Research progress of non-noble metal catalysts based on electrocatalytic oxygen evolution reaction
-
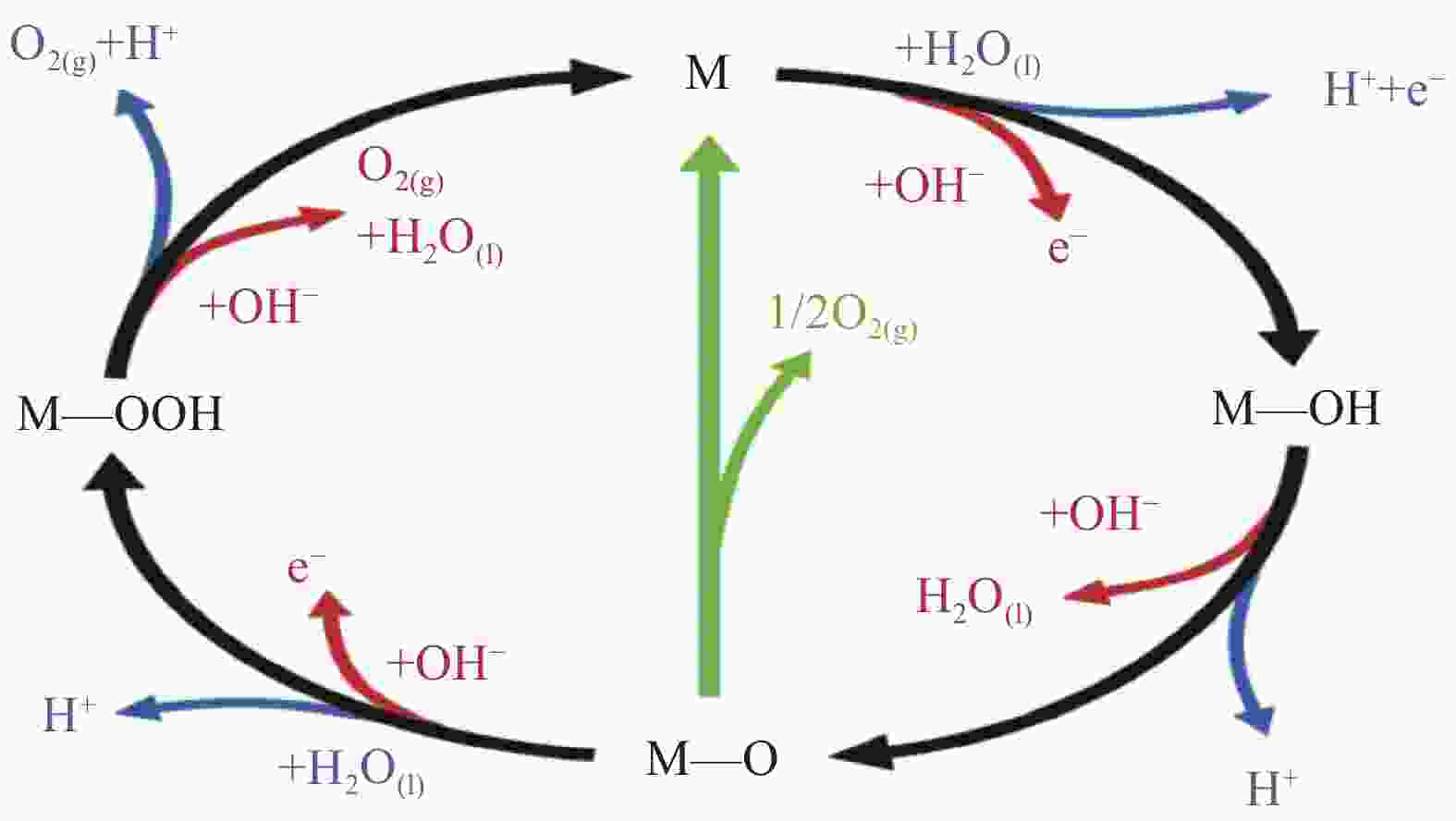
摘要: 在全球变暖和能源危机的背景下,能源问题已成为全球各国战略安全的重要组成部分。氢能作为可持续的新型可再生清洁能源,对缓解全球性能源短缺具有重要意义。在众多制氢候选方案中,电解水制备氢气被认为是最可靠、最可行的途径之一。但在电解过程中,反应动力学极为迟缓的阳极析氧反应(Oxygen evolution reaction,OER)严重制约着整体反应效率。因此,开发成本相对低廉、催化剂性能优异、耐久性好的高效OER电催化剂,从而提高电解水制氢工艺技术的能源转换效果受到了广泛关注。本文首先简要阐述了析氧反应的反应机制及其性能的评价参数,接着对非贵金属催化剂的研究进行分类讨论,并列举了提高催化性能的策略和方法,最后对设计新型催化剂进行展望。Abstract: In the context of global warming and energy crisis, energy issues have become an important part of the strategic security of countries around the world. As a sustainable new renewable clean energy, hydrogen energy is of great significance to alleviate the global energy shortage. Among many hydrogen production candidate schemes, electrolysis of water to produce hydrogen is considered to be one of the most reliable and feasible ways. However, in the electrolysis process, the anodic oxygen evolution reaction (OER) with extremely slow reaction kinetics seriously restricts the overall reaction efficiency. Therefore, the development of high-efficiency OER electrocatalyst with relatively low cost, excellent catalyst performance and good durability, so as to improve the energy conversion effect of electrolytic water hydrogen production technology, has attracted widespread attention. This paper first briefly expounds the reaction mechanism of oxygen evolution reaction and the evaluation parameters of its performance, then discusses the research of non-noble metal catalysts, lists the strategies and methods to improve the catalytic performance, and finally prospects the design of new catalysts.
-
Key words:
- electrocatalysis /
- oxygen evolution reaction /
- non-noble metals /
- transition metals /
- composite
-
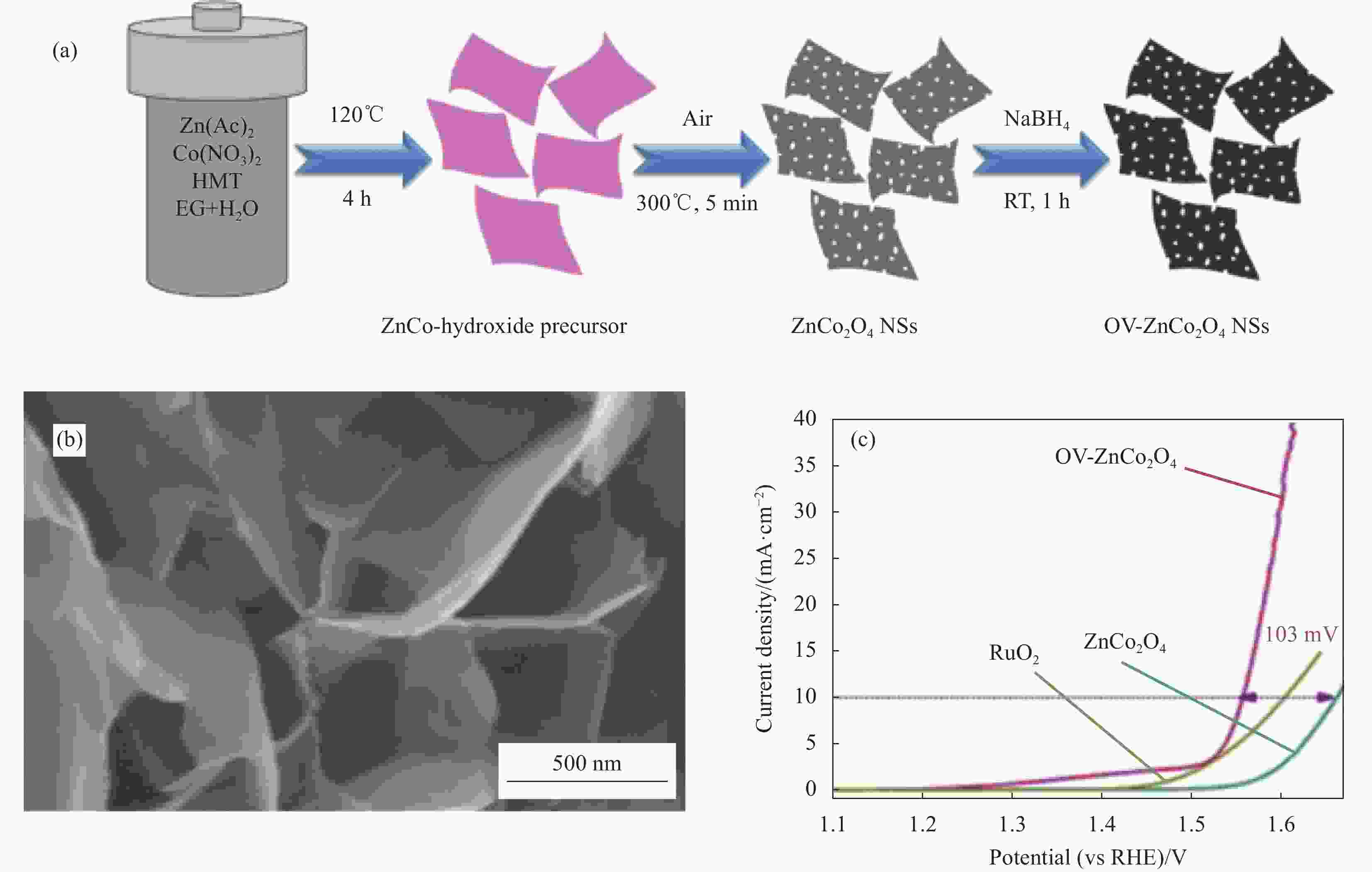
图 5 (a) 含氧空位ZnCo2O4纳米片(OV-ZnCo2O4)合成流程示意图;(b) OV-ZnCo2O4纳米片的SEM图像;(c) ZnCo2O4纳米片、OV-ZnCo2O4纳米片、RuO2的极化曲线[40]
Figure 5. (a) Schematic diagram of synthesis process of oxygen-containing vacancy ZnCo2O4 nanosheets (OV-ZnCo2O4); (b) SEM image of OV-ZnCo2O4 nanosheets; (c) Polarization curves of ZnCo2O4 nanosheets, OV-ZnCo2O4 nanosheets and RuO2[40]
HMT—Hexamethylenetetramine; RT—Room temperature; EG—Ethylene glycol; NSs—Nanosheets
图 7 (a) Ni-Fe层状双氢氧化物(NixFey-LDH)合成流程示意图;(b) Ni2Fe1-LDH(Ni/Fe原子比为2:1)的SEM图像;(c) 不同Ni-Fe比例的氢氧化物与IrO2的极化曲线;(d) 相应的Tafel曲线[45]
Figure 7. (a) Schematic diagram of synthesis process of Ni-Fe layered double hydroxide (NixFey-LDH); (b) SEM image of Ni2Fe1-LDH (Ni/Fe atom ratio of 2:1); (c) Polarization curves of hydroxides with different Ni-Fe ratios and IrO2; (d) Corresponding Tafel curves[45]
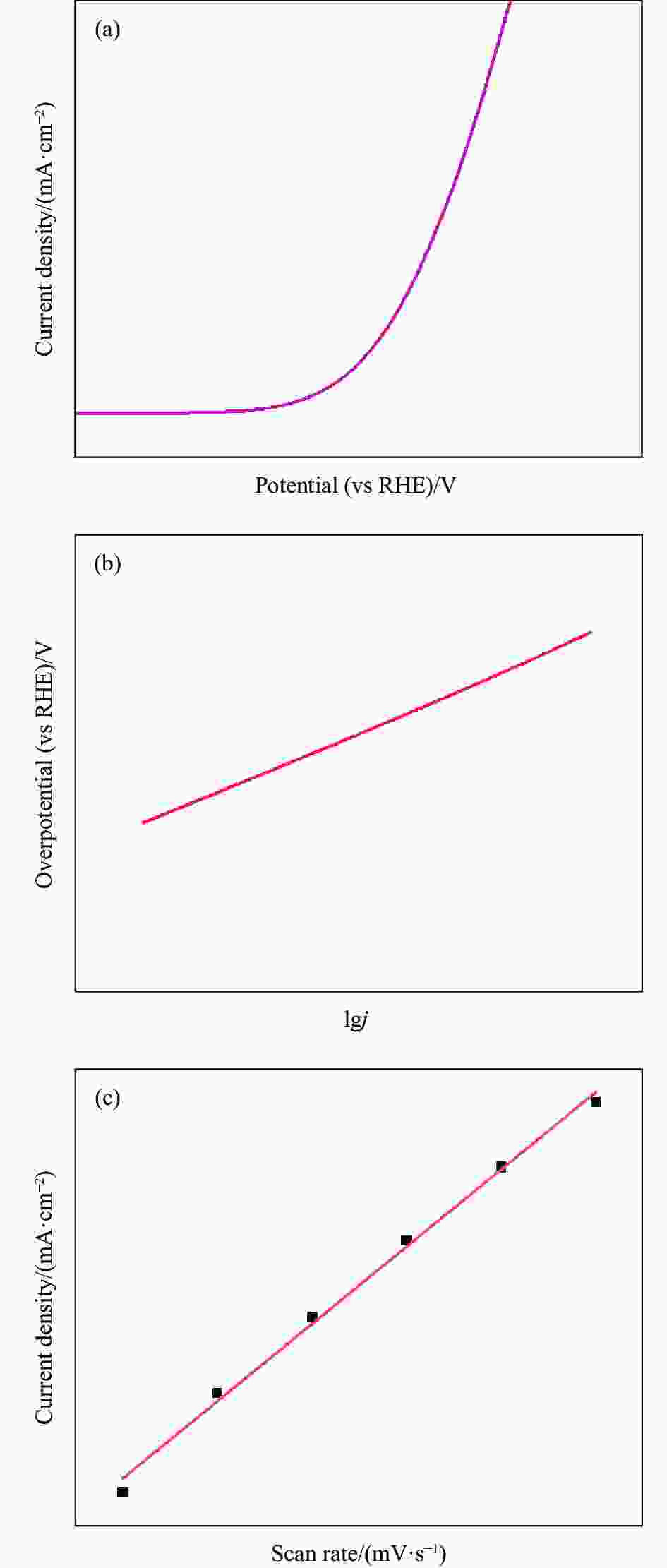
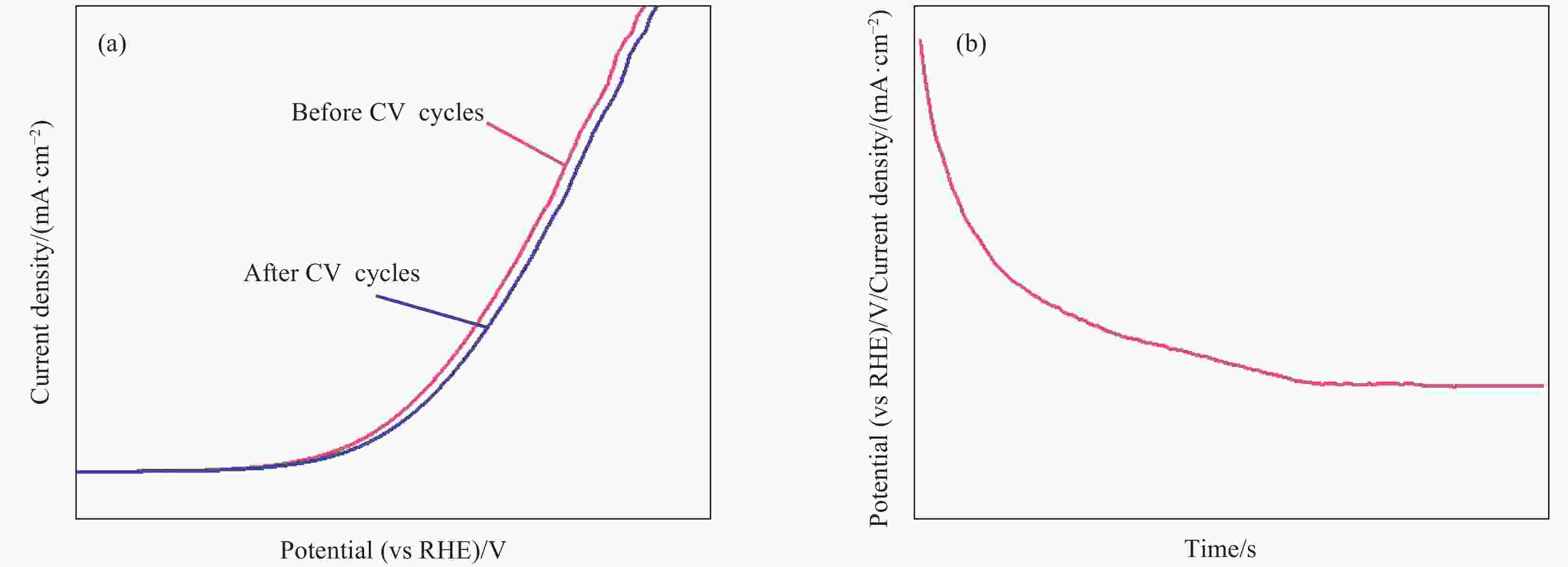
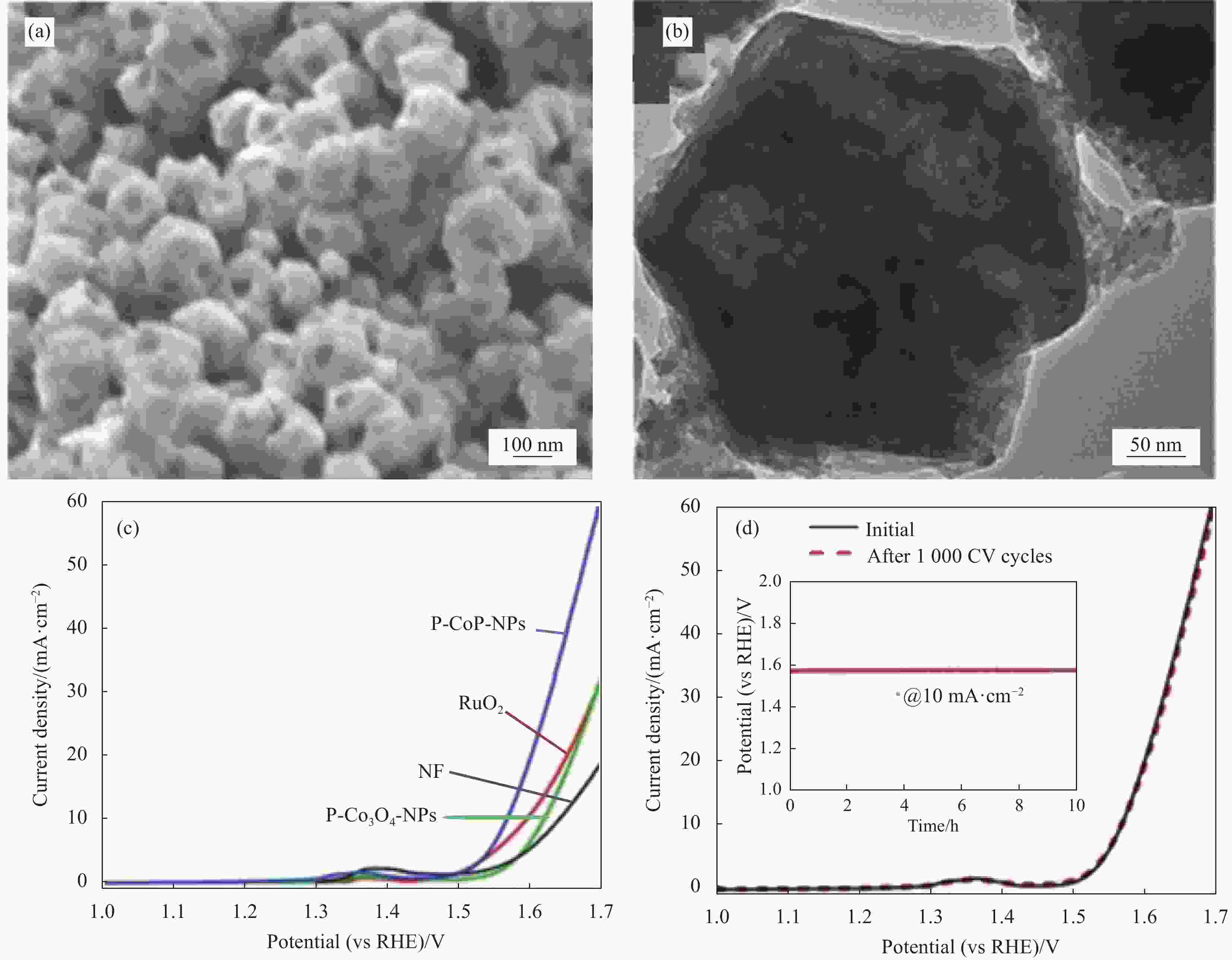
图 8 ((a), (b)) 多孔磷化钴纳米颗粒(P-CoP-NPs)的SEM图像及TEM图像;(c) 磷化后的多孔Co3O4纳米颗粒(P-Co3O4-NPs)、P-CoP-NPs、泡沫镍(NF)与RuO2的极化曲线;(d) 1000次CV循环前后P-CoP-NPs的极化曲线(插图:10 mA·cm-2电流密度下P-CoP-NPs的计时电位曲线)[49]
Figure 8. ((a), (b)) SEM image and TEM image of porous cobalt phosphide nanoparticles (P-CoP-NPs); (c) Polarization curves of porous Co3O4 nanoparticles after phosphating (P-Co3O4-NPs), P-CoP-NPs, nickle foam (NF) and RuO2; (d) Polarization curve of P-CoP-NPs before and after 1000 CV cycles (Inset: Chronopotentiometric curve of P-CoP-NPs at the current density of 10 mA·cm-2)[49]
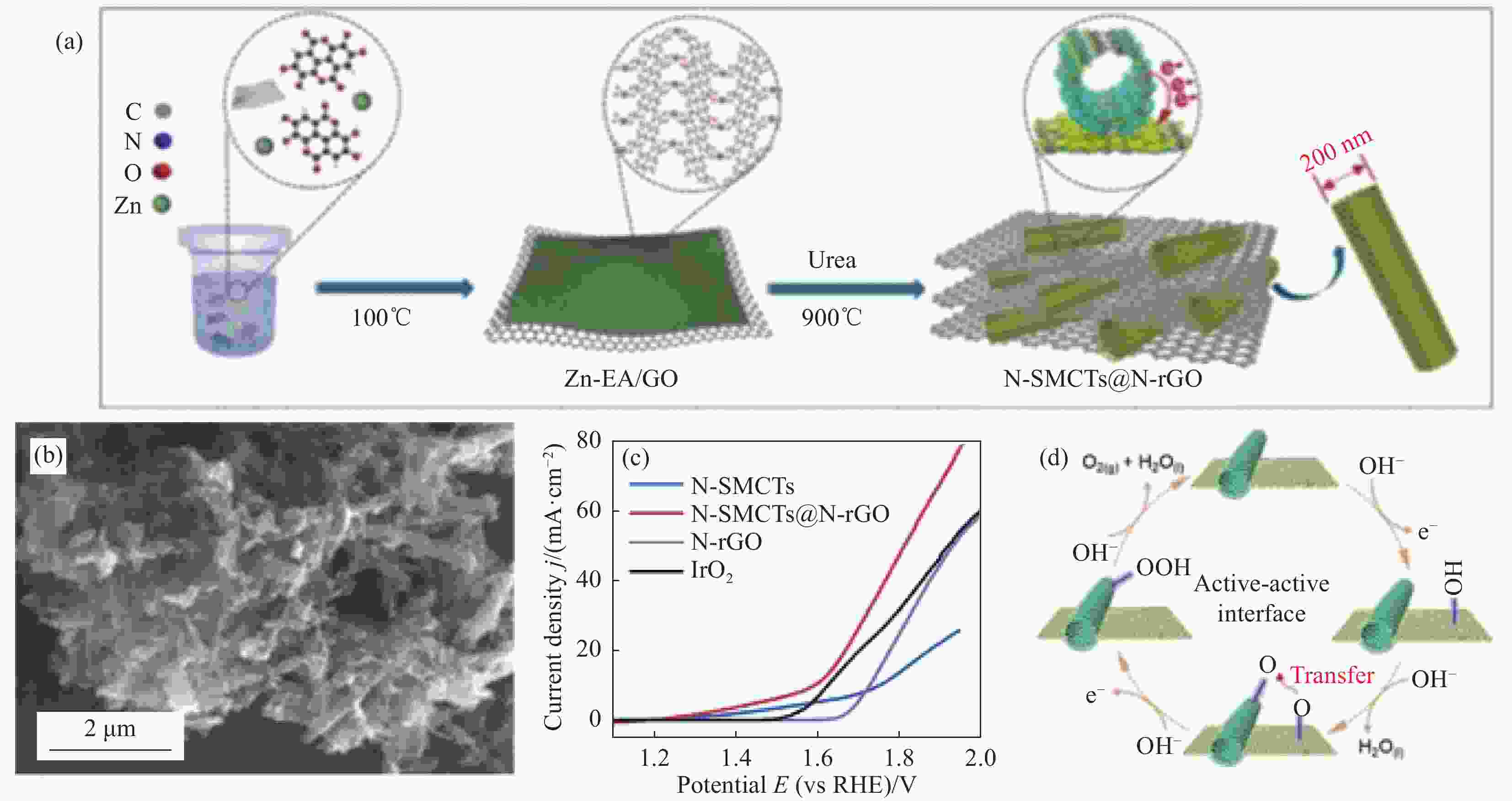
图 9 (a) N掺杂亚微米碳管@N掺杂还原氧化石墨烯(N-SMCTs@N-rGO)合成流程示意图;(b) N-SMCTs@N-rGO的SEM图像;(c) N-SMCTs、N-rGO、N-SMCTs@N-rGO与IrO2的极化曲线;(d) 活性界面双位点机制图[56]
Figure 9. (a) Schematic diagram of the synthesis process of N-doped submicron carbon tube@N-doped reduced graphene oxide (N-SMCTs@N-rGO); (b) SEM image of N-SMCTs@N-rGO; (c) Polarization curves of N-SMCTs, N-rGO, N-SMCTs@N-rGO and IrO2; (d) Mechanism diagram of active-active interface with dual-site mechanism[56]
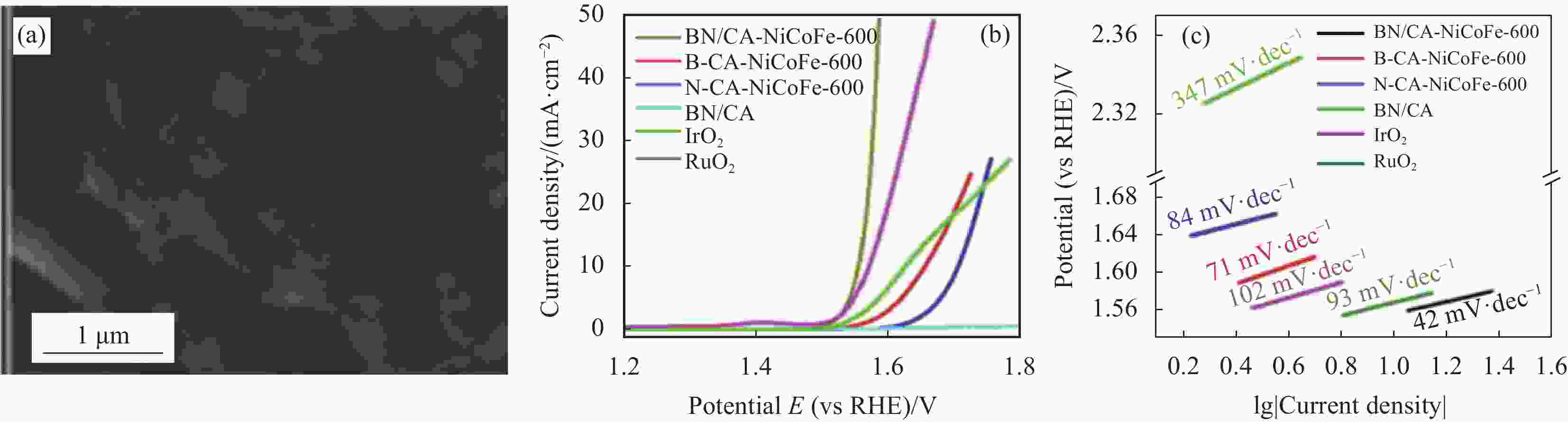
图 10 (a) NiCoFe合金锚定的硼氮共掺杂碳气凝胶(BN/CA-NiCoFe-600)的SEM图像;(b) BN/CA-NiCoFe-600、NiCoFe合金锚定的硼掺杂碳气凝胶(B-CA-NiCoFe-600)、NiCoFe合金锚定的氮掺杂碳气凝胶(N-CA-NiCoFe-600)、氮化硼碳气凝胶(BN/CA)、IrO2 和 RuO2的极化曲线;(c) 相应的Tafel曲线[68]
Figure 10. (a) SEM image of B, N co-doped carbon aerogel anchored with NiCoFe alloy (BN/CA-NiCoFe-600); (b) Polarization curves of BN/CA-NiCoFe-600, B doped carbon aerogel anchored with NiCoFe alloy (B-CA-NiCoFe-600), N doped carbon aerogel anchored with NiCoFe alloy (N-CA-NiCoFe-600), boron nitride carbon aeroge (BN/CA), IrO2 and RuO2; (c) Corresponding Tafel curves[68]
表 1 催化剂的电催化OER活性
Table 1. Electrocatalytic OER activity of catalyst
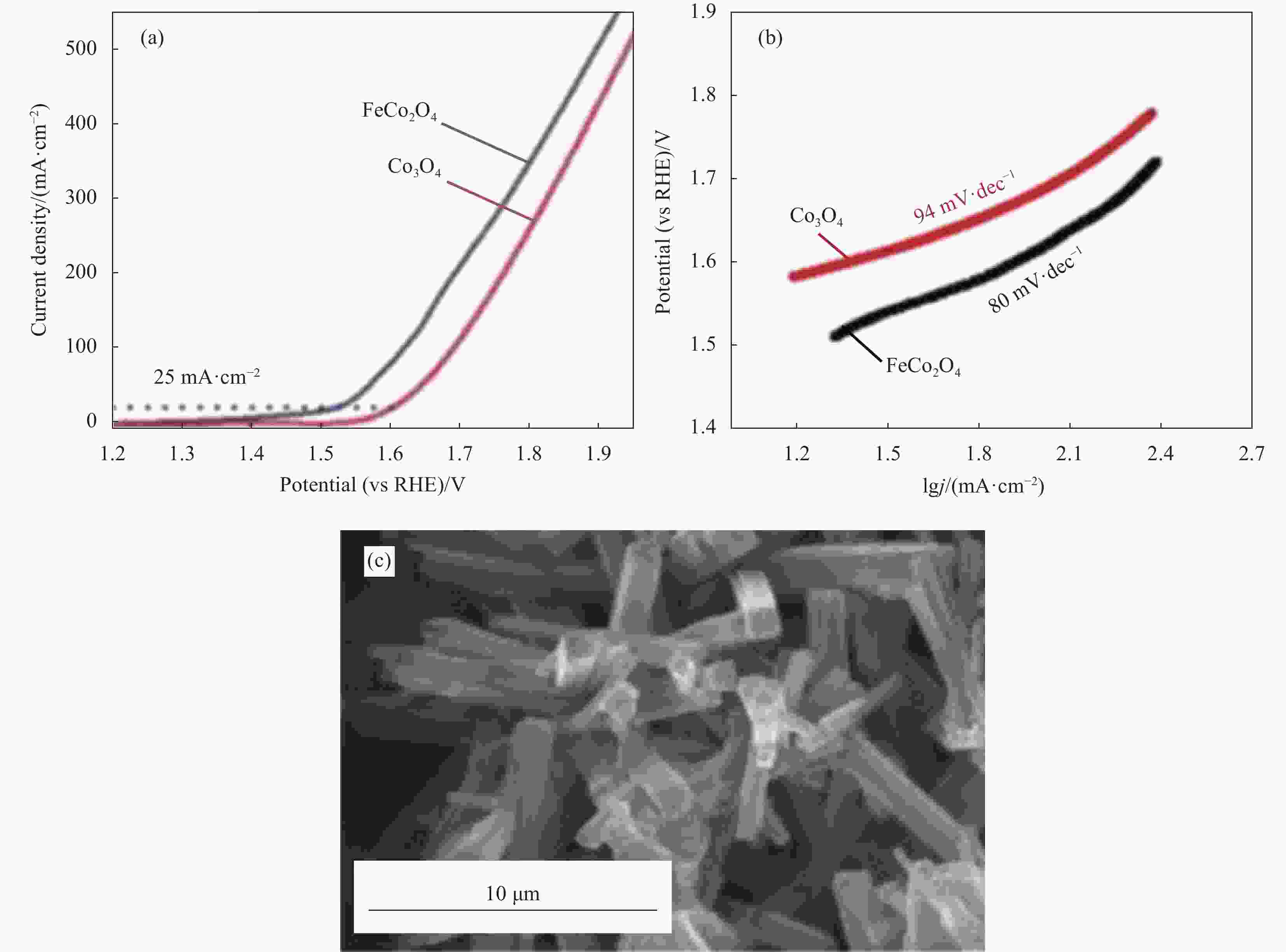
Catalyst Electrolyte Overpotential at a specific current density Tafel slope/(mV·dec−1) Reference NiCo2O4-OV-400 1.0 mol/L KOH 325 mV @ 10 mA·cm−2 71 [38] OV-ZnCo2O4 0.1 mol/L KOH 324 mV @ 10 mA·cm−2 56.9 [40] CuCo2O4/NF 1.0 mol/L KOH 220 mV @ 10 mA·cm−2 92.5 [41] FeCo2O4 1.0 mol/L KOH 290 mV @ 25 mA·cm−2 80 [42] Ni2Fe1-LDHs 1.0 mol/L KOH 245 mV @ 10 mA·cm−2 57 [45] Fe-CoNi-LDHs 1.0 mol/L KOH 260 mV @ 10 mA·cm−2 70 [46] Fe-Co-LDHs/GS 1.0 mol/L KOH 370 mV @ 10 mA·cm−2 44 [47] CoSx/Co 1.0 mol/L KOH 284 mV @ 10 mA·cm−2 75.8 [48] P-CoP-NPs-350 1.0 mol/L KOH 320 mV @ 10 mA·cm−2 73.7 [49] W-Fe-Ni-B/NF 1.0 mol/L KOH 223 mV @ 10 mA·cm−2 38.3 [50] T-GO 0.5 mol/L KOH 176 mV @ 2 mA·cm−2 69 [53] CNT@NCNT 0.1 mol/L KOH 260 mV @ 10 mA·cm−2 − [54] N-MWCNTs 1.0 mol/L NaOH 320 mV @ 10 mA·cm−2 68 [55] N-SMCTs@N-rGO 1.0 mol/L KOH 351 mV @ 10 mA·cm−2 − [56] D-NSOCs 1.0 mol/L KOH 350 mV @ 10 mA·cm−2 69 [61] N, S-MWCNTs 1.0 mol/L KOH 360 mV @ 10 mA·cm−2 56 [62] PN-CC 1.0 mol/L KOH 340 mV @ 10 mA·cm−2 54.9 [63] PA-PPy/CC 1.0 mol/L KOH 340 mV @ 10 mA·cm−2 54.9 [64] 1D g-CN 1.0 mol/L KOH 316 mV @ 10 mA·cm−2 125 [65] BRP-g-C3N4 1.0 mol/L KOH 430 mV @ 10 mA·cm−2 93 [66] Ni-MWCNTs 1.0 mol/L KOH 320 mV @ 10 mA·cm−2 30.08 [67] BN/CA-NiCoFe-600 1.0 mol/L KOH 321 mV @ 10 mA·cm−2 42 [68] Co-CoO/BC 1.0 mol/L KOH 300 mV @ 10 mA·cm−2 73.3 [69] Co-NCNT 1.0 mol/L KOH 310 mV @ 10 mA·cm−2 74.67 [70] Notes: OV—Oxygen-containing vacancy; CNT—Carbon nanotube; MWCNTs—Multiwalled carbon nanotube; NF—Nickel foam; GS—Graphene sheet; BRP—Black red phosphorus; g-C3N4—Graphite phase carbon nitride; T-GO—Amine group functionalizes graphene oxide; g-CN—Graphitic carbon nitride nanorods; NCNT—Nitrogen-doped carbon nanotube; D-NSOCs—N and S co-doped and oxygen-functionalized carbon materials; PN-CC—Plasma and acid treated carbon cloth. -
[1] CHEN Y, YU Y. Research on the relationship between China's energy consumption and economic growth based on big data[J]. Fresenius Environmental Bulletin,2021,30(9):10652-10658. [2] 王则鉴, 洪佳佳, NG Sue-faye, 等. 氧化物钙钛矿的光催化研究进展: CO2还原、水裂解、固氮[J]. 物理化学学报, 2021, 37(6):84-114.WANG Zejian, HONG Jiajia, NG Sue-faye, et al. Recent progress of perovskite oxide in emerging photocatalysis landscape: Water splitting, CO2 reduction, and N2 fixation[J]. Acta Physico-Chimica Sinica,2021,37(6):84-114(in Chinese). [3] QADIR S A, AL-MOTAIRI H, TAHIR F, et al. Incentives and strategies for financing the renewable energy transition: A review[J]. Energy Reports,2021,7:3590-3606. doi: 10.1016/j.egyr.2021.06.041 [4] BORETTI A. Continued fossil fuel emissions and cognition impairment[J]. International Journal of Global Warming,2021,24(1):86-90. doi: 10.1504/IJGW.2021.115116 [5] OGUNKUNLE O, AHMED N A. Overview of biodiesel combustion in mitigating the adverse impacts of engine emissions on the sustainable human-environment scenario[J]. Sustainability,2021,13(10):5465. doi: 10.3390/su13105465 [6] PRYKHODKO Y, FATYEYEVA K, HESPEL L, et al. Progress in hybrid composite nafion (R)-based membranes for proton exchange fuel cell application[J]. Chemical Engineering Journal,2021,409:127329. doi: 10.1016/j.cej.2020.127329 [7] MEGIA P J, VIZCAINO A J, CALLES J A, et al. Hydrogen production technologies: From fossil fuels toward renewable sources. A mini review[J]. Energy & Fuels,2021,35(20):16403-16415. [8] HAEGEL N, KURTZ S. Global progress toward renewable electricity: Tracking the role of solar[J]. IEEE Journal of Photovoltaics,2021,11(6):1335-1342. doi: 10.1109/JPHOTOV.2021.3104149 [9] PENG X K, LIU Z C, JIANG D. A review of multiphase energy conversion in wind power generation[J]. Renewable & Sustainable Energy Reviews,2021,147:111172. [10] ZHAN L, BO Y, LIN T, et al. Development and outlook of advanced nuclear energy technology[J]. Energy Strategy Reviews,2021,34:100630. doi: 10.1016/j.esr.2021.100630 [11] AMJITH L R, BAVANISH B. A review on biomass and wind as renewable energy for sustainable environment[J]. Chemosphere,2022,293:133579. doi: 10.1016/j.chemosphere.2022.133579 [12] BUNDSCHUH J, KACZMARCZYK M, GHAFFOUR N, et al. State-of-the-art of renewable energy sources used in water desalination: Present and future prospects[J]. Desalination,2021,508:115035. doi: 10.1016/j.desal.2021.115035 [13] YANG Y C, JAVANROODI K, NIK V M. Climate change and renewable energy generation in Europe-long-term impact assessment on solar and wind energy using high-resolution future climate data and considering climate uncertainties[J]. Energies,2022,15(1):302. doi: 10.3390/en15010302 [14] JIANG S Q, SUO H L, ZHANG T, et al. Recent advances in seawater electrolysis[J]. Catalysts,2022,12(2):123. doi: 10.3390/catal12020123 [15] CUI P Z, YAO D, MA Z Y, et al. Life cycle water footprint comparison of biomass-to-hydrogen and coal-to-hydrogen processes[J]. Science of the Total Environment,2021,773:145056. doi: 10.1016/j.scitotenv.2021.145056 [16] NGUYEN H L. Metal-organic frameworks for photocatalytic water splitting[J]. Solar RRL,2021,5(7):2100198. doi: 10.1002/solr.202100198 [17] LI Z X, HU M L, WANG P, et al. Heterojunction catalyst in electrocatalytic water splitting[J]. Coordination Che-mistry Reviews,2021,439:213953. doi: 10.1016/j.ccr.2021.213953 [18] LIU X M, CUI X Y, DASTAFKAN K, et al. Recent advances in spinel-type electrocatalysts for bifunctional oxygen reduction and oxygen evolution reactions[J]. Journal of Energy Chemistry,2021,53:290-302. doi: 10.1016/j.jechem.2020.04.012 [19] 李孟婷, 郑星群, 李莉, 等. 碱性介质中氢氧化和析氢反应机理研究现状[J]. 物理化学学报, 2021, 37(9):21-37.LI Mengting, ZHENG Xingqun, LI Li, et al. Research progress of hydrogen oxidation and hydrogen evolution reaction mechanism in alkaline media[J]. Acta Physico-Chimica Sinica,2021,37(9):21-37(in Chinese). [20] LIANG Q H, BROCKS G, BIEBERLE-HUTTER A. Oxygen evolution reaction (OER) mechanism under alkaline and acidic conditions[J]. Journal of Physics-Energy,2021,3(2):026001. doi: 10.1088/2515-7655/abdc85 [21] SUEN N T, HUNG S F, QUAN Q, et al. Electrocatalysis for the oxygen evolution reaction: Recent development and future perspectives[J]. Chemical Society Reviews,2017,46(2):337-365. doi: 10.1039/C6CS00328A [22] WANG J J, YUE X Y, YANG Y Y, et al. Earth-abundant transition-metal-based bifunctional catalysts for overall electrochemical water splitting: A review[J]. Journal of Alloys and Compounds,2020,819:153346. doi: 10.1016/j.jallcom.2019.153346 [23] SINGH C, LIBERMAN I, SHIMONI R, et al. Pristine versus pyrolyzed metal-organic framework-based oxygen evolution electrocatalysts: Evaluation of intrinsic activity using electrochemical impedance spectroscopy[J]. Journal of Physical Chemistry Letters,2019,10(13):3630-3636. doi: 10.1021/acs.jpclett.9b01232 [24] TAHIR M, PAN L, IDREES F, et al. Electrocatalytic oxygen evolution reaction for energy conversion and storage: A comprehensive review[J]. Nano Energy,2017,37:136-157. doi: 10.1016/j.nanoen.2017.05.022 [25] JAMESH M I, HARB M. Tuning the electronic structure of the earth-abundant electrocatalysts for oxygen evolution reaction (OER) to achieve efficient alkaline water splitting-A review[J]. Journal of Energy Chemistry,2021,56:299-342. doi: 10.1016/j.jechem.2020.08.001 [26] ANANTHARAJ S, EDE S R, KARTHICK K, et al. Precision and correctness in the evaluation of electrocatalytic water splitting: Revisiting activity parameters with a critical assessment[J]. Energy & Environmental Science,2018,11(4):744-771. [27] CAI Z Y, BU X M, WANG P, et al. Recent advances in layered double hydroxide electrocatalysts for the oxygen evolution reaction[J]. Journal of Materials Chemistry A,2019,7(10):5069-5089. doi: 10.1039/C8TA11273H [28] CAO L M, LU D, ZHONG D C, et al. Prussian blue analogues and their derived nanomaterials for electrocatalytic water splitting[J]. Coordination Chemistry Reviews,2020,407:213156. doi: 10.1016/j.ccr.2019.213156 [29] HU W K, LIU Q, LV T X, et al. Impact of interfacial CoOOH on OER catalytic activities and electrochemical behaviors of bimetallic CoxNi-LDH nanosheet catalysts[J]. Electrochimica Acta,2021,381:138276. doi: 10.1016/j.electacta.2021.138276 [30] BENCK J D, HELLSTERN T R, KIBSGAARD J, et al. Catalyzing the hydrogen evolution reaction (HER) with molybdenum sulfide nanomaterials[J]. ACS Catalysis,2014,4(11):3957-3971. doi: 10.1021/cs500923c [31] ANANTHARAJ S, KARTHIK P E, KUNDU S. Petal-like hierarchical array of ultrathin Ni(OH)2 nanosheets decorated with Ni(OH)2 nanoburls: A highly efficient OER electrocatalyst[J]. Catalysis Science & Technology,2017,7(4):882-893. [32] CHEN F Y, WU Z Y, ADLER Z, et al. Stability challenges of electrocatalytic oxygen evolution reaction: From mecha-nistic understanding to reactor design[J]. Joule,2021,5(7):1704-1731. doi: 10.1016/j.joule.2021.05.005 [33] BOCKRIS J O, OTAGAWA T. The electrocatalysis of oxygen evolution on perovskites[J]. Journal of the Electrochemi-cal Society,1984,131(2):290-302. doi: 10.1149/1.2115565 [34] SUNTIVICH J, MAY K J, GASTEIGER H A, et al. A perovskite oxide optimized for oxygen evolution catalysis from molecular orbital principles[J]. Science,2011,334(6061):1383-1385. doi: 10.1126/science.1212858 [35] DIAS J A, ANDRADE M A S, SANTOS H L S, et al. Lanthanum-based perovskites for catalytic oxygen evolution reaction[J]. Chemelectrochem,2020,7(15):3173-3192. doi: 10.1002/celc.202000451 [36] MATIENZO D D, KUTLUSOY T, DIVANIS S, et al. Benchmarking perovskite electrocatalysts' OER activity as candidate materials for industrial alkaline water electrolysis[J]. Catalysts,2020,10(12):1387. doi: 10.3390/catal10121387 [37] LIU W, KAMIKO M, YAMADA I, et al. Electrochemical deposition of amorphous cobalt oxides for oxygen evolution catalysis[J]. RSC Advances,2022,12(14):8731-8736. doi: 10.1039/D2RA00492E [38] YAO S Z, WEI H S, ZHANG Y, et al. Controlled growth of porous oxygen-deficient NiCo2O4 nanobelts as high-efficiency electrocatalysts for oxygen evolution reaction[J]. Catalysis Science & Technology,2021,11(1):264-271. [39] HARADA M, KOTEGAWA F, KUWA M. Structural changes of spinel MCo2O4 (M=Mn, Fe, Co, Ni, and Zn) electrocatalysts during the oxygen evolution reaction investigated by in situ X-ray absorption spectroscopy[J]. ACS Applied Energy Materials,2022,5(1):278-294. doi: 10.1021/acsaem.1c02824 [40] XIANG K, WU D, FAN Y, et al. Enhancing bifunctional electrodes of oxygen vacancy abundant ZnCo2O4 nanosheets for supercapacitor and oxygen evolution[J]. Chemical Engineering Journal,2021,425:130583. doi: 10.1016/j.cej.2021.130583 [41] ZHANG P, HE H W. Rational rope-like CuCo2O4 nanosheets directly on ni foam as multifunctional electrodes for supercapacitor and oxygen evolution reaction[J]. Journal of Alloys and Compounds,2020,826:153993. doi: 10.1016/j.jallcom.2020.153993 [42] BABAR P, PATIL K, BHOITE P, et al. 1D iron cobaltite electrode for efficient electrochemical water oxidation[J]. Materials Letters,2022,312:131663. doi: 10.1016/j.matlet.2022.131663 [43] YU J, YU F, YUEN M F, et al. Two-dimensional layered double hydroxides as a platform for electrocatalytic oxygen evolution[J]. Journal of Materials Chemistry A,2021,9(15):9389-9430. doi: 10.1039/D0TA11910E [44] ZHOU D J, LI P S, LIN X, et al. Layered double hydroxide-based electrocatalysts for the oxygen evolution reaction: Identification and tailoring of active sites, and superaerophobic nanoarray electrode assembly[J]. Chemical Society Reviews,2021,50(15):8790-8817. doi: 10.1039/D1CS00186H [45] CHEN F W, ZHANG S N, LI J J, et al. Precursor-mediated synthesis of interconnected ultrathin NiFe-layered double hydroxides nanosheets for efficient oxygen evolution electrocatalysis[J]. Materials Letters,2022,309:131470. doi: 10.1016/j.matlet.2021.131470 [46] SHI Y L, LI J Q, ZHANG B Y, et al. Tuning electronic structure of CoNi LDHs via surface Fe doping for achieving effective oxygen evolution reaction[J]. Applied Surface Science,2021,565:150506. doi: 10.1016/j.apsusc.2021.150506 [47] ENSAFI A A, MANDIAN-TALKHOONCHE B, HEYDARI-SOURESHJANI E, et al. Graphene-like sheets supported Fe-Co layered double hydroxides nanoflakes as an efficient electrocatalyst for both hydrogen and oxygen evolution reaction, a green investigation[J]. Chemosphere,2022,299:134251. doi: 10.1016/j.chemosphere.2022.134251 [48] BIAN H D, CHEN T Y, CHEN Z X, et al. One-step synthesis of mesoporous cobalt sulfides (CoSx) on the metal substrate as an efficient bifunctional electrode for overall water splitting[J]. Electrochimica Acta,2021,389:138786. doi: 10.1016/j.electacta.2021.138786 [49] LI Y X, ZHANG Z Q, XIE M G, et al. A facile templating fabrication of porous CoP nanoparticles towards electrocatalytic oxygen evolution[J]. Applied Surface Science,2022,583:152402. doi: 10.1016/j.apsusc.2021.152402 [50] SUN Y H, ZHAO Y, DENG X Y, et al. An efficient amorphous ternary transition metal boride (WFeNiB) electrocatalyst for oxygen evolution from water[J]. Sustainable Energy & Fuels,2022,6(5):1345-1352. [51] WANG J, KONG H, ZHANG J Y, et al. Carbon-based electrocatalysts for sustainable energy applications[J]. Progress in Materials Science,2021,116:100717. doi: 10.1016/j.pmatsci.2020.100717 [52] LIU H M, LIU Q L, WANG Y R, et al. Bifunctional carbon-based cathode catalysts for zinc-air battery: A review[J]. Chinese Chemical Letters,2022,33(2):683-692. doi: 10.1016/j.cclet.2021.07.038 [53] SAPNER V S, MULIK B B, DIGRASKAR R, et al. Enhanced oxygen evolution reaction on amine functionalized graphene oxide in alkaline medium[J]. RSC Advances,2019,9(12):6444-6451. doi: 10.1039/C8RA10286D [54] TIAN G L, ZHANG Q, ZHANG B S, et al. Toward full expo-sure of "active sites": Nanocarbon electrocatalyst with surface enriched nitrogen for superior oxygen reduction and evolution reactivity[J]. Advanced Functional Materials,2014,24(38):5956-5961. doi: 10.1002/adfm.201401264 [55] PAVODI F, TAVAKKOLI M, LAHTINEN J, et al. Straightforward synthesis of nitrogen-doped carbon nanotubes as highly active bifunctional electrocatalysts for full water splitting[J]. Journal of Catalysis,2017,353:19-27. doi: 10.1016/j.jcat.2017.07.001 [56] ZHAO J, LI Q Q, ZHANG Q C, et al. Carbon tube-graphene heterostructure with different n-doping configurations induces an electrochemically active-active interface for efficient oxygen electrocatalysis[J]. Chemical Engineering Journal,2022,431:133730. doi: 10.1016/j.cej.2021.133730 [57] ARAFAT Y, AZHAR M R, ZHONG Y J, et al. Metal-free carbon based air electrodes for zn-air batteries: Recent advances and perspective[J]. Materials Research Bulletin,2021,140:111315. doi: 10.1016/j.materresbull.2021.111315 [58] THAKUR A K, KURTYKA K, MAJUMDER M, et al. Recent advances in boron- and nitrogen-doped carbon-based materials and their various applications[J]. Advanced Materials Interfaces,2022,9(11):2101964. doi: 10.1002/admi.202101964 [59] CHATTOPADHYAY J, PATHAK T S, PAK D. Heteroatom-doped metal-free carbon nanomaterials as potential electrocatalysts[J]. Molecules,2022,27(3):670. doi: 10.3390/molecules27030670 [60] LU Y, SHI H J, SU Y F, et al. Application of element-doped carbonaceous materials in lithium-sulfur batteries[J]. Progress in Chemistry,2021,33(9):1598-1613. [61] ZHOU S Y, ZANG J B, GAO H W, et al. Deflagration method synthesizing N, S co-doped oxygen-functionalized carbons as a bifunctional catalyst for oxygen reduction and oxygen evolution reaction[J]. Carbon,2021,181:234-245. doi: 10.1016/j.carbon.2021.05.034 [62] QU K, ZHENG Y, JIAO Y, et al. Polydopamine-inspired, dual heteroatom-doped carbon nanotubes for highly efficient overall water splitting[J]. Advanced Energy Materials,2016,7(9):1602068. [63] MEI T T, GAO M, LIU D N, et al. Enhanced electrocatalytic activity of carbon cloth by synergetic effect of plasma and acid treatment[J]. Plasma Science & Technology,2021,23(2):025504. [64] HU Q, LI G M, LIU X F, et al. Superhydrophilic phytic-acid-doped conductive hydrogels as metal-free and binder-free electrocatalysts for efficient water oxidation[J]. Angewandte Chemie-International Edition,2019,58(13):4318-4322. doi: 10.1002/anie.201900109 [65] DESALEGN B Z, JADHAV H S, SEO J G. Highly efficient g-C3N4 nanorods with dual active sites as an electrocatalyst for the oxygen evolution reaction[J]. Chemcatchem,2019,11(12):2870-2878. doi: 10.1002/cctc.201900330 [66] SHIRAVANI F, TASHKHOURIAN J, HAGHIGHI B. One-step synthesis of graphitic carbon-nitride doped with black-red phosphorus as a novel, efficient and free-metal bifunctional catalyst and its application for electrochemical overall water splitting[J]. Sustainable Energy & Fuels,2021,5(12):3229-3239. [67] KANG S, HAN H, MHIN S, et al. Ni-doped carbon nano-tubes fabricated by pulsed laser ablation in liquid as efficient electrocatalysts for oxygen evolution reaction[J]. Applied Surface Science,2021,547:149197. doi: 10.1016/j.apsusc.2021.149197 [68] LU R Q, SAM D K, WANG W B, et al. Boron, nitrogen co-doped biomass-derived carbon aerogel embedded nickel-cobalt-iron nanoparticles as a promising electrocatalyst for oxygen evolution reaction[J]. Journal of Colloid and Interface Science,2022,613:126-135. doi: 10.1016/j.jcis.2022.01.029 [69] YANG M J, WU D F, CHENG D J. Biomass-derived porous carbon supported Co-CoO yolk-shell nanoparticles as enhanced multifunctional electrocatalysts[J]. International Journal of Hydrogen Energy,2019,44(13):6525-6534. doi: 10.1016/j.ijhydene.2019.01.155 [70] SINGH S K, TAKEYASU K, PAUL B, et al. CoOx electro-catalysts anchored on nitrogen-doped carbon nanotubes for the oxygen evolution reaction[J]. Sustainable Energy & Fuels,2021,5(3):820-827. [71] CUI H J, ZHOU Z, JIA D Z. Heteroatom-doped graphene as electrocatalysts for air cathodes[J]. Materials Horizons,2017,4(1):7-19. doi: 10.1039/C6MH00358C [72] TANG C, ZHANG Q. Nanocarbon for oxygen reduction electrocatalysis: Dopants, edges, and defects[J]. Advanced Materials,2017,29(13):1604103. doi: 10.1002/adma.201604103 [73] ZHANG Y, ZHU X J, ZHANG G L, et al. Rational catalyst design for oxygen evolution under acidic conditions: Strategies toward enhanced electrocatalytic performance[J]. Journal of Materials Chemistry A,2021,9(10):5890-5914. doi: 10.1039/D0TA11982B [74] KARMAKAR A, KARTHICK K, SANKAR S S, et al. A vast exploration of improvising synthetic strategies for enhancing the OER kinetics of LDH structures: A review[J]. Journal of Materials Chemistry A,2021,9(3):1314-1352. doi: 10.1039/D0TA09788H [75] ZHU K Y, SHI F, ZHU X F, et al. The roles of oxygen vacancies in electrocatalytic oxygen evolution reaction[J]. Nano Energy,2020,73:104761. doi: 10.1016/j.nanoen.2020.104761 [76] CHAUHAN D S, QURAISHI M A, ANSARI K R, et al. Graphene and graphene oxide as new class of materials for corrosion control and protection: Present status and future scenario[J]. Progress in Organic Coatings,2020,147:105741. doi: 10.1016/j.porgcoat.2020.105741 [77] NASREEN F, ANWAR A W, AHMAD M A, et al. A facile improved oxidation method for ecological production of graphene oxide[J]. Digest Journal of Nanomaterials and Biostructures,2021,16(1):119-124. [78] DONNET J B, DHAMI T L, DONG S, et al. Microwave plasma treatment effect on the surface-energy of carbon-fibers[J]. Journal of Physics D-Applied Physics,1987,20(3):269-275. doi: 10.1088/0022-3727/20/3/005 [79] 张海宝, 陈强. 非热等离子体材料表面处理及功能化研究进展[J]. 物理学报, 2021, 70(9):22-38.ZHANG H B, CHEN Q. Recent progress of non-thermal plasma material surface treatment and functionalization[J]. Acta Physica Sinica,2021,70(9):22-38(in Chinese). [80] TIAN W L, ZHENG D C, SUN X, et al. Structural and interfacial engineering of Ni2P/Fe3O4 porous nanosheet arrays for efficient oxygen evolution reaction[J]. Inorganic Che-mistry,2021,60(19):14786-14792. doi: 10.1021/acs.inorgchem.1c02028 [81] WEISSENBERGER T, MACHOKE A G F, REIPRICH B, et al. Preparation and potential catalytic applications of hierarchically structured zeolites with macropores[J]. Advanced Materials Interfaces,2021,8(4):2001653. doi: 10.1002/admi.202001653 [82] TACHIBANA N, IKEDA S, YUKAWA Y, et al. Highly porous nitrogen-doped carbon nanoparticles synthesized via simple thermal treatment and their electrocatalytic activity for oxygen reduction reaction[J]. Carbon,2017,115:515-525. doi: 10.1016/j.carbon.2017.01.034 -





 下载:
下载: