Injectable nanomotors-hydrogel system with high cellular uptake for targeted cancer gene therapy
-
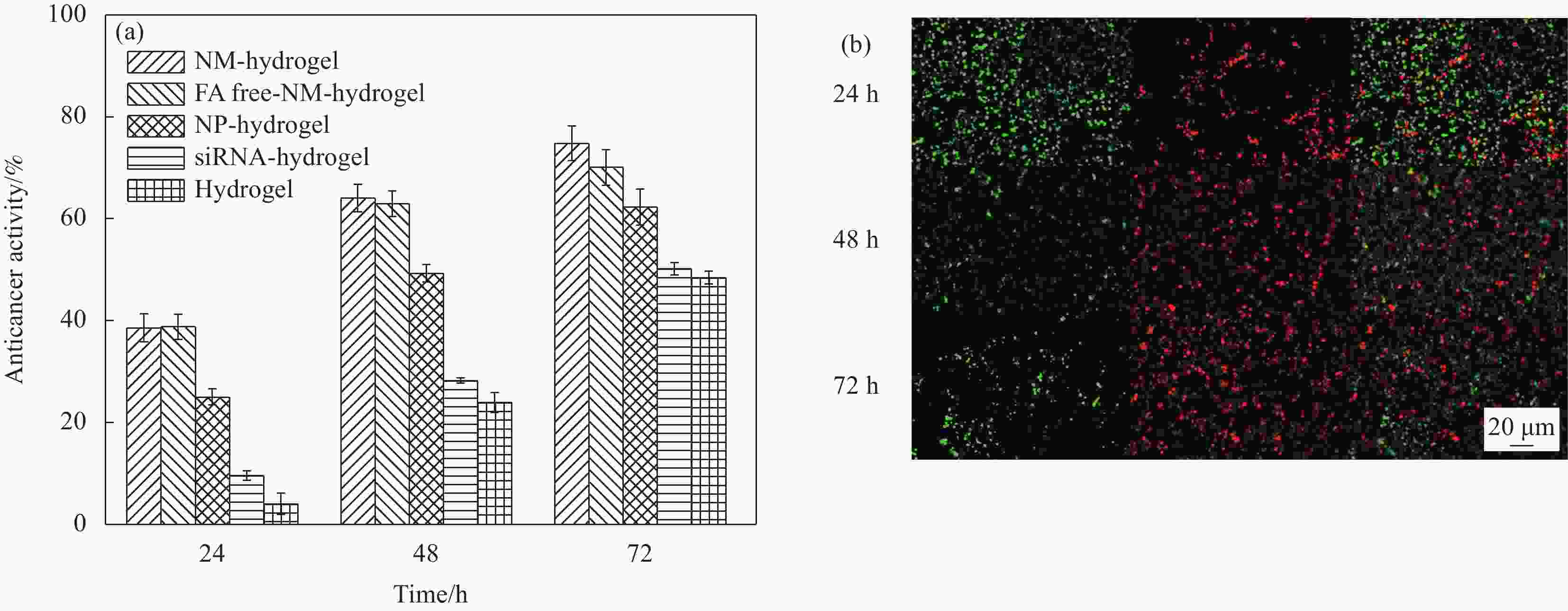
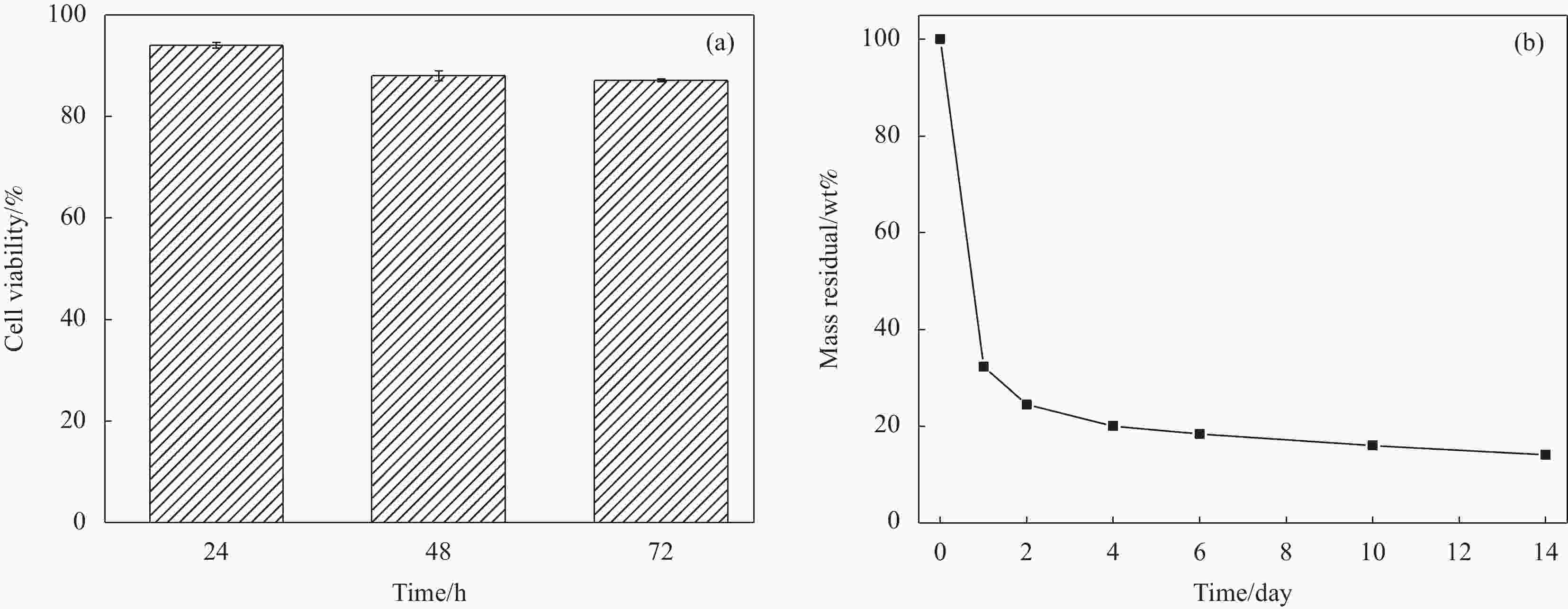
摘要: 小干扰RNA(siRNA)由于其降低基因表达的能力,通常在基因治疗中充当重要的治疗剂。然而,siRNA的低细胞摄取限制了其在癌症治疗中的功效。本文报告了一种生物相容的纳米马达-水凝胶递送系统,使siRNA可在癌症靶向治疗中获得高细胞摄取率。首先通过层层自组装技术,以铂纳米粒子为核,使用聚乙烯亚胺(PEI)和聚苯乙烯磺酸钠(PSS)制备载有siRNA的纳米马达(NM)。为了瘤内给药和缓释,将纳米马达装载在席夫碱水凝胶中,构建NM-hydrogel系统。肿瘤微环境具有弱酸性和高H2O2含量的特点。水凝胶响应弱酸性微环境释放纳米马达,释放的纳米马达可以通过过氧化氢的催化分解实现自驱动,其运动速度在1wt%H2O2下为1.78 µm/s(每秒约22.25个体长)。纳米马达的自驱动性能和由纳米马达上修饰的叶酸(FA)介导的特定内吞作用使NM-hydrogel系统具有63.8%的高细胞摄取率。同时,自驱动性能和PEI引起的质子海绵效应促进了纳米马达在肿瘤细胞中的深度渗透和长时间滞留,从而促进了NM-hydrogel系统的抗癌作用。结果表明:该体系在72 h时的抗癌活性为74.8%。同时,NM-hydrogel系统具有良好的生物相容性和生物降解性,为其未来在体内基因治疗中的应用奠定了基础。Abstract: Small interfering RNA (siRNA) often acts as an important therapeutic agent in gene therapy due to its ability of reducing gene expression. However, the low cell uptake rate of siRNA limits its efficacy in cancer treatment. Here, we report a biocompatible nanomotors-hydrogel system to obtain high cell uptake rate for targeted cancer therapy. Nanomotors (NM) loaded with siRNA were prepared employing polyethyleneimine (PEI) and sodium polystyrene sulfonate (PSS) via layer-by-layer self-assembly technology. For the sake of intratumoral administration and slow release, the nanomotors were loaded in Schiff-base hydrogel to build NM-hydrogel system. Taking advantage of the tumor microenvironment featured with an acidic pH and a high content of hydrogen peroxide, the hydrogel releases nanomotors in response to slightly acidic tumor matrix, and the released nanomotors can autonomously move through catalytic decomposition of hydrogen peroxide, the speed of which can be maintained at 1.78 µm/s at 1wt%H2O2 (22.25 body lengths per second). The autonomous motion of nanomotors and specific endocytosis which is mediated by folic acid (FA) modified on the nanomotors gives the NM-hydrogel system a high cell uptake rate of 63.8%. The proton sponge effect caused by PEI and autonomous motion promote the deep penetration and long-time retention of namomotors in tumor cells, which facilitates the anticancer effect of the NM-hydrogel system. The results show that the anticancer activity of the system is 74.8% at 72 h. Meanwhile, the NM-hydrogel system has good biocompatibility and biodegradability, which lays the foundation for its future application in gene therapy in vivo.
-
Key words:
- nanoparticle /
- self-propulsion /
- cell uptake /
- hydrogel /
- gene therapy /
- cancer treatment
-
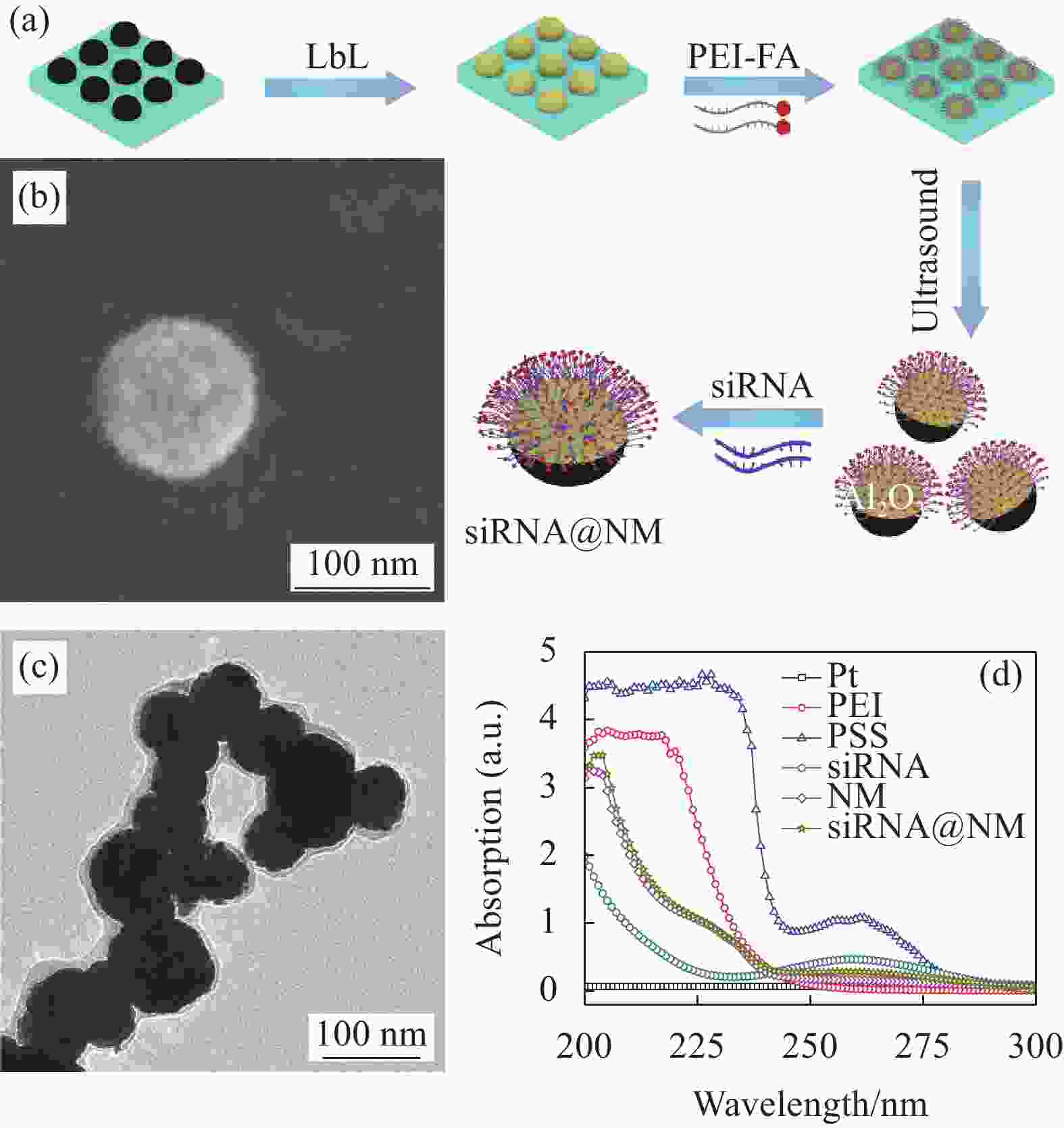
图 1 (a) 负载小干扰RNA的纳米马达(siRNA@NM)合成示意图;siRNA@NM的SEM图像 (b)、TEM图像 (c) 和紫外-可见光谱 (d)
Figure 1. (a) Schematic fabrication process of nanomotors loaded with small interfering RNA (siRNA) (siRNA@NM); SEM image (c), TEM image (d) and UV-visible transmission spectra (b) of siRNA@NM
LbL—Layer by layer; PEI—Polyethyleneimine; PSS—Poly sodium styrene sulfonate; FA—Folic acid
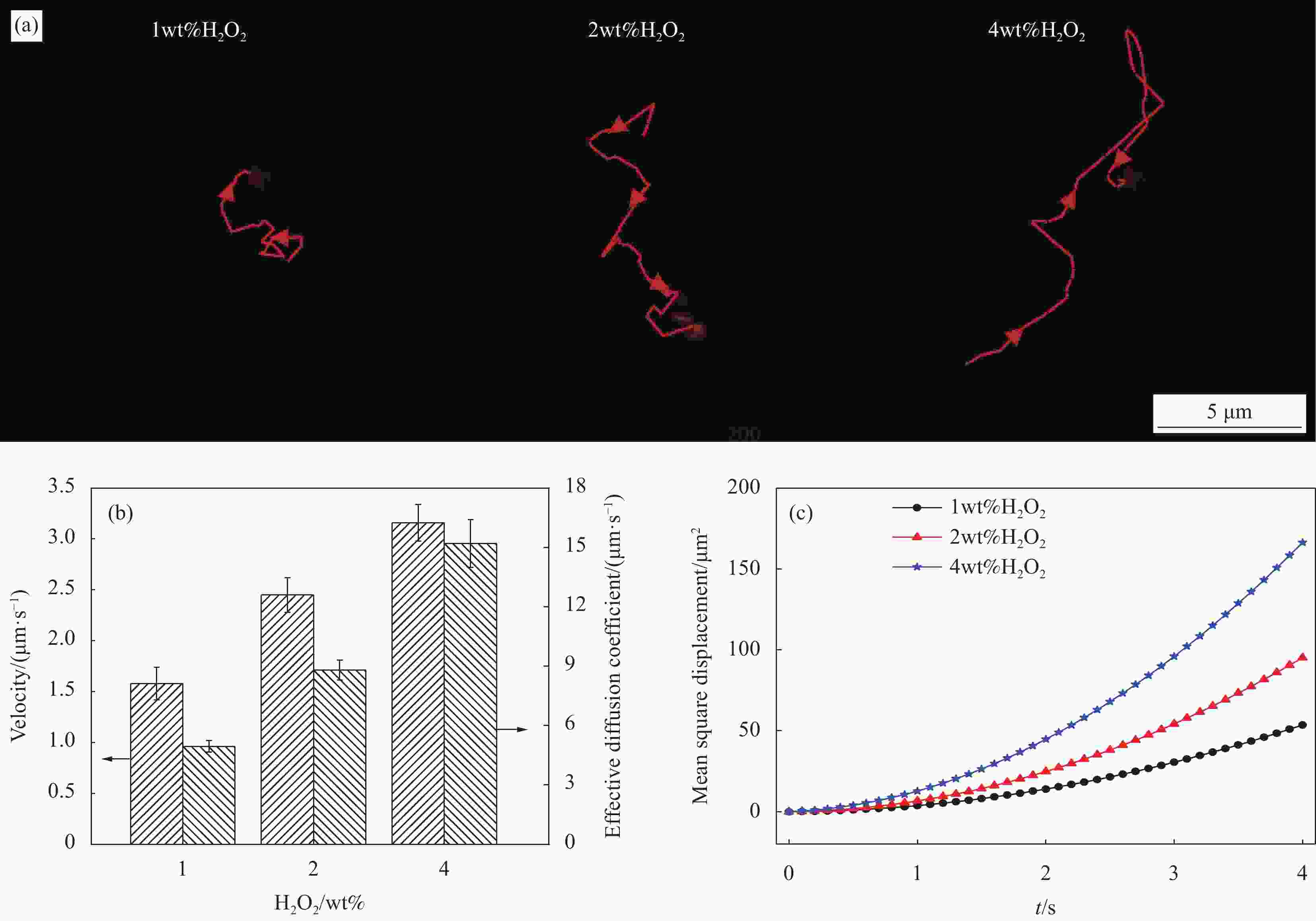
图 2 (a) siRNA@NM在不同H2O2溶液中的运动轨迹;(b) siRNA@NM在不同H2O2溶液中的速度和有效扩散系数;(c) siRNA@NM在不同H2O2溶液中的均方位移-时间(MSD-Δt)曲线
Figure 2. (a) Motion trajectories of siRNA@NM in different H2O2 solution; (b) Velocity and the effective diffusion coefficients of the siRNA@NM in different H2O2 solution; (c) Mean azimuth shift-time (MSD-Δt) plot of siRNA@NM in different H2O2 solution
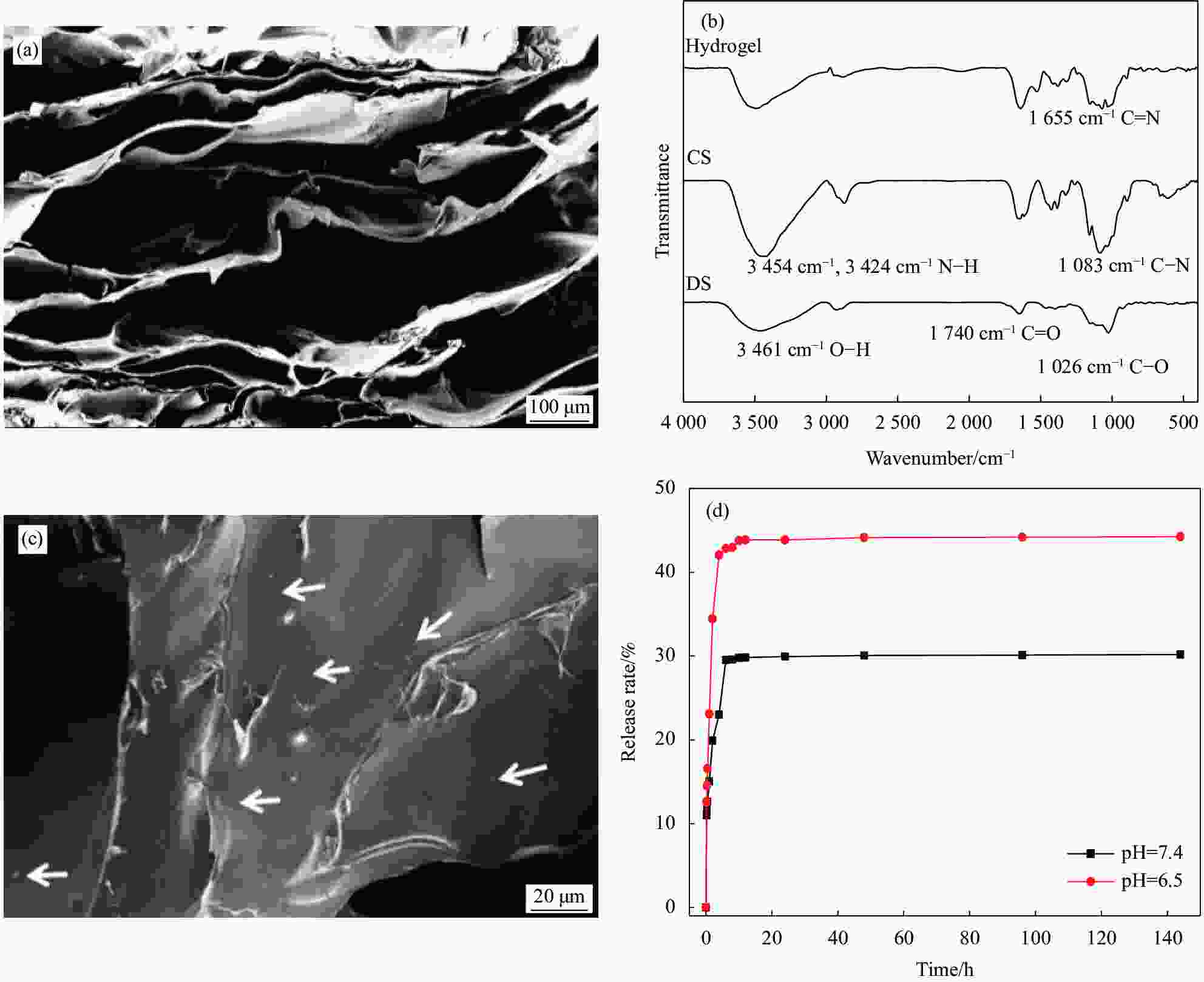
图 3 (a) 席夫碱水凝胶的SEM图像;(b) 双醛淀粉(DS)、壳聚糖(CS)和席夫碱水凝胶的FTIR图谱;(c) NM-hydrogel系统的SEM图像;(d) siRNA@NMs在PBS溶液(pH=6.5、pH=7.4)中的释放曲线
Figure 3. (a) SEM image of Schiff-base hydrogel; (b) FTIR spectra of dialdehyde starch (DS), chitosan (CS) and Schiff-base hydrogel; (c) SEM image of NM-hydrogel system; (d) Time-dependent release of siRNA@NMs from the NM-hydrogel system in PBS solution (pH=6.5, pH=7.4)
-
[1] FAN H, DEMIRCI U, CHEN P. Emerging organoid models: Leaping forward in cancer research[J]. Journal of Hematology & Oncology,2019,12(1):142. [2] SIHOE A D L. Video-assistedthoracoscopic surgery as the gold standard for lung cancer surgery[J]. Respirology,2020,25:49-60. [3] WANG W, HAO Y, LIU Y, et al. Nanomedicine in lung cancer: Current states of overcoming drug resistance and improving cancer immunotherapy[J]. Wiley Interdisciplinary Reviews-Nanomedicine and Nanobiotechnology,2020,13(1):e1654. [4] HAO M, CHEN B, ZHAO X, et al. Organic/inorganic nanocomposites for cancer immunotherapy[J]. Materials Chemistry Frontiers,2020,4(9):2571-2609. doi: 10.1039/D0QM00323A [5] SHI S, VISSAPRAGADA R, JAOUDE J A, et al. Evolving role of biomaterials in diagnostic and therapeutic radiation oncology[J]. Bioactive Materials,2020,5(2):233-240. doi: 10.1016/j.bioactmat.2020.01.011 [6] LEVITZKI A, KLEIN S. My journey from tyrosine phosphorylation inhibitors to targeted immune therapy as strategies to combat cancer[J]. Proceedings of the National Academy of Sciences of the United States of America,2019,116(24):11579-11586. doi: 10.1073/pnas.1816012116 [7] LEI Z, DING L, YAO C, et al. A Highly efficient tumor-targeting nanoprobe with a novel cell membrane permeability mechanism[J]. Advanced Materials,2019,31(12):1807456. doi: 10.1002/adma.201807456 [8] LEDFORD H. Gene therapy’s toughest test[J]. Nature,2019,576(7785):22-25. doi: 10.1038/d41586-019-03698-8 [9] MA C C, WANG Z L, XU T, et al. The approved gene therapy drugs worldwide: From 1998 to 2019[J]. Biotechnology Advances,2020,40:107502. doi: 10.1016/j.biotechadv.2019.107502 [10] WAYNE E C, LONG C, HANEY M J, et al. Targeted delivery of siRNA lipoplexes to cancer cells using macrophage transient horizontal gene transfer[J]. Advanced Science,2019,6(21):1900582. [11] ZHANG M J, WENG Y H, CAO Z Y, et al. ROS-activatable sirna-engineered polyplex for NIR-triggered synergistic cancer treatment[J]. ACS Applied Materials & Interfaces,2020,12(29):32289-32300. [12] MI Y, HAGAN C T, VINCENT B G, et al. Emerging nano-/microapproaches for cancer immunotherapy[J]. Advanced Science,2019,6(6):1801874. [13] WANG Z H, MA Y, WANG H, et al. CXCR4-enriched nano-trap targeting CXCL12 in lung for early prevention and enhanced photodynamic therapy of breast cancer metastasis[J]. Advanced Functional Materials,2019,29(45):1905480. [14] WANG H, PUMERA M. Coordinated behaviors of artificial micro/nanomachines: from mutual interactions to interactions with the environment[J]. Chemical Society Reviews,2020,49(10):3211-3230. doi: 10.1039/C9CS00877B [15] WANG H, PUMERA M. Micro/nanomachines and living biosystems: From simple interactions to microcyborgs[J]. Advanced Functional Materials,2018,28(25):1705421. doi: 10.1002/adfm.201705421 [16] MOU F Z, LI X F, XIE Q, et al. Active micromotor systems built from passive particles with biomimetic predator-prey interactions[J]. ACS Nano,2020,14(1):406-414. doi: 10.1021/acsnano.9b05996 [17] ZHOU M, XING Y, LI X, et al. Cancer cell membrane camouflaged semi-yolk@spiky-shell nanomotor for enhanced cell adhesion and synergistic therapy[J]. Small,2020,16(39):2003834. doi: 10.1002/smll.202003834 [18] WAN M, WANG Q, LI X, et al. Systematic research and evaluation models of nanomotors for cancer combined therapy[J]. Angewandte Chemie-International Edition,2020,59(34):14458-14465. doi: 10.1002/anie.202002452 [19] VENUGOPALAN P L, DE AVILA B E F, PAL M, et al. Fantastic voyage of nanomotors into the cell[J]. ACS Nano,2020,14(8):9423-9439. doi: 10.1021/acsnano.0c05217 [20] WU Y J, SI T Y, SHAO J X, et al. Near-infrared light-driven Janus capsule motors: Fabrication, propulsion, and simulation[J]. Nano Research,2016,9(12):3747-3756. doi: 10.1007/s12274-016-1245-0 [21] ROY S, ZHU D, PARAK W J, et al. Lysosomal proton buffering of poly(ethylenimine) measured in situ by fluorescent pH-sensor microcapsules[J]. ACS Nano,2020,14(7):8012-8023. doi: 10.1021/acsnano.9b10219 [22] SALIM L, ISLAM G, DESAULNIERS J P. Targeted delivery and enhanced gene-silencing activity of centrally modified folic acid-siRNA conjugates[J]. Nucleic Acids Research,2020,48(1):75-85. doi: 10.1093/nar/gkz1115 [23] HORMIGOS R M, SANCHEZ B J, ESCARPA A. Multi-light-responsive quantum dot sensitized hybrid micromotors with dual-mode propulsion[J]. Angewandte Chemie-International Edition,2019,58(10):3128-3132. doi: 10.1002/anie.201811050 [24] ZENG M X, YUAN S, HUANG D L, et al. Accelerated design of catalytic water-cleaning nanomotors via machine learning[J]. ACS Applied Materials& Interfaces,2019,11(43):40099-40106. [25] HOWSE J R, JONES R A L, RYAN A J, et al. Self-motile colloidal particles: From directed propulsion to random walk[J]. Physical Review Letters,2007,99(4):048102. doi: 10.1103/PhysRevLett.99.048102 [26] DUNDERDALE G, EBBENS S, FAIRCLOUGH P, et al. Importance of particle tracking and calculating the mean-squared displacement in distinguishing nanopropulsion from other processes[J]. Langmuir,2012,28(30):10997-11006. doi: 10.1021/la301370y [27] REN J Y, XUAN H Y, DAI W, et al. Double network self-healing film based on metal chelation and Schiff-base interaction and its biological activities[J]. Applied Surface Science,2018,448:609-617. doi: 10.1016/j.apsusc.2018.04.122 [28] REN J Y, XUAN H Y, GE L Q. Double network self-healing chitosan/dialdehyde starch-polyvinyl alcohol film for gas separation[J]. Applied Surface Science,2019,469:213-219. doi: 10.1016/j.apsusc.2018.11.001 [29] ARAGHI S O, KIEFTE-DE J J C, VAN DIJK S C, et al. Folic acid and vitamin B12 supplementation and the risk of cancer: Long-term follow-up of the B vitamins for the prevention of osteoporotic fractures (B-PROOF) trial[J]. Cancer Epidemiology Biomarkers Prevention,2019,28(2):275-282. doi: 10.1158/1055-9965.EPI-17-1198 [30] AKBAL O, BOLAT G, YAMAN Y T, et al. Folic acid conjugated Prussian blue nanoparticles: Synthesis, physicochemical characterization and targeted cancer cell sensing[J]. Colloids and Surfaces B: Biointerfaces,2020,187:110655. doi: 10.1016/j.colsurfb.2019.110655 [31] CAO Q, YANG J, ZHANG H, et al. Traceable in-cell synthesis and cytoplasm-to-nucleus translocation of a zinc Schiff base complex as a simple and economical anticancer strategy[J]. Chemical Communication,2019,55(54):7852-7855. doi: 10.1039/C9CC03480C [32] PARVEEN S. Recent advances in anticancer ruthenium Schiff base complexes[J]. Applied Organometallic Che-mistry,2020,34(8):e5687. -





 下载:
下载: