Controllable construction of ZnO@SnO2 heterojunction composite nanotubes and their photocatalytic properties
-
摘要: 性能优异的功能纳米材料的设计构筑对于光催化应用而言至关重要。基于模板自刻蚀机制,利用两步溶剂热技术,以一维ZnO纳米棒为模板,在无需附加酸刻蚀的条件下成功制备一维圆顶状ZnO@SnO2异质结纳米管复合材料(Heterojunction domed nanotubes,HDNs)。由于ZnO与SnO2具有匹配的能级结构,在纳米管界面处可形成促进载流子分离的内建电场,赋予该材料优异的光催化与稳定性能。通过控制实验过程中自产生的碱性强弱,实现两性氧化物ZnO的自刻蚀,从而实现ZnO@SnO2 HDNs的管壁厚度可控调控与催化性能的调节。借助SEM、TEM、STEM及PL等表征手段对材料的微观形貌、元素组成、生长机制与性能进行了考察。以甲基橙、亚甲基蓝、曙红等为污染物模型,光催化污染物降解实验结果表明,获得的ZnO@SnO2 HDNs具有优良的光催化性能,光照60 min内对亚甲基蓝、曙红的降解率可达到95%,表明构筑的纳米管异质结极大地促进了载流子的分离,抑制其复合,提高了光催化性能。同时,循环稳定性能测试说明构建的异质结纳米管催化剂具有良好的稳定性能,在染料降解方面具有广阔的应用前景。Abstract:
The design and construction of functional nanomaterials with excellent properties is very important for photocatalytic applications. The ZnO@SnO2 heterojunction domed nanotubes (HDNs) were successfully prepared without additional acid etching step using one-dimensional ZnO nanorods as templates by two-step solvothermal technology based on the template self-etching mechanism. The built-in electric field which can promote carrier separation can be formed at the nanotube interface owing to the matching energy level structure between ZnO and SnO2, endowing the material excellent photocatalytic and stability properties. By controlling the intensity of self-generated alkaline during the experimental processes, the amphoteric oxide ZnO can be etched, achieving the controllable regulation of the thickness of tubes and their photocatalytic performance. The morphology, element composition, growth mechanism and properties of the ZnO@SnO2 HDNs were investigated by means of SEM, TEM, STEM and PL. Taking methyl orange, methylene blue and eosin as pollutant models, the experimental results of photocatalytic pollutant degradation showed that ZnO@SnO2 HDNs had excellent photocatalytic performance, and the degradation rate of methylene blue and eosin can reach 95% within 60 min. These results indicate that the constructed one-dimensional heterojunction can greatly promote the separation of carriers and inhibit their recombination, thereby improving the photocatalytic performance. At the same time, the cycle stability test showes that the heterojunction nanotube photocatalyst has great stability and broad application prospects in dye degradation. -
Key words:
- ZnO@SnO2 /
- heterojunction /
- charge separation /
- photocatalysis /
- dye degradation
-
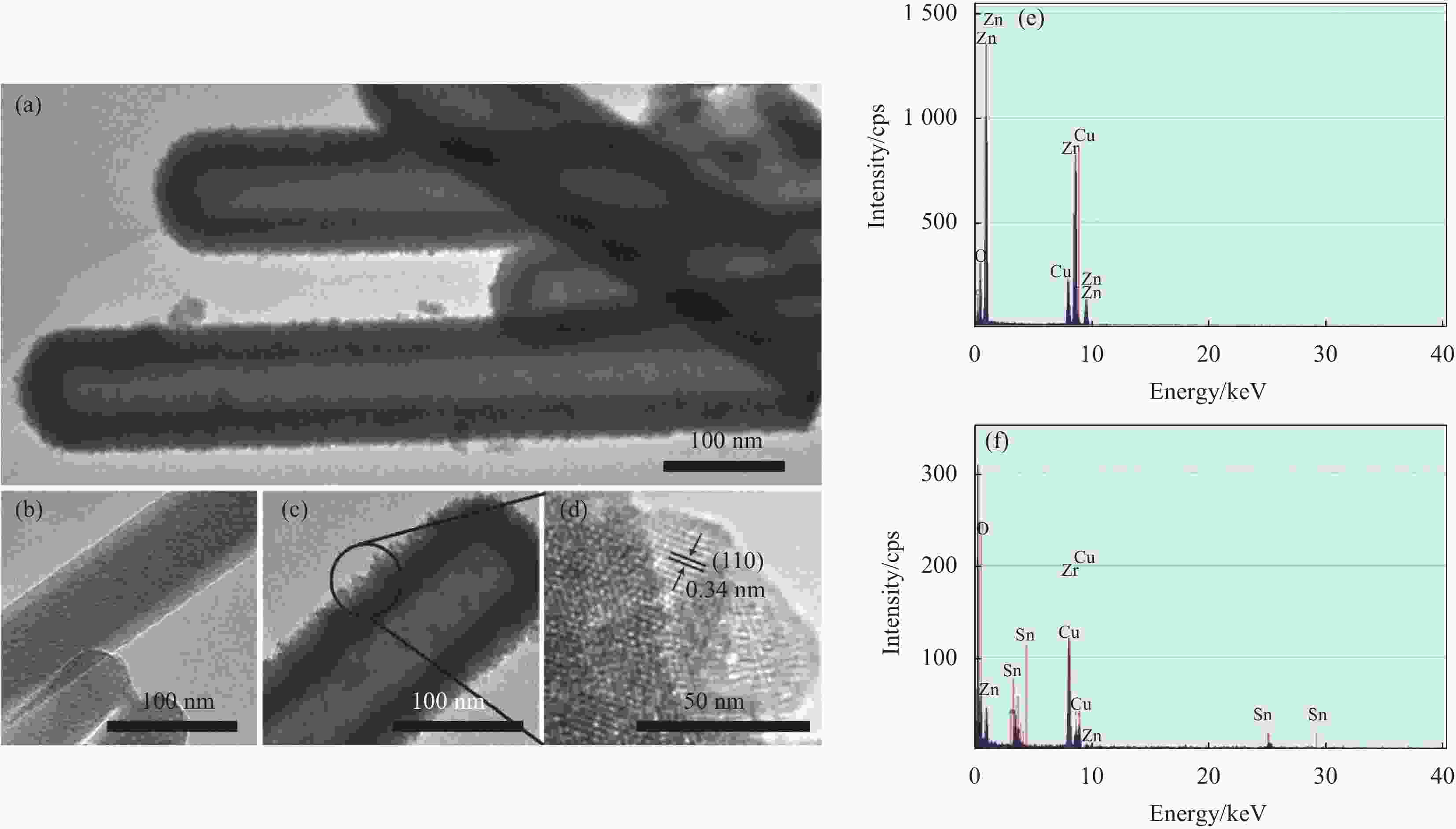
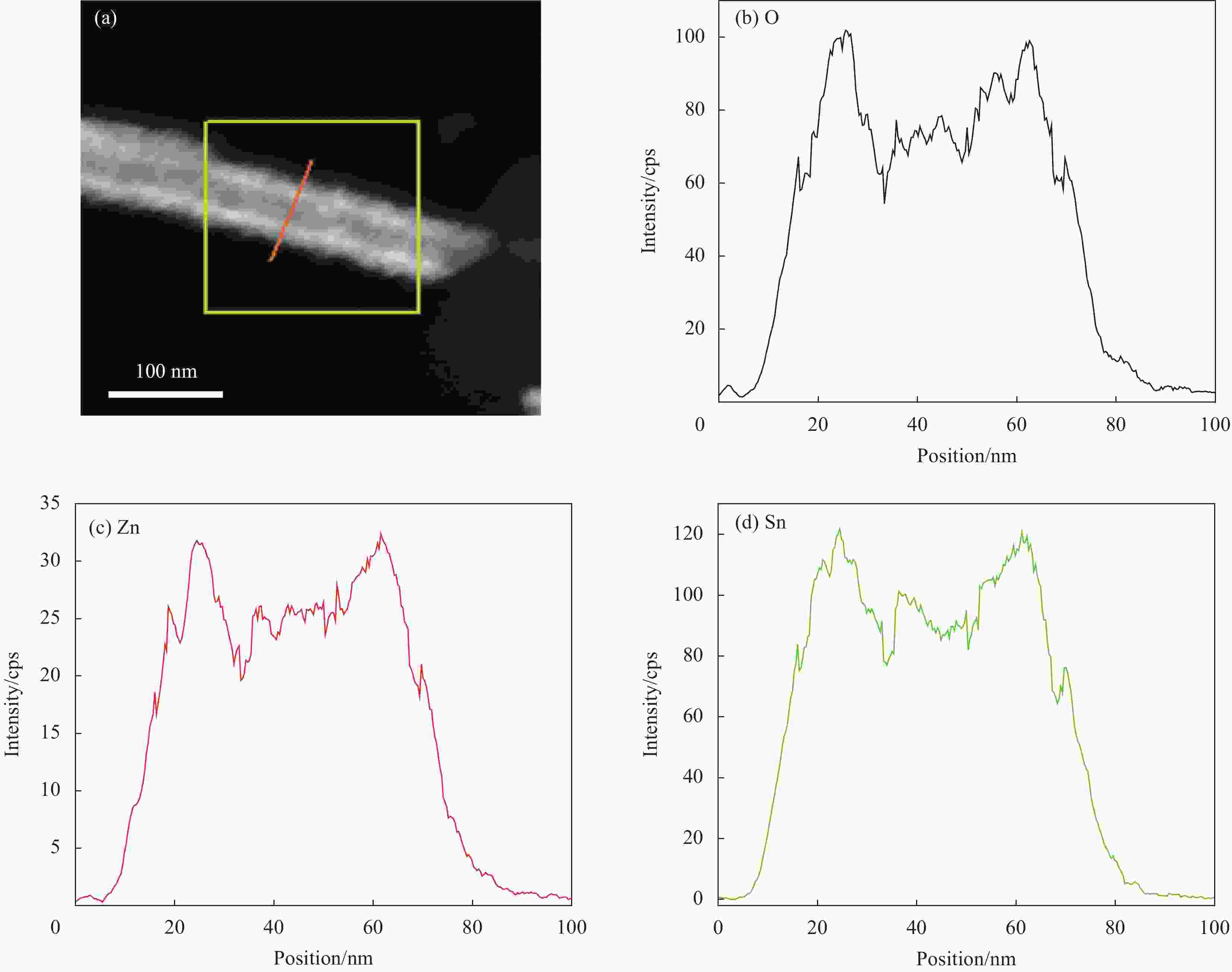
图 5 ZnO NRs在溶剂热生长SnO2壳前后的SEM图像:当SnO2前驱体浓度为0 mmol/L、2 mmol/L, 7 mmol/L和15 mmol/L时样品的SEM图像 ((a)~(d)) 及相应的断面SEM图像 ((e)~(h))
Figure 5. SEM images of ZnO NRs before and after SnO2 shell coating with hydrothermal method: SnO2 precursors concentration are set at 0 mmol/L (as-prepared ZnO NRs), 2 mmol/L, 7 mmol/L, 15 mmol/L, and the plane SEM images are shown in ((a)-(d)) and present the corresponding cross-sectional SEM images, respectively ((e)-(h))
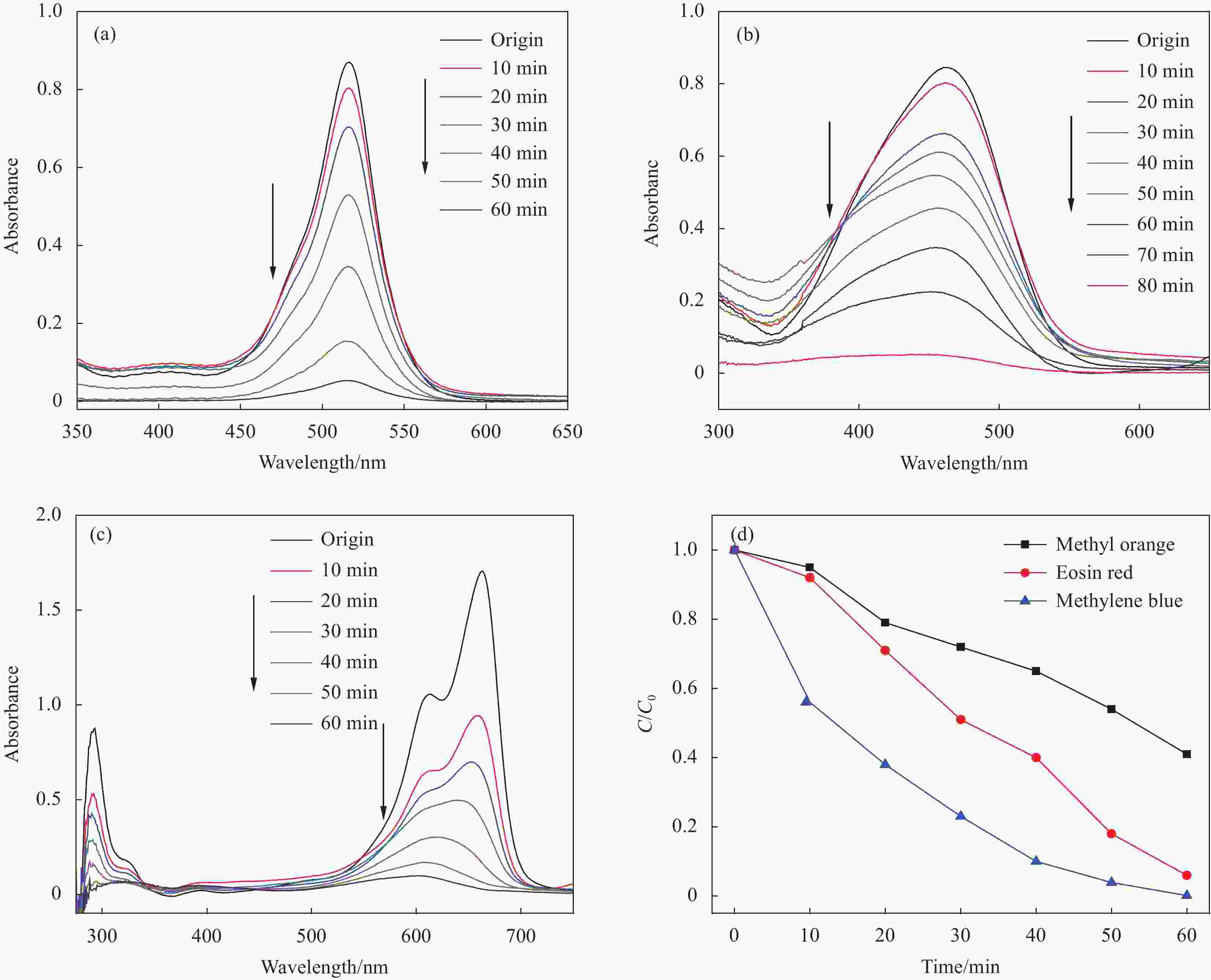
图 9 ZnO@SnO2 HDNs对曙红 (a)、甲基橙 (b) 和亚甲基蓝 (c) 的降解效果和上述3种有机染料的光催化降解速率图 (d)
Figure 9. UV-vis absorption spectra of eosin red (a), methyl orange (b) and methylene blue (c) in the presence of ZnO@SnO2 HDNs and the photocatalysis degradation rate of the three organic dyes (d)
C—Real-time concentration of organic dyes; C0—Initial concentration of an organic dye
表 1 本工作中ZnO@SnO2 HDNs与以往报道复合光催化剂对甲基橙光催化降解性能的比较
Table 1. Comparison of photocatalytic performance of ZnO@SnO2 HDNs toward methyl orange in this work with previously reported composite photocatalysts
Sample Light source Experimental condition Degradation time Ref. ZnO/SnO2 nanocomposites 300 W Hg lamp 200 mg; 100 mL, 6×10−5 mol/L 100 min/56% [31] SnO2 aerogel/rGO nanocomposite 40 W UV lamp
(370 nm)100 mg L−1; 1×10−5 mol/L 60 min/84% [32] SnO2-CNT nanocomposites 9 W eight UV-vis
lamps (365 nm)200 mg; 100 mL, 3×10−5 mol/L 180 min/79% [33] Chitosan-SnO2 8 W mercury lamp 100 mg; 5×10−5 mol/L 100 min/92% [34] Cu-doped SnO2 QDs 200 W xenon lamp 100 mg; 100 mL, 6×10−5 mol/L 180 min/99% [35] ZnO@SnO2 HDNs 500 W xenon lamp 50 mg 80 min/95% This work Notes: rGO—reduced graphene oxide; CNT—carbon nanotube; QDs—quantum dot. -
[1] 衣晓虹, 王崇臣. 铁基金属-有机骨架及其复合物高级氧化降解水中新兴有机污染物[J]. 化学进展, 2021, 33(3):471-489.YI Xiaohong, WANG Chongchen. Elimination of emerging organic contaminants in wastewater by advanced oxidation process over iron-based mofs and their composites[J]. Progress in chemistry,2021,33(3):471-489(in Chinese). [2] LIU Y T, ZHAGN Q P, XU M, et al. Novel and efficient synthesis of Ag-ZnO nanoparticles for the sunlight-induced photocatalytic degradation[J]. Applied Surface Science,2019,476:632-640. doi: 10.1016/j.apsusc.2019.01.137 [3] ZOU W X, GUO B, OK Y S, et al. Integrated adsorption and photocatalytic degradation of volatile organic compounds (VOCs) using carbon-based nanocomposites: A critical review[J]. Chemosphere,2019,218:845-859. doi: 10.1016/j.chemosphere.2018.11.175 [4] CHEN F, MA, T, ZHANG, T, et al. Atomic-level charge separation strategies in semiconductor-based photocatalysts[J]. Advanced Materials,2021,33:2005256. doi: 10.1002/adma.202005256 [5] 孟培媛, 郭明媛, 乔勋. WS2/g-C3N4异质结光催化分解水制氢性能及机制[J]. 复合材料学报, 2021, 38(2):591-600.MENG Peiyuan, GUO Mingyuan, QIAO Xun. H2 production performance of photocatalyst and mechanism of WS2/g-C3N4 heterojunction[J]. Acta Materiae Compositae Sinica,2021,38(2):591-600(in Chinese). [6] ZHANG L, JARONIEC M. Toward designing semiconductor-semiconductor heterojunctions for photocatalytic applications[J]. Applied Surface Science,2017,430(1):2-17. [7] ZHANG Y F, ZHU Y K, LV C X, et al. Enhanced visible-light photoelectrochemical performance via chemical vapor deposition of Fe2O3 on a WO3 film to form a heterojunction[J]. Rare Metals,2020,39(7):841-849. doi: 10.1007/s12598-019-01311-5 [8] YANG L Q, HUANG J F, SHI L, et al. Efficient hydrogen evolution over Sb doped SnO2 photocatalyst sensitized by Eosin Y under visible light irradiation[J]. Nano Energy,2017,36:331-340. doi: 10.1016/j.nanoen.2017.04.039 [9] LI D G, HUANG J X, LI R B, et al. Synthesis of a carbon dots modified g-C3N4/SnO2 Z-scheme photocatalyst with superior photocatalytic activity for PPCPs degradation under visible light irradiation[J]. Journal of Hazardous Materials,2021,401(5):123257. [10] SUN Y K, ZHU Q, BAI B, et al. Novel all-solid-state Z-scheme SnO2/Pt/In2O3 photocatalyst with boosted photocatalytic performance on water splitting and 2, 4-dichlorophenol degradation under visible light[J]. Che-mical Engineering Journal,2020,390(15):124518. [11] LIU H J, DU C W, LI M, et al. One-pot hydrothermal synthesis of SnO2/BiOBr heterojunction photocatalysts for the efficient degradation of organic pollutants under visible light[J]. ACS Applied Materials & Interfaces,2018,10(34):28686-28694. [12] HU R, ZHANG H, LIU J, et al. Deformable fibrous carbon supported ultrafine nano-SnO2 as a high volumetric capacity and cyclic durable anode for Li storage[J]. Journal of Materials Chemistry A,2015,3(29):15097-15107. doi: 10.1039/C5TA03401A [13] SHEN R, HONG Y, STANKOVICH J J, et al. Synthesis of cambered nano-walls of SnO2/rGO composites using a recyclable melamine template for lithium-ion batteries[J]. Journal of Materials Chemistry A,2015,3(34):17635-17643. doi: 10.1039/C5TA03166D [14] LWIN H M, ZHAN W Q, SONG S X, et al. Visible-light photocatalytic degradation pathway of tetracycline hydrochloride with cubic structured ZnO/SnO2 heterojunction nanocatalyst[J]. Chemical Physics Letters,2019,736:136806. doi: 10.1016/j.cplett.2019.136806 [15] HE Z L, ZHANG J, LI X, et al. 1D/2D heterostructured photocatalysts: From design and unique properties to their environmental applications[J]. Small,2020,16(46):2005051. doi: 10.1002/smll.202005051 [16] HOU H L, ZHANG X W. Rational design of 1D/2D heterostructured photocatalyst for energy and environmental applications[J]. Chemical Engineering Journal,2020,395:125030. doi: 10.1016/j.cej.2020.125030 [17] ZHANG Y, PENG C D, HE Z T, et al. Interface engineering of heterojunction photocatalysts based on 1D nanomate-rials[J]. Catalysis Science & Technology,2021,11:27-42. [18] HAN Z H, HONG W Z, XING W N, et al. Shockley partial dislocation-induced self-rectified 1D hydrogen evolution photocatalyst[J]. ACS Applied Materials & Interfaces,2019,11(22):20521-20527. [19] WANG W J, NIU Q Y, ZENG G M, et al. 1D porous tubular g-C3N4 capture black phosphorus quantum dots as 1D/0D metal-free photocatalysts for oxytetracycline hydrochloride degradation and hexavalent chromium reduction[J]. Applied Catalysis B: Environmental,2020,273:119051. doi: 10.1016/j.apcatb.2020.119051 [20] MAO L, CAI X Y, ZHU M S, Hierarchically 1D CdS decorated on 2D perovskite-type La2Ti2O7 nanosheet hybrids with enhanced photocatalytic performance[J]. Rare Metals, 2021, 40: 1067-1076. [21] CHEN X, ZHANG F, WANG Q, et al. The synthesis of ZnO/SnO2 porous nanofibers for dye adsorption and degradation[J]. Dalton Transactions,2015,44(7):3034-3042. doi: 10.1039/C4DT03382E [22] WANG C, HU L, CHAI B, et al. Enhanced photocatalytic activity of electrospun nanofibrous TiO2/g-C3N4 heterojunction photocatalyst under simulated solar light[J]. Applied Surface Science,2017,430(1):243-252. [23] WANG J Z, DU N, ZHANG H, et al. Large-scale synthesis of SnO2 nanotube arrays as high-performance anode mate-rials of Li-ion batteries[J]. The Journal of Physical Chemistry C,2011,115(22):11302-11305. doi: 10.1021/jp203168p [24] MONIZ S J, SHEVLIN S A, MARTIN D J, et al. Visible-light driven heterojunction photocatalysts for water splitting-A critical review[J]. Energy & Environmental Science,2015,8(3):731-759. [25] WANG X, SUN F, DUAN Y, et al. Highly sensitive, tempera-ture-dependent gas sensor based on hierarchical ZnO nanorod arrays[J]. Journal of Materials Chemistry C,2015,3(43):11397-11405. doi: 10.1039/C5TC02187A [26] JUTTUKONDA V PADDOCK R L, RAYMOND J E, et al. Facile synthesis of tin oxide nanoparticles stabilized by dendritic polymers[J]. Journal of the American Chemical Society,2006,128:420-421. doi: 10.1021/ja056902n [27] GAO C M, WEI T, ZHANG Y Y, et al. A Photoresponsive rutile TiO2 heterojunction with enhanced electron-hole separation for high-performance hydrogen evolution[J]. Advanced Materials,2019,31(8):1806596. doi: 10.1002/adma.201806596 [28] WANG C, WANG K W, FENG Y B, et al. Co and Pt dual-single-atoms with oxygen-coordinated Co-O-Pt dimer sites for ultrahigh photocatalytic hydrogen evolution efficiency[J]. Advanced Materials,2021,33:2003327. doi: 10.1002/adma.202003327 [29] ZHUANG T T, LIU Y, LI Y, et al. Integration of semiconducting sulfides for full-spectrum solar energy absorption and efficient charge separation[J]. Angewandte Chemie International Edition,2016,55:6396-6400. doi: 10.1002/anie.201601865 [30] FENG N D, LIN H W, DENG F, et al. Interfacial-bonding Ti-N-C boosts efficient photocatalytic H2 evolution in close coupling g-C3N4/TiO2[J]. The Journal of Physical Chemistry C,2021,125(22):12012-12018. doi: 10.1021/acs.jpcc.1c02606 [31] ALI W, ULLAH H, ZADA A, et al. Effect of calcination temperature on the photoactivities of ZnO/SnO2 nanocompo-sites for the degradation of methyl orange[J]. Materials Chemistry and Physics,2018,213(1):259-266. [32] KIM T, PARALE V G, JUNG H N, et al. Facile synthesis of SnO2 aerogel/reduced graphene oxide nanocomposites via in situ annealing for the photocatalytic degradation of methyl orange[J]. Nanomaterials,2019,9(3):358. doi: 10.3390/nano9030358 [33] KIM S P, CHOI M Y, CHOI H C, Characterization and photocatalytic performance of SnO2-CNT nanocomposites[J]. Applied Surface Science, 2015, 357: 302-308. [34] GUPTA V K, SARAVANAN R, AGARWAL S, et al. Degradation of azo dyes under different wavelengths of UV light with chitosan-SnO2 nanocomposites[J]. Journal of Molecular Liquids,2017,232:423-430. doi: 10.1016/j.molliq.2017.02.095 [35] BABU B, KADAM A, RAVIKUMAR R, et al. Enhanced visible light photocatalytic activity of Cu-doped SnO2 quantum dots by solution combustion synthesis[J]. Jour-nal of Alloys and Compounds,2017,703(5):330-336. -






 下载:
下载: