Preparation of fibrous magnetic titania composites and their adsorption behavior for La3+
-
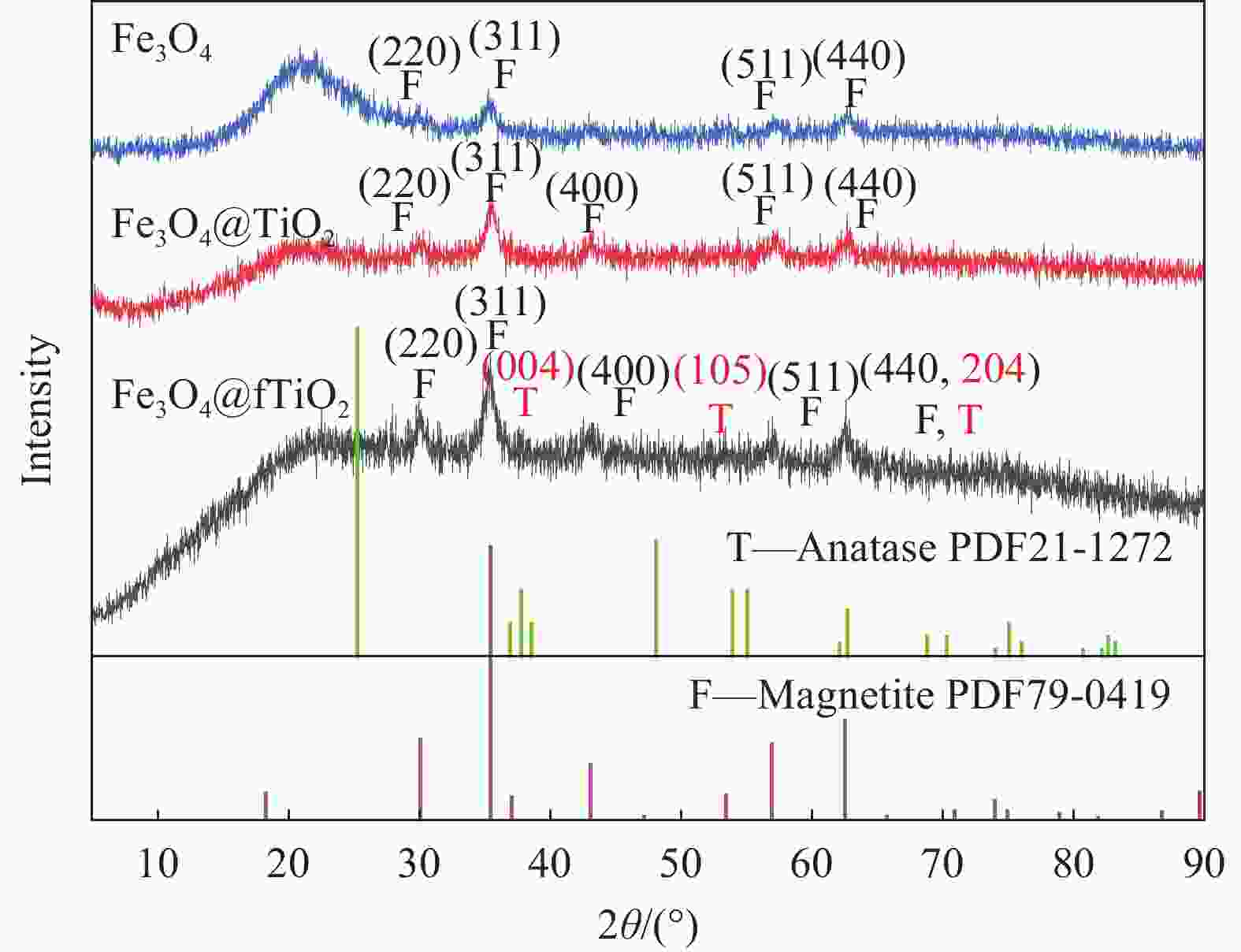
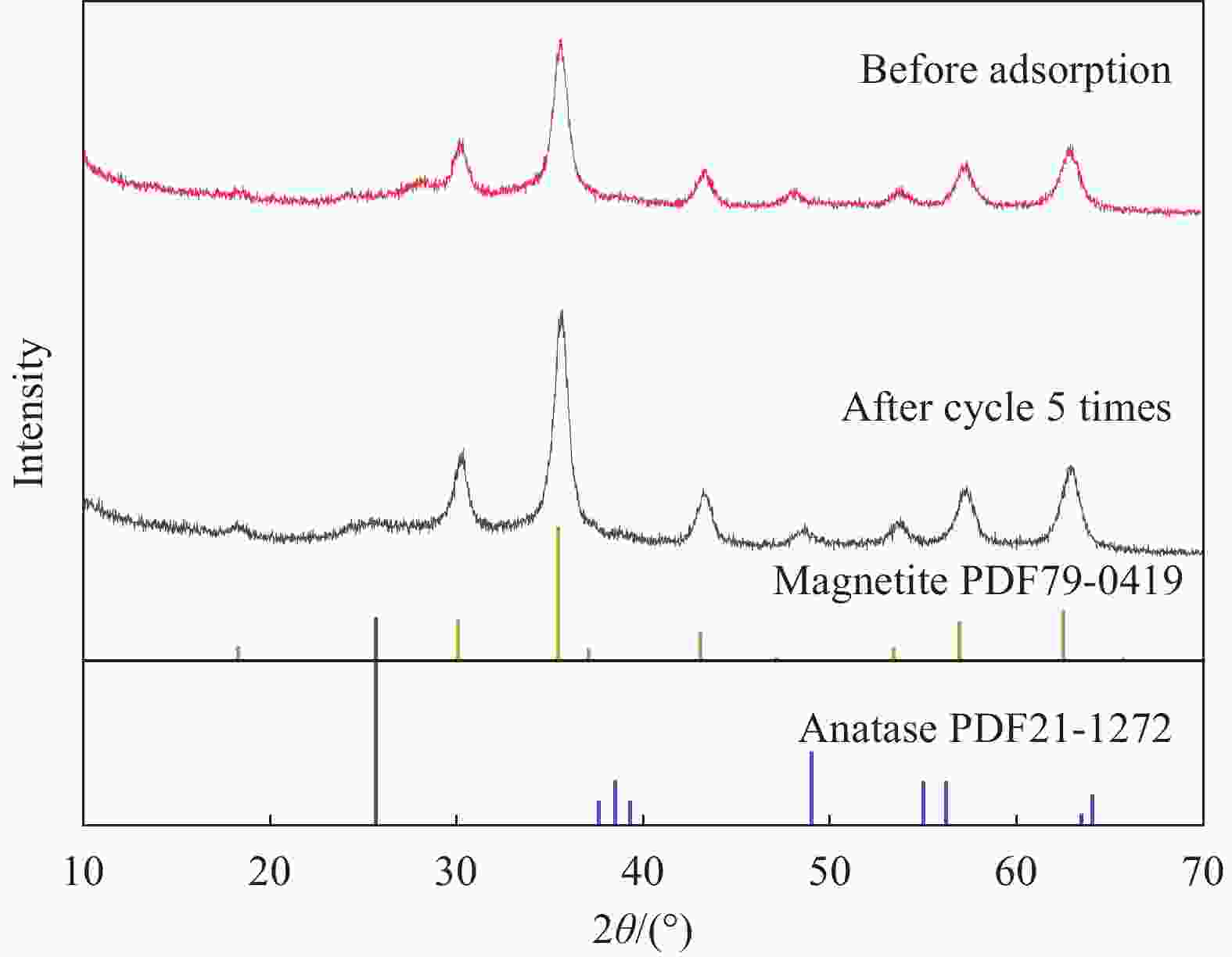
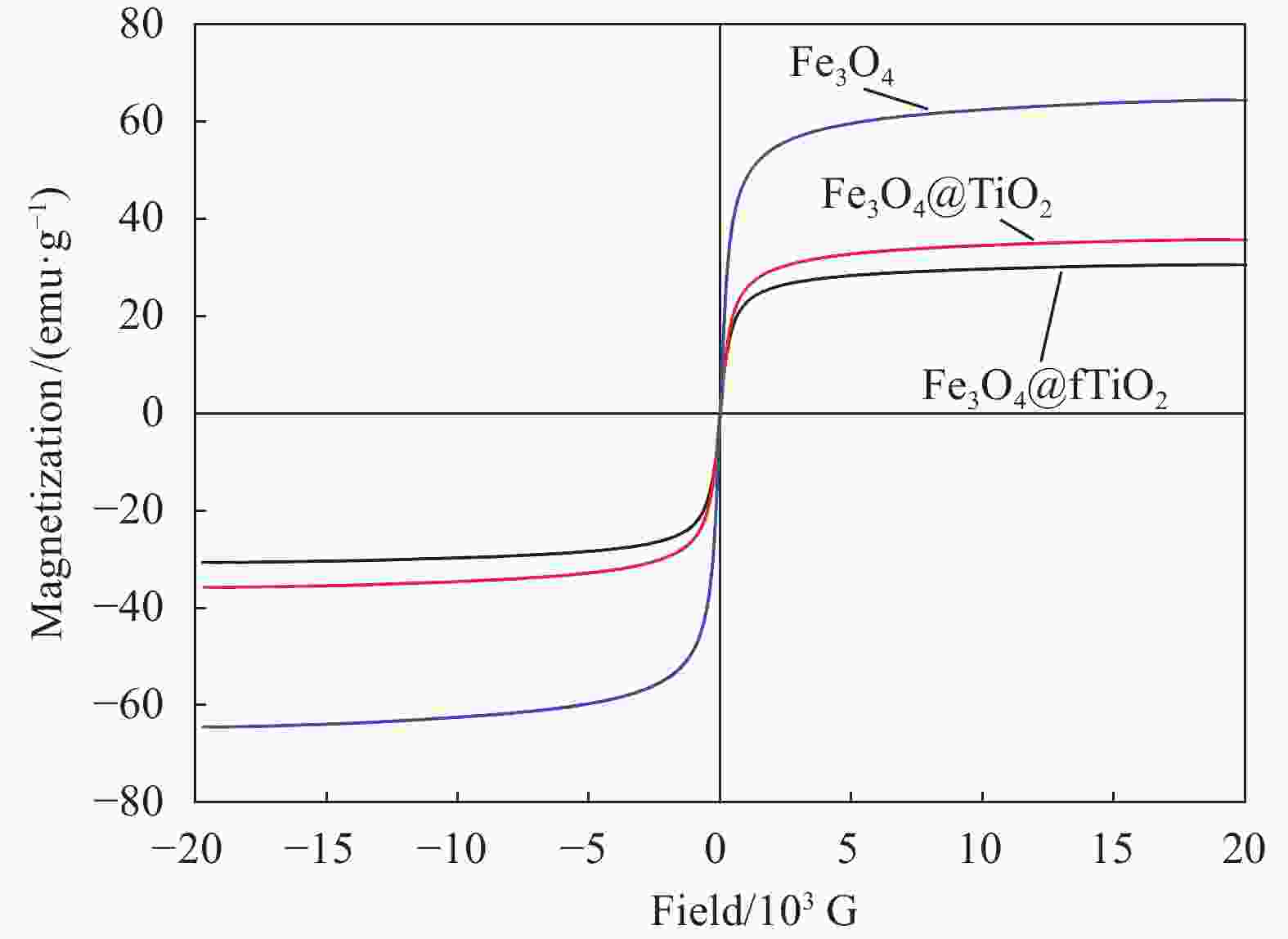
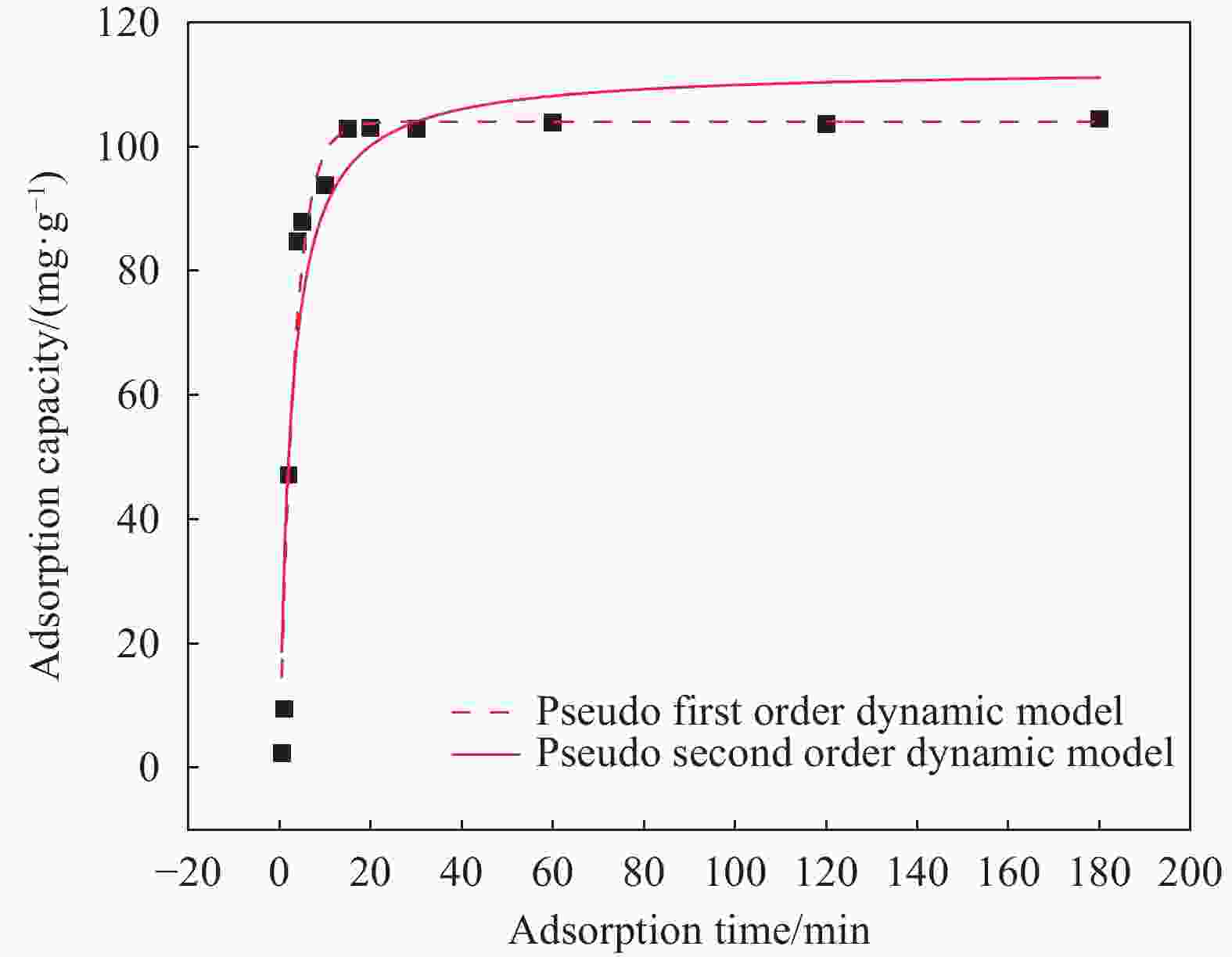
摘要: 为富集回收低浓度矿山尾水中稀土资源,采用溶胶-凝胶法和水热法制备了纤维状外壳的磁性二氧化钛复合材料Fe3O4@fTiO2,利用SEM、TEM、XPS、FTIR和XRD对材料进行表征,考察了Fe3O4@fTiO2对稀土La3+的吸附行为。结果表明:Fe3O4@fTiO2是外壳为纤维状的核壳结构磁性复合材料;吸附剂具有良好的超顺磁性,饱和磁化强度高达30.81 emu·g−1;在15℃、pH=5的酸性条件下,Fe3O4@fTiO2对稀土La3+在15 min内达到吸附平衡,且符合伪一级动力学模型;Langmuir等温吸附模型能较好地描述吸附La3+过程,理论吸附容量为142.88 mg·g−1;Fe3O4@fTiO2在NaOH的再生下循环利用5次时的吸附量为110 mg·g−1,是首次吸附容量的73.8%,表现出良好的循环利用性。Abstract: In order to enrich and recover rare earth resources in low-concentration mine tail water, a fibrous shell magnetic titanium dioxide composite Fe3O4@fTiO2 was prepared by using sol-gel method and hydrothermal methods. The material was analyzed by SEM, TEM, XPS, FTIR and XRD. The adsorption behavior of Fe3O4@fTiO2 for rare earth La3+ was investigated. The results show that Fe3O4@fTiO2 is a core-shell magnetic composite with a fibrous shell. The adsorbent has good superparamagnetic properties, and the saturation magnetization is as high as 30.81 emu·g−1. At pH=5 and 15℃, Fe3O4@fTiO2 reaches the adsorption equilibrium for rare earth La3+ within 15 min, and conformes to the pseudo-first-order kinetic model. The Langmuir isotherm adsorption model can describe the adsorption process of La3+ well, with the theoretical adsorption capacity of 142.88 mg·g−1. With NaOH solution as the regeneration reagent, the adsorption capacity of Fe3O4@fTiO2 is 110 mg·g−1 after cyclic adsorption/desorption for 5 times, which is 73.8% of the initial value, showing the good cyclic utilization.
-
Key words:
- fibrous /
- magnetic /
- titanium dioxide /
- adsorption /
- rare earth
-
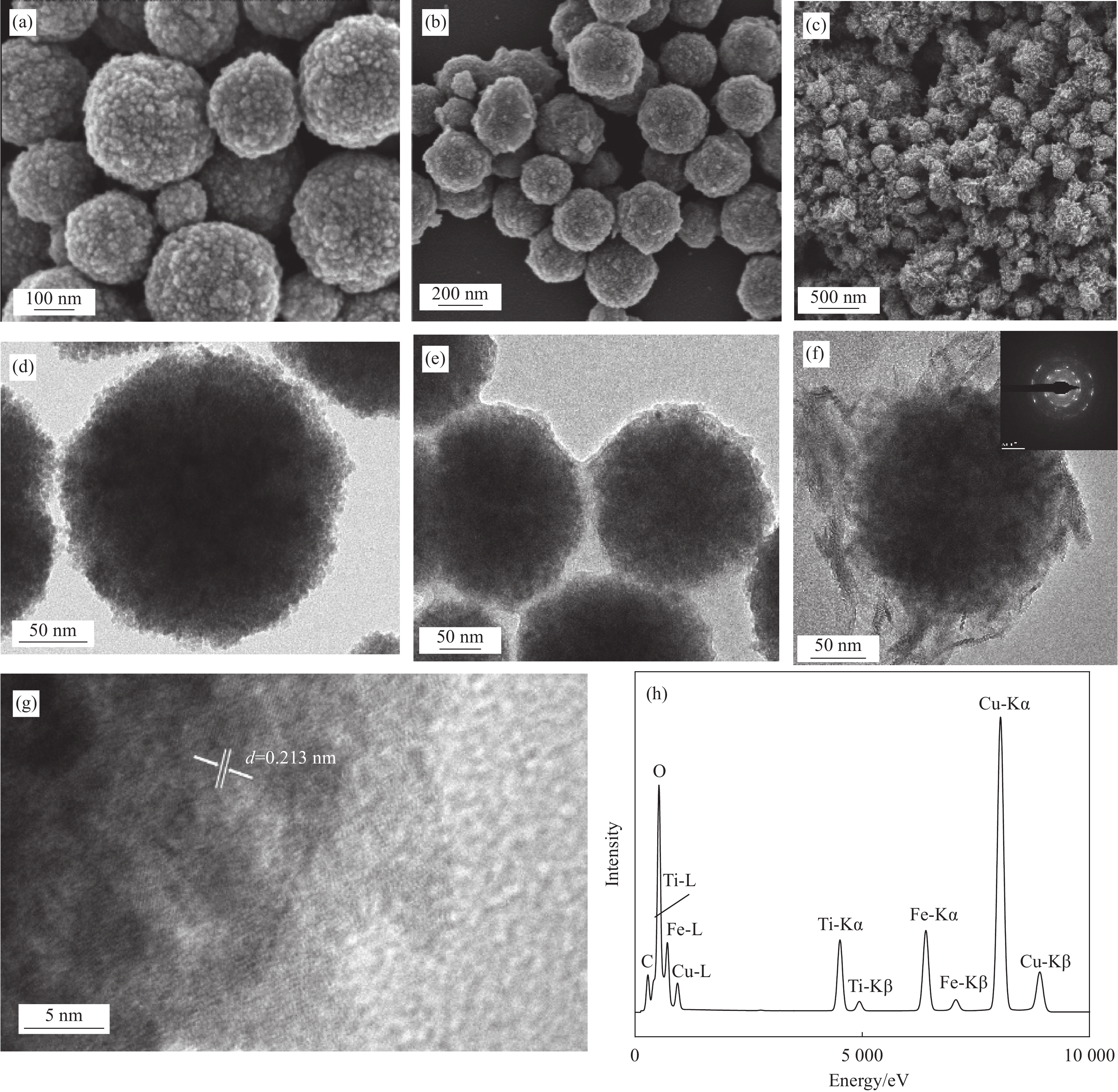
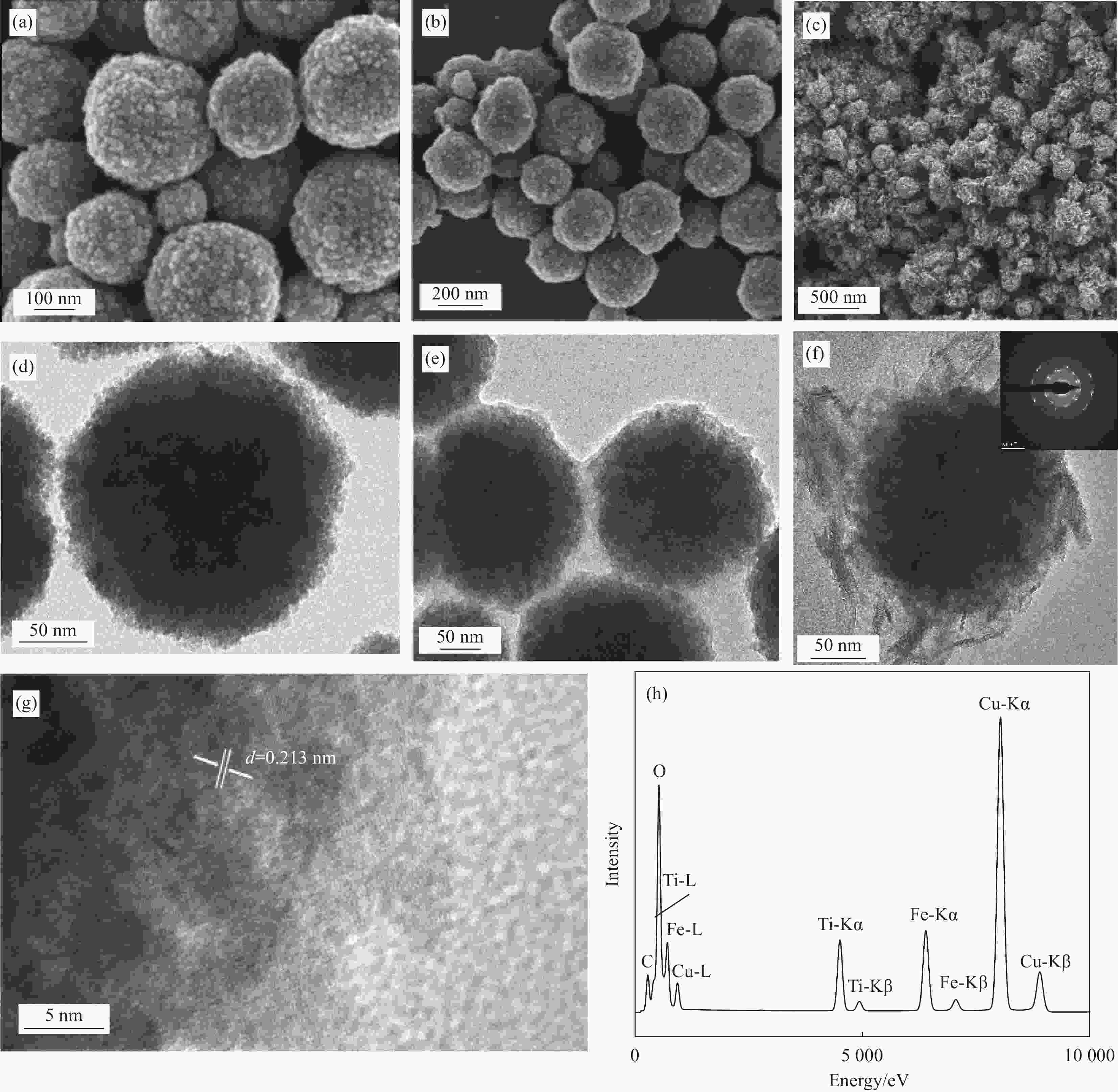
图 1 Fe3O4 ((a), (d))、Fe3O4@TiO2 ((b), (e)) 和Fe3O4@fTiO2 ((c), (f)) 的SEM及TEM图像;(g) Fe3O4@fTiO2的HRTEM图像;(h) Fe3O4@fTiO2的EDS图谱
d—Interlayer spacing
Figure 1. SEM and TEM images of Fe3O4 ((a), (d)), Fe3O4@TiO2 ((b), (e)) and Fe3O4@fTiO2 ((c), (f)); (g) HRTEM image of Fe3O4@fTiO2; (h) EDS pattern of Fe3O4@fTiO2
表 1 Fe3O4@fTiO2吸附La3+动力学模型的拟合参数
Table 1. Fitting parameters for kinetic models of La3+ adsorption onto Fe3O4@fTiO2
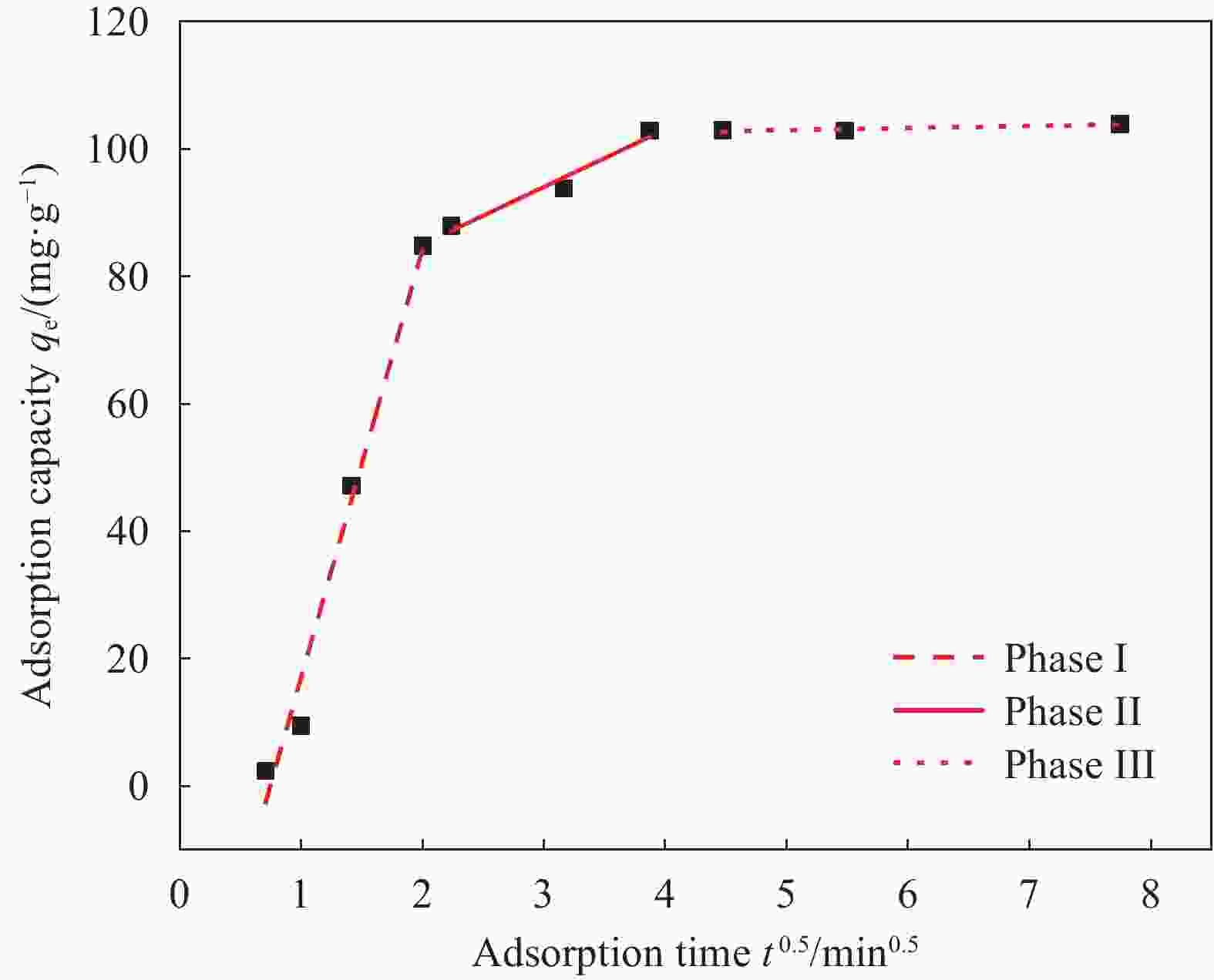
Model k/min−1 qe/(mg·g−1) R2 Pseudo-first-order 0.3036 104.001 0.957 Pseudo-second-order 0.0036 112.663 0.912 Notes: k—Pseudo-first-order kinetic and Pseudo-second-order kinetic constants; R2—Correlation coefficient. 表 2 Fe3O4@fTiO2吸附La3+颗粒的内扩散模型的拟合参数
Table 2. Intra-particle diffusion model parameters of adsorption La3+ on Fe3O4@fTiO2
Model Parameter kid/(mg·min1/2·g−1) cid/(mg·g−1) R2 Boundary layer
diffusion stage67.12 −49.99 0.980 Intragranular
diffusion stage9.01 67.01 0.962 Equilibrium stage 0.33 101.30 0.870 Notes: kid—Intraparticle diffusion rate constant; cid—Constant related to boundary layer thickness. 表 3 Fe3O4@fTiO2在不同pH下上清液的离子组分
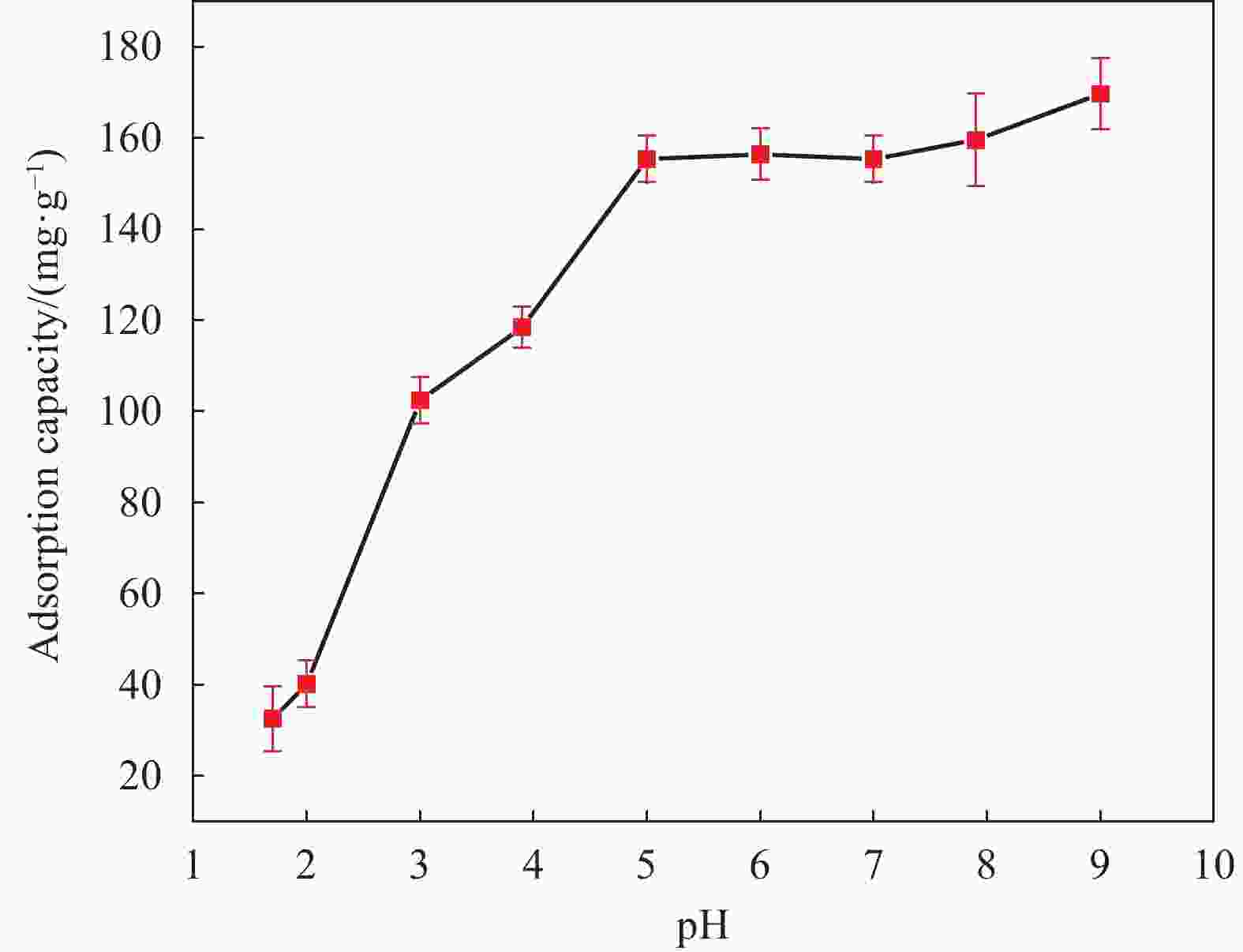
Table 3. Ionic components of supernatants of Fe3O4@fTiO2 at different pH
mg/L pH Fe Ti Na La 2 0.93 1.72 25.40 259.82 4 − 0.12 48.50 181.58 6 − 0.13 44.50 143.58 表 4 Fe3O4@fTiO2吸附La3+等温线模型的拟合参数
Table 4. Fitting parameters for isotherm models of La3+ adsorption onto Fe3O4@fTiO2
Langmuir Freundlich KL/min−1 qm/(mg·g−1) R2 Kf/(mg·g−1) n R2 0.01846 142.88 0.979 18.6652 3.276 0.937 Notes: KL—Langmuir coefficient of distribution of the adsorption; qm—Langmuir adsorption maximum; Kf—Freundlich coefficient of distribution of the adsorption; n—Freundlich constant related to adsorption strength. 表 5 La3+在Fe3O4@fTiO2上吸附的热力学参数
Table 5. Thermodynamic parameters of adsorption of La3+ on Fe3O4@fTiO2
Temperature
T/KKT ΔGθ/
(kJ·mol−1)ΔHθ/
(kJ·mol−1)ΔSθ/
(J·mol−1·K−1)288 1.42×105 −28.41 14.88 149.82 303 1.59×105 −30.18 318 2.57×105 −32.94 Notes: KT—A dimensionless parameter calculated using the Langmuir constant KL according to equation (11); ΔGθ—Gibbs free energy; ΔHθ—Enthalpy; ΔSθ—Entropy. 表 6 Fe3O4@fTiO2吸附La3+前后表面元素的原子分数参数
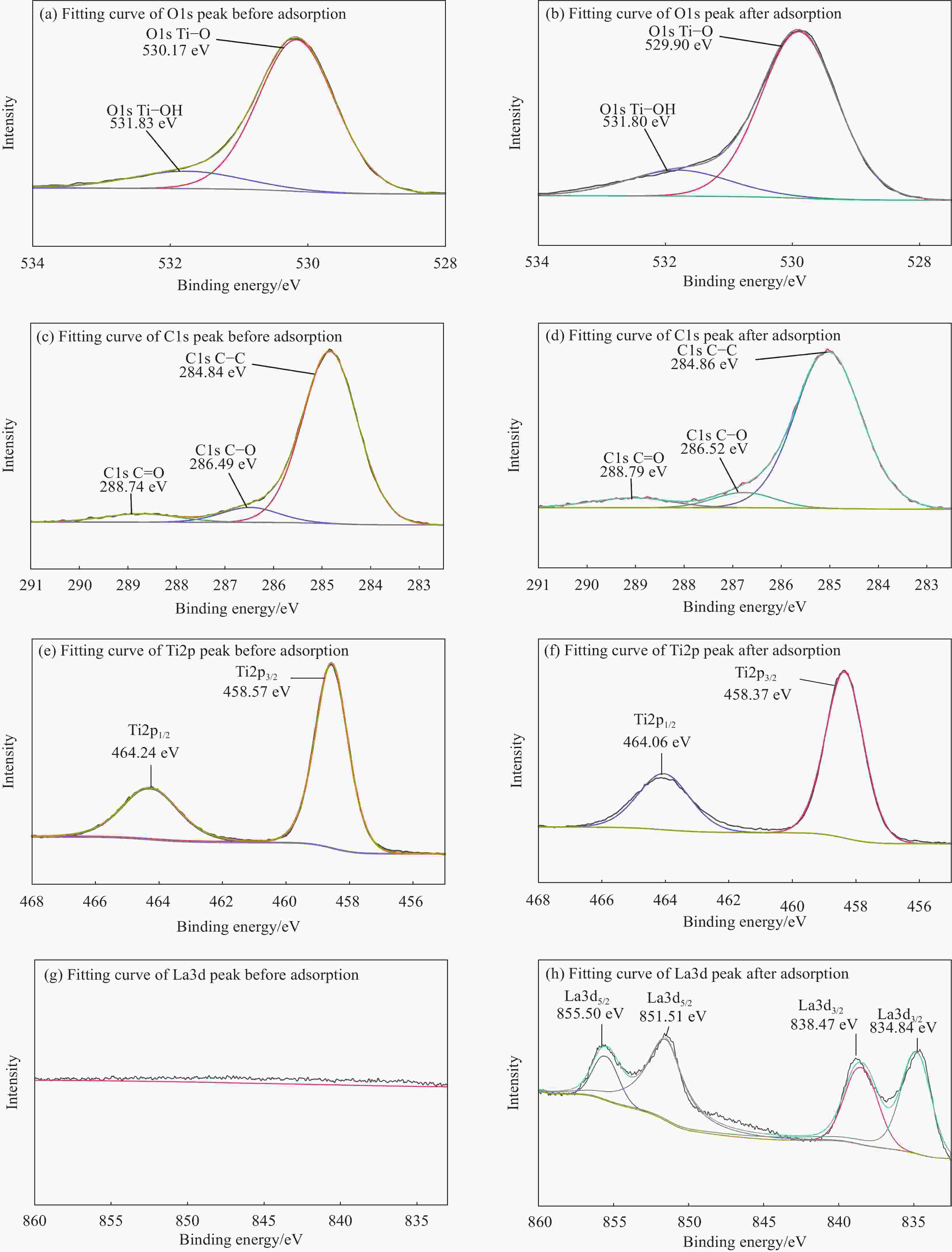
Table 6. Atomic fraction of elements on the surface of Fe3O4@fTiO2 before and after La3+ adsorption
Element La/at% Ti/at% O/at% C/at% Fe/at% Na/at% Before adsorption 0.00 16.60 41.30 26.04 4.00 12.07 After adsorption 2.79 17.34 44.54 31.84 3.49 0.00 表 7 不同吸附剂对La3+吸附能力的比较
Table 7. Comparison of La3+ adsorption capacity among different adsorbents
Materials qm/(mg·g−1) t/min pH Ref. PAMAM 50.12 240 7 [45] LA-IIP 62.80 30 4 [46] ZnO NPs 58.80 70 5 [47] SnO2-TiO2 NPs 65.60 60 5 [48] Activated carbon 175.40 60 3.5 [49] Fe3O4@fTiO2 142.90 15 5 This work Notes: t—Reaction time; PAMAM—Straw grafted with polyamide amine dendritic macromolecules; LA-IIP—Novel La(III) imprinted polymers; NPs—Nanocomposite. -
[1] 程建忠, 车丽萍. 中国稀土资源开采现状及发展趋势[J]. 稀土, 2010, 31(2):65-69, 85. doi: 10.3969/j.issn.1004-0277.2010.02.015CHENG Jianzhong, CHE Liping. The current situation and development trend of rare earth resource extraction in China[J]. Chinese Rare Earths,2010,31(2):65-69, 85(in Chinese). doi: 10.3969/j.issn.1004-0277.2010.02.015 [2] 池汝安, 刘雪梅. 风化壳淋积型稀土矿开发的现状及展望[J]. 中国稀土学报, 2019, 37(2):129-140.CHI Ru'an, LIU Xuemei. Prospect and development of weathered crust elution-deposited rare earth ore[J]. Jour-nal of the Chinese Society of Rare Earths,2019,37(2):129-140(in Chinese). [3] 邓国庆, 杨幼明. 离子型稀土矿开采提取工艺发展述评[J]. 稀土, 2016, 37(3):129-133.DENG Gouqing, YANG Youming. A review of the mining technologies of ion-absorbed rare earth mineral[J]. Chinese Rare Earths,2016,37(3):129-133(in Chinese). [4] 秦磊, 胡世丽, 宋晨曦, 等. 离子型稀土尾矿的氨氮淋洗去除[J]. 中国有色金属学报, 2021, 31(5):1395-1404.QIN Lei, HU Shili, SONG Chenxi, et al. Removal of ammonia nitrogen from ion type tailings[J]. The Chinses Journal of Nonferrous Metals,2021,31(5):1395-1404(in Chinese). [5] 高国华, 赖富国, 徐耗祥, 等. 氧化钙沉淀富集低浓度硫酸稀土溶液中稀土的研究[J]. 稀有金属, 2019, 43(4):409-419. doi: 10.13373/j.cnki.cjrm.xy18010036GAO Guohua, LAI Fuguo, XU Haoxiang, et al. Enrichment of rare earth from low concentration rare earth sulfate solution by calcium oxide precipitation[J]. Chinese Journal of Rare Metals,2019,43(4):409-419(in Chinese). doi: 10.13373/j.cnki.cjrm.xy18010036 [6] 徐光宪, 高松, 李标国, 等. 稀土萃取分离工艺研究新进展[J]. 中国稀土学报, 1993(3):193-198.XU Guangxian, GAO Song, LI Biaoguo, et al. Research progress of rare earth extraction and separation technology[J]. Journal of the Chinese Society of Rare Earths,1993(3):193-198(in Chinese). [7] 钟宜航, 彭陈亮, 王观石, 等. 蒙脱石对Y3+的吸附特性及机制研究[J]. 中国稀土学报, 2019, 37(6):713-723.ZHONG Yihang, PENG Chenliang, WANG Guanshi, et al. Adsorption characteristics and mechanism of Y3+ by montmorillonite[J]. Journal of the Chinese Society of Rare Earths,2019,37(6):713-723(in Chinese). [8] 陈灿, 杨黎彬, 周雪飞. 纳米材料在水环境中对微藻毒性的效应及机理研究进展[J]. 净水技术, 2022, 41(5):5-13.CHEN Can, YANG Libin, ZHOU Xuefei. Research progress of the effect and mechanism of nanomaterials on microalgae toxicity in water environment[J]. Water Purification Technology,2022,41(5):5-13(in Chinese). [9] 任家丰, 陆晓赟, 徐珂凡, 等. 自掺杂TiO2纳米材料在能源和环境领域的应用进展[J]. 化工新型材料, 2022, 50(2):15-19.REN Jiafeng, LU Xiaoyun, XU Kefan, et al. Application progress on self-doped TiO2 nanomaterial in the fields of energy and environment[J]. New Chemical Materials,2022,50(2):15-19(in Chinese). [10] KHIN M M, NAIR A S, BABU V J, et al. A review on nanomaterials for environmental remediation[J]. Energy & Environmental Science,2012,5(8):8075-8109. [11] LIU X, HU Q, FANG Z, et al. Magnetic chitosan nanocomposites: A useful recyclable tool for heavy metal ion removal[J]. Langmuir,2009,25(1):3-8. doi: 10.1021/la802754t [12] 王子帅, 王耀强, 肖刚, 等. 磁性纳米Fe3O4@TiO2可见光下光催化还原Cr(Ⅵ)[J]. 化工学报, 2019, 70(10):4062-4071.WANG Zishuai, WANG Yaoqiang, XIAO Gang, et al. Photocatalytic reduction of Cr(Ⅵ) by magnetic nanomaterial Fe3O4@TiO2 under visible light[J]. CIESC Journal,2019,70(10):4062-4071(in Chinese). [13] 王耀强, 赵怡琳, 李玲慧, 等. 海胆状Fe3O4@TiO2磁性纳米介质对Pb2+的选择吸附特性[J]. 化工学报, 2018, 69(1):446-454.WANG Yaoqiang, ZHAO Yilin, LI Linghui, et al. Selective adsorption of Pb2+ by sea urchin magnetic nano- Fe3O4@TiO2[J]. CIESC Journal,2018,69(1):446-454(in Chinese). [14] MA W F, ZHANG Y, LI L L, et al. Tailor-made magnetic Fe3O4@mTiO2 microspheres with a tunable mesoporous anatase shell for highly selective and effective enrichment of phosphopeptides[J]. ACS Nano,2012,6(4):3179-3188. doi: 10.1021/nn3009646 [15] DENG H, LI X, PENG Q, et al. Monodisperse magnetic single-crystal ferrite microspheres[J]. Angewandte Chemie,2005,117(18):2842-2845. doi: 10.1002/ange.200462551 [16] ZHU Y, YONEDA K, KANAMORI K, et al. Hierarchically porous titanium phosphate monoliths and their crystallization behavior in ethylene glycol[J]. New Journal of Chemistry,2016,40(5):4153-4159. doi: 10.1039/C5NJ02820E [17] MAHMOUD M E, NABIL G M, KHALIFA M A, et al. Effective removal of crystal violet and methylene blue dyes from water by surface functionalized zirconium silicate nanocomposite[J]. Journal of Environmental Chemical Engineering, 2019, 7(2): 103009. [18] WANG F, ZHAO J, PAN F, et al. Adsorption properties toward trivalent rare earths by alginate beads doping with silica[J]. Industrial & Engineering Chemistry Research,2013,52(9):3453-3461. [19] MALLAKPOUR S, TABESH F. Tragacanth gum based hydrogel nanocomposites for the adsorption of methylene blue: Comparison of linear and non-linear forms of different adsorption isotherm and kinetics models[J]. International Journal of Biological Macromolecules,2019,133:754-766. doi: 10.1016/j.ijbiomac.2019.04.129 [20] ZHAO M H, XU Y, ZHANG C S, et al. New trends in removing heavy metals from wastewater[J]. Applied Microbiology and Biotechnology,2016,100(15):6509-6518. doi: 10.1007/s00253-016-7646-x [21] CAO H, WU X, SYED-HASSAN S S A, et al. Characteristics and mechanisms of phosphorous adsorption by rape straw-derived biochar functionalized with calcium from eggshell[J]. Bioresource Technology,2020 , 318:124063. [22] TANG Y J, YANG R, MA D, et al. Removal of methyl orange from aqueous solution by adsorption onto a hydrogel composite[J]. Polymers & Polymer Composites,2018,26(2):161-168. [23] SONG Z, LIAN F, YU Z, et al. Synthesis and characterization of a novel MnOx-loaded biochar and its adsorption properties for Cu2+ in aqueous solution[J]. Chemical Engineering Journal,2014,242:36-42. doi: 10.1016/j.cej.2013.12.061 [24] PARK J O, RHEE K Y, PARK S J. Silane treatment of Fe3O4 and its effect on the magnetic and wear properties of Fe3O4/epoxy nanocomposites[J]. Applied Surface Science,2010,256(23):6945-6950. doi: 10.1016/j.apsusc.2010.04.110 [25] WU Q H, CHEN M, CHEN K Y, et al. Fe3O4-based core/shell nanocomposites for high-performance electrochemical supercapacitors[J]. Journal of Materials Science,2016,51(3):1572-1580. doi: 10.1007/s10853-015-9480-4 [26] TAN L C, ZHANG X F, LIU Q, et al. Synthesis of Fe3O4@TiO2 core-shell magnetic composites for highly efficient sorption of uranium (VI)[J]. Colloids and Surfaces A-Physicochemical and Engineering Aspects,2015,469:279-286. [27] ALQADAMI A A, KHAN M A, OTERO M, et al. A magnetic nanocomposite produced from camel bones for an efficient adsorption of toxic metals from water[J]. Journal of Cleaner Production,2018,178:293-304. doi: 10.1016/j.jclepro.2018.01.023 [28] 高立波, 张强, 李俊漾, 等. Fe3O4@SiO2/TiO2核壳结构纳米颗粒降解TNT溶液[J]. 环境工程学报, 2014, 8(6):2413-2416.GAO Libo, ZHANG Qiang, LI Junyang, et al. Degradation of TNT waste solution by Fe3O4@SiO2/TiO2 magnetic core-shell nanoparticles[J]. Chinese Journal of Environmental Engineering,2014,8(6):2413-2416(in Chinese). [29] ZHANG Z, FANG Y, ZHUO L, et al. Reduced graphene oxide wrapped Fe3O4@TiO2 yolk-shell nanostructures as a magnetic recyclable photocatalytic antibacterial agent[J]. Journal of Alloys and Compounds,2022,904:164001. [30] 宋海南, 李国喜, 周建庆, 等. Fe3O4/TiO2磁性催化剂的制备及在污水治理中的应用[J]. 分子催化, 2011, 25(6):557-562.SONG Hainan, LI Guoxi, ZHOU Jianqing, et al. Preparation of Fe3O4/TiO2 mgnetic ctalyst and application in swage teatment[J]. Journal of Molecular Catalysis,2011,25(6):557-562(in Chinese). [31] LIU Z, FENG Y, LI H. Application of titanium phosphate prepared from acidic titanium dioxide wastewater to remove cerium(III) in aqueous solution[J]. Colloids and Surfaces A-Physicochemical and Engineering Aspects,2021,630:127613. [32] 张玲, 乌东北, 朱宝辉, 等. 磁性海藻酸钙-壳聚糖凝胶珠吸附La3+性能研究[J]. 分析化学, 2010, 38(12):1732-1736.ZHANG Ling, WU Dongbei, ZHU Baohui, et al. Adsorption properties of magnetic Ca-alginate-chitosan gel bead for La3+ ions[J]. Chinese Journal of Analytical Chemistry,2010,38(12):1732-1736(in Chinese). [33] 唐林旺, 陆柳鲜, 李雪蓉, 等. 低浓度稀土废水资源回收技术研究进展[J]. 现代矿业, 2021, 37(7):97-102. doi: 10.3969/j.issn.1674-6082.2021.07.024TANG Linwang, LU Liuxian, LI Xuerong, et al. Research progress and prospect of recovery technology applied in low concentration rare earth wastewater resource[J]. Modern Mining,2021,37(7):97-102(in Chinese). doi: 10.3969/j.issn.1674-6082.2021.07.024 [34] 邹成龙, 徐志威, 聂发辉, 等. Fe3O4@SA/GO凝胶球的制备及对亚甲基蓝的吸附性能[J]. 环境工程学报, 2022, 16(1): 121-132.ZOU Chenglong, XU Zhiwei, NIE Fahui, et al. Preparation of Fe3O4@SA/GO gel ball and its adsorption performance towards methylene blue[J]. Chinese Journal of Environmental Engineering, 2022, 16(1): 121-132(in Chinese). [35] 王忠凯, 季军荣, 汤睿, 等. 双有机改性磁性膨润土对Cu(II)和Zn(II)的吸附研究[J]. 高校化学工程学报, 2022, 36(2): 276-286.WANG Zhongkai, JI Junrong, TANG Rui, et al. Preparation of dual organic modified magnetic bentonite for Cu(II) and Zn(II) adsorption[J]. Journal of Chemical Engineering of Chinese Universities, 2022, 36(2): 276-286(in Chinese). [36] 王莉, 李亮, 杨幼明. 电解质溶液对离子型稀土矿表面离子吸附的影响研究[J]. 稀土, 2021, 42(6):49-56. doi: 10.16533/J.CNKI.15-1099/TF.202106007WANG Li, LI Liang, YANG Youming. Study on the effect of electrolyte solution on the ions adsorption on the surface of ionic type rare earth ore[J]. Chinese Rare Earths,2021,42(6):49-56(in Chinese). doi: 10.16533/J.CNKI.15-1099/TF.202106007 [37] 沈州, 罗仙平, 周丹, 等. 生物炭对离子型稀土矿山尾水中氨氮的吸附特性研究[J]. 中国稀土学报, 2021, 39(6):916-926.SHEN Zhou, LUO Xianping, ZHOU Dan, et al. Study on the adsorption characteristics of biochar on ammonia nitrogen in tailwater of ionic rare earth mines[J]. Journal of the Chinese Society of Rare Earths,2021,39(6):916-926(in Chinese). [38] SARUCHI, KUMAR V. Adsorption kinetics and isotherms for the removal of rhodamine B dye and Pb2+ ions from aqueous solutions by a hybrid ion-exchanger[J]. Arabian Journal of Chemistry,2019,12(3):316-329. doi: 10.1016/j.arabjc.2016.11.009 [39] 李玲慧. Fe3O4@TiO2磁性纳米粒子的制备及吸附重金属性能研究[D]. 北京: 北京化工大学, 2017.LI Linghui. Preparation of Fe3O4@TiO2 magnetic nanoparticles and study on the adsorption of heavy metals[D]. Beijing: Beijing University of Chemical Technology, 2017(in Chinese). [40] 查汝华. 不同形貌纳米氧化物的制备及性能研究[D]. 武汉: 华中科技大学, 2015.ZHA Ruhua. Preparation and properties of nano-oxides with different morphologies[D]. Wuhan: Huazhong University of Science and Technology, 2015(in Chinese). [41] HE Z, XU X, SONG S, et al. A visible light-driven titanium dioxide photocatalyst codoped with lanthanum and iodine: An application in the degradation of oxalic acid[J]. Journal of Physical Chemistry C,2008,112(42):16431-16437. doi: 10.1021/jp803291c [42] JIA K, PAN B C, ZHANG Q R, et al. Adsorption of Pb2+, Zn2+, and Cd2+ from waters by amorphous titanium phosphate[J]. Journal of Colloid and Interface Science,2008,318(2):160-166. doi: 10.1016/j.jcis.2007.10.043 [43] YU B Z, HU Z Y, ZHOU F, et al. Lanthanum(Ⅲ) and Yttrium(Ⅲ) adsorption on montmorillonite: The role of aluminum ion in solution and minerals[J]. Mineral Processing and Extractive Metallurgy Review,2020,41(2):107-116. doi: 10.1080/08827508.2019.1575215 [44] 胡振宇, 余秉哲, 冯健, 等. 高岭土在La-Al和Y-Al二元体系中的吸附特性[J]. 矿冶工程, 2018, 38(5):87-91. doi: 10.3969/j.issn.0253-6099.2018.05.023HU Zhenyu, YU Bingzhe, FENG Jian, et al. Adsorption characteristics of Kaoline in La-Al and Y-Al system[J]. Mining and Metallurgical Engineering,2018,38(5):87-91(in Chinese). doi: 10.3969/j.issn.0253-6099.2018.05.023 [45] 章昌华, 郭明, 涂伟萍, 等. PAMAM接枝稻草对Nd3+、Sm3+、La3+的吸附特性[J]. 精细化工, 2020, 37(3):584-589, 597.ZHANG Changhua, GUO Ming, TU Weiping, et al. Adsorption characteristics of PAMAM grafted rice straw for Nd3+, Sm3+ and La3+[J]. Fine Chemicals,2020,37(3):584-589, 597(in Chinese). [46] NI C Q, LIU Q M, REN Z, et al. Selective removal and recovery of La(III) using a phosphonic-based ion imprinted polymer: Adsorption performance, regeneration, and mechanism[J]. Journal of Environmental Chemical Engi-neering, 2021, 9(6): 106701. [47] ZHENG X, WU D, SU T, et al. Magnetic nanocomposite hydrogel prepared by ZnO-initiated photopolymerization for La(III) adsorption[J]. ACS Applied Materials & Interfaces,2014,6(22):19840-19849. [48] RAHMAN M M, KHAN S B, MARWANI H M, et al. SnO2-TiO2 nanocomposites as new adsorbent for efficient removal of La(III) ions from aqueous solutions[J]. Journal of the Taiwan Institute of Chemical Engineers,2014,45(4):1964-1974. doi: 10.1016/j.jtice.2014.03.018 [49] AWWAD N S, GAD H M H, AHMAD M I, et al. Sorption of lanthanum and erbium from aqueous solution by activated carbon prepared from rice husk[J]. Colloids and Surfaces B-Biointerfaces,2010,81(2):593-599. doi: 10.1016/j.colsurfb.2010.08.002 -





 下载:
下载: