气的温差变化的户外装置图;(c) CNC/PEG-30%-CA、CNC/PEG-30%与空气的温差图Figure 10. ((a), (b)) Diagram of an outdoor installation for testing the change in temperature difference between CNC/PEG-30%-CA, CNC/PEG-30% and air; (c) Temperature comparison of CNC/PEG-30%-CA, CNC/PEG-30% and air
图 10 ((a), (b))测试CNC/PEG-30%-CA、CNC/PEG-30%和空气的温差变化的户外装置图;(c) CNC/PEG-30%-CA、CNC/PEG-30%与空气的温差图Figure 10. ((a), (b)) Diagram of an outdoor installation for testing the change in temperature difference between CNC/PEG-30%-CA, CNC/PEG-30% and air; (c) Temperature comparison of CNC/PEG-30%-CA, CNC/PEG-30% and air3. 结 论
本文将纤维素纳米晶体(CNC)与聚乙二醇(PEG)以不同比例混合,采用自组装的方法制备了具有辐射制冷性能的结构色复合薄膜,将结构色复合薄膜与具有多孔结构的醋酸纤维素膜(CA)相结合,制备具有辐射制冷和结构色特性的双层复合膜。分别对复合薄膜和双层复合膜的性能进行分析,得出的结论如下:
(1) CNC/PEG复合薄膜具有手性向列结构和鲜艳的结构色,复合薄膜出现明显的双折射特性,随着PEG含量的增加,复合薄膜手性向列结构的螺距增大,反射波长随之发生红移,最终导致薄膜结构色的变化;
(2)对CNC/PEG复合薄膜和CNC/PEG-CA双层复合膜进行FTIR和UV-vis测试可知,复合薄膜在0.25~2.5 μm的波长范围内的反射率高达93.0%,双层复合膜反射率可达68.0%,复合薄膜在“大气窗口”(8~13 μm)范围内的发射率可达68.5%,双层复合膜发射率高达91.8%;
(3)在氙灯照射下,CNC/PEG结构色复合薄膜具有辐射制冷性能,与装置内空气温度对比,平均降温可达3.4℃左右。与具有多孔结构的醋酸纤维素膜结合,双层结构色复合薄膜的辐射制冷性能得到提升,平均降温可达14.3℃左右。在户外降温性能测试中,复合薄膜可以达到平均2℃左右的降温效果,双层复合膜可以达到平均6℃左右的降温效果。
-
图 1 二维层状MXene相应元素、结构以及刻蚀示意图:(a) 4种MXene的结构图[33];(b) M、X、T、A在元素周期表中的对应元素[33];(c) MAX相的结构和相应的MXene[34];(d) Ti3AlC2颗粒(d1)与HF刻蚀的 Ti3C2Tx (d2)、TiNbCTx (d3)、Ta4C3Tx (d4)、Ti3CNTx (d5)、Ti2CTx (d6)的SEM图像[37]
Figure 1. Schematic diagrams of the corresponding elements, structures, and etching of the 2D layered MXene: (a) Structural diagrams of the four MXene[33]; (b) Corresponding elements of M, X, T, and A in the periodic table[33]; (c) Structures of the MAX phases and the corresponding MXene[34]; (d) SEM images of Ti3AlC2 (d1) and HF-etched Ti3C2Tx (d2), TiNbCTx (d3), Ta4C3Tx (d4), Ti3CNTx (d5), Ti2CTx (d6)[37]
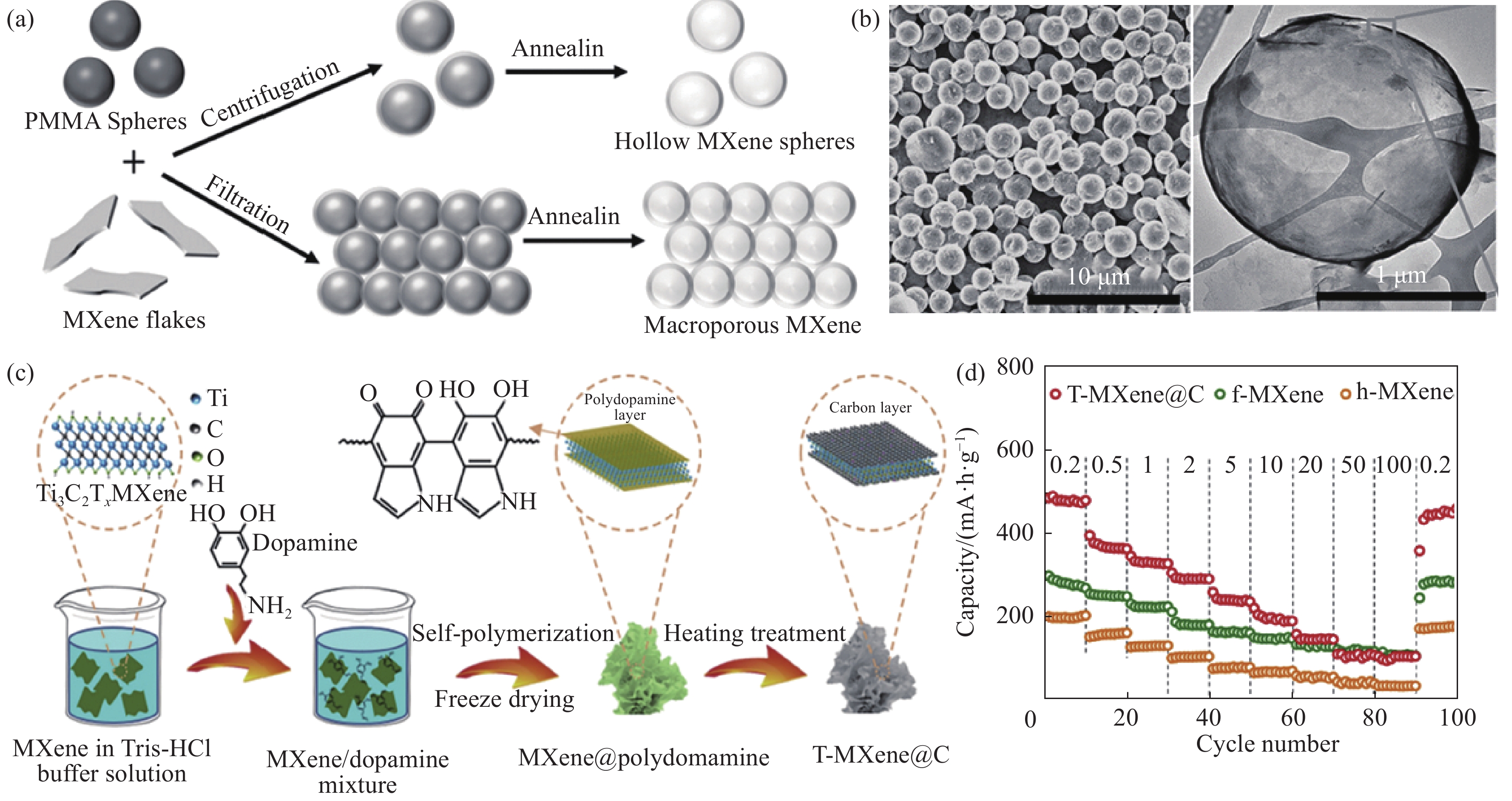
图 3 MXene形貌与结构优化:(a) 3D Ti3C2Tx制备原理图[65];(b) 3D Ti3C2Tx透射电子显微镜(TEM)图像[65];(c) T-MXene@C制备原理图[67];(d) T-MXene@C相对于f-MXene和h-MXene的倍率性能[67]
Figure 3. MXene morphology and structure optimization: (a) 3D Ti3C2Tx preparation schematic diagram[65]; (b) 3D Ti3C2Tx TEM images[65]; (c) T-MXene@C preparation schematic diagram[67]; (d) Multiplier performance of T-MXene@C relative to f-MXene and h-MXene[67]
PMMA—Methyl polymethylpropionate
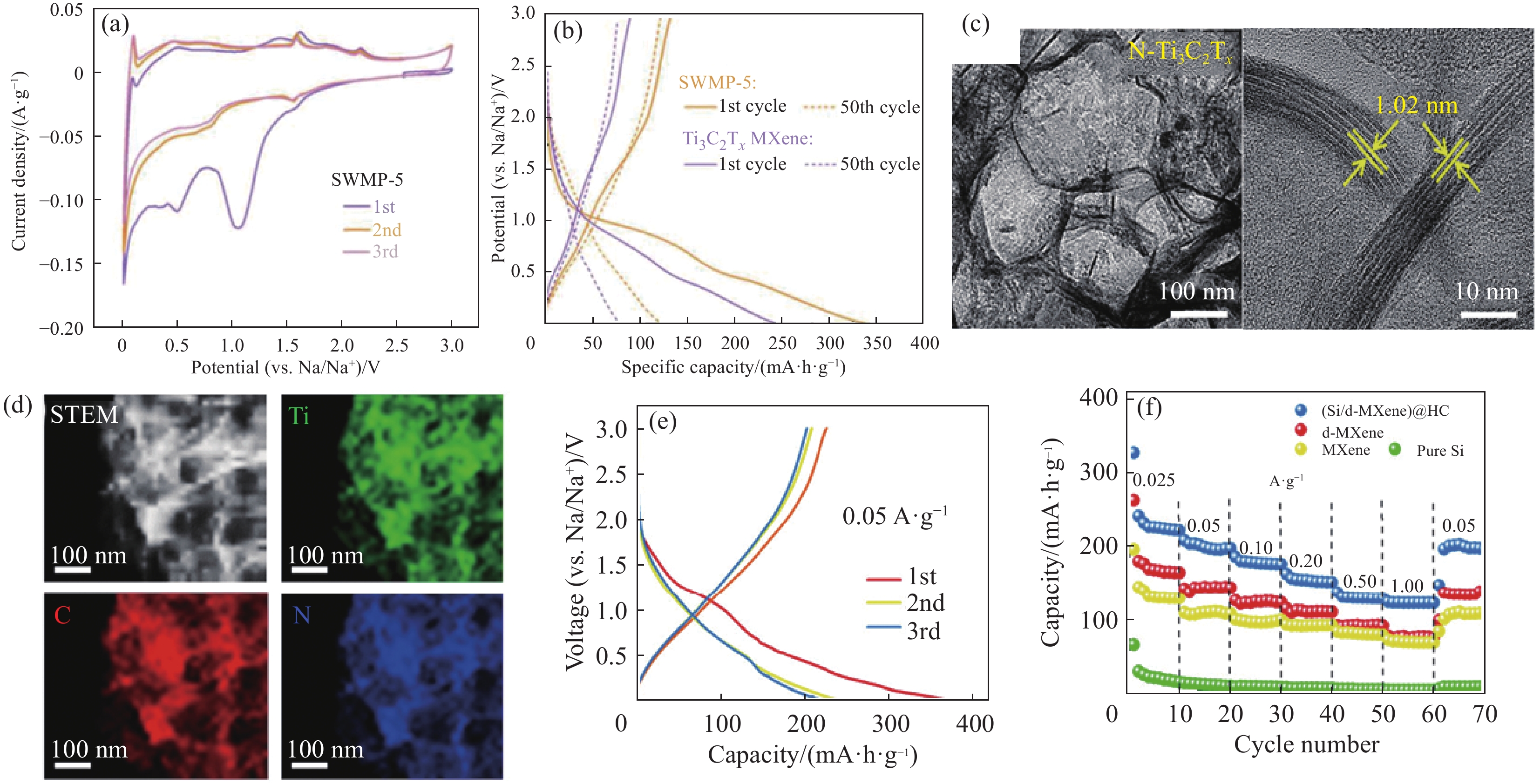
图 4 杂原子掺杂MXene复合材料:(a) N-Ti3C2 MXene在1 mV·s−1时的循环伏安曲线[69];(b) N-Ti3C2 MXene和Ti3C2Tx MXene的恒流充放电曲线[69];(c)多孔N-Ti3C2Tx TEM图像[70];(d)多孔N-Ti3C2Tx相应的EDS[70];(e)电流速率为0.05 A·g−1时(Si/d-MXene)@HC前3个循环的充放电曲线[73];(f) (Si/d-MXene)@HC、d-MXene、MXene和纯硅的倍率性能[73]
Figure 4. Heteroatom doped MXene composites: (a) Cyclic voltammetry curves of N-Ti3C2 MXene at 1 mV·s−1 [69]; (b) Constant-current charge-discharge curves of N-Ti3C2 and Ti3C2Tx[69]; (c) TEM images of porous N-Ti3C2Tx [70]; (d) EDS images of porous N-Ti3C2Tx corresponding [70]; (e) Charge-discharge profile of the (Si/d-MXene)@HC electrode at 0.05 A·g−1 for the first three cycles[73]; (f) Rate capability of (Si/d-MXene)@HC, d-MXene, MXene, and pure Si electrodes[73]
SWMP-5—Polyethyleneimine composite MXene; STEM—Scanning transmission electron microscope; HC—Hard carbon
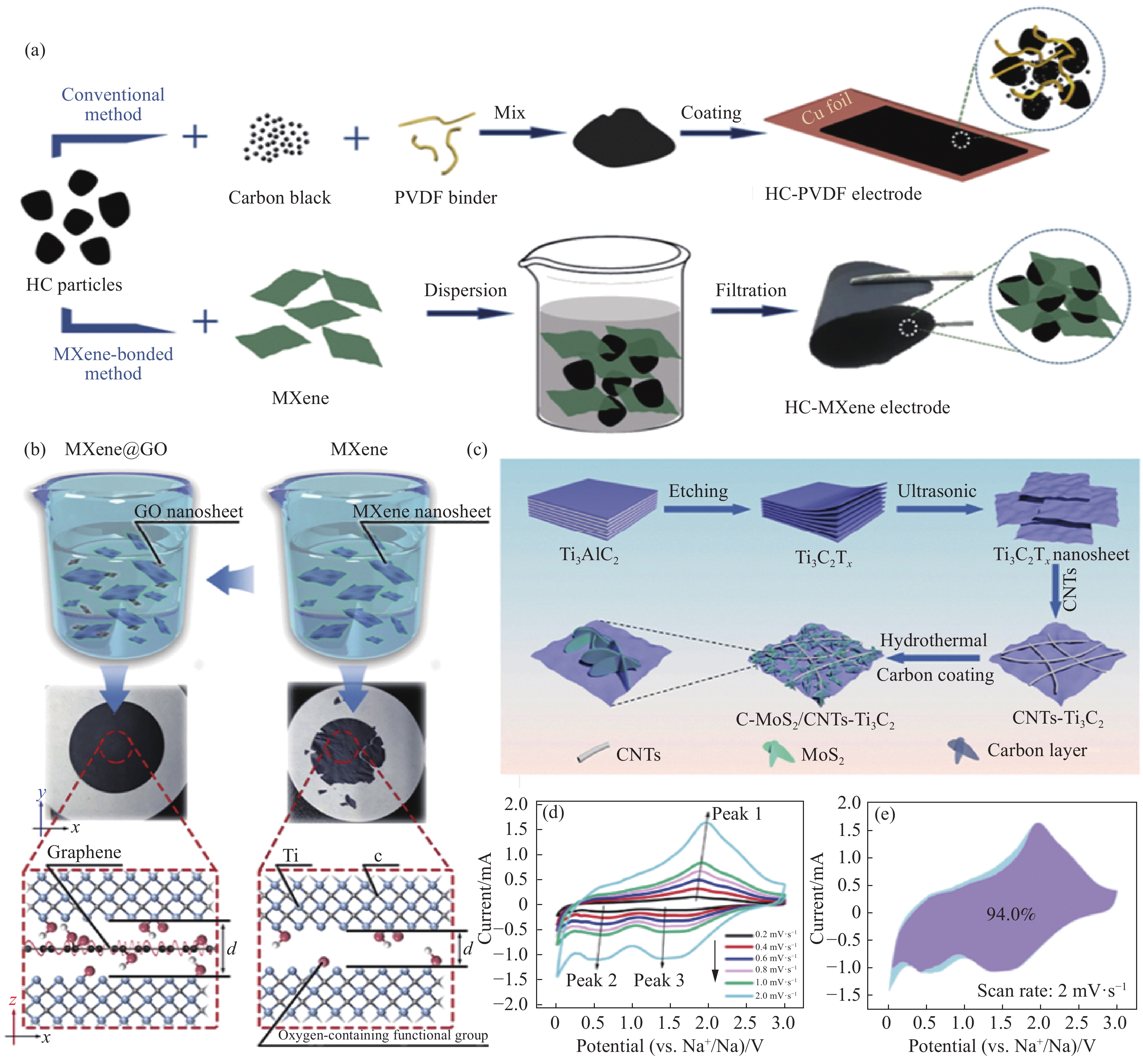
图 5 MXene基复合材料:(a)传统聚偏二氟乙烯(PVDF)粘合和HC-MXene电极制备原理图[74];(b)制备MXene@氧化石墨烯薄膜(GO)的机制示意图[60];(c) C-MoS2/CNTs-Ti3C2复合材料的制备过程示意图[86];(d)不同扫描速度下的循环伏安曲线[86];(e) 2.0 mV·s−1下复合材料对电荷存储的贡献[86]
Figure 5. MXene-based composites: (a) Schematic diagram of the preparation of conventional polyvinylidene difluoride (PVDF) bonded and HC-MXene electrodes[74]; (b) Schematic diagram of the mechanism for the preparation of MXene@graphene oxide thin films[60]; (c) Schematic diagram of the preparation process of C-MoS2/CNTs-Ti3C2 composites[86]; (d) Cyclic voltammetry curves at different scanning speeds[86]; (e) Contribution of composites to charge storage at 2.0 mV·s−1 [86]
c—Carbon skeleton; d—Interval between floors
图 6 MXene的储钠机制示意图:(a) Ti3C2Tx电极高角度环形暗场(HAADF)图像[106];(b) 20 mA·g−1下Ti3C2Tx的充放电曲线[107];(c)在初始两个循环中O1s的XPS光谱[107];(d)初始两个循环中Ti的X射线吸收近边结构(XANES)谱图[107];(e) Na+嵌入和脱出的机制示意图[107]
Figure 6. Schematic of the sodium storage mechanism of MXene: (a) High-angle annular dark-field (HAADF) of Ti3C2Tx electrode[106]; (b) Charge-discharge curves of Ti3C2Tx at 20 mA·g−1 [107]; (c) XPS spectra of O1s during the initial two cycles[107]; (d) X-ray absorption near edge structure (XANES) spectra of Ti during the initial two cycles[107]; (e) Schematic of the mechanism for the embeddedness and detachment of Na+ [107]
K—X-ray absorption spectroscopy
图 7 Na+在MXene中原子尺度变化的示意图:(a) Na+嵌入Ti3C2Tx的原位XRD图谱[106];(b) Na+脱出Ti3C2Tx的原位XRD图谱[106];(c) V2CTx在扫描速率为1 mV·s−1时的循环伏安曲线[110];(d)在10 mA·g−1下0.1~3.0 V电压范围内的恒流充放电曲线[110];(e) V2CTx的XANES光谱[110];(f)在相应的电压分布下钒的能量变化示意图[110]
Figure 7. Schematic representation of the atomic scale changes of Na+ in MXene: (a) In situ XRD pattern of Na+ embedded in Ti3C2Tx[106]; (b) In situ XRD pattern of Na+ de-embedded from Ti3C2Tx[106]; (c) Cyclic voltammetry curves of V2CTx at a scan rate of 1 mV·s−1 [110]; (d) Constant-current in the voltage range of 0.1-3.0 V at 10 mA·g−1 charge/discharge curves[110]; (e) XANES spectra of V2CTx[110]; (f) Schematic diagram of vanadium energy changes at the corresponding voltage distribution[110]
Disc.—Discharge; Cha.—Charge; OCV—Open circuit voltage; E0.5—Voltage 0.5 V
表 1 钠离子电池正极、负极材料优缺点比较
Table 1 Comparison of advantages and disadvantages of cathode and anode materials for sodium ion batteries
表 2 MXene各制备方法形貌以及优缺点比较
Table 2 Comparisons of the morphologies, advantages and disadvantages of each preparation method of MXene
MAX MXene 制备方法 SEM图像 优点 缺点 参考文献 Ti3AlC2 Ti3C2Tx 酸刻蚀法 
制备方法简便
适用范围广危险
环境有害[54] Ti3AlC2 Ti3C2Tx 碱刻蚀法 
末端羟基丰富
环境友好危险
实验条件严苛[43] Ti3AlC2 Ti2CTx 电化学刻蚀法 
末端基团种类丰富
方法温和完全刻蚀难
易过度腐蚀[47] – Ti2CCl2 化学气相沉积法 
比表面较大
成核位点多无表面反应终止
反应温度较高[48] Ti3AlC2 Ti3C2Tx 熔盐法 
适用于氮化物MXene 难以完全刻蚀 [52] Ti3AlC2 Ti3C2Cl2 置换反应法 
获得氯端基团 制备方法难 [53] 表 3 MXene基复合材料的储钠性能比较
Table 3 Comparison of sodium storage properties of MXene matrix composites
Material First
dischargeMultiplier performance Cyclic performance Ref. Capacity/
(mA·h·g−1)Current density/
(A·g−1)Capacity/
(mA·h·g−1)Current density/
(A·g−1)Number of cycles Capacity/
(mA·h·g−1)Pure MXene 100 0.8 53.7 0.2 1000 68.3 [56] Topography and structure TMAOH/MXene 340 1 260 0.5 800 280 [62] DMSO/MXene 120 1 70 0.1 500 103 [63] 3D Ti3C2Tx 330 5 120 0.5 1000 295 [65] T-MXene@C 268.9 10 77.8 1 3000 139.5 [67] Heteroatom doping N-Ti3C2Tx 264.4 5 180.5 1 1000 284.2 [70] N-Ti3C2 MXene 338.5 2 105 0.5 3000 148.4 [69] SPM 487.8 10 213 5 2480 256.1 [71] S@Ti3C2Tx – 5 136.6 2 1000 135 [72] Si/MXene@HC 212 1 122.5 0.05 500 208.5 [73] Composite with carbon materials HC-MXene 368.5 10 166.7 0.2 1500 272.3 [74] HCS/MXene 346 0.2 239 0.5 1000 155 [75] H-MXG – 1 179.2 0.5 2000 100 [77] Composite
with metal
oxidesTiO2@Ti3C2Tx 223.9 0.06 196 0.96 5000 110 [79] NC-TiO2/MXene 205 10 100.1 2 1900 157.5 [80] Sb2O3/Ti3C2Tx 471 2 295 0.05 50 470 [82] SnO2/MXene 363.2 0.2 300 0.4 100 208.6 [84] Complexed with metal
sulfidesMXene/MoS2@CS – 5 310.6 1 800 394 [85] C-MoS2/CNTs-Ti3C2 594 2 475 1 500 442 [86] 1T-2H MoS2/Ti3C2 663.4 5 439.3 1 600 582 [87] MoS2/MXene/CKF 583.5 0.5 372 – 300 298 [88] MXene/SnS2 460 0.1 377 0.1 200 322 [89] SnS/Ti3C2Tx 348.4 1 255.9 0.5 50 320 [90] Sb2S3@m-Ti3C2Tx 669 1 255 0.1 100 156 [91] Complexed with metal selenide CoSe2@CNTs-MXene 324.6 5 347.5 2 200 400 [93] SnSe2/Ti3C2Tx – 5 192 1 445 245 [92] MoSe2/Mo2CTx/C 558 10 330.6 2 2200 238.4 [94] Ti3C2/Co0.5Ni0.5Se2 452 3 337 1 600 338 [95] Composite with organic materials Fe2O3@C@Ti3C2Tx 370 5 98 1 1000 197 [97] SWMP-5 338.5 2 105 1 5000 123.4 [69] PANI/MXene 305 5 142 2 10000 135.4 [98] Composite with nanomaterials m-Sb/C@MXene – 5 258.3 0.5 200 285.8 [99] Sb/Sb2S3@Ti3C2Tx@C 366 2 225 1 2500 250 [81] FL-Si/MXene 364 2 163 0.1 500 376 [68] Composite with other
materialsBP/Ti3C2 331.9 1 67.3 0.1 200 100 [102] PDDA-BP/Ti3C2 1780 2 461 1 2000 658 [103] BPQD/TNS 723 – – – – – [104] MXene/Na2Ti3O7@C 208 0.2 173 2 200 119 [105] Notes: TMAOH—Tetramethylammonium hydroxide; DMSO—Dimethyl sulfoxide; CS—Carbon sphere; CKF—Hollow carbonized wood fiber; PANI—Polyaniline; BP—Black phosphate; PDDA—Poly dimethyl diallyl ammonium chloride; T-MXene@C—Three-dimensional plexiform MXene; SPM—Sulfur-doped three-dimensional porous MXene; H-MXG—Graphene-assisted MXene; m-Sb—Antimony particles; FL-Si—Florid silica; BPQD/TNS—Black phosphorus composite with MXene. -
[1] YU Z Q, WANG Q, ZHU K, et al. N-doped carbon coated MoO3/MoS2 integrated MXene nanosheets with ultra-long cycle stability for sodium-ion batteries[J]. Applied Surface Science, 2024, 652(11): 159294.
[2] AN R F, NIU H K, TAN Q, et al. Enhancing sodium-ion battery performance with MXene@CoSe2/ZnSe@NC 0D2D micro-/nanostructures[J]. Journal of Energy Storage, 2024, 82(8): 110526.
[3] MING F W, LIANG H F, HUANG G, et al. MXenes for rechargeable batteries beyond the lithium-ion[J]. Advanced Materials, 2021, 33(1): 2004039. DOI: 10.1002/adma.202004039
[4] 陈淋, 李星, 王明珊, 等. 基于Ti3C2T x材料在钠离子电池中的应用进展[J]. 材料工程, 2023, 51(2): 1-14. DOI: 10.11868/j.issn.1001-4381.2022.000094 CHEN Lin, LI Xing, WANG Mingshan, et al. Application of Ti3C2T x in sodium-ion batteries[J]. Journal of Materials Engineering, 2023, 51(2): 1-14(in Chinese). DOI: 10.11868/j.issn.1001-4381.2022.000094
[5] 程颖博. 宽温域凝胶态水系钠离子电解质的研制[D]. 上海: 中国科学院大学(中国科学院上海硅酸盐研究所), 2021. CHENG Yingbo. Preparation of gel aqueous sodium ion electrolyte with wide temperature range[D]. Shanghai: Shanghai Institute of Silicate, University of Chinese Academy of Sciences, 2021(in Chinese).
[6] LIU Y C, SHEN Q Y, ZHAO X D, et al. Hierarchical engineering of porous P2-Na2/3Ni1/3Mn2/3O2 nanofibers assembled by nanoparticles enables superior sodium-ion storage cathodes[J]. Advanced Functional Materials, 2020, 30(6): 1907837. DOI: 10.1002/adfm.201907837
[7] SHEN Q Y, LIU Y C, ZHAO X D, et al. Transition-metal vacancy manufacturing and sodium-site doping enable a high-performance layered oxide cathode through cationic and anionic redox chemistry[J]. Advanced Functional Materials, 2021, 31(51): 2106923. DOI: 10.1002/adfm.202106923
[8] JIN T, LI H X, ZHU K J, et al. Polyanion-type cathode materials for sodium-ion batteries[J]. Chemical Society Reviews, 2020, 49(8): 2342-2377. DOI: 10.1039/C9CS00846B
[9] LIU X Y, CAO Y, SUN J. Defect engineering in Prussian blue analogs for high-performance sodium-ion batteries[J]. Advanced Energy Materials, 2022, 12(46): 2202532. DOI: 10.1002/aenm.202202532
[10] XIE B X, SUN B Y, GAO T Y, et al. Recent progress of Prussian blue analogues as cathode materials for nonaqueous sodium-ion batteries[J]. Coordination Chemistry Reviews, 2022, 460(11): 214478.
[11] RAJAGOPALAN R, TANG Y G, JIA C K, et al. Understanding the sodium storage mechanisms of organic electrodes in sodium ion batteries: Issues and solutions[J]. Energy & Environmental Science, 2020, 13(6): 1568-1592.
[12] CHEN X, FENG X, REN B, et al. High rate and long lifespan sodium-organic batteries using pseudocapacitive porphyrin complexes-based cathode[J]. Nano-Micro Letters, 2021, 13(1): 71.
[13] NWANYA A C, NDIPINGWI M M, IKPO C O, et al. Biomass mediated multi layered NaNi xCo1− xO2 (x=0.4) and α-Fe2O3 nanoparticles for aqueous sodium ion battery[J]. Journal of Electroanalytical Chemistry, 2020, 858(3): 113809.
[14] GU Z Y, GUO J Z, CAO J M, et al. An advanced high-entropy fluorophosphate cathode for sodium-ion batteries with increased working voltage and energy density[J]. Advanced Materials, 2022, 34(14): 2110108. DOI: 10.1002/adma.202110108
[15] KANG H L, HAN L S, CHEN S L, et al. Research progress on two-dimensional layered MXene/elastomer nanocomposites[J]. Polymers, 2022, 14(19): 4094. DOI: 10.3390/polym14194094
[16] 刘鑫龙. MXene基复合材料的制备及钠离子电池性能研究[D]. 广州: 广州大学, 2021. LIU Xinlong. Preparation of MXene based composites and study on performance of sodium-ion batteries[D]. Guangzhou: Guangzhou University, 2021(in Chinese).
[17] 张鼎, 叶子贤, 刘镇铭, 等. 钠离子电池黑磷基负极材料研究进展[J]. 储能科学与技术, 2023, 12(8): 2482-2490. ZHANG Ding, YE Zixian, LIU Zhenming, et al. Research progress of black phosphorus-based anode materials for sodium ion batteries[J]. Energy Storage Science and Technology, 2023, 12(8): 2482-2490(in Chinese).
[18] LIN F, LIU Y J, YU X Q, et al. Synchrotron X-ray analytical techniques for studying materials electrochemistry in rechargeable batteries[J]. Chemical Reviews, 2017, 117(21): 13123-13186. DOI: 10.1021/acs.chemrev.7b00007
[19] XU H, YAN Q, YAO W J, et al. Mainstream optimization strategies for cathode materials of sodium-ion batteries[J]. Small Structures, 2022, 3(4): 2100217. DOI: 10.1002/sstr.202100217
[20] 赵毅伟, 张福华, 颜顺, 等. 普鲁士蓝类钠离子电池正极材料导电性研究进展[J]. 储能科学与技术, 2024, 13(5): 1474-1486. ZHAO Yiwei, ZHANG Fuhua, YAN Shun, et al. Research progress on conductivity of positive electrode materials of Prussian blue sodium-ion batteries[J]. Energy Storage Science and Technology, 2024, 13(5): 1474-1486(in Chinese).
[21] KANG H Y, LIU Y C, CAO K Z, et al. Update on anode materials for Na-ion batteries[J]. Journal of Materials Chemistry A, 2015, 3(35): 17899-17913. DOI: 10.1039/C5TA03181H
[22] SHEN L Y, SHI S S, ROY S, et al. Recent advances and optimization strategies on the electrolytes for hard carbon and P-based sodium-ion batteries[J]. Advanced Functional Materials, 2021, 31(4): 2006066. DOI: 10.1002/adfm.202006066
[23] 丁玉寅, 祝鹏浩, 陆继鑫, 等. 钠离子电池正、负极材料研究进展[J]. 化工科技, 2022, 30(1): 57-62. DOI: 10.3969/j.issn.1008-0511.2022.01.012 DING Yuyin, ZHU Penghao, LU Jixin, et al. Research progress of anode and cathode materials for sodium ion batteries[J]. Chemical Science and Technology, 2022, 30(1): 57-62(in Chinese). DOI: 10.3969/j.issn.1008-0511.2022.01.012
[24] 刘金玉, 范凤兰, 焦连升, 等. 钠离子电池负极材料的研究现状及展望[J]. 辽宁化工, 2023, 52(2): 238-245. DOI: 10.3969/j.issn.1004-0935.2023.02.023 LIU Jinyu, FAN Fenglan, JIAO Liansheng, et al. Research status and prospect of anode materials for sodium ion batteries[J]. Liaoning Chemical Industry, 2023, 52(2): 238-245(in Chinese). DOI: 10.3969/j.issn.1004-0935.2023.02.023
[25] LIANG J L, WEI C B, HUO D X, et al. Research progress on modification and application of two-dimensional anode materials for sodium ion batteries[J]. Journal of Energy Storage, 2024, 85(12): 111044.
[26] CAI K D, JING X Q, ZHANG Y T, et al. A novel reed-leaves like aluminum-doped manganese oxide presetting sodium-ion constructed by coprecipitation method for high electrochemical performance sodium-ion battery[J]. International Journal of Energy Research, 2022, 46(10): 14570-14580. DOI: 10.1002/er.8090
[27] NAYAK P K, YANG L T, BREHM W, et al. From lithium-ion to sodium-ion batteries: Advantages, challenges, and surprises[J]. Angewandte Chemie International Edition, 2018, 57(1): 102-120. DOI: 10.1002/anie.201703772
[28] KUMAR PRAJAPATI A, BHATNAGAR A. A review on anode materials for lithium/sodium-ion batteries[J]. Journal of Energy Chemistry, 2023, 83(8): 509-540.
[29] WANG X, WANG Y Z, NI K, et al. Fullerene intercalation of MXene toward super-long-cycle sodium ion storage[J]. Advanced Functional Materials, 2024, 34(21): 2400185.
[30] VENKATESHALU S, SUBASHINI G, BHARDWAJ P, et al. Phosphorene, antimonene, silicene and siloxene based novel 2D electrode materials for supercapacitors—A brief review[J]. Journal of Energy Storage, 2022, 48(4): 104027.
[31] LI T, SHANG D W, GAO S W, et al. Two-dimensional material-based electrochemical sensors/biosensors for food safety and biomolecular detection[J]. Biosensors, 2022, 12(5): 314. DOI: 10.3390/bios12050314
[32] LI K, LIANG M Y, WANG H, et al. 3D MXene architectures for efficient energy storage and conversion[J]. Advanced Functional Materials, 2020, 30(47): 2000842. DOI: 10.1002/adfm.202000842
[33] NAGUIB M, MOCHALIN V N, BARSOUM M W, et al. 25th anniversary article: MXenes: A new family of two-dimensional materials[J]. Advanced Materials, 2014, 26(7): 992-1005. DOI: 10.1002/adma.201304138
[34] WEI Y, ZHANG P, SOOMRO R A, et al. Advances in the synthesis of 2D MXenes[J]. Advanced Materials, 2021, 33(39): e2103148. DOI: 10.1002/adma.202103148
[35] SU D, ZHANG H, ZHANG J W, et al. Design and synthesis strategy of MXenes-based anode materials for sodium-ion batteries and progress of first-principles research[J]. Molecules, 2023, 28(17): 6292. DOI: 10.3390/molecules28176292
[36] 胡睿. 二维层状碳化钛基复合材料的制备及其电化学性能研究[D]. 杭州: 浙江大学, 2020. HU Rui. Preparation and electrochemical properties of two-dimensional layered titanium carbide composites[D]. Hangzhou: Zhejiang University, 2020(in Chinese).
[37] NAGUIB M, MASHTALIR O, CARLE J, et al. Two-dimensional transition metal carbides[J]. ACS Nano, 2012, 6(2): 1322-1331. DOI: 10.1021/nn204153h
[38] LEI Y J, YAN Z C, LAI W H, et al. Tailoring MXene-based materials for sodium-ion storage: Synthesis, mechanisms, and applications[J]. Electrochemical Energy Reviews, 2020, 3(4): 766-792. DOI: 10.1007/s41918-020-00079-y
[39] MA P, FANG D L, LIU Y L, et al. MXene-based materials for electrochemical sodium-ion storage[J]. Advanced Science, 2021, 8(11): 2003185. DOI: 10.1002/advs.202003185
[40] HAN M K, SHUCK C E, RAKHMANOV R, et al. Beyond Ti3C2T x: MXenes for electromagnetic interference shielding[J]. ACS Nano, 2020, 14(4): 5008-5016. DOI: 10.1021/acsnano.0c01312
[41] MATHIS T S, MALESKI K, GOAD A, et al. Modified MAX phase synthesis for environmentally stable and highly conductive Ti3C2 MXene[J]. ACS Nano, 2021, 15(4): 6420-6429. DOI: 10.1021/acsnano.0c08357
[42] LIPATOV A, GOAD A, LOES M J, et al. High electrical conductivity and breakdown current density of individual monolayer Ti3C2Tx MXene flakes[J]. Matter, 2021, 4(4): 1413-1427.
[43] LI G N, TAN L, ZHANG Y M, et al. Highly efficiently delaminated single-layered MXene nanosheets with large lateral size[J]. Langmuir, 2017, 33(36): 9000-9006. DOI: 10.1021/acs.langmuir.7b01339
[44] ZHANG B, ZHU J F, SHI P, et al. Fluoride-free synthesis and microstructure evolution of novel two-dimensional Ti3C2(OH)2 nanoribbons as high-performance anode materials for lithium-ion batteries[J]. Ceramics International, 2019, 45(7): 8395-8405.
[45] LI T F, YAO L L, LIU Q L, et al. Fluorine-free synthesis of high-purity Ti3C2T x (T=OH, O) via alkali treatment[J]. Angewandte Chemie International Edition, 2018, 57(21): 6115-6119. DOI: 10.1002/anie.201800887
[46] ZHANG L, SONG W W, LIU H S, et al. Influencing factors on synthesis and properties of MXene: A review[J]. Processes, 2022, 10(9): 1744. DOI: 10.3390/pr10091744
[47] YANG S, ZHANG P P, WANG F X, et al. Fluoride-free synthesis of two-dimensional titanium carbide (MXene) using a binary aqueous system[J]. Angewandte Chemie International Edition, 2018, 57(47): 15491-15495. DOI: 10.1002/anie.201809662
[48] WANG D, ZHOU C K, FILATOV A S, et al. Direct synthesis and chemical vapor deposition of 2D carbide and nitride MXenes[J]. Science, 2023, 379(6638): 1242-1247. DOI: 10.1126/science.add9204
[49] XU C, WANG L B, LIU Z B, et al. Large-area high-quality 2D ultrathin Mo2C superconducting crystals[J]. Nature Materials, 2015, 14(11): 1135-1141. DOI: 10.1038/nmat4374
[50] KAMYSBAYEV V, FILATOV A S, HU H C, et al. Covalent surface modifications and superconductivity of two-dimensional metal carbide MXenes[J]. Science, 2020, 369(6506): 979-983. DOI: 10.1126/science.aba8311
[51] LI X L, LI M, YANG Q, et al. Vertically aligned Sn4+ preintercalated Ti2CT x MXene sphere with enhanced Zn ion transportation and superior cycle lifespan[J]. Advanced Energy Materials, 2020, 10(35): 2001394. DOI: 10.1002/aenm.202001394
[52] LI S Q, ZHU J H, LU W, et al. Iron nanoparticles surface decorated MXene via molten salts etching as selenium host for ultrafast sodium ion storage[J]. Journal of Colloid and Interface Science, 2024, 663(11): 801-809.
[53] LI M, LU J, LUO K, et al. Element replacement approach by reaction with lewis acidic molten salts to synthesize nanolaminated MAX phases and MXenes[J]. Journal of the American Chemical Society, 2019, 141(11): 4730-4737. DOI: 10.1021/jacs.9b00574
[54] WANG T, YAO K, HUA Y B, et al. Rational design of MXene-MoS2 heterostructure with rapid ion transport rate as an advanced anode for sodium-ion batteries[J]. Chemical Engineering Journal, 2023, 457(7): 141363.
[55] CAO J M, MA M Y, LIU H H, et al. Interfacial-confined isochronous conversion to biphasic selenide heterostructure with enhanced adsorption behaviors for robust high-rate Na-ion storage[J]. Small, 2024, 20(11): 2311024. DOI: 10.1002/smll.202311024
[56] XU E Z, ZHANG Y, WANG H, et al. Ultrafast kinetics net electrode assembled via MoSe2/MXene heterojunction for high-performance sodium-ion batteries[J]. Chemical Engineering Journal, 2020, 385(7): 123839.
[57] XIONG D B, LI X F, BAI Z M, et al. Recent advances in layered Ti3C2T x MXene for electrochemical energy storage[J]. Small, 2018, 14(17): 1703419. DOI: 10.1002/smll.201703419
[58] ZHANG X Y, WANG J, JIANG Y T, et al. A 3D crinkled MXene/TiO2 heterostructure with interfacial coupling for ultra-fast and reversible potassium storage[J]. Journal of Materials Chemistry A, 2024, 12(13): 7598-7604. DOI: 10.1039/D3TA07682B
[59] ZHENG X Z, YUAN M W, GUO D H, et al. Theoretical design and structural modulation of a surface-functionalized Ti3C2T x MXene-based heterojunction electrocatalyst for a Li-oxygen battery[J]. ACS Nano, 2022, 16(3): 4487-4499. DOI: 10.1021/acsnano.1c10890
[60] OUYANG Y X, QIU L, BAI Y Y, et al. Synergistical thermal modulation function of 2D Ti3C2 MXene composite nanosheets via interfacial structure modification[J]. iScience, 2022, 25(8): 104825. DOI: 10.1016/j.isci.2022.104825
[61] ZHANG S L, HAN W Q. Recent advances in MXenes and their composites in lithium/sodium batteries from the viewpoints of components and interlayer engineering[J]. Physical Chemistry Chemical Physics, 2020, 22(29): 16482-16526. DOI: 10.1039/D0CP02275F
[62] SONG X L, WANG H, JIN S M, et al. Oligolayered Ti3C2T x MXene towards high performance lithium/sodium storage[J]. Nano Research, 2020, 13(6): 1659-1667. DOI: 10.1007/s12274-020-2789-6
[63] LYU G X, WANG J, SHI Z Q, et al. Intercalation and delamination of two-dimensional MXene (Ti3C2T x) and application in sodium-ion batteries[J]. Materials Letters, 2018, 219(10): 45-50.
[64] SHI C J, LIU Z J, TIAN Z, et al. Regulated layer spacing and functional surface group of MXene film by hexamethylenetetramine for high-performance supercapacitors[J]. Applied Surface Science, 2022, 596(26): 153632.
[65] ZHAO M Q, XIE X Q, REN C E, et al. Hollow MXene spheres and 3D macroporous MXene frameworks for Na-ion storage[J]. Advanced Materials, 2017, 29(37): 1702410. DOI: 10.1002/adma.201702410
[66] XIU L Y, WANG Z Y, YU M Z, et al. Aggregation-resistant 3D MXene-based architecture as efficient bifunctional electrocatalyst for overall water splitting[J]. ACS Nano, 2018, 12(8): 8017-8028. DOI: 10.1021/acsnano.8b02849
[67] ZHANG P, SOOMRO R A, GUAN Z R X, et al. 3D carbon-coated MXene architectures with high and ultrafast lithium/sodium-ion storage[J]. Energy Storage Materials, 2020, 29(6): 163-171.
[68] WANG H Q, ZHAO Y X, GOU L, et al. Rational construction of densely packed Si/MXene composite microspheres enables favorable sodium storage[J]. Rare Metals, 2022, 41(5): 1626-1636. DOI: 10.1007/s12598-021-01895-x
[69] JIN X, ZHANG W S, LIU S Y, et al. Highly stable Ti3C2T x MXene-based sandwich-like structure via interfacial self-assembly of nitrogen-rich polymer network for superior sodium-ion storage performance[J]. Chemical Engineering Journal, 2023, 451(1): 138763.
[70] FAN Z D, WEI C H, YU L H, et al. 3D printing of porous nitrogen-doped Ti3C2 MXene scaffolds for high-performance sodium-ion hybrid capacitors[J]. ACS Nano, 2020, 14(1): 867-876. DOI: 10.1021/acsnano.9b08030
[71] ZHANG L L, CHEN Y G, YU L H, et al. Efficient sulfur atom-doped three-dimensional porous MXene-assisted sodium ion batteries[J]. Dalton Transactions, 2024, 53(15): 6583-6591. DOI: 10.1039/D3DT04312F
[72] SUN S J, XIE Z L, YAN Y R, et al. Hybrid energy storage mechanisms for sulfur-decorated Ti3C2 MXene anode material for high-rate and long-life sodium-ion batteries[J]. Chemical Engineering Journal, 2019, 366(12): 460-467.
[73] GOU L, JING W F, LI Y, et al. Lattice-coupled Si/MXene confined by hard carbon for fast sodium-ion conduction[J]. ACS Applied Energy Materials, 2021, 4(7): 7268-7277. DOI: 10.1021/acsaem.1c01396
[74] SUN N, ZHU Q Z, ANASORI B, et al. MXene-bonded flexible hard carbon film as anode for stable Na/K-ion storage[J]. Advanced Functional Materials, 2019, 29(51): 1906282. DOI: 10.1002/adfm.201906282
[75] CAO H L, YANG L T, ZHAO M, et al. A flexible hard carbon microsphere/MXene film as a high-performance anode for sodium-ion storage[J]. New Carbon Materials, 2022, 37(6): 1154-1160. DOI: 10.1016/S1872-5805(22)60616-4
[76] CHEN T D, WANG J Q, WU X Z, et al. Ethanediamine induced self-assembly of long-range ordered GO/MXene composite aerogel and its piezoresistive sensing performances[J]. Applied Surface Science, 2021, 566(32): 150719.
[77] WEI C, CAO H L, DENG W, et al. Graphene-mediated dense integration of Ti3C2T x MXene monoliths for compact energy storage: Balancing kinetics and packing density[J]. Applied Surface Science, 2022, 604(34): 154565.
[78] ASLAM M K, ALGARNI T S, JAVED M S, et al. 2D MXene materials for sodium ion batteries: A review on energy storage[J]. Journal of Energy Storage, 2021, 37(5): 102478.
[79] GUO X, ZHANG J Q, SONG J J, et al. MXene encapsulated titanium oxide nanospheres for ultra-stable and fast sodium storage[J]. Energy Storage Materials, 2018, 14(5): 306-313.
[80] WANG F, MA X D, ZOU P J, et al. Nitrogen-doped carbon decorated TiO2/Ti3C2T x MXene composites as anode material for high-performance sodium-ion batteries[J]. Surface and Coatings Technology, 2021, 422(18): 127568.
[81] WANG D, MA Q, HE H, et al. Double-confined nanoheterostructure Sb/Sb2S3@Ti3C2T x@C toward ultra-stable Li-/Na-ion batteries[J]. Rare Metals, 2024, 43(1): 2067-2079.
[82] GUO X, XIE X Q, CHOI S H, et al. Sb2O3/MXene(Ti3C2T x) hybrid anode materials with enhanced performance for sodium-ion batteries[J]. Journal of Materials Chemistry A, 2017, 5(24): 12445-12452. DOI: 10.1039/C7TA02689G
[83] LIAO A, PAN Y, LEI W X, et al. Carbon aerogel/SnO2 as an advanced anode for sodium-ion batteries[J]. Functional Materials Letters, 2020, 13(6): 2051026. DOI: 10.1142/S1793604720510261
[84] CHEN Y Z, SUN Y D, GENG M, et al. SnO2/MXene nanoparticles as a superior high-rate and cycling-stable anode for sodium ion batteries[J]. Materials Letters, 2021, 304(23): 130704.
[85] YUAN Z Y, CAO J M, VALERII S, et al. MXene-bonded hollow MoS2/carbon sphere strategy for high-performance flexible sodium ion storage[J]. Chemical Engineering Journal, 2022, 430(4): 132755.
[86] HE F Y, TANG C, LIU Y D, et al. Carbon-coated MoS2 nanosheets@CNTs-Ti3C2 MXene quaternary composite with the superior rate performance for sodium-ion batteries[J]. Journal of Materials Science & Technology, 2022, 100(5): 101-109.
[87] WEN B J, KONG N Z, HUANG M, et al. 1T-2H MoS2/Ti3C2 MXene heterostructure with high-rate and high-capacity performance for sodium-ion batteries[J]. Energy & Fuels, 2022, 36(18): 11234-11244.
[88] ZHANG X Y, SHI H T, LIU L S, et al. Construction of MoS2/Mxene heterostructure on stress-modulated kapok fiber for high-rate sodium-ion batteries[J]. Journal of Colloid and Interface Science, 2022, 605(1): 472-482.
[89] WU Y T, NIE P, WU L Y, et al. 2D MXene/SnS2 composites as high-performance anodes for sodium ion batteries[J]. Chemical Engineering Journal, 2018, 334(4): 932-938.
[90] ZHANG Y Q, GUO B S, HU L Y, et al. Synthesis of SnS nanoparticle-modified MXene (Ti3C2T x) composites for enhanced sodium storage[J]. Journal of Alloys and Compounds, 2018, 732(3): 448-453.
[91] ZHANG H N, REN M X, JIANG W, et al. Hierarchical Sb2S3@m-Ti3C2T x composite anode with enhanced Na-ion storage properties[J]. Journal of Alloys and Compounds, 2021, 887(38): 161318.
[92] FAN T X, WU Y K, LI J, et al. Sheet-to-layer structure of SnSe2/MXene composite materials for advanced sodium ion battery anodes[J]. New Journal of Chemistry, 2021, 45(4): 1944-1952. DOI: 10.1039/D0NJ04788K
[93] XU E Z, LI P C, QUAN J J, et al. Dimensional gradient structure of CoSe2@CNTs-MXene anode assisted by ether for high-capacity, stable sodium storage[J]. Nano-Micro Letters, 2021, 13: 40. DOI: 10.1007/s40820-020-00562-7
[94] TAN Y B, YI M J, ZHU Z Y, et al. Carbon-coated MoSe2/MXene heterostructures as active materials for high-performance Na+ batteries[J]. Materials Today Communications, 2022, 31(2): 103740.
[95] DENG Q X, WANG M Q, LIU X L, et al. Ultrathin cobalt nickel selenides (Co0.5Ni0.5Se2) nanosheet arrays anchoring on Ti3C2 MXene for high-performance Na+/K+ batteries[J]. Journal of Colloid and Interface Science, 2022, 626(22): 700-709.
[96] JO Y M, JO Y K, LEE J H, et al. MOF-based chemiresistive gas sensors: Toward new functionalities[J]. Advanced Materials, 2023, 35(43): 2206842. DOI: 10.1002/adma.202206842
[97] LI Z C, LONG Z W, DAI H, et al. Ti3C2T x MXene@metal-organic frameworks-derived bead-like carbon nanofibers heterostructure aerogel for enhanced performance lithium/sodium storage[J]. Journal of Power Sources, 2024, 606(18): 234586.
[98] WANG X, WANG J, QIN J W, et al. Surface charge engineering for covalently assembling three-dimensional MXene network for all-climate sodium ion batteries[J]. ACS Applied Materials & Interfaces, 2020, 12(35): 39181-39194.
[99] LIANG Y W, WANG Z S, XU Z Y, et al. Highly-confined, micro-Sb/C@MXene 3D architectures with strengthened interfacial bonding for high volumetric sodium-ion storage[J]. Applied Surface Science, 2024, 651(10): 159234.
[100] MA L, WANG Y D, ZHU Z M. Interface engineering of MXene containing phosphorus towards for enhancing the flame retardance and toughness of epoxy resins[J]. Progress in Organic Coatings, 2024, 191(6): 108414.
[101] LIANG Y, WANG Z, XU Z, et al. Highly-confined, micro-Sb/C@MXene 3D architectures with strengthened interfacial bonding for high volumetric sodium-ion storage[J]. Applied Surface Science, 2024, 651: 159234. DOI: 10.1016/j.apsusc.2023.159234
[102] LI H, LIU A M, REN X F, et al. A black phosphorus/Ti3C2 MXene nanocomposite for sodium-ion batteries: A combined experimental and theoretical study[J]. Nanoscale, 2019, 11(42): 19862-19869. DOI: 10.1039/C9NR04790E
[103] ZHAO R Z, QIAN Z, LIU Z Y, et al. Molecular-level heterostructures assembled from layered black phosphorene and Ti3C2 MXene as superior anodes for high-performance sodium ion batteries[J]. Nano Energy, 2019, 65(11): 104037.
[104] MENG R J, HUANG J M, FENG Y T, et al. Black phosphorus quantum dot/Ti3C2 MXene nanosheet composites for efficient electrochemical lithium/sodium-ion storage[J]. Advanced Energy Materials, 2018, 8(26): 1801514. DOI: 10.1002/aenm.201801514
[105] ZHONG W, TAO M L, TANG W W, et al. MXene-derivative pompon-like Na2Ti3O7@C anode material for advanced sodium ion batteries[J]. Chemical Engineering Journal, 2019, 378(24): 122209.
[106] WANG X F, SHEN X, GAO Y R, et al. Atomic-scale recognition of surface structure and intercalation mechanism of Ti3C2X[J]. Journal of the American Chemical Society, 2015, 137(7): 2715-2721. DOI: 10.1021/ja512820k
[107] KAJIYAMA S, SZABOVA L, SODEYAMA K, et al. Sodium-ion intercalation mechanism in MXene nanosheets[J]. ACS Nano, 2016, 10(3): 3334-3341. DOI: 10.1021/acsnano.5b06958
[108] LUKATSKAYA M R, BAK S M, YU X Q, et al. Probing the mechanism of high capacitance in 2D titanium carbide using in situ X-ray absorption spectroscopy[J]. Advanced Energy Materials, 2015, 5(15): 1500589. DOI: 10.1002/aenm.201500589
[109] FAN K Q, WEI C L, FENG J K. Recent progress of MXene-based materials as anodes in sodium-ion batteries[J]. Journal of Electronic Materials, 2023, 52(2): 847-863. DOI: 10.1007/s11664-022-10142-7
[110] BAK S M, QIAO R M, YANG W L, et al. Na-ion intercalation and charge storage mechanism in 2D vanadium carbide[J]. Advanced Energy Materials, 2017, 7(20): 1700959. DOI: 10.1002/aenm.201700959
-
目的
钠离子的离子半径和标准电极电势高,阻碍了其实际应用。因此,寻找合适的负极材料是钠离子电池应用的关键。二维层状结构材料MXene具有高导电性和大比表面积,在储能方面具有明显的优势,可以为Na提供更多的附着点,且化学稳定性好、可塑性强,是钠离子电池负极材料的优良选择。近年来,MXene材料在钠离子电池中应用广泛,并取得了重大进展。本文综述了MXene材料在钠离子电池中的研究进展,总结了MXene的制备方法,重点介绍了MXene材料的优化改性方法以及储钠机理,同时对应用前景展开了分析。
方法通过知网,web of science以及学校图书馆等文献检索工具的数据库中检索,选择了近年来质量优异的文章进行综述。通过文章发表年份、复合材料类型及制备方法对文章进行梳理分类,确定了文章的整体脉络和关键词,即MXene、钠离子电池、负极材料和储钠机理。
结果通过对上述文章的梳理分析和总结,将文章主要划分为三个方面:(1)二维层状MXene材料:MXene是由相应的MAX相选择性刻蚀A层元素,破坏M-A键保留M-X键,获得二维层状的MXene。MXene材料常见的制备法包括酸刻蚀法、碱刻蚀法、电化学刻蚀法和化学气相沉积法等;(2)MXene基钠离子电池负极材料:MXene材料易氧化,且片层易再次堆叠,因此需要在保持原有优势的基础上强化结构稳定性、提高比容量。优化改性方法主要包括调控形貌结构、杂原子掺杂、碳材料或金属氧族化物等多种材料复合等;(3)MXene的储钠机理:MXene对Na的存储主要是通过Na在表层的嵌入和脱出,随后进入MXene主体,表现为双钠层的电容式电荷存储机理。最后,本文提出了MXene材料在钠离子电池中面对的挑战和未来的研究方向,期望为MXene材料在钠离子电池中的工业化应用提供一定的参考价值。
结论本文综述了MXene在钠离子电池负极材料中的研究进展,介绍了近年来MXene材料的制备方法,包括含氟刻蚀法和不含氟制备法,总结了MXene材料的优化和改进措施,包括形貌调控、杂原子掺杂以及材料复合等,同时详细阐释了MXene的储钠机理,即双钠层的电容式电荷存储机理。MXene面临着制备方法对环境不友好、材料易氧化、片层易再次塌陷等挑战。针对上述问题,本文从以下四个方面进行了对未来研究方向的展望:(1)改进合成
方法改进MXene材料的制备方法和制备过程,发展环境友好的刻蚀方法,避免高浓度氟离子对MXene侵害;(2)提高稳定性:进一步设计MXene的结构和形貌,同时控制MXene的储存条件或封装MXene;(3)提高实际容量:进一步发展合成MXene基复合材料;(4)调控表面官能团:通过化学修饰或其他手段,精确控制MXene表面的官能团类型。




 下载:
下载: