Preparation and performance characterization of primary secondary amine-modified Fe3O4 magnetic materials
-
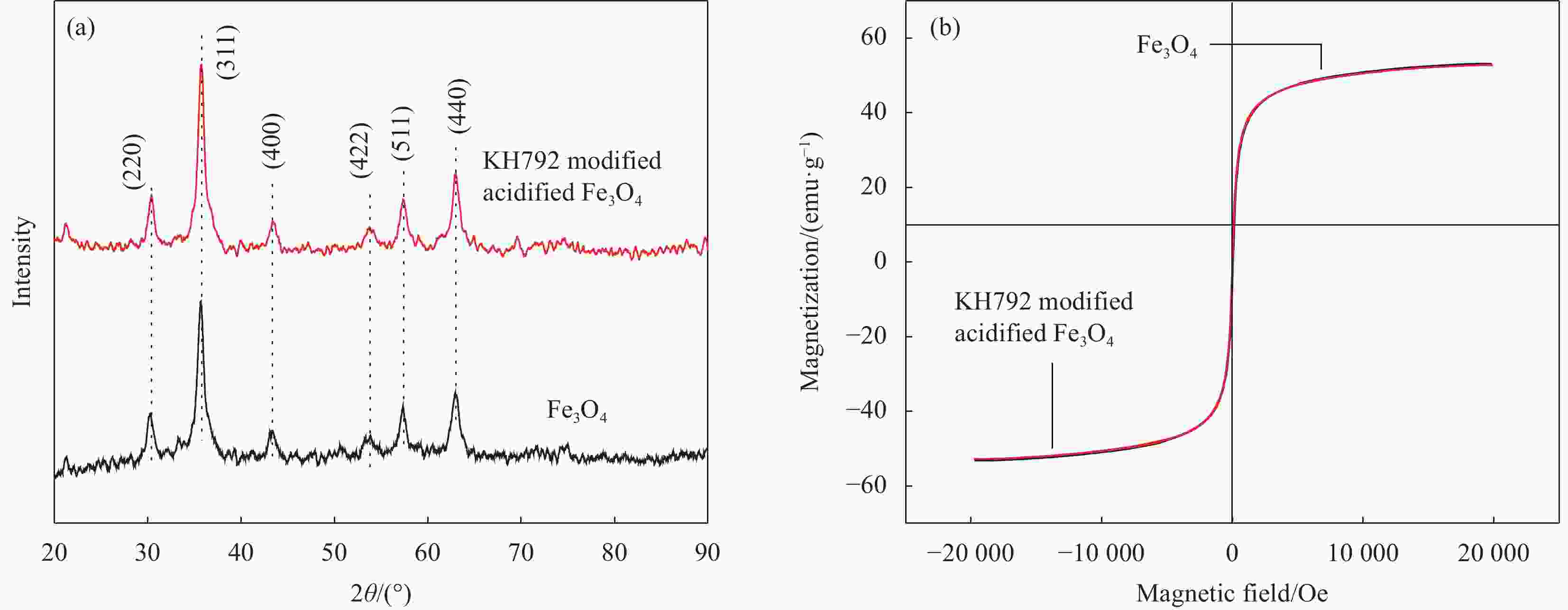
摘要: 食品中的有机酸等内源性物质会干扰其中痕量残留物的检测,为消除此影响,以N-β-(氨乙基)-γ-氨丙基三甲氧基硅烷(KH792)为氨基化试剂,直接修饰酸化后的Fe3O4得到磁性吸附材料。经响应面试验优化得到KH792对酸化Fe3O4的最佳修饰条件为温度 82.5℃、pH=4.9,KH792添加量 1.8 mL,在最佳条件下,KH792直接修饰的酸化Fe3O4对没食子酸吸附量为22.8 mg/g,比市售Fe3O4提高了188%,且具备快速固液分离的能力、良好的稳定性及分散性。通过BET、Zeta电位、FTIR和XPS对直接修饰的产物性能进行了表征,结果表明,KH792与Fe3O4表面羟基通过Fe—O—Si键结合,酸化提高了Fe3O4表面羟基含量,因此也增强了KH792对Fe3O4的修饰效果,本文制备的磁性产物可用于净化复杂样品基质中的有机酸。
-
关键词:
- N-β-(氨乙基)-γ-氨丙基三甲氧基硅烷 /
- 酸化Fe3O4 /
- 直接修饰法 /
- 磁性吸附材料 /
- 修饰机制
Abstract: Endogenous substances, such as organic acids in food, can interfere with the detection of trace residues. To eliminate this effect, the magnetic adsorbent material was obtained by directly modifying acidified Fe3O4 with N-β-(aminoethyl)-γ-aminopropyltrimethoxysilane (KH792) as an amination reagent. The optimal modification condition of KH792 for acidified Fe3O4 was optimized by response surface methodology, which was 82.5℃, pH=4.9, and KH792 addition of 1.8 mL. Under these optimal conditions, the acidified Fe3O4 directly modified by KH792 has an adsorption capacity of 22.8 mg/g and shows a 188% increase in the adsorption capacity of the gallic acid compared to commercially available Fe3O4. Additionally, the modified Fe3O4 exhibits excellent performances of rapid solid-liquid separation, good stability, and dispersibility. The properties of the directly modified products were characterized by BET, Zeta potential, FTIR, and XPS. The results show that KH792 bonds on Fe3O4 surface hydroxyl group's by Fe—O—Si bonding, and acidification improves the number of hydroxyl groups on Fe3O4 surface, thus improving the KH792 modification for Fe3O4. The magnetic products prepared in this work can be used for the purification of organic acids in complex sample matrices. -
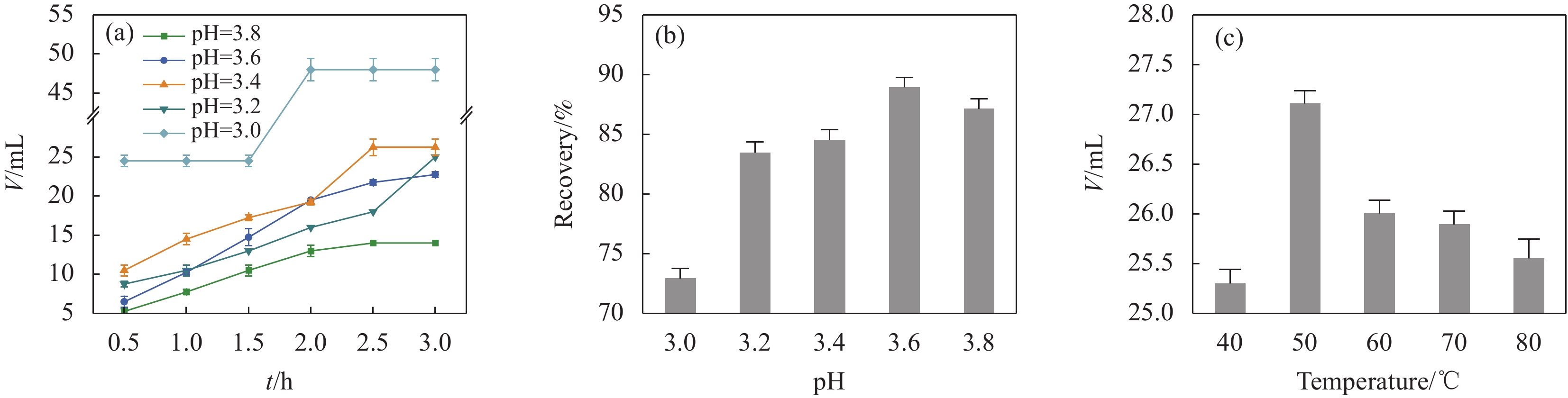
图 1 不同pH下Fe3O4消耗盐酸量随时间的变化(a)、回收率的变化(b)、不同温度下Fe3O4消耗盐酸量(c)
Figure 1. Variation of hydrochloric acid consumption by Fe3O4 with time (a), the recovery at different pH (b), the consumption of hydrochloric acid by Fe3O4 at different temperatures (c)
V—Consumption of hydrochloric acid by Fe3O4; t—Acidification time
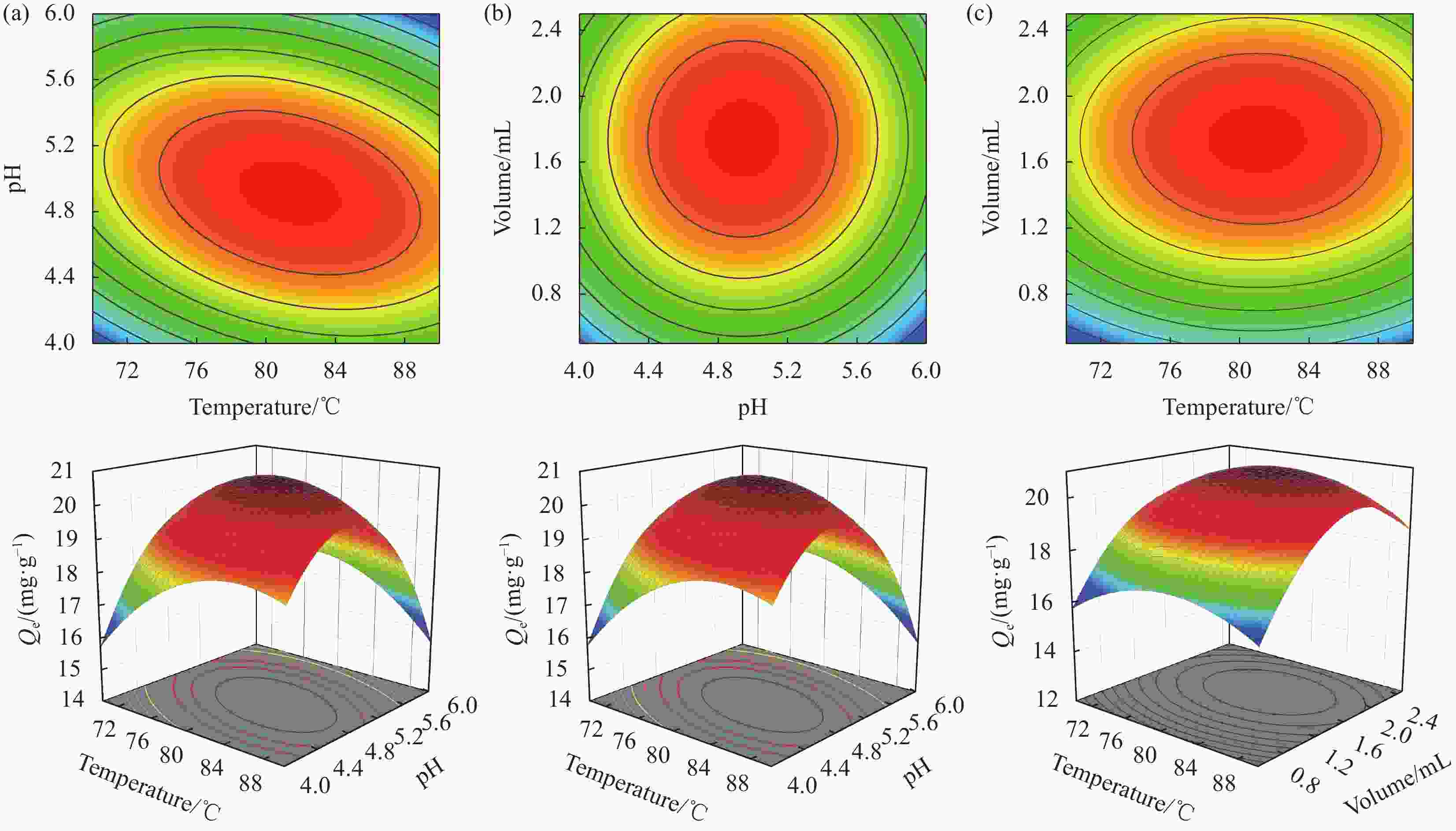
图 2 自变量和没食子酸吸附量的等高线和响应面图:(a) 温度和pH;(b) pH和KH792添加量;(c) 温度和KH792添加量
Figure 2. Contour and response surface plots of the independent variables and gallic acid sorption: (a) Temperature and pH; (b) pH and KH792 addition level; (c) Temperature and KH792 addition level
Qe—Adsorption capacity of Fe3O4-2NH2 for gallic acid; Volume is KH792 addition level
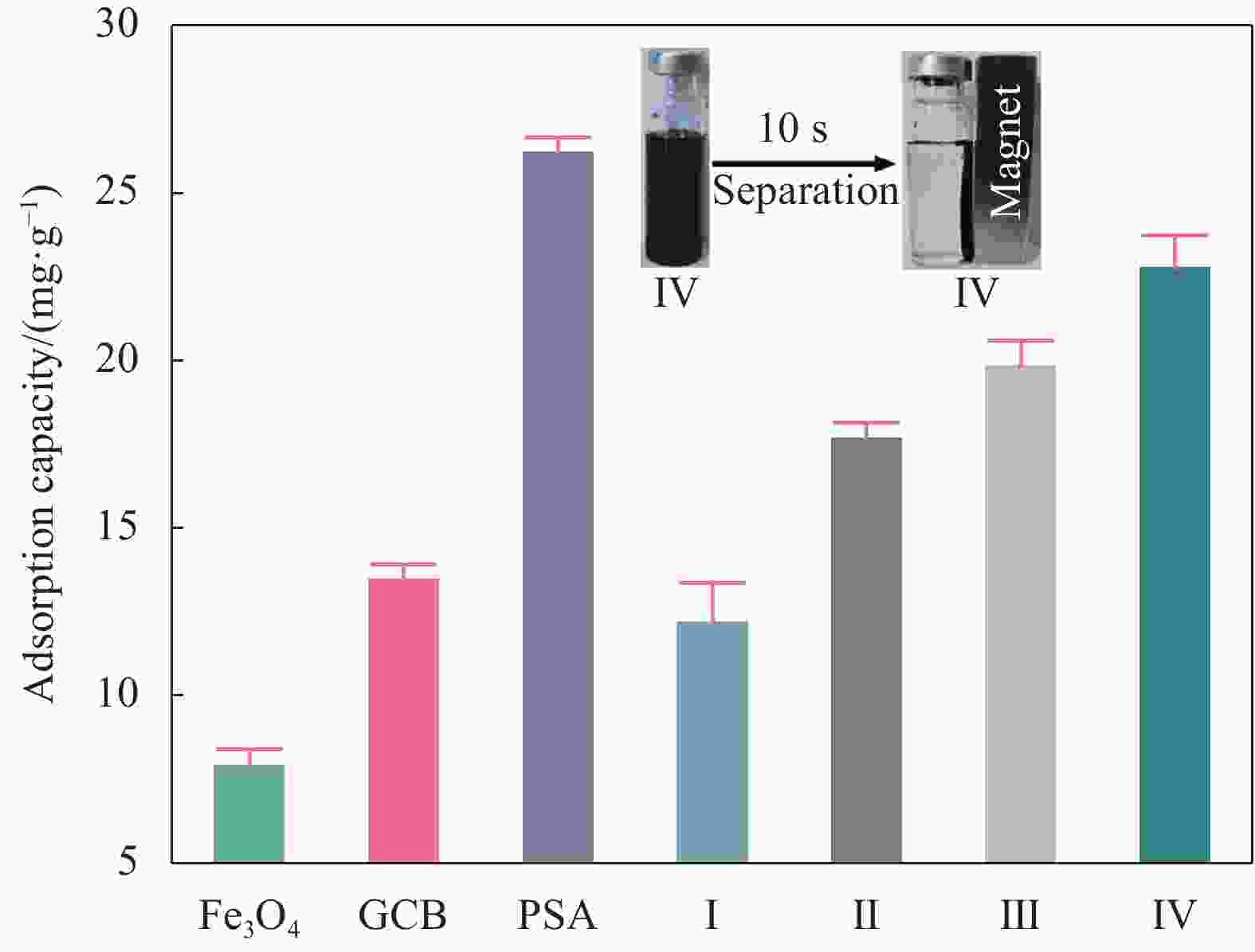
图 3 不同材料的没食子酸吸附量
Figure 3. Adsorption capacity of different materials for gallic acid
GCB—Graphitized carbon black; PSA—Primary secondary amine; I—KH792 post-modification product; II—KH792 modified unacidified Fe3O4; III—KH792 modified acidified Fe3O4; IV—KH792 acidified Fe3O4 modified by optimized conditions
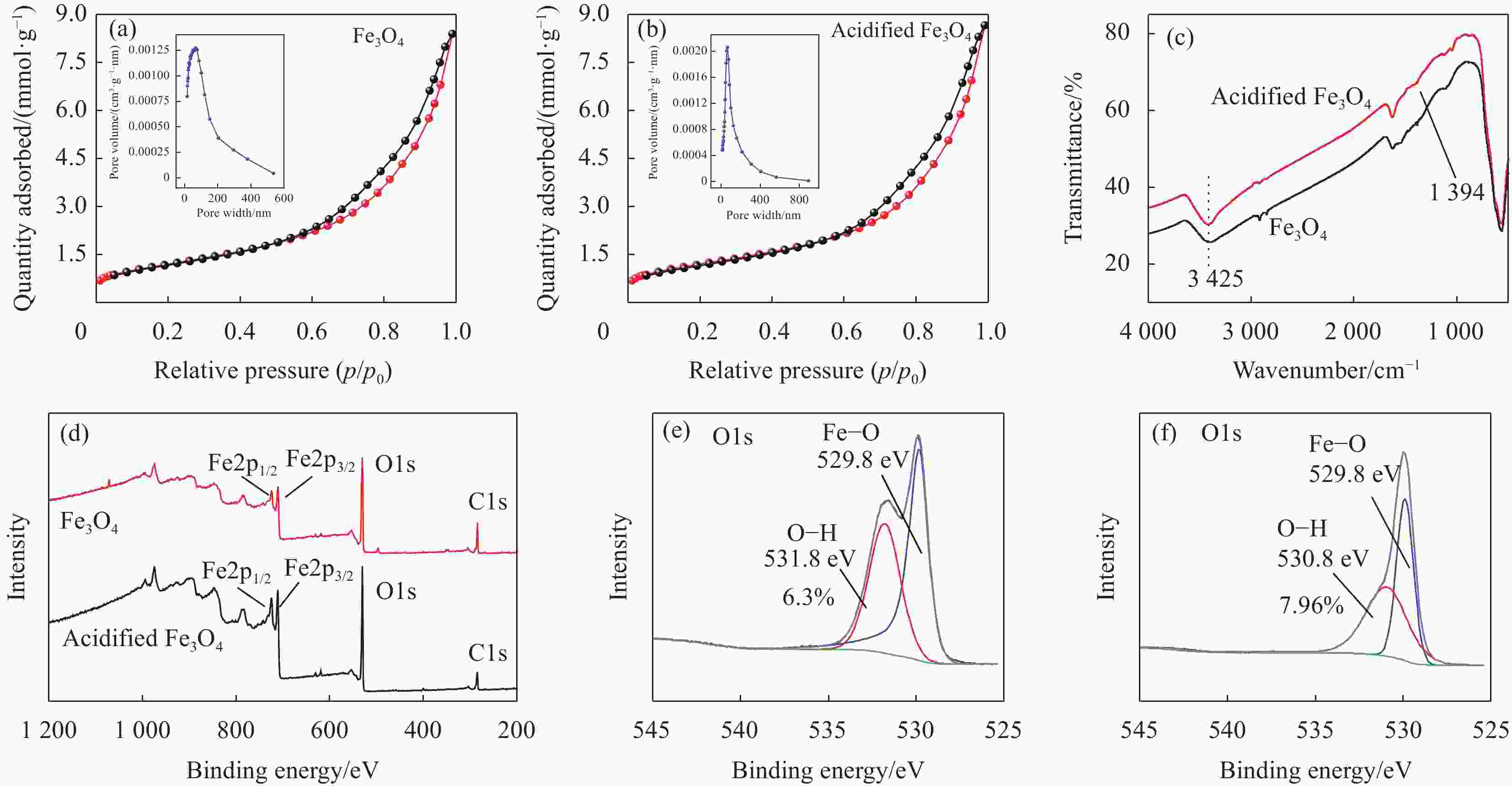
图 4 Fe3O4 (a)和酸化的Fe3O4 (b)的N2吸附-解吸等温线;Fe3O4和酸化的Fe3O4的红外图谱(c)和XPS全谱(d); Fe3O4 (e)和酸化的Fe3O4(f)的O1s图谱
Figure 4. N2 adsorption-desorption isotherms of Fe3O4 (a) and acidified Fe3O4 (b); Infrared spectra (c) and XPS wide-scan spectra (d) of Fe3O4 and acidified Fe3O4; O1s XPS spectra of Fe3O4 (e) and acidified Fe3O4 (f)
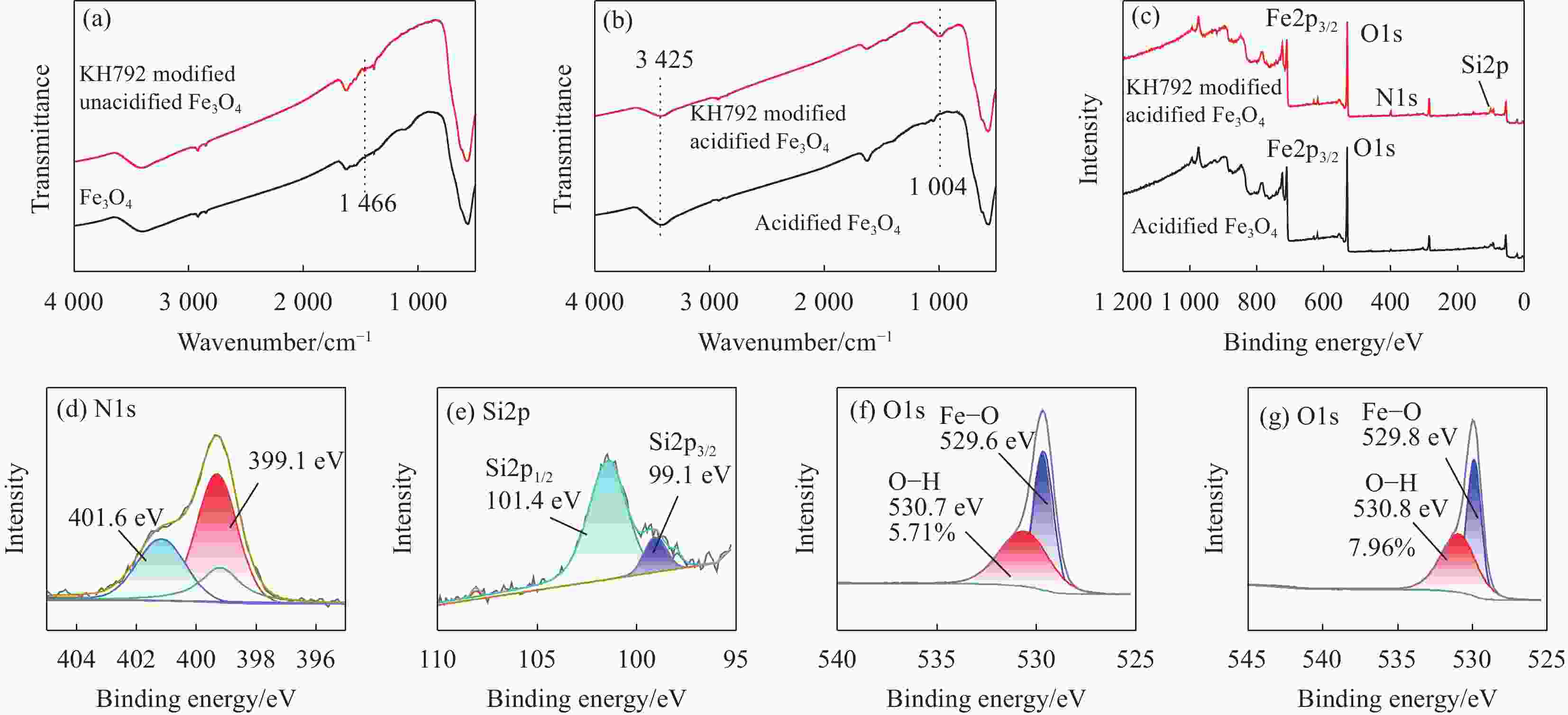
图 5 (a) Fe3O4和KH792修饰的未酸化Fe3O4的红外图谱;酸化的Fe3O4和KH792修饰的酸化Fe3O4的红外图谱(b)和XPS全谱(c);KH792修饰的酸化Fe3O4的N1s (d)、Si2p (e)、O1s (f) XPS图谱;(g) 酸化的Fe3O4的O1s XPS图谱
Figure 5. (a) Infrared spectra of Fe3O4 and KH792 modified unacidified Fe3O4; Infrared (b) and XPS full spectra (c) of acidified Fe3O4 and KH792 modified acidified Fe3O4; N1s (d), Si2p (e), O1s (f) XPS spectra of KH792 modified acidified Fe3O4; (g) O1 XPS spectra of acidified Fe3O4
表 1 响应面试验设计、结果及方差分析
Table 1. Response surface experimental design, result and analysis of variance
No. A: Temperature/℃ B: pH C: KH792 addition level/mL Y: Adsorption capacity for
gallic acid/(mg·g−1)1 70.0 4.0 1.5 16.33 2 90.0 4.0 1.5 19.39 3 70.0 6.0 1.5 17.04 4 90.0 6.0 1.5 15.78 5 70.0 5.0 0.5 15.45 6 90.0 5.0 0.5 16.01 7 70.0 5.0 2.5 18.92 8 90.0 5.0 2.5 19.93 9 80.0 4.0 0.5 15.21 10 80.0 6.0 0.5 14.68 11 80.0 4.0 2.5 17.56 12 80.0 6.0 2.5 16.45 13 80.0 5.0 1.5 21.54 14 80.0 5.0 1.5 21.35 15 80.0 5.0 1.5 21.56 16 80.0 5.0 1.5 20.11 17 80.0 5.0 1.5 21.66 Equation Y=21.25+0.4204A−0.5677B+1.44C−1.08AB+0.1126AC−0.1437BC−1.25A2−2.86B2−2.41C2 Note: KH792—N-β-(aminoethyl)-γ-aminopropyltrimethoxysilane. 表 2 不同修饰方法制得产物的Zeta电位
Table 2. Zeta potential of products made by different modification methods
Product Zeta potential/mV Fe3O4 19.02 KH792 post-modification product 33.99 KH792 modified unacidified Fe3O4 30.59 KH792 modified acidified Fe3O4 42.92 -
[1] ZHANG Z Z, XIA K, PAN Z W, et al. Removal of mercury by magnetic nanomaterial with bifunctional groups and core-shell structure: Synthesis, characterization and optimization of adsorption parameters[J]. Applied Surface Science,2020,500:143970. [2] EBRAHIMI M, EBRAHIMITALAB A, ES'HAGHI Z, et al. Magnetized silane-coupling agent KH-570 based solid-phase extraction followed by gas chromatography-flame ionization detection to determine venlafaxine in human hair and aqueous environmental samples[J]. Archives of Environmental Contamination and Toxicology,2015,68(2):412-420. doi: 10.1007/s00244-014-0100-0 [3] 周书威, 傅红, 杨方. 磁性碳材料的制备及其在食品农药残留检测中的应用[J]. 分析测试学报, 2023, 42(2):241-250. doi: 10.19969/j.fxcsxb.22092602ZHOU Shuwei, FU Hong, YANG Fang. Preparation of magnetic carbon materials and their application in the detection of pesticide residues in food[J]. Journal of Instrumental Analysis,2023,42(2):241-250(in Chinese). doi: 10.19969/j.fxcsxb.22092602 [4] JIANG X, PAN W Y, XIONG Z L, et al. Facile synthesis of layer-by-layer decorated graphene oxide based magnetic nanocomposites for β-agonists/dyes adsorption removal and bacterial inactivation in wastewater[J]. Journal of Alloys and Compounds,2021,870:159414. doi: 10.1016/j.jallcom.2021.159414 [5] LI F, HUANG Y, HUANG K, et al. Functional magnetic graphene composites for biosensing[J]. International Journal of Molecular Sciences,2020,21(2):390. doi: 10.3390/ijms21020390 [6] LI N, JIANG H L, WANG X L, et al. Recent advances in graphene-based magnetic composites for magnetic solid-phase extraction[J]. TrAC Trends in Analytical Chemistry,2018,102:60-74. doi: 10.1016/j.trac.2018.01.009 [7] AHANGARAN F, NAVARCHIAN A H. Recent advances in chemical surface modification of metal oxide nanoparticles with silane coupling agents: A review[J]. Advances in Colloid and Interface Science,2020,286:102298. doi: 10.1016/j.cis.2020.102298 [8] WANG C S, XU F, HE M, et al. Castor oil-based polyurethane/silica nanocomposites: Morphology, thermal and mechanical properties[J]. Polymer Composites,2018,39(S3):E1800-E1806. doi: 10.1002/pc.24798 [9] SHIMPI N, MALI A, HANSORA D P, et al. Synthesis and surface modification of calcium carbonate nanoparticles using ultrasound cavitation technique[J]. Nanoscience and Nanoengineering,2015,3(1):8-12. doi: 10.13189/nn.2015.030102 [10] ZHAO M, REN Z Z, YANG M B, et al. Effects of modified nano-silica on the microstructure of PVDF and its microporous membranes[J]. Journal of Polymer Research,2019,26(2):28. doi: 10.1007/s10965-018-1681-5 [11] SAEN P, ATAI M, NODEHI A, et al. Physical characterization of unfilled and nanofilled dental resins: Static versus dynamic mechanical properties[J]. Dental Materials,2016,32(8):e185-e197. doi: 10.1016/j.dental.2016.06.001 [12] DENG X J, LIN K, CHEN X P, et al. Preparation of magnetic Fe3O4/Au composites for extraction of benzo[a]pyrene from aqueous solution[J]. Chemical Engineering Journal,2013,225:656-663. doi: 10.1016/j.cej.2013.04.004 [13] 袁鹏园, 袁梅, 刘宇, 等. 钢铁基体上KH792改性TEOS聚硅氧薄膜的制备及其耐蚀性能[J]. 材料保护, 2018, 51(11):75-79. doi: 10.16577/j.cnki.42-1215/tb.2018.11.016YUAN Pengyuan, YUAN Mei, LIU Yu, et al. Preparation and corrosion resistance of polysilicon oxide conversion film modified by KH792 on steel matrix[J]. Materials Protection,2018,51(11):75-79(in Chinese). doi: 10.16577/j.cnki.42-1215/tb.2018.11.016 [14] 周书威, 傅红, 薛晓康, 等. 基于晶体生长法的磁性石墨化碳黑的制备及在茶叶农药残留检测中的应用[J]. 分析化学, 2022, 50(3):472-481. doi: 10.19756/j.issn.0253-3820.210844ZHOU Shuwei, FU Hong, XUE Xiaokang, et al. Preparation of magnetic graphitized carbon black based on crystal growth method and its application in detection of pesticide residues in tea[J]. Chinese Journal of Analytical Chemistry,2022,50(3):472-481(in Chinese). doi: 10.19756/j.issn.0253-3820.210844 [15] ZHENG H B, ZHAO Q, MO J Z, et al. Quick, easy, cheap, effective, rugged and safe method with magnetic graphitized carbon black and primary secondary amine as adsorbent and its application in pesticide residue analysis[J]. Journal of Chromatography A,2013,1300:127-133. doi: 10.1016/j.chroma.2013.04.040 [16] YU C C, HAO D Y, CHU Q, et al. A one adsorbent QuEChERS method coupled with LC-MS/MS for simultaneous determination of 10 organophosphorus pesticide residues in tea[J]. Food Chemistry,2020,321:126657. doi: 10.1016/j.foodchem.2020.126657 [17] HAYWARD D G, WONG J W, SHI F, et al. Multiresidue pesticide analysis of botanical dietary supplements using salt-out acetonitrile extraction, solid-phase extraction cleanup column, and gas chromatography-triple quadrupole mass spectrometry[J]. Analytical Chemistry,2013,85(9):4686-4693. doi: 10.1021/ac400481w [18] CUI H C, ZHANG J J, LU J J, et al. Research on modification of Fe3O4 magnetic nanoparticles with two silane coupling agents[J]. Magnetochemistry,2022,9(1):1-13. doi: 10.3390/magnetochemistry9010001 [19] QI P P, WANG Z W, YANG G L, et al. Removal of acidic interferences in multi-pesticides residue analysis of fruits using modified magnetic nanoparticles prior to determination via ultra-HPLC-MS/MS[J]. Microchimica Acta,2015,182(15):2521-2528. [20] 冯雨琛. 硅烷偶联剂改性Kevlar纤维-水泥基材料性能研究[D]. 北京: 北京建筑大学, 2022.FENG Yuchen. Study on properties of Kevlar fiber-cement based material modified by silane coupling agent[D]. Beijing: Beijing University of Civil Engineering and Architecture, 2022(in Chinese). [21] YAMAURA M, CAMILO R L, SAMPAIO L C, et al. Preparation and characterization of (3-aminopropyl)triethoxysilane-coated magnetite nanoparticles[J]. Journal of Magnetism and Magnetic Materials,2004,279(2-3):210-217. doi: 10.1016/j.jmmm.2004.01.094 [22] 胡玮, 娄兆文. 四氧化三铁磁性纳米微粒表面的氨基化修饰[J]. 化学研究, 2013, 24(2):144-148. doi: 10.3969/j.issn.1008-1011.2013.02.007HU Wei, LOU Zhaowen. Amination surface-modification of ferriferous oxide magnetic nanoparticles[J]. Chemical Research,2013,24(2):144-148(in Chinese). doi: 10.3969/j.issn.1008-1011.2013.02.007 [23] 中华人民共和国国家质量监督检验检疫总局. 茶叶中茶多酚和儿茶素类含量的检测方法: GB/T 8313—2008[S]. 北京: 中国标准出版社, 2008.General Administration of Quality Supervision, Inspection and Quarantine of the People's Republic of China. Determination of total polyphenols and catechins content in tea: GB/T 8313—2008[S]. Beijing: Standards Press of China, 2008(in Chinese). [24] KHABIBULLIN A, BHANGAONKAR K, MAHONEY C, et al. Grafting PMMA brushes from α-alumina nanoparticles via SI-ATRP[J]. ACS Applied Materials & Interfaces,2016,8(8):5458-5465. [25] WILKOWSKA A, BIZIUK M. Determination of pesticide residues in food matrices using the QuEChERS methodology[J]. Food Chemistry,2011,125(3):803-812. doi: 10.1016/j.foodchem.2010.09.094 [26] 李媛, 肖乐辉, 周乃元, 等. 在茶叶农药残留测定中用四氧化三铁纳米粒子去除样品中的色素[J]. 分析化学, 2013, 41(1):63-68.LI Yuan, XIAO Lehui, ZHOU Naiyuan, et al. Purification of pigments by iron oxide nanoparticles for analysis of pesticide residues in tea[J]. Chinese Journal of Analytical Chemistry,2013,41(1):63-68(in Chinese). [27] SAKEYE M, SMÅTT J H. Comparison of different amino-functionalization procedures on a selection of metal oxide microparticles: Degree of modification and hydrolytic stability[J]. Langmuir,2012,28(49):16941-16950. doi: 10.1021/la303925x [28] 王帆, 张金才, 赵沛祯, 等. 分散剂对煅烧高岭土和钛白粉分散性的影响[J]. 非金属矿, 2023, 46(1):55-59. doi: 10.3969/j.issn.1000-8098.2023.01.014WANG Fan, ZHANG Jincai, ZHAO Peizhen, et al. Effect of dispersant on dispersibility of calcined Kaolin and titanium dioxide[J]. Non-Metallic Mines,2023,46(1):55-59(in Chinese). doi: 10.3969/j.issn.1000-8098.2023.01.014 [29] WANJERI V W O, SHEPPARD C J, PRINSLOO A R E, et al. Isotherm and kinetic investigations on the adsorption of organophosphorus pesticides on graphene oxide based silica coated magnetic nanoparticles functionalized with 2-phenylethylamine[J]. Journal of Environmental Chemical Engineering,2018,6(1):1333-1346. doi: 10.1016/j.jece.2018.01.064 [30] 刘约权. 现代仪器分析[M]. 北京: 高等教育出版社, 2014: 110-111.LIU Yuequan. Modern instrumental analysis[M]. Beijing: Higher Education Press, 2014: 110-111(in Chinese). [31] YAMASHITA T, HAYES P. Analysis of XPS spectra of Fe2+ and Fe3+ ions in oxide materials[J]. Applied Surface Science,2008,254(8):2441-2449. doi: 10.1016/j.apsusc.2007.09.063 [32] MATINLINNA J P, LUNG C Y K, TSOI J K H. Silane adhesion mechanism in dental applications and surface treatments: A review[J]. Dental Materials,2018,34(1):13-28. doi: 10.1016/j.dental.2017.09.002 [33] FENG B, HONG R Y, WANG L S, et al. Synthesis of Fe3O4/APTES/PEG diacid functionalized magnetic nanoparticles for MR imaging[J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects,2008,328(1-3):52-59. [34] RAHMAN O U, MOHAPATRA S C, AHMAD S. Fe3O4 inverse spinal super paramagnetic nanoparticles[J]. Materials Chemistry and Physics,2012,132(1):196-202. doi: 10.1016/j.matchemphys.2011.11.032 [35] RAHIMI R, MALEKI A, MALEKI S. Preparation of magnetic fluorochromate hybrid nanomaterials with triphenylphosphine surface modified iron oxide nanoparticles and their characterization[J]. Journal of Magnetism and Magnetic Materials,2014,355:300-305. doi: 10.1016/j.jmmm.2013.12.021 [36] BRINKER C J. Hydrolysis and condensation of silicates: Effects on structure[J]. Journal of Non-Crystalline Solids,1988,100(1-3):31-50. [37] MALLAKPOUR S, MADANI M. A review of current coupling agents for modification of metal oxide nanoparticles[J]. Progress in Organic Coatings,2015,86:194-207. doi: 10.1016/j.porgcoat.2015.05.023 -






 下载:
下载: