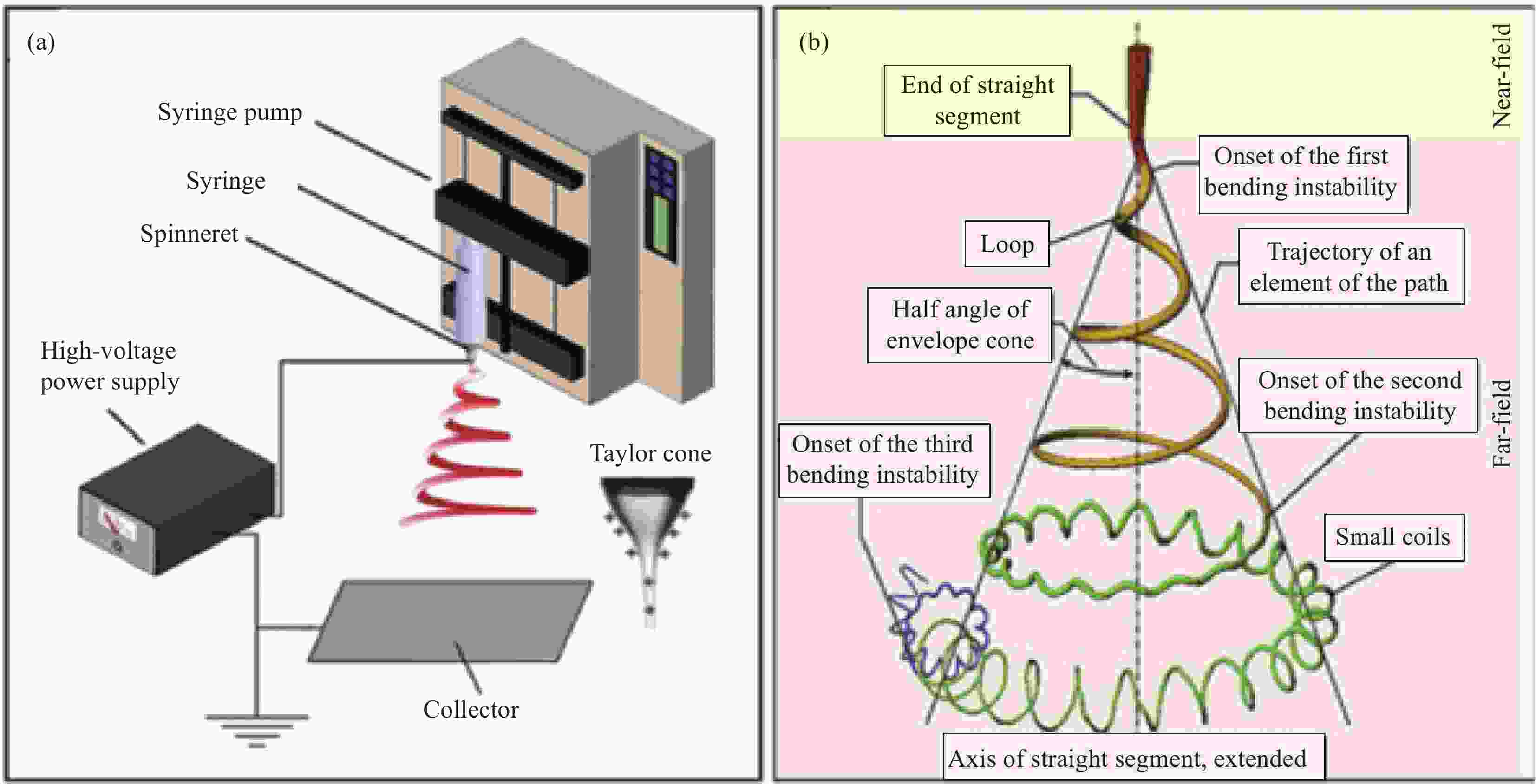
Preparation of surface morphology of electrospun nanofibers and their biomedical applications
-
摘要: 通过模仿天然细胞外基质的成分和结构特性对材料表面形貌进行设计与调控,可获得新型仿生材料,并广泛应用于生物医学领域。其中,静电纺纳米纤维通过调控孔隙率、比表面积及微纳米结构等,可以模拟天然细胞外基质的结构,实现其生物功能。本文将提供不同表面形貌电纺纤维的概述。首先介绍静电纺丝的原理、设备和参数,然后讨论电纺纤维的4种表面形貌:纳米孔、串晶、沟槽和皱缩结构的制备原理、方法及在生物医学领域的应用,并对该领域相关研究和研究现状进行评价。Abstract: By emulating the composition and structural features of the natural extracellular matrix, the design and regulation of material surface morphologies can yield novel biomimetic materials that find extensive applications in the field of biomedical science. Among these, electrospun nanofibers, by manipulating parameters like porosity, surface area, and micro/nanoarchitecture, can replicate the structure of the native extracellular matrix, thus achieving corresponding biological functionalities. This article offers an overview of various surface morphologies of electrospun fibers. It begins by outlining the principles, equipment, and parameters of electrospinning, and subsequently delves into the preparation principles and methods of four types of surface morphologies of electrospun fibers: Nanopores, crystal arrays, grooves, and wrinkled structures. It explores their applications in the biomedical field, evaluates pertinent research, and provides an up-to-date assessment of the most relevant studies in this domain.
-
Key words:
- electrospun /
- fiber /
- material surface morphology /
- biomedical applications /
- tissue engineering
-
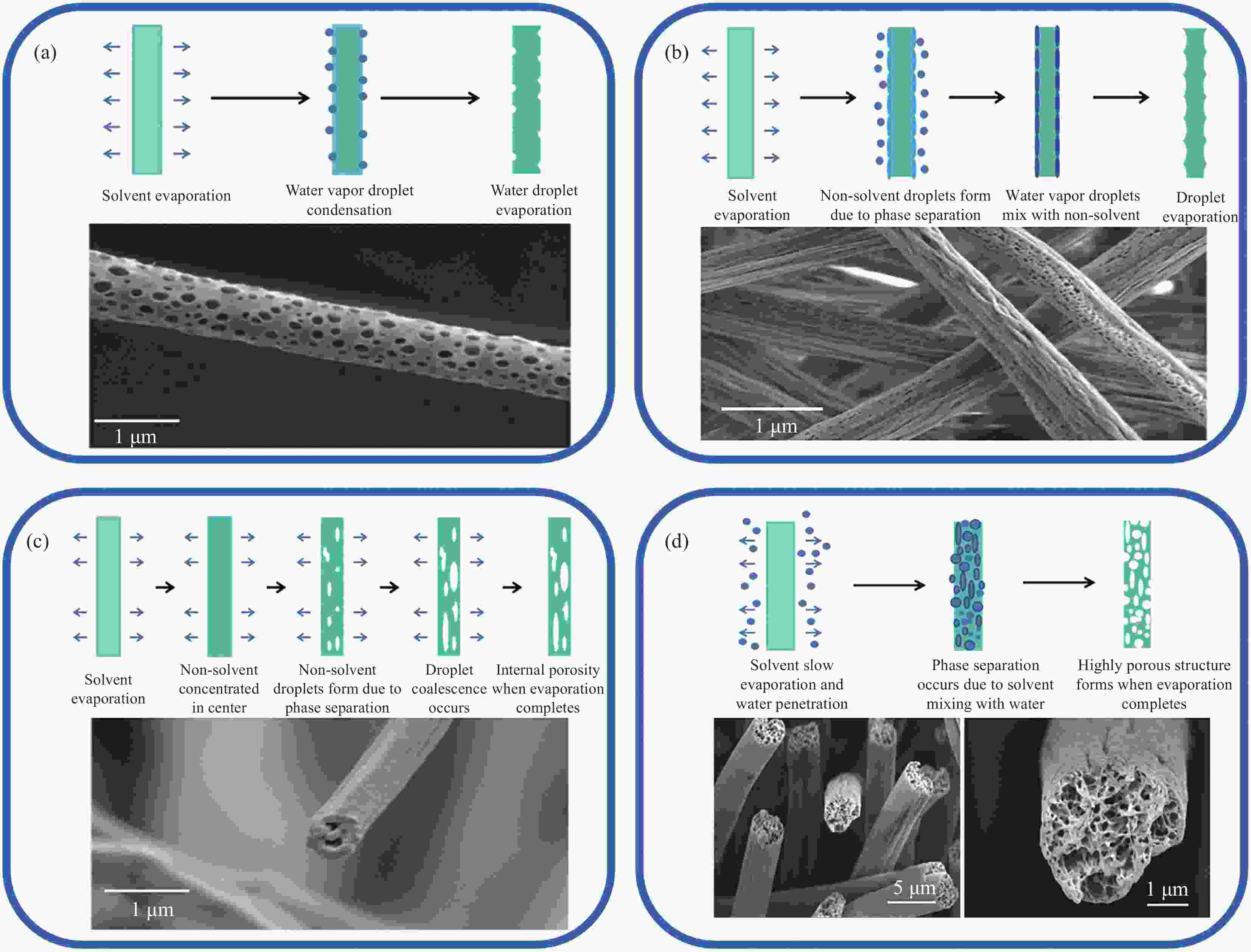
图 3 (a) Breath figure机制引起的表面孔隙形成示意图和 PLA纤维SEM图像[37];(b)非溶剂诱导相分离(NIPS)引起表面孔的示意图[37]和聚氰基丙烯酸乙酯(PECA)-聚己内酯(PCL)纤维SEM图像[42];(c) NIPS引起内部孔的示意图和 PLA纤维SEM图像[37];(d)蒸汽诱导相分离(VIPS)引起的孔隙示意图和聚苯乙烯(PS)纤维SEM图像[46]
Figure 3. (a) Schematic diagram of surface pore formation due to Breath figure mechanism and SEM image of PLA fibers[37]; (b) Schematic diagram of surface pores due to nonsolventinduce phase separation (NIPS)[37] and SEM image of poly(ethyl cyanoacrylate) (PECA)-polycaprolactone (PCL) fibers[42]; (c) Schematic diagram of internal pores due to NIPS and SEM image of PLA fibers[37]; (d) Schematic diagram of porosity due to vapour induced phase separation (VIPS) and SEM images of polystyrene (PS) fibers[46]
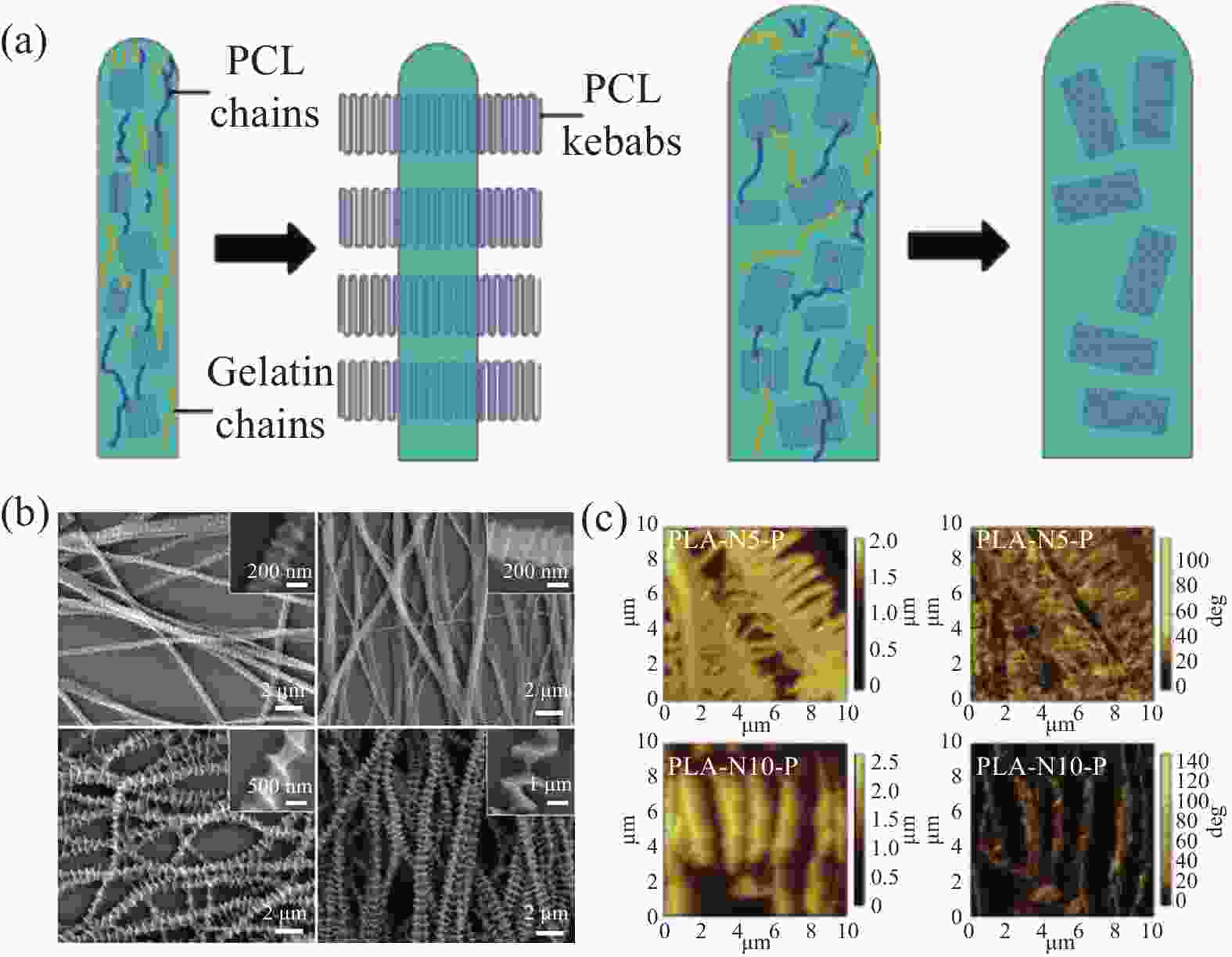
图 4 (a)直径和纤维取向影响串晶方向的示意图[50];(b) PCL纳米纤维在1%PCL/乙酸戊酯中孵育5、15、30和60 min的SEM图像[52];(c) 聚乳酸(PLA)-N5-P和 PLA-N10-P 样品的表面形貌和相图[53]
PLA-N5-P, PLA-N10-P—Poly(lactic acid) nanofibers modified in 0.1 mol/L sodium hydroxide solution for 5 min and 10 min, respectively, then immersed in aqueous polycaprolactone acetate solution for 30 min
Figure 4. (a) Schematic diagram of diameter and fiber orientation affect the orientation of the clusters[50]; (b) SEM images of PCL nanofibers incubated in 1%PCL/pentyl acetate for 5, 15, 30 and 60 min[52]; (c) Surface morphology and phase diagrams of PLA-N5-P and PLA-N10-P samples[53]
图 5 (a) 孔洞伸长法形成沟槽纤维机制示意图[66]和15wt%左旋聚乳酸 (PLLA) (DCM/N, N-二甲基甲酰胺(DMF)体积比为3∶1)沟槽纤维SEM图像[63];(b) 皱缩伸长法形成沟槽纤维机制示意图[66]和30wt%醋酸丁酸纤维素 (CAB) (丙酮/二甲基乙酰胺(DMAc)体积比为2∶1)沟槽纤维SEM图像[61];(c) 塌陷喷射伸长法形成沟槽纤维机制示意图[66]和15wt%PS (DCM/DMF体积比为1∶1)沟槽纤维SEM图像;(d) 选择性溶解法形成沟槽纤维机制示意图和PCL光滑纤维(PCL/聚乙烯吡咯烷酮(PVP)体积比为1∶2、1∶1)沟槽纤维SEM图像[64]
Figure 5. (a) Schematic diagram of groove fiber formation mechanism by hole elongation[66] and 15wt% PLLA (Volume ratio of DCM/N, N-dimethylformamide (DMF) is 3∶1) groove fiber electron microscopy[63]; (b) Schematic diagram of groove fiber formation mechanism by crinkle elongation[66] and 30wt% CAB (Volume ratio of acetone/dimethylacetamide (DMAc) is 2∶1) groove fiber electron microscopy[61]; (c) Schematic diagram of groove fiber formation mechanism by collapse injection elongation[66] and 15wt% PS (Volume ratio of DCM/DMF is 1∶1) groove fiber electron micrographs; (d) Schematic diagram of groove fiber formation mechanism by selective dissolution and PCL smooth fiber (Volume ratio of PCL/PVP is 1∶2, 1∶1) groove fiber electron micrographs[64]
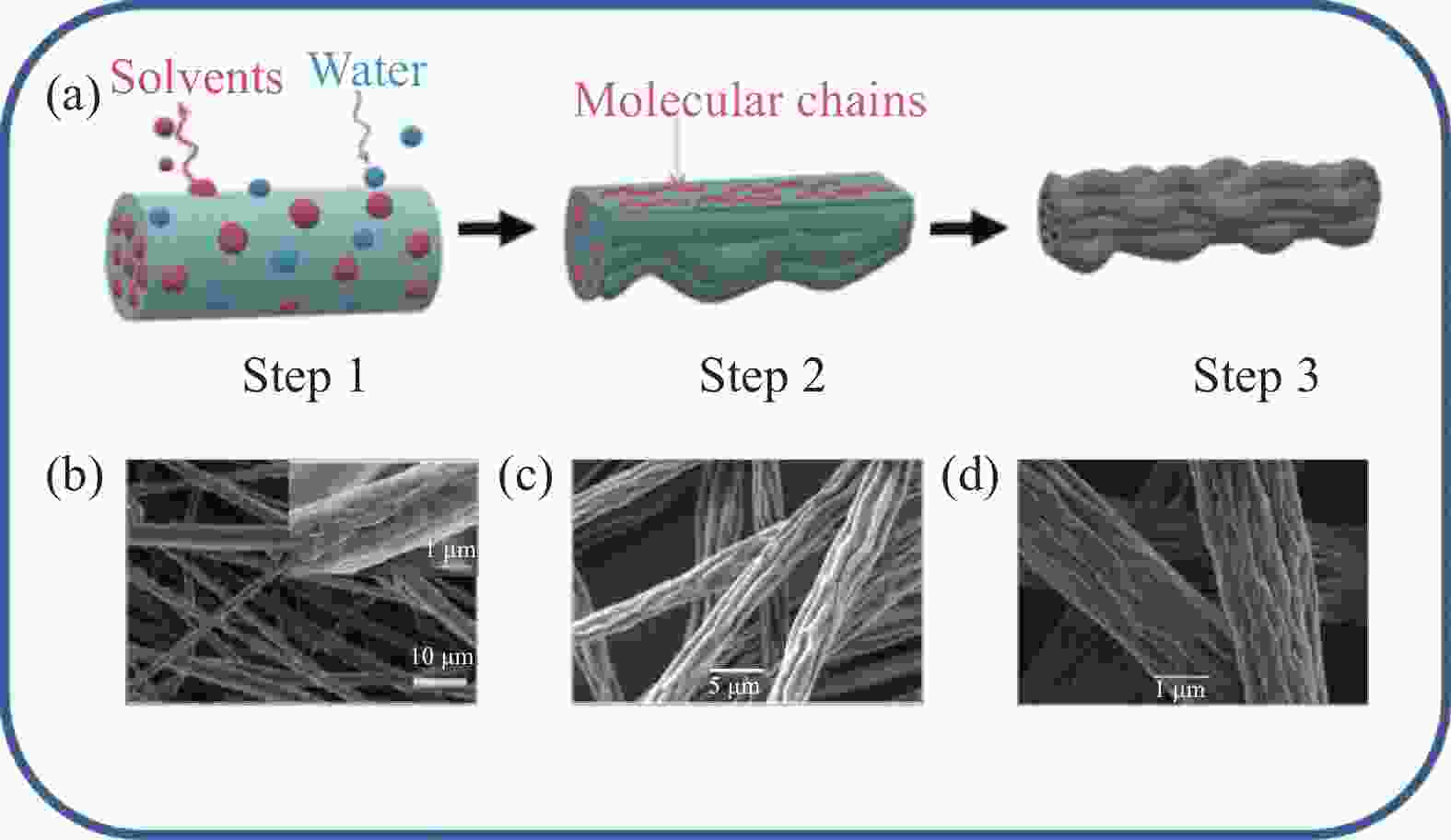
图 6 (a)皱缩纤维形成机制示意图[17];(b)分子量为180×103的聚偏氟乙烯(PVDF)皱缩纤维电镜图[17];(c) 30wt%PS/DMF溶液在相对湿度为15%时的静电纺纳米纤维电镜图[72];(d) 20%聚甲基丙烯酸甲酯(PMMA)/四氢呋喃(THF)溶液在电压为12 kV时的静电纺纳米纤维[73]
Figure 6. (a) Schematic diagram of crinkle fiber formation mechanism[17]; (b) Electron micrograph of polyvinylidene fluoride (PVDF) crinkle fiber with molecular weight of 180×103[17]; (c) Electron micrograph of electrostatically spun nanofibers with 30wt%PS/DMF solution at 15% relative humidity[72]; (d) Electrostatically spun nanofibers with 20% polymethyl methacrylate (PMMA)/tetrahydrofuran (THF) solution at a voltage of 12 kV[73]
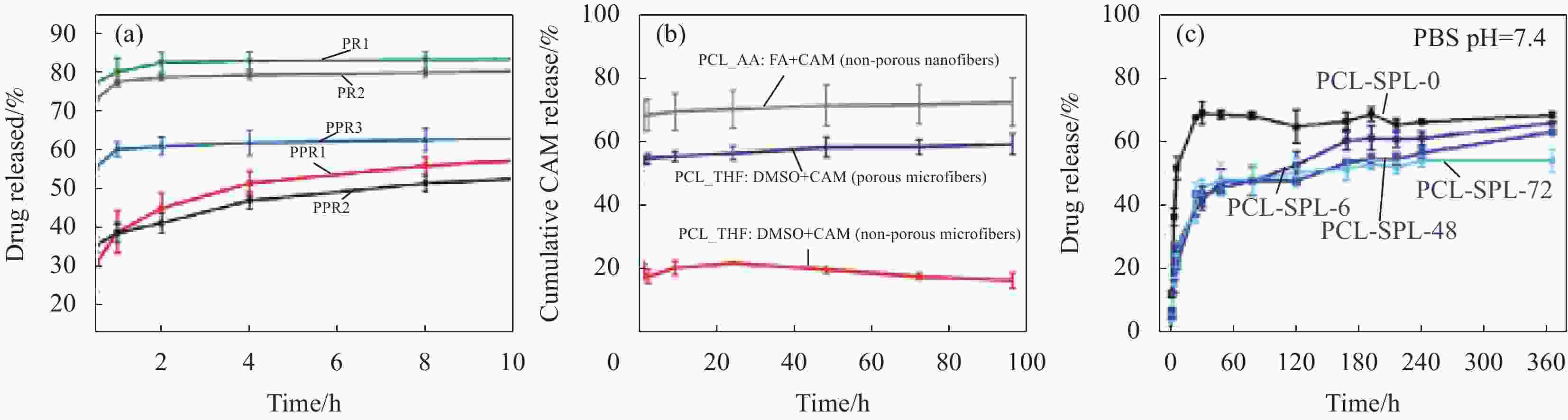
图 8 (a)同轴多孔纳米纤维PPR1、PPR2和PPR3及单轴纳米纤维PR1、PR2的药物释放曲线[82];(b)在37℃ 磷酸盐缓冲液(PBS)中多孔微纤维、非多孔微纤维和非多孔纳米纤维的累积药物释放特征[81];(c)负载小分子螺内酯(SPL)的PCL纳米纤维的体外药物释放情况[84]
PPR1, PPR2 and PPR3—Roxithromycin loaded PCL/PLA nanofibers with various solvent types; PR1, PR2—Roxithromycin loaded PCL nanofibers with different solvents; PCL-SPL-0, PCL-SPL-6, PCL-SPL-48 and PCL-SPL-72—Annealing time of 0, 6, 48, 72 h, respectively; CAM—Chloramphenicol; PCL—Polycaprolactone; THF—Tetrahydrofurane; DMSO—Dimethylsulfoxide; AA—Acetic acid; FA—Formic acid
Figure 8. (a) Drug release profiles of porous coaxial nanofibers PPR1, PPR2 and PPR3 and uniaxial nanofibers PR1 and PR2[82]; (b) Cumulative drug release characteristics of porous microfibers, non-porous microfibers and non-porous nanofibers in phosphate buffer (PBS) at 37℃[81]; (c) In vitro drug release from polycaprolactone (PCL) nanofibers loaded with small-molecule spironolactone (SPL)[84]
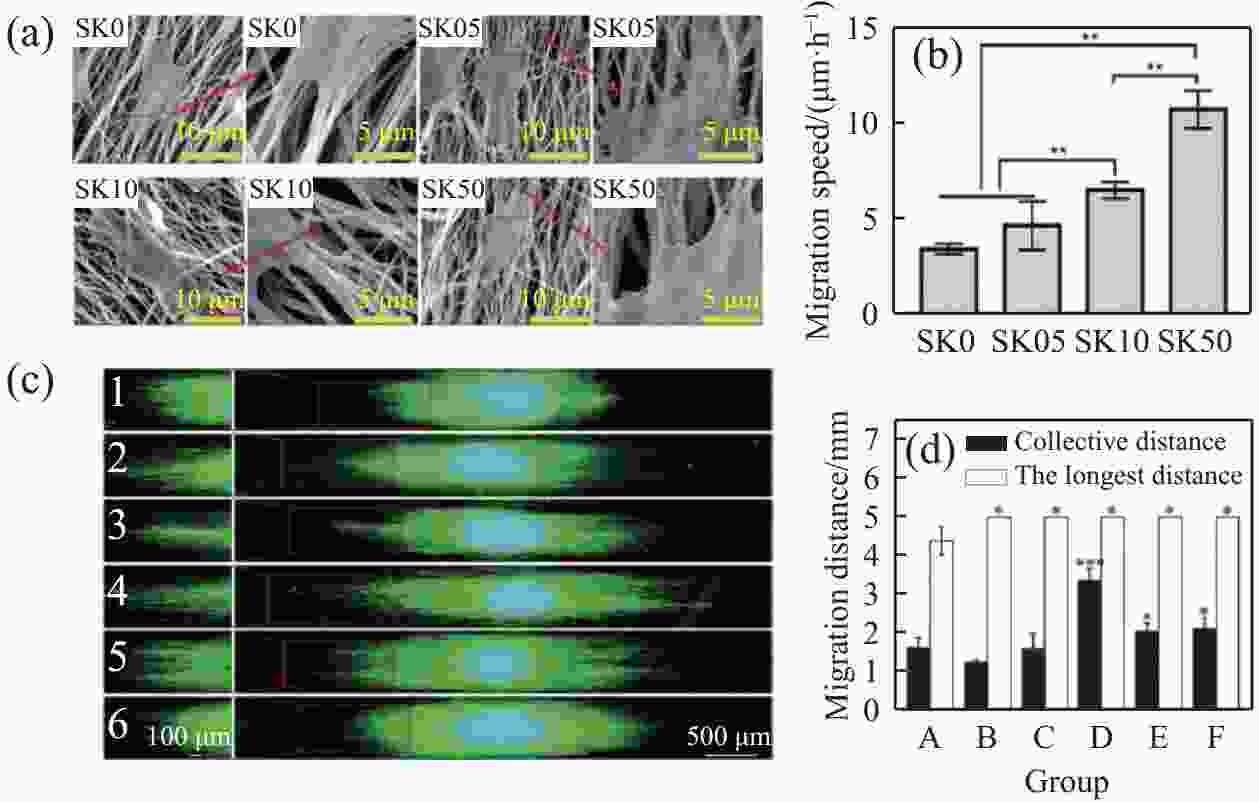
图 9 (a)串晶结构纤维上人脐静脉内皮细胞(HUVECs) 的细胞形貌;(b)单个细胞的迁移速度[92];(c)鸡胚背根神经节小体(DRG) 的神经突生长;(d)施旺细胞在不同组的迁移距离:(A)光滑PCL微纤维;(B~E)具有纳米级沟槽的微纤维;(F)具有纳米孔的微纤维[64]
SK0—Samples of PCL nanofibers treated with pure acrtic acid/deionized water; SK05, SK10 and SK50—Sample with PCL concentrations set to 0.05wt%, 0.10wt%, 0.50wt%; *—Significance level p<0.05; **—p<0.01; ***—p<0.001
Figure 9. (a) Cellular morphology of human umbilic vein endothelial cells (HUVECs) on string-crystal structured fibers; (b) Migration velocity of individual cells[92]; (c) Neurite growth of chick embryonic dorsal root ganglion (DRG) vesicles; (d) Migration distance of Schwann cells in different groups: (A) Smooth PCL microfibers; (B-E) Microfibers with nanoscale grooves; (F) Microfibers with nanopores[64]
表 1 静电纺纳米纤维的表面形貌、材料、优缺点和应用总结
Table 1. Summary of surface morphology, materials, advantages and disadvantages and applications of electrospun nanofibers
Material surface morphology Material Application Advantage and disadvantage Ref. Nanopores PLA Oil/water separation, antibacterial Advantages: The process is facile cost-effective, increase in surface area and porosity;
Disadvantages: Limited to specific polymers and solvent, uncontrollable nanopores size[34, 37, 97] PLLA Tissue engineering [35] PDLLA Tissue engineering [36] PVDF Electrodes materials [40] SA/PEG/PLA Drug release, wound-healing [41] PLA Tissue engineering, antibacterial [44, 96] Ipp Tissue engineering [48] PECA/PCL Environmental and energy related applications [49] PS Oil/water separation [78] PCL Drug release, tissue engineering [81] PCL/PLA Drug release, antibacterial [82] PLGA Tissue engineering [87] CA Antibacterial, wound-healing [95] Crystal
arraysPCL Drug release Advantages: Increase in surface area, high roughness, simple process, easily functionalized;
Disadvantages: Unclear molding mechanism[84, 89, 91, 92, 93] PCL/CS Tissue engineering [88] PCL/nHA Tissue engineering [90] PCL/PVA Drug release, tissue engineering [85] Grooves CAB Tissue engineering Advantages: Increase in surface area, high roughness;
Disadvantages: Limited to specific polymers and solvent[61] PCL/PVP Tissue engineering, antibacterial [64, 99] PLLA Tissue engineering [98] PVA/PAN Tissue engineering [68] PS Tissue engineering [66] Wrinkled PVDF Energy harvesting, oil/water separation Advantages: Increase in surface area, good mechanical properties, high roughness;
Disadvantages: Limited to specific polymers and solvent, complex operation[19, 20] PMMA Tissue engineering [69] PS Tissue engineering [72] PLA Tissue engineering [34] CA Tissue engineering [70] Ipp Tissue engineering [71] PCL/PEO Drug release [83] Notes: PLA—Polylactic acid; PLLA—Poly(L-lactic acid); PDLLA—Poly(D, L-lactide); PVDF—Polyvinylidene difluoride; SA—Salicylic acid; PEG—Poly(ethylene glycol); Ipp—Isotactic polypropylene; PECA—Poly(ethyl cyanoacrylate); PCL—Polycaprolactone; PS—Polystyrene; PLGA—Poly(lactic-co-glycolic acid); CA—Cellulose acetate; CS—Chitosan; HA—Hualuronic acid; PVA—Poly(lactic-co-glycolic acid); CAB—Cellulose acetate butyrate; PVP—Polyvinyl pyrrolidone; PAN—Polyacrylonitrile; PMMA—Polymethyl methacrylate; PEO—Polyethylene oxide. -
[1] ANSELME K, DAVIDSON P, POPA A M, et al. The interaction of cells and bacteria with surfaces structured at the nanometre scale[J]. Acta Biomaterialia, 2010, 6(10): 3824-3846. doi: 10.1016/j.actbio.2010.04.001 [2] SHI X, WANG Y, LI D, et al. Cell adhesion on a POEGMA-modified topographical surface[J]. Langmuir, 2012, 28(49): 17011-17018. doi: 10.1021/la303042d [3] XUE J J, WU T, DAI Y Q, et al. Electrospinning and electrospun nanofibers: Methods, materials, and applications[J]. Chemical Reviews, 2019, 119(8): 5298-5415. doi: 10.1021/acs.chemrev.8b00593 [4] LIU W, THOMOPOULOS S, XIA Y. Electrospun nanofibers for regenerative medicine[J]. Advanced Healthcare Materials, 2012, 1(1): 10-25. doi: 10.1002/adhm.201100021 [5] LIU Y, WANG C. Advanced nanofibrous materials manufacture technology based on electrospinning[M]. Boca Raton: CRC Press, 2019: 121-140. [6] LI J, LEMSTRA P J, MA P. Chapter 7: Can high-performance fibers be(come) bio-based and also biocompostable?[J]. Advanced Industrial and Engineering Polymer Research, 2022, 5(2): 117-132. doi: 10.1016/j.aiepr.2022.03.002 [7] XUE J, XIE J, LIU W, et al. Electrospun nanofibers: New concepts, materials, and applications[J]. Accounts of Chemical Research, 2017, 50(8): 1976-1987. doi: 10.1021/acs.accounts.7b00218 [8] GAO X, WEN M, LIU Y, et al. Synthesis and characterization of PU/PLCL/CMCS electrospun scaffolds for skin tissue engineering[J]. Polymers, 2022, 14(22): 5029. doi: 10.3390/polym14225029 [9] YOO H S, KIM T G, PARK T G. Surface-functionalized electrospun nanofibers for tissue engineering and drug delivery[J]. Advanced Drug Delivery Reviews, 2009, 61(12): 1033-1042. doi: 10.1016/j.addr.2009.07.007 [10] HABELITZ S, BALOOCH M, MARSHALL S J, et al. In situ atomic force microscopy of partially demineralized human dentin collagen fibrils[J]. Journal of Structural Biology, 2002, 138(3): 227-236. doi: 10.1016/S1047-8477(02)00029-1 [11] CLARK P, CONNOLLY P, CURTIS A S, et al. Cell guidance by ultrafine topography in vitro[J]. Journal of Cell Science, 1991, 99: 73-77. [12] BETTINGER C J, LANGER R, BORENSTEIN J T. Engineering substrate topography at the micro- and nanoscale to control cell function[J]. Angewandte Chemie International Edition, 2009, 48(30): 5406-5415. doi: 10.1002/anie.200805179 [13] DIEHL K A, FOLEY J D, NEALEY P F, et al. Nanoscale topography modulates corneal epithelial cell migration[J]. Journal of Biomedical Materials Research Part A, 2005, 75(3): 603-611. [14] 常晓华. 结晶调控的聚乳酸及其共聚物在溶液中的多层次聚集结构与性能[D]. 杭州: 浙江大学, 2019.CHANG Xiaohua. Multilevel aggregated structure and properties of poly(lactic acid) and its copolymers in solution tuned by crystallization[D]. Hangzhou: Zhejiang University, 2019(in Chinese). [15] ZAAROUR B, ZHU L, JIN X. A review on the secondary surface morphology of electrospun nanofibers: Formation mechanisms, characterizations, and applications[J]. ChemistrySelect, 2020, 5(4): 1335-1348. doi: 10.1002/slct.201903981 [16] JIN S A, SPONTAK R J. Fundamentals of and advances in nanocellulose and nanochitin systems[J]. Advanced Industrial and Engineering Polymer Research, 2023, 6(4): 356-381. doi: 10.1016/j.aiepr.2023.04.003 [17] ZAAROUR B, ZHANG W, ZHU L, et al. Maneuvering surface structures of polyvinylidene fluoride nanofibers by controlling solvent systems and polymer concentration[J]. Textile Research Journal, 2018, 89(12): 2406-2422. [18] HUAN S, LIU G, HAN G, et al. Effect of experimental parameters on morphological, mechanical and hydrophobic properties of electrospun polystyrene fibers[J]. Materials, 2015, 8(5): 2718-2734. doi: 10.3390/ma8052718 [19] ZAAROUR B, ZHU L, JIN X. Controlling the surface structure, mechanical properties, crystallinity, and piezoelectric properties of electrospun PVDF nanofibers by maneuvering molecular weight[J]. Soft Materials, 2019, 17(8): 1-9. [20] ZAAROUR B, ZHU L, HUANG C, et al. Fabrication of a polyvinylidene fluoride cactus-like nanofiber through one-step electrospinning[J]. RSC Advances, 2018, 8(74): 42353-42360. doi: 10.1039/C8RA09257E [21] JIAN S, ZHU J, JIANG S, et al. Nanofibers with diameter below one nanometer from electrospinning[J]. RSC Advances, 2018, 8(9): 4794-4802. doi: 10.1039/C7RA13444D [22] BAKAR S S S, FONG K C, ELEYAS A, et al. Effect of voltage and flow rate electrospinning parameters on polyacrylonitrile electrospun fibers[J]. IOP Conference Series: Materials Science and Engineering, 2018, 318: 012076. doi: 10.1088/1757-899X/318/1/012076 [23] MENG N, ZHENG Y, XIN B. Surface morphologies of electrospun polystyrene fibers induced by an electric field[J]. Textile Research Journal, 2019, 89(18): 3850-3859. doi: 10.1177/0040517518824843 [24] ACIK G, KAMACI M, CANSOY C E J C, et al. Superhydrophobic EVA copolymer fibers: The impact of chemical composition on wettability and photophysical properties[J]. Colloid and Polymer Science, 2018, 296: 1759-1766. doi: 10.1007/s00396-018-4395-7 [25] LI Z, WANG C. One-dimensional nanostructures: Electrospinning technique and unique nanofibers[M]. Heidelberg: Springer, 2013: 40-60. [26] ZAAROUR B, ZHU L, HUANG C, et al. Enhanced piezoelectric properties of randomly oriented and aligned electrospun PVDF fibers by regulating the surface morphology[J]. Journal of Applied Polymer Science, 2019, 136(6): 47049. [27] ZAAROUR B, ZHU L, HUANG C, et al. Controlling the secondary surface morphology of electrospun PVDF nanofibers by regulating the solvent and relative humidity[J]. Nanoscale Research Letters, 2018, 13(1): 285. doi: 10.1186/s11671-018-2705-0 [28] BADMUS M, LIU J, WANG N, et al. Hierarchically electrospun nanofibers and their applications: A review[J]. Nano Materials Science, 2021, 3(3): 213-232. doi: 10.1016/j.nanoms.2020.11.003 [29] HUANG C, THOMAS N L. Fabrication of porous fibers via electrospinning: Strategies and applications[J]. Polymer Reviews, 2019, 60(4): 595-647. [30] ŞİMŞEK M. Altering surface topography of electrospun fibers[J]. Natural and Applied Sciences Journal, 2019, 2(1): 8-14. doi: 10.38061/idunas.569788 [31] WANG P, LYU H, CAO X, et al. Recent progress of the preparation and application of electrospun porous nanofibers[J]. Polymers, 2023, 15(4): 921. doi: 10.3390/polym15040921 [32] TAN S H, INAI R, KOTAKI M, et al. Systematic parameter study for ultra-fine fiber fabrication via electrospinning process[J]. Polymer, 2005, 46(16): 6128-6134. doi: 10.1016/j.polymer.2005.05.068 [33] LI L, HASHAIKEH R, ARAFAT H A. Development of eco-efficient micro-porous membranes via electrospinning and annealing of poly(lactic acid)[J]. Journal of Membrane Science, 2013, 436: 57-67. doi: 10.1016/j.memsci.2013.02.037 [34] CASASOLA R, THOMAS N, TRYBAŁA A, et al. Electrospun poly lactic acid (PLA) fibres: Effect of different solvent systems on fibre morphology and diameter[J]. Polymer, 2014, 55(18): 4728-4737. doi: 10.1016/j.polymer.2014.06.032 [35] BOGNITZKI M, CZADO W, FRESE T, et al. Nanostructured fibers via electrospinning[J]. Advanced Materials, 2001, 13(1): 70-72. doi: 10.1002/1521-4095(200101)13:1<70::AID-ADMA70>3.0.CO;2-H [36] BRANCIFORTI M C, CUSTODIO T A, GUERRINI L M, et al. Characterization of nano-structured poly(D, L-lactic acid) nonwoven mats obtained from different solutions by electrospinning[J]. Journal of Macromolecular Science Part B: Physics, 2009, 48(6): 1222-1240. doi: 10.1080/10408390903060970 [37] HUANG C, THOMAS N L. Fabricating porous poly(lactic acid) fibres via electrospinning[J]. European Polymer Journal, 2018, 99: 464-476. doi: 10.1016/j.eurpolymj.2017.12.025 [38] KATSOGIANNIS K A G, VLADISAVLJEVIĆ G T, GEORGIADOU S. Porous electrospun polycaprolactone (PCL) fibres by phase separation[J]. European Polymer Journal, 2015, 69: 284-295. doi: 10.1016/j.eurpolymj.2015.01.028 [39] QI Z, YU H, CHEN Y, et al. Highly porous fibers prepared by electrospinning a ternary system of nonsolvent/solvent/poly(L-lactic acid)[J]. Materials Letters, 2009, 63(3): 415-418. [40] YANG Y, CENTRONE A, CHEN L, et al. Highly porous electrospun polyvinylidene fluoride (PVDF)-based carbon fiber[J]. Carbon, 2011, 49(11): 3395-3403. doi: 10.1016/j.carbon.2011.04.015 [41] NGUYEN T T T, GHOSH C, HWANG S G, et al. Porous core/sheath composite nanofibers fabricated by coaxial electrospinning as a potential mat for drug release system[J]. International Journal of Pharmaceutics, 2012, 439(1): 296-306. [42] ZHANG W, MELE E. Phase separation events induce the coexistence of distinct nanofeatures in electrospun fibres of poly(ethyl cyanoacrylate) and polycaprolactone[J]. Materials Today Communications, 2018, 16: 135-141. doi: 10.1016/j.mtcomm.2018.05.005 [43] REZABEIGI E, STA M, SWAIN M, et al. Electrospinning of porous polylactic acid fibers during nonsolvent induced phase separation[J]. Journal of Applied Polymer Science, 2017, 134(20): 44862. doi: 10.1002/app.44862 [44] KOUPARITSAS I K, MELE E, RONCA S. Synthesis and electrospinning of polycaprolactone from an aluminium-based catalyst: Influence of the ancillary ligand and initiators on catalytic efficiency and fibre structure[J]. Polymers (Basel), 2019, 11(4): 677. doi: 10.3390/polym11040677 [45] ABADI F J H, TEHRAN M A, ZAMANI F, et al. Effect of nanoporous fibers on growth and proliferation of cells on electrospun poly(ε-caprolactone) scaffolds[J]. International Journal of Polymeric Materials and Polymeric Biomaterials, 2014, 63(2): 57-64. doi: 10.1080/00914037.2013.769248 [46] ZHENG J, ZHANG H, ZHAO Z, et al. Construction of hierarchical structures by electrospinning or electrospraying[J]. Polymer, 2012, 53(2): 546-554. doi: 10.1016/j.polymer.2011.12.018 [47] MCCANN J T, MARQUEZ M, XIA Y. Highly porous fibers by electrospinning into a cryogenic liquid[J]. Journal of the American Chemical Society, 2006, 128(5): 1436-1437. doi: 10.1021/ja056810y [48] LI W, SHI L, ZHANG X, et al. Electrospinning of polycaprolactone nanofibers using H2O as benign additive in polycaprolactone/glacial acetic acid solution[J]. Journal of Applied Polymer Science, 2018, 135(3): 45578. doi: 10.1002/app.45578 [49] LEONG M F, RASHEED M Z, LIM T C, et al. In vitro cell infiltration and in vivo cell infiltration and vascularization in a fibrous, highly porous poly(D, L-lactide) scaffold fabricated by cryogenic electrospinning technique[J]. Journal of Biomedical Materials Research Part A, 2009, 91(1): 231-240. [50] YE X Y, LIN F W, HUANG X J, et al. Polymer fibers with hierarchically porous structure: Combination of high temperature electrospinning and thermally induced phase separation[J]. RSC Advances, 2013, 3(33): 13851. doi: 10.1039/c3ra41315b [51] GLEESON S E, KIM S, YU T, et al. Size-dependent soft epitaxial crystallization in the formation of blend nanofiber shish kebabs[J]. Polymer, 2020, 202: 122644. doi: 10.1016/j.polymer.2020.122644 [52] CHEN X, DONG B, WANG B, et al. Crystalline block copolymer decorated, hierarchically ordered polymer nanofibers[J]. Macromolecules, 2010, 43(23): 9918-9927. doi: 10.1021/ma101900n [53] CHANG Z H, WANG D F, XU Y Y, et al. Structure and morphology of poly(ε-caprolactone) heterogeneous Shish-Kebab structure induced by poly(lactic acid) nanofibers[J]. Chinese Journal of Polymer Science, 2022, 40: 1223-1232. doi: 10.1007/s10118-022-2747-8 [54] LIU C L, WANG M J, WU G, et al. Preparation of core-shell nanofibers with selectively localized CNTs from Shish Kebab-like hierarchical composite micelles[J]. Macromolecular Rapid Communications, 2014, 35(16): 1450-1457. doi: 10.1002/marc.201400261 [55] XU X, WANG H, JIANG L, et al. Comparison between cellulose nanocrystal and cellulose nanofibril reinforced poly(ethylene oxide) nanofibers and their novel Shish-Kebab-like crystalline structures[J]. Macromolecules, 2014, 47(10): 3409-3416. doi: 10.1021/ma402627j [56] LIU T, LIAN X, LI L, et al. Facile fabrication of fully biodegradable and biorenewable poly (lactic acid)/poly (butylene adipate-co-terephthalate) in-situ nanofibrillar composites with high strength, good toughness and excellent heat resistance[J]. Polymer Degradation and Stability, 2020, 171: 109044. doi: 10.1016/j.polymdegradstab.2019.109044 [57] LIANG Y, ZHENG G, HAN W, et al. Nano-hybrid Shish-Kebab: Isotactic polypropylene epitaxial growth on electrospun polyamide 66 nanofibers via isothermal crystallization[J]. Materials Letters, 2011, 65(4): 653-656. doi: 10.1016/j.matlet.2010.11.034 [58] WANG Z, MAO Y M, LI X K, et al. The influence of ethyl branch on formation of Shish-Kebab crystals in bimodal polyethylene under shear at low temperature[J]. Chinese Journal of Polymer Science, 2021, 39(8): 1050-1058. doi: 10.1007/s10118-021-2568-1 [59] WANG L, WANG J H, YANG B, et al. A novel hierarchical crystalline structure of injection-molded bars of linear polymer: Co-existence of bending and normal Shish-Kebab structure[J]. Colloid and Polymer Science, 2013, 291(6): 1503-1511. doi: 10.1007/s00396-012-2887-4 [60] ZHAN L, DENG J, KE Q, et al. Grooved fibers: Preparation principles through electrospinning and potential applications[J]. Advanced Fiber Materials, 2022, 4(2): 203-213. doi: 10.1007/s42765-021-00116-5 [61] HUANG C, TANG Y, LIU X, et al. Electrospinning of nanofibres with parallel line surface texture for improvement of nerve cell growth[J]. Soft Matter, 2011, 7(22): 10812-10817. doi: 10.1039/c1sm06430d [62] LIU W, HUANG C, JIN X. Tailoring the grooved texture of electrospun polystyrene nanofibers by controlling the solvent system and relative humidity[J]. Nanoscale Research Letters, 2014, 9(1): 350. doi: 10.1186/1556-276X-9-350 [63] LU Z, ZHANG B, GONG H, et al. Fabrication of hierarchical porous poly(L-lactide) (PLLA) fibrous membrane by electrospinning[J]. Polymer, 2021, 226: 123797. doi: 10.1016/j.polymer.2021.123797 [64] WU T, XUE J, XIA Y. Engraving the surface of electrospun microfibers with nanoscale grooves promotes the outgrowth of neurites and the migration of schwann cells[J]. Angewandte Chemie-International Edition, 2020, 59(36): 15626-15632. doi: 10.1002/anie.202002593 [65] EKRAM B, ABD EL-HADY B M, EL-KADY A M, et al. Enhanced mesenchymal stem cells growth on antibacterial microgrooved electrospun zinc chloride/polycaprolactone conduits for peripheral nerve regeneration[J]. Journal of Bioactive and Compatible Polymers, 2021, 36(2): 152-168. doi: 10.1177/0883911520988305 [66] LIU W, HUANG C, JIN X. Electrospinning of grooved polystyrene fibers: Effect of solvent systems[J]. Nanoscale Research Letters, 2015, 10(1): 949. [67] LIANG M, CHEN X, XU Y, et al. Double-grooved nanofibre surfaces with enhanced anisotropic hydrophobicity[J]. Nanoscale, 2017, 9(42): 16214-16222. doi: 10.1039/C7NR05188C [68] KIM M N, KOH J, LEE Y, et al. Preparation of PVA/PAN bicomponent nanofiber via electrospinning and selective dissolution[J]. Journal of Applied Polymer Science, 2009, 113(1): 274-282. doi: 10.1002/app.29553 [69] LI L, LI R, LI M, et al. Theoretical selection of solvent for production of electrospun PMMA fibers with wrinkled surfaces[J]. RSC Advances, 2014, 4(53): 27914. doi: 10.1039/c4ra03657c [70] WU X, WANG L, YU H, et al. Effect of solvent on morphology of electrospinning ethyl cellulose fibers[J]. Journal of Applied Polymer Science, 2005, 97(3): 1292-1297. doi: 10.1002/app.21818 [71] PAI C, BOYCE M, RUTLEDGE G C. Morphology of porous and wrinkled fibers of polystyrene electrospun from dimethylformamide[J]. Macromolecules, 2009, 42: 2102-2114. doi: 10.1021/MA802529H [72] PAI C L, BOYCE M C, RUTLEDGE G C J M. Morphology of porous and wrinkled fibers of polystyrene electrospun from dimethylformamide[J]. Macromolecules, 2009, 42(6): 2102-2114. doi: 10.1021/ma802529h [73] LI L, JIANG Z, LI M, et al. Hierarchically structured PMMA fibers fabricated by electrospinning[J]. RSC Advances, 2014, 4(95): 52973-52985. doi: 10.1039/C4RA05385K [74] NEO Y P, RAY S, PERERA C O. Fabrication of functional electrospun nanostructures for food applications[M]//GRUMEZESCU A M, HOLBAN A M. Role of Materials Science in Food Bioengineering. New York: Academic Press, 2018: 109-146. [75] LEIDY R, XIMENA Q. Use of electrospinning technique to produce nanofibres for food industries: A perspective from regulations to characterisations[J]. Trends in Food Science & Technology, 2019, 85: 92-106. [76] PAVINATTO A, MERCANTE L A, LEANDRO C S, et al. Layer-by-layer assembled films of chitosan and multi-walled carbon nanotubes for the electrochemical detection of 17α-ethinylestradiol[J]. Journal of Electroanalytical Chemistry, 2015, 755(15): 215-220. [77] PRIYA S, INMAN D J J S U. Energy harvesting technologies[M]. NewYork: Springer, 2009: 243-280. [78] WANG X, YU J, SUN G, et al. Electrospun nanofibrous materials: A versatile medium for effective oil/water separation[J]. Materials Today, 2016, 19(7): 403-414. doi: 10.1016/j.mattod.2015.11.010 [79] ZARE M R, KHORRAM M, BARZEGAR S, et al. Antimicrobial core-shell electrospun nanofibers containing ajwain essential oil for accelerating infected wound healing[J]. International Journal of Pharmacology, 2021, 603(15): 120698. [80] TOPUZ F, UYAR T. Electrospinning of cyclodextrin functional nanofibers for drug delivery applications[J]. Pharmaceutics, 2018, 11(1): 6. [81] RAMOS C, LANNO G M, LAIDMÄE I, et al. High humidity electrospinning of porous fibers for tuning the release of drug delivery systems[J]. International Journal of Polymeric Materials and Polymeric Biomaterials, 2020, 70(12): 880-892. doi: DOI:10.1080/00914037.2020.1765361 [82] CHEN X, LI H, LU W, et al. Antibacterial porous coaxial drug-carrying nanofibers for sustained drug-releasing applications[J]. Nanomaterials, 2021, 11(5): 1316. doi: 10.3390/nano11051316 [83] CHEN R, LIN L, WANG H, et al. Effects of morphologies of thermosensitive electrospun nanofibers on controllable drug release[J]. Tissue Engineering Part A, 2021, 27(11-12): 724-732. doi: 10.1089/ten.tea.2020.0258 [84] CHIU Y J, ZHANG Z, DZIEMIDOWICZ K, et al. The effect of solvent vapor annealing on drug-loaded electrospun polymer fibers[J]. Pharmaceutics, 2020, 12(2): 139. doi: 10.3390/pharmaceutics12020139 [85] HUANG C, YANG G, ZHOU S, et al. Controlled delivery of growth factor by hierarchical nanostructured core-shell nanofibers for the efficient repair of critical-sized rat calvarial defect[J]. ACS Biomaterials Science & Engineering, 2020, 6(10): 5758-5770. [86] EL-FIQI A, PARK J H. Novel large-volume and highly porous scaffold of poly(ε-caprolactone) microfibers/collagen nanofibers for regenerative medicine[J]. Materials Letters, 2022, 322: 132474. doi: 10.1016/j.matlet.2022.132474 [87] LIU W, JIAO T, SU Y, et al. Electrospun porous poly(3-hydroxybutyrate-co-4-hydroxybutyrate)/lecithin scaffold for bone tissue engineering[J]. RSC Advances, 2022, 12(19): 11913-11922. doi: 10.1039/D2RA01398C [88] ZAMANI F, AMANI-TEHRAN M, LATIFI M, et al. The influence of surface nanoroughness of electrospun PLGA nanofibrous scaffold on nerve cell adhesion and proliferation[J]. Journal of Materials Science: Materials in Medicine, 2013, 24(6): 1551-1560. doi: 10.1007/s10856-013-4905-6 [89] JING X, MI H Y, WANG X C, et al. Shish-Kebab-structured poly(ε-caprolactone) nanofibers hierarchically decorated with chitosan-poly(ε-caprolactone) copolymers for bone tissue engineering[J]. ACS Applied Materials & Interfaces, 2015, 7(12): 6955-6965. [90] YU T, GLEESON S E, LI C Y, et al. Electrospun poly(ε-caprolactone) nanofiber shish kebabs mimic mineralized bony surface features[J]. Journal of Biomedical Materials Research Part B: Applied Biomaterials, 2019, 107(4): 1141-1149. doi: 10.1002/jbm.b.34207 [91] JING X, JIN E, MI H Y, et al. Hierarchically decorated electrospun poly(ε-caprolactone)/nanohydroxyapatite composite nanofibers for bone tissue engineering[J]. Journal of Materials Science, 2015, 50(12): 4174-4186. doi: 10.1007/s10853-015-8933-0 [92] GUO X, WANG X, LI X, et al. Endothelial cell migration on poly(ε-caprolactone) nanofibers coated with a nanohybrid Shish-Kebab structure mimicking collagen fibrils[J]. Biomacromolecules, 2020, 21(3): 1202-1213. doi: 10.1021/acs.biomac.9b01638 [93] JIANG L, WANG L, WANG N, et al. Fabrication of polycaprolactone electrospun fibers with different hierarchical structures mimicking collagen fibrils for tissue engineering scaffolds[J]. Applied Surface Science, 2018, 427: 311-325. doi: 10.1016/j.apsusc.2017.08.005 [94] YIN J, XU L. Batch preparation of electrospun polycaprolactone/chitosan/aloe vera blended nanofiber membranes for novel wound dressing[J]. International Journal of Biological Macromolecules, 2020, 160(1): 352-363. [95] CHEN Y, MENSAH A, WANG Q, et al. Hierarchical porous nanofibers containing thymol/beta-cyclodextrin: Physico-chemical characterization and potential biomedical applications[J]. Materials Science & Engineering C: Materials for Biological Applications, 2020, 115: 111155. [96] SUN Z, FAN C, TANG X, et al. Characterization and antibacterial properties of porous fibers containing silver ions[J]. Applied Surface Science, 2016, 387: 828-838. doi: 10.1016/j.apsusc.2016.07.015 [97] SU X, ZHAI Y, JIA C, et al. Improved antibacterial properties of polylactic acid-based nanofibers loaded with ZnO-Ag nanoparticles through pore engineering[J]. ACS Applied Materials & Interfaces, 2023, 15(36): 42920-42929. [98] ZHAN L, WANG L, DENG J, et al. Enhanced cellular infiltration of tissue-engineered scaffolds fabricated by PLLA nanogrooved microfibers[J]. Nano Research, 2022, 16(1): 1614-1625. [99] QI L, HUANG Y, SUN D, et al. Guiding the path to healing: CuO2-laden nanocomposite membrane for diabetic wound treatment[J]. Small, 2023, 20(3): e2305100. -





 下载:
下载: