Preparation and photocatalytic properties of SnS2-Ag/g-C3N4 composites
-
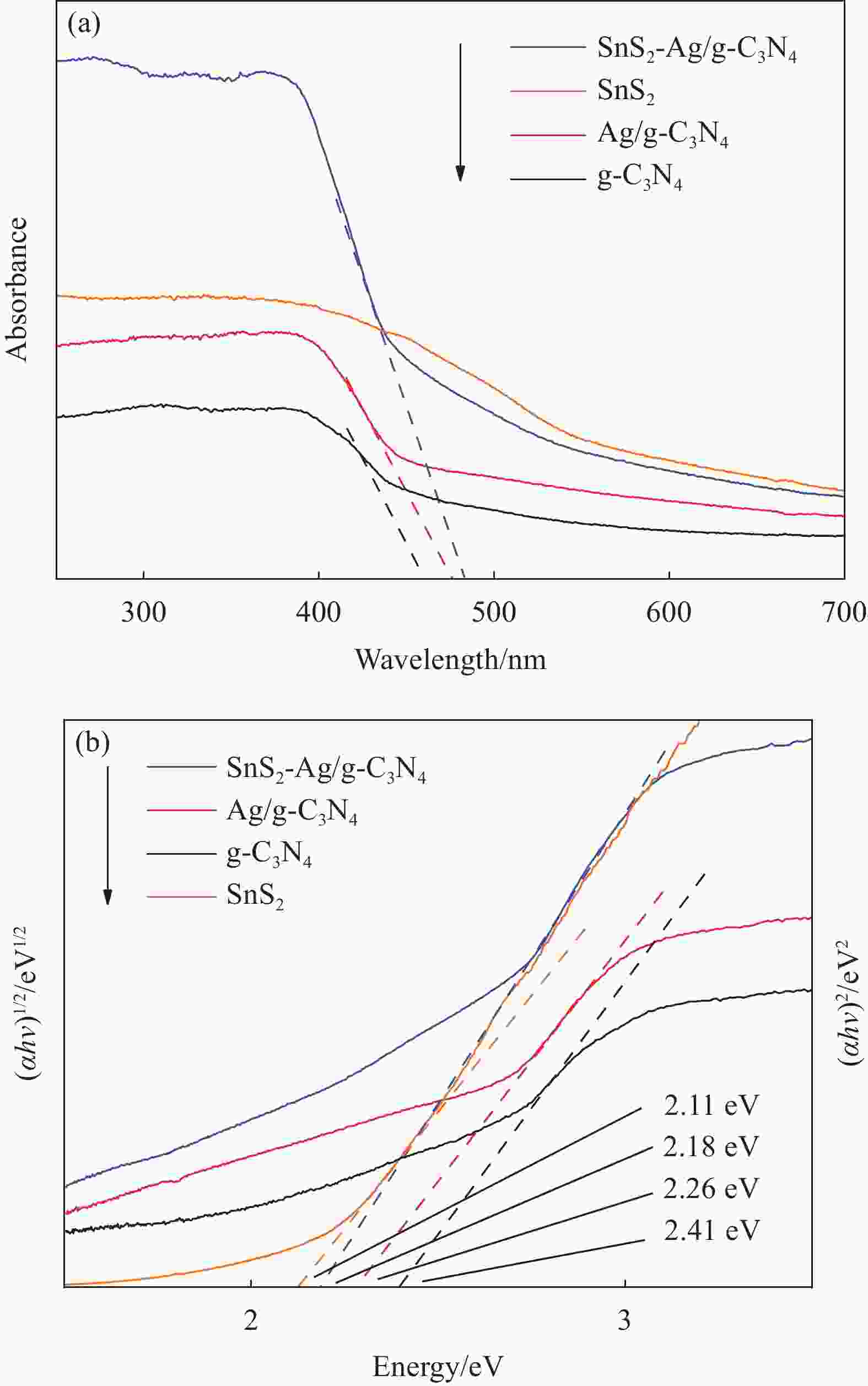
摘要: 为解决单相光催化材料结构和性能上的缺陷,通过二次煅烧法获得二维石墨相氮化碳g-C3N4,通过光沉积法获得Ag/g-C3N4,选择SnS2与Ag/g-C3N4通过简单的超声和蒸发溶剂的方法制备了三相复合材料SnS2-Ag/g-C3N4,成功构建了n-n型异质结,并对材料的微观形貌、相结构、光响应能力和孔隙结构等进行了详尽表征。结果表明:材料依然保留了片层状结构并构建了浪花状形貌,各相结晶度较高且界面构建良好,形成了类似三明治结构的2D-0D-2D形貌,复合材料较单相材料具有更高的比表面积和更强的可见光响应性能。当SnS2的含量为10wt%时,所合成SnS2-Ag/g-C3N4复合材料对罗丹明B的光催化降解效率达到最高的95.6%,降解速率最快且为g-C3N4的3.5倍,经4次循环后材料的光催化效率仍然保持在85.3%以上。Abstract: In order to solve the defects in the structure and performance of single-phase photocatalytic materials, the 2D graphitic carbon nitride (g-C3N4) was synthesized by secondary calcining, and Ag/g-C3N4 was prepared via photodeposition method. SnS2-Ag/g-C3N4 with n-n type heterojunction structure was prepared through simple ultrasonic and solvent evaporation methods. The microstructure, phase structure, light response ability and pore structure of the materials were characterized. The results showed that the lamellar structure and the spray shape coexist in the composite. The 2D-0D-2D morphology similar to sandwich structure is formed. The composite has high crystallinity and well-constructed interface, which give it a higher specific surface area and stronger visible light response performance. When the mass fraction of SnS2 is 10wt%, the photocatalytic degradation efficiency of the SnS2-Ag/g-C3N4 composite is up to 95.6%, and the degradation rate is 3.5 times as that of g-C3N4. After 4 cycles, the photocatalytic efficiency remained above 85.3%.
-
Key words:
- graphitic carbon nitride /
- SnS2 /
- Ag /
- composite /
- photocatalysis /
- degradation
-
图 9 不同SnS2含量的SnS2-Ag/g-C3N4对RhB的光催化降解速率 (a) 和动力学图 (b);(c) 不同pH条件下催化剂的降解效率; (d) 催化材料的稳定性
Figure 9. Photocatalytic degradation rate (a) and kinetic (b) diagram of RhB by SnS2-Ag/g-C3N4 with different SnS2 content; (c) Degradation efficiency of catalyst under different pH conditions; (d) Stability of catalytic materials
k—Reaction rate constant; C—Concentration; C0—Original concentration
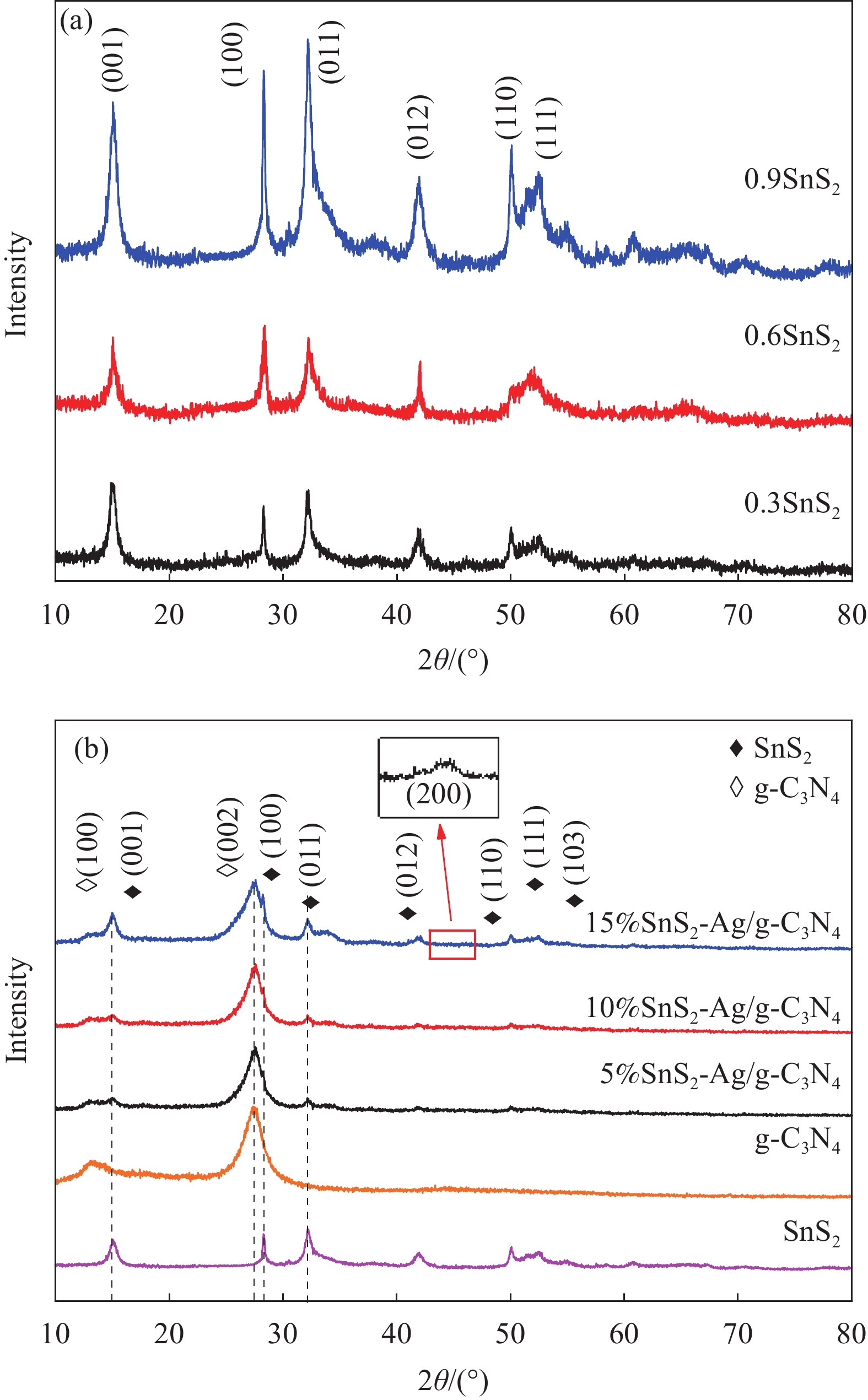
表 1 层状SnS2的配比
Table 1. Ratio of layered SnS2
Sample PEG-6000/g Sn/mmol S/mmol 0.3SnS2 0.3 1 2 0.6SnS2 0.6 1 2 0.9SnS2 0.9 1 2 Note: PEG—Polyethylene glycol. 表 2 SnS2-Ag/石墨相氮化碳(g-C3N4)样品名及其成分配比
Table 2. Sample name of SnS2-Ag/graphite phase carbon nitride (g-C3N4) and composition ratio
Sample 0.3SnS2/wt% Ag/wt% 5%SnS2-Ag/g-C3N4 5 5 10%SnS2-Ag/g-C3N4 10 15%SnS2-Ag/g-C3N4 15 -
[1] FUJISHIMA Akira, HONDA Kenichi. Electrochemical photolysis of water at a semiconductor electrode[J]. Nature,1972,238(5358):37-38. doi: 10.1038/238037a0 [2] WEN Jiuqing, XIE Jun, CHEN Xiaobo, et al. A Review on g-C3N4-based photocatalysts[J]. Applied Surface Science,2017,391:72-123. [3] WANG Chuang, ZHAI Jiali, JIANG Huan, et al. CdS/Ag2S nanocomposites photocatalyst with enhanced visible light photocatalysis activity[J]. Solid State Sciences,2019,98:106020. doi: 10.1016/j.solidstatesciences.2019.106020 [4] GUO Rui, YAN Aiguo, XU Juanjuan, et al. Effects of morphology on the visible-light-driven photocatalytic and bactericidal properties of BiVO4/CdS heterojunctions: A discussion on photocatalysis mechanism[J]. Journal of Alloys and Compounds,2020,817:153246. doi: 10.1016/j.jallcom.2019.153246 [5] MATIMIN J, MOHAMAD A J, HAZWANEE O, et al. Photochemical synthesis of nanosheet Tin Di/sulfide with sunlight response on water pollutant degradation[J]. Nanomaterials (Basel),2019,9(2):264. doi: 10.3390/nano9020264 [6] HUO Yao, YANG Yang, DAI Kai, et al. Construction of 2D/2D porous graphitic C3N4/SnS2 composite as a direct Z-scheme system for efficient visible photocatalytic activity[J]. Applied Surface Science,2019,481:1260-1269. doi: 10.1016/j.apsusc.2019.03.221 [7] GUO Shengqi, ZHANG Haijun, HU Zhengzhong, et al. Composition-dependent micro-structure and photocatalytic performance of g-C3N4 quantum dots@SnS2 heterojunction[J]. Nano Research, 2021, 14(11): 4188-4196. [8] FENG Juan, CHEN Jibing, MU Jianglong, et al. A facile in situ solvothermal method for two-dimensional layered g-C3N4/SnS2 p-n heterojunction composites with efficient visible-light photocatalytic activity[J]. Journal of Nanoparticle Research,2018,20:38. doi: 10.1007/s11051-018-4143-4 [9] DI Tingmin, ZHU Bicheng, CHENG Bei, et al. A direct Z-scheme g-C3N4/SnS2 photocatalyst with superior visible-light CO2 reduction performance[J]. Journal of Catalysis,2017,352:532-541. [10] KIM S C, KIM S M, YOON G J, et al. Gelatin-based sponge with Ag nanoparticles prepared by solution plasma: Fabrication, characteristics, and their bactericidal effect[J]. Current Applied Physics,2014,14:S172-S179. doi: 10.1016/j.cap.2013.12.032 [11] ZHAO Wei, LI Yajuan, ZHAO Pushu, et al. Novel Z-scheme Ag-C3N4/SnS2 plasmonic heterojunction photocatalyst for degradation of tetracycline and H2 production[J]. Chemi-cal Engineering Journal,2021,405:126555. doi: 10.1016/j.cej.2020.126555 [12] ZHANG Zhenyi, HUANG Jindou, ZHANG Mingyi, et al. Ultrathin hexagonal SnS2 nanosheets coupled with g-C3N4 nanosheets as 2D/2D heterojunction photocatalysts toward high photocatalytic activity[J]. Applied Catalysis B: Environmental,2015,163:298-305. doi: 10.1016/j.apcatb.2014.08.013 [13] LIANG Mingxi, YU Yajing, WANG Ying, et al. Remarkably efficient charge transfer through a double heterojunction mechanism by a CdS-SnS-SnS2/rGO composite with excellent photocatalytic performance under visible light[J]. Journal of Hazardous Materials,2020,391:121016. doi: 10.1016/j.jhazmat.2019.121016 [14] KUMAR G M, CHO H D, ILANCHEZHIYAN P, et al. Evidencing enhanced charge-transfer with superior photocatalytic degradation and photoelectrochemical water splitting in Mg modified few-layered SnS2[J]. Journal of Colloid and Interface Science,2019,540:476-485. doi: 10.1016/j.jcis.2019.01.039 [15] DENG Lu, ZHU Zhenfeng, LIU Liu, et al. Synthesis of Ag2O and Ag co-modified flower-like SnS2 composites with enhanced photocatalytic activity under solar light irradiation[J]. Solid State Sciences,2017,63:76-83. doi: 10.1016/j.solidstatesciences.2016.11.016 [16] PHAM M T, NGUYEN T M, BUI D T, et al. Enhancing quantum efficiency at Ag/g-C3N4 interfaces for rapid removal of nitric oxide under visible light[J]. Sustainable Chemistry and Pharmacy,2022,25:100596. doi: 10.1016/j.scp.2021.100596 [17] XU Ge, XU Yanheng, ZHOU Zhicun, et al. Facile hydrothermal preparation of graphitic carbon nitride supercell structures with enhanced photodegradation activity[J]. Diamond and Related Materials,2019,97:107461. doi: 10.1016/j.diamond.2019.107461 [18] NAZNEEN A, KHAN M I, NAEEM M A, et al. Structural, morphological, optical, and photocatalytic properties of Ag-doped MoS2 nanoparticles[J]. Journal of Molecular Structure,2020,1220:128735. doi: 10.1016/j.molstruc.2020.128735 [19] FANG Yanfen, MA Wanhong, HUANG Yingping, et al. Exploring the reactivity of multicomponent photocatalysts: Insight into the complex valence band of BiOBr[J]. Che-mistry,2013,19(9):3224-3229. doi: 10.1002/chem.201202602 [20] SINGH Jasminder, BASU Soumen. Synthesis of mesoporous magnetic Fe2O3/g-C3N4 monoliths for Rhodamine B removal[J]. Microporous and Mesoporous Materials,2020,303:110299-110308. doi: 10.1016/j.micromeso.2020.110299 [21] JIANG Qiongji, GAN Huihui, HUANG Yin, et al. Peroxymonosulfate activation on carbon nano-onions modified graphitic carbon nitride via light-tuning radical and nonradical pathways[J]. Journal of Environmental Chemical Engineering,2021,9(6):106592. doi: 10.1016/j.jece.2021.106592 [22] XU Y, SCHOONEN M A A. The absolute energy positions of conduction and valence bands of selected semiconducting minerals[J]. American Mineralogist,2000,85(3-4):543-556. doi: 10.2138/am-2000-0416 [23] 郑小刚, 杜京城, 付孝锦, 等. 双金属位催化剂Ag-Ni/g-C3N4可见光催化降解亚甲基蓝[J]. 硅酸盐学报, 2018, 46(1):85-92.ZHENG Xiaogang, DU Jingcheng, FU Xiaojin, et al. Carbon nitride bimetallic Ag-Ni/g-C3N4 catalysts for photocatalytic degradation of methylene blue under visible-light irradiation[J]. Journal of the Chinese Ceramic Society,2018,46(1):85-92(in Chinese). -






 下载:
下载: