Research progress in synthesis and catalytic application of carbon/metal oxide composites
-
摘要: 碳材料和金属氧化物为常用的多相催化剂载体,其中碳材料具有巨大的比表面积、良好的吸附性能,但力学性能较差。金属氧化物载体则具有较高的机械强度和热稳定性,但常常会与活性金属作用太强使其催化活性降低。碳/金属氧化物复合材料作为一类新型材料受到了科学家的广泛关注,由于碳与金属氧化物(如氧化钛、氧化铝等)之间的协同作用,该复合材料表现出不同于单一组分的独特物理化学性质,重点综述了该类复合材料的制备方法及其在催化领域近年来的研究进展。Abstract: Carbon materials and metal oxides are commonly used heterogeneous catalyst supports. Among them, carbon materials have large specific surface area and good adsorption performance, but have poor mechanical properties. Metal oxide supports have good mechanical and thermal stability, but they usually have a strong interaction with active metals leading to lower catalytic activity. As a new type of material, carbon/metal oxide composites have attracted extensive attention from scientists. Because of the synergistic effect between carbon and metal oxides (such as titania, alumina, etc.), the composites exhibit unique physical and chemical properties different from a single component. In this review, the preparation methods of the composites and the research progress in the field of catalysis in recent years were summarized.
-
Key words:
- carbon/metal oxide composites /
- synthesis /
- synergistic effect /
- catalysis /
- carbon materials /
- application
-
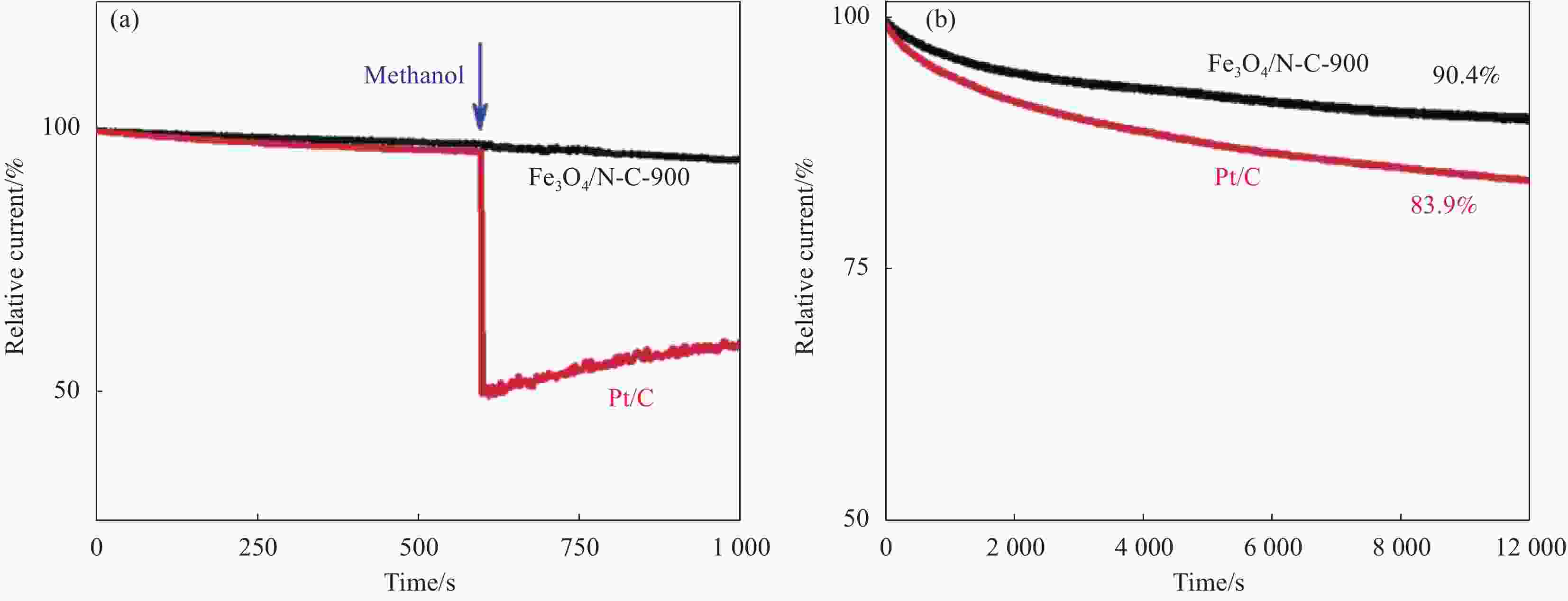
图 4 在−0.4 V、O2饱和的0.1 mol/L KOH中加入 (a) 、不加入 (b) 甲醇(1.0 mol/L)时Fe3O4/N-C-900和Pt/C的电流-时间(I-t)图[51]
Figure 4. Current-time (I–t) plots of Fe3O4/N-C-900 and Pt/C at −0.4 V in O2-saturated 0.1 mol/L KOH with (a) and without (b) the addition of methanol (1.0 mol/L)[51]
Fe3O4/N-C-900—Pyrolyzed Fe3O4/N-C at 900℃
表 1 N, N-二甲基胺-N′, N′-二甲氨基甲酸盐(DIMCARB)与CO2/H2合成N,N-二甲基甲酰胺(DMF)a的反应结果[38]
Table 1. Results for N, N-dimethylformamide (DMF)a synthesis from N, N-dimethylamine-N ', N '-dimethyl carbamate (DIMCARB) and CO2/H2[38]
Entry Ligand/10% Yied/%b Content/% DMF N(CH3)3 1 None 93.7 41.8 58.2 2 Et3N 90.0 30.3 69.7 3 PhNMe2 89.4 30.3 69.7 4 Py 99.0 44.0 56.0 5 Bipy 91.3 43.6 56.4 6 TMEDA 98.2 52.6 47.4 7 DMEDA 92.1 77.9 22.1 8 1,10-Phenanthro-line 89.7 86.7 13.3 9c 1,10-Phenanthro-line 93.0 78.4 21.6 10d 1,10-Phenanthro-line 98.3 92.0 8.0 11e 1,10-Phenanthro-line 95.0 97.3 2.7 12f 1,10-Phenanthro-line 89.6 97.3 2.7 Notes: a—DIMCARB (0.5 mmol, equal to 1 mmol HNMe2), CuAlOx (100 mg, 9 mmol% Cu), ligand (10 mol%), 1, 2-Dimethoxyethane (4 mL), 3 MPa CO2, 7 MPa H2, 160 °C, 24 h;
b—Combined yield of DMF and N(CH3)3 were determined by GC-FID using 1, 4-dioxane as the internal standard material;
c—1, 10-Phen (5 mol%);
d—1, 10-Phen (20 mol%);
e—1, 10-Phen (30 mol%);
f—Catalyst was reused at the third run; Bipy—2, 2′-bipyridine; Py—pyridine; TMEDA—N, N, N′, N′-Tetramethylethyleneediamine; DMEDA—N, N′-di- methylethylenediamine.表 2 NiZG-70 NCs与已有文献对MB的光降解效果的对比
Table 2. Comparison of photodegradation of MB by NiZG-70 NCs and some previous literatures
-
[1] JIN X, GU T H, KWON N H, et al. Synergetic advantages of atomically coupled 2D inorganic and graphene nanosheets as versatile building blocks for diverse functional nanohybrids[J]. Advanced Materials,2021,164:126176. [2] VISSERS J P R, MERCX F P M, BOUWENS S M A M, et al. Carbon-covered alumina as a support for sulfide catalysts[J]. Journal of Catalysis,1988,114(2):291-302. doi: 10.1016/0021-9517(88)90033-4 [3] SAKTHIVEL S, KISCH H. Daylight photocatalysis by carbon-modified titanium dioxide[J]. Angewandte Chemie International Edition,2010,42(40):4908-4911. [4] MU J, CHEN B, GUO Z, et al. Highly dispersed Fe3O4 nanosheets on one-dimensional carbon nanofibers: Synthesis, formation mechanism, and electrochemical performance as supercapacitor electrode materials[J]. Nanoscale,2011,3(12):5034-5040. doi: 10.1039/c1nr10972c [5] WANG J G, YANG Y, HUANG Z H, et al. Incorporation of nanostructured manganese dioxide into carbon nanofibers and its electrochemical performance[J]. Materials Letters,2012,72:18-21. doi: 10.1016/j.matlet.2011.12.071 [6] LV P, ZHAO H, ZENG Z, et al. Facile preparation and electrochemical properties of carbon coated Fe3O4 as anode material for lithium-ion batteries[J]. Journal of Power Sources,2014,259:92-97. doi: 10.1016/j.jpowsour.2014.02.081 [7] LI L, LAFDI K. Composite films prepared by immersion deposition of manganese oxide in carbon nanotubes grown on graphite for supercapacitors[J]. Journal of Materials Science,2011,46(22):7328-7334. doi: 10.1007/s10853-011-5694-2 [8] VELLACHERI R, PILLAI V K, KURUNGOT S. Hydrous RuO2-carbon nanofiber electrodes with high mass and electrode-specific capacitance for efficient energy storage[J]. Nanoscale,2012,4(3):890-896. doi: 10.1039/C2NR11479H [9] HOANG D L, PREISS H, PARLITZ B, et al. Zirconia/carbon composites as monofunctional catalysts in C6+ alkane aromatization[J]. Applied Catalysis A,1999,182(2):385-397. doi: 10.1016/S0926-860X(99)00033-2 [10] ZHANG L, ZHOU M, WANG A, et al. Selective hydrogenation over supported metal catalysts: From nanoparticles to single atoms[J]. Chemical Reviews,2020,120(2):683-733. doi: 10.1021/acs.chemrev.9b00230 [11] 俞铁铭, 李艳, 徐晓玲, 等. 掺杂Co对CNTs负载Pt催化剂上肉桂醛选择性催化加氢性能的影响[J]. 化工学报, 2009, 60(7):1667-1672.YU Tieming, LI Yan, XU Xiaoling, et al. Effect of Co on selective hydrogenation of cinnamaldehyde to cinnamyl alcohol over Pt/CNTs catalyst[J]. Journal of Chemical Industry and Engineering(China),2009,60(7):1667-1672(in Chinese). [12] HANDJANI S, MARCEAU E, BLANCHARD J, et al. Influence of the support composition and acidity on the catalytic properties of mesoporous SBA-15, Al-SBA-15, and Al2O3-supported Pt catalysts for cinnamaldehyde hydrogenation[J]. Journal of Catalysis,2011,282(1):228-236. doi: 10.1016/j.jcat.2011.06.017 [13] WU H C, CHEN T C, WU J H, et al. Influence of sodium-modified Ni/SiO2 catalysts on the tunable selectivity of CO2 hydrogenation: Effect of the CH4 selectivity, reaction pathway and mechanism on the catalytic reaction[J]. Journal of Colloid and Interface Science,2020,586:514-527. [14] KOSTYNIUK A, BAJEC D, LIKOZAR B. Catalytic hydrogenation, hydrocracking and isomerization reactions of biomass tar model compound mixture over Ni-modified zeolite catalysts in packed bed reactor[J]. Renewable Energy,2020,167:409-424. [15] CHEN W, CHENG B, SUN Q, et al. Preparation of MOF confined Ag nanoparticles for the highly active, size selective hydrogenation of olefins[J]. ChemCatChem,2018,10(17):3659-3665. doi: 10.1002/cctc.201800744 [16] NOYORI R, HASHIGUCHI S. Asymmetric transfer hydrogenation catalyzed by chiral ruthenium complexes[J]. Accounts of Chemical Research,1997,30(2):97-102. doi: 10.1021/ar9502341 [17] CLIMENT M J, CORMA A, IBORRA S. Heterogeneous catalysts for the one-pot synthesis of chemicals and fine chemicals[J]. Chemical Reviews,2011,111(2):1072-1133. doi: 10.1021/cr1002084 [18] GIANVITO V, ALBANI D, NEYVIS A B, et al. Advances in the design of nanostructured catalysts for selective hydrogenation[J]. ChemCatChem,2016,8(1):21-33. doi: 10.1002/cctc.201501269 [19] GALLEZOT P, RICHARD D. Selective hydrogenation of α, β-unsaturated aldehydes[J]. Catalysis Reviews,1998,40(1):81-126. [20] YANG Y, RAO D, CHEN Y, et al. Selective hydrogenation of cinnamaldehyde over Co-based intermetallic compounds derived from layered double hydroxides[J]. ACS Catalysis,2018,8(12):11749-11760. doi: 10.1021/acscatal.8b02755 [21] ZHANG Z, SONG J, JIANG Z, et al. Direct synthesis of ultrasmall ruthenium nanoparticles on porous supports using natural sources for highly efficient and selective furfural hydrogenation[J]. ChemCatChem,2017,9(13):2448-2452. doi: 10.1002/cctc.201700262 [22] WANG G H, DENG X, GU D, et al. Co3O4 nanoparticles supported on mesoporous carbon for selective transfer hydrogenation of α, β-unsaturated aldehydes[J]. Angewandte Chemie International Edition,2016,128(37):11267-11271. [23] LI C, KE C, HAN R, et al. The remarkable promotion of in situ formed Pt-cobalt oxide interfacial sites on the carbonyl reduction to allylic alcohols[J]. Molecular Catalysis,2018,455:78-87. doi: 10.1016/j.mcat.2018.05.028 [24] CRESPO-QUESADA M, CÁRDENAS-LIZANA F, DESSIMOZ A L, et al. Modern trends in catalyst and process design for alkyne hydrogenations[J]. ACS Catalysis,2012,2(8):1773-1786. doi: 10.1021/cs300284r [25] BRIDIER B, JAVIER P. Cooperative effects in ternary Cu−Ni−Fe catalysts lead to enhanced alkene selectivity in alkyne hydrogenation[J]. Journal of the American Chemical Society,2010,132(12):4321-4327. doi: 10.1021/ja9101997 [26] YAN X L, BAO J H, YUAN C, et al. Gold on carbon and titanium oxides composites: Highly efficient and stable acetylene hydrogenation in large excess of ethylene[J]. Journal of Catalysis,2016,344:194-201. doi: 10.1016/j.jcat.2016.09.018 [27] CORMA A, SERNA P. Chemoselective hydrogenation of nitro compounds with supported gold catalysts[J]. Science,2006,313(5785):332-334. doi: 10.1126/science.1128383 [28] WEI H, WEI X, YANG X, et al. Supported Au-Ni nano-alloy catalysts for the chemoselective hydrogenation of nitroarenes[J]. Chinese Journal of Catalysis,2015,32(2):160-167. [29] TIAN H, LIU X, DONG L, et al. Submicroreactors: Enhanced hydrogenation performance over hollow structured Co-CoOx@N-C Capsules[J]. Advanced Science,2019,6(22):1970135. doi: 10.1002/advs.201970135 [30] CHEN B, LI F, HUANG Z, et al. Recyclable and selective nitroarene hydrogenation catalysts based on carbon-coated cobalt oxide nanoparticles[J]. ChemCatChem,2016,8(6):1132-1138. doi: 10.1002/cctc.201501265 [31] ZHANG J, JIANG H, LIU Y, et al. Tuning surface properties of N-doped carbon with TiO2 nano-islands for enhanced phenol hydrogenation to cyclohexanone[J]. Applied Surface Science,2019,488(15):555-564. [32] SONG T, MA Z, YANG Y. Chemoselective hydrogenation of α, β-unsaturated carbonyls catalyzed by biomass-derived cobalt nanoparticles in water[J]. ChemCatChem,2019,11(4):1313-1319. doi: 10.1002/cctc.201801987 [33] XIONG H, PHAM H N, DATYE A K. A facile approach for the synthesis of niobia/carbon composites having improved hydrothermal stability for aqueous-phase reactions[J]. Journal of Catalysis,2013,302:93-100. doi: 10.1016/j.jcat.2013.03.007 [34] TRUNSCHKE A, HOANG D L, RADNIK J, et al. Transition metal oxide/carbon composite catalysts for N-alkane aromatization: Structure and catalytic properties[J]. Applied Catalysis A, General,2001,208(1-2):381-392. doi: 10.1016/S0926-860X(00)00736-5 [35] ISHIHARA A, ANDOU A, HASHIMOTO T, et al. Steam reforming of ethanol using novel carbon-oxide composite-supported Ni, Co and Fe catalysts[J]. Fuel Processing Technology,2020,197:106203. doi: 10.1016/j.fuproc.2019.106203 [36] XIONG H F, THOMAS J S, NALIN I A, et al. Graphitic-carbon layers on oxides: Toward stable heterogeneous catalysts for biomass conversion reactions[J]. Angewandte Chemie International Edition,2015,54(27):7939-7943. doi: 10.1002/anie.201502206 [37] LIU C, HE Y, WEI L, et al. Hydrothermal carbon-coated TiO2 as support for Co-based catalyst in fischer–tropsch synthesis[J]. ACS Catalysis,2018,8(2):1591-1600. doi: 10.1021/acscatal.7b03887 [38] WU Y, WANG T, WANG H, et al. Active catalyst construction for CO2 recycling via catalytic synthesis of N-doped carbon on supported Cu[J]. Nature Communications,2019,10(1):2599. doi: 10.1038/s41467-019-10633-y [39] WANG Q, HISATOMI T, JIA Q, et al. Scalable water splitting on particulate photocatalyst sheets with a solar-to-hydrogen energy conversion efficiency exceeding 1%[J]. Nature Materials,2016,15(6):611-615. doi: 10.1038/nmat4589 [40] YU D, YU G. A bio-inspired, heavy-metal-free, dual-electrolyte liquid battery towards sustainable energy storage[J]. Angew Chem Int Ed Engl,2016,55(15):4772-4776. doi: 10.1002/anie.201600705 [41] ZHAO P, XU W, HUA X, et al. Facile synthesis N-doped Fe3C@CNT/porous carbon hybrid for an advanced oxygen reduction and water oxidation electrocatalyst[J]. The Journal of Physical Chemistry C,2016,120:11006-11013. doi: 10.1021/acs.jpcc.6b03070 [42] 郭亚肖, 商昌帅, 李敬, 等. 电催化析氢、析氧及氧还原研究进展[J]. 中国科学: 化学, 2018, 48(8):926-940. doi: 10.1360/N032018-00063GUO Yaxiao, SHANG Changshuai, LI Jing, et al. Research progress of electrocatalytic hydrogen evolution, oxygen evolution and oxygen reduction[J]. Scientia Sinica Chimica,2018,48(8):926-940(in Chinese). doi: 10.1360/N032018-00063 [43] 康启平, 张国强, 张志芸, 等. PEMFC非金属催化剂的研究进展[J]. 电池, 2019, 49(1):76-78.KANG Qiping, ZHANG Guoqiang, ZHANG Zhiyun, et al. Research progress in metal-free catalysts for PEMFC[J]. Battery,2019,49(1):76-78(in Chinese). [44] ZHAO C, LIU J, WANG J, et al. Recent advances of noble-metal-free bifunctional oxygen reduction and evolution electrocatalysts.[J]. Chemical Society Reviews,2021,50(13):135. doi: 10.1039/D1CS00135C [45] CHEN M, WANG L, YANG H, et al. Nanocarbon/oxide composite catalysts for bifunctional oxygen reduction and evolution in reversible alkaline fuel cells: A mini review[J]. Journal of Power Sources,2017,375(31):277-290. [46] 付永胜, 毕敏, 李春, 等. 非贵金属/碳氮复合材料电催化析氧反应的研究进展[J/OL]. 无机材料学报, 2021.DOI: 10.15541/jim20210015.FU Y S, BI M, LI C, et al. Research progress on non-noble metal/nitrogen-doped carbon composite materials in electrocatalytic oxygen evolution reaction[J/OL]. Journal of Inorganic Materials, 2021.DOI: 10.15541/jim20210015. [47] POUX T, NAPOLSKIY F S, DINTZER T, et al. Dual role of carbon in the catalytic layers of perovskite/carbon composites for the electrocatalytic oxygen reduction reaction[J]. Catalysis Today,2012,189(1):83-92. doi: 10.1016/j.cattod.2012.04.046 [48] RYABOVA A S, BONNEFONT A, SIMONOV P A, et al. Further insights into the role of carbon in manganese oxide/carbon composites in the oxygen reduction reaction in alkaline media[J]. Electrochimica Acta,2017,246:643-653. doi: 10.1016/j.electacta.2017.06.017 [49] ANDERSEN N I, SEROV A, ATANASSOV P. Metal oxides/CNT nano-composite catalysts for oxygen reduction/oxygen evolution in alkaline media[J]. Applied Catalysis B: Environmental,2015,163:623-627. doi: 10.1016/j.apcatb.2014.08.033 [50] TAN Y, XU C, CHEN G, et al. Facile synthesis of manganese-oxide-containing mesoporous nitrogen-doped carbon for efficient oxygen reduction[J]. Advanced Functional Materials,2012,22(21):4584-4591. doi: 10.1002/adfm.201201244 [51] SU Y, JIANG H, ZHU Y, et al. Enriched graphitic N-doped carbon-supported Fe3O4 nanoparticles as efficient electrocatalysts for oxygen reduction reaction[J]. Journal of Materials Chemistry A,2014,2(20):7281-7287. doi: 10.1039/C4TA00029C [52] LU Z W, CHEN J P, WANG W L, et al. Electrocatalytic, kinetic, and mechanism insights into the oxygen-reduction catalyzed based on the biomass-derived FeOx@N-doped porous carbon composites[J]. Small,2021,17(19):2007326. doi: 10.1002/smll.202007326 [53] LIU D, CHEN S, LI R, et al. Review of Z-scheme heterojunctions for photocatalytic energy conversion[J]. Acta Physico-Chimica Sinica,2020,37(6):2010017. [54] MUGUNTHAN E, SAIDUTTA M B, JAGADEESHBABU P E. Photocatalytic degradation of diclofenac using TiO2–SnO2 mixed oxide catalysts[J]. Environmental Technology,2019,40(7):929-941. doi: 10.1080/09593330.2017.1411398 [55] WANG X, KHAN M A, XIA M, et al. Synthesis of RGO and g-C3N4 hybrid with WO3/Bi2WO6 to boost degradation of nitroguanidine under visible light irradiation[J]. Journal of Materials Science Materials in Electronics,2019,30:5503-5515. doi: 10.1007/s10854-019-00844-w [56] HARIWAL R, MALIK H K, NEGI A, et al. Controlling room temperature ferromagnetism and band gap in ZnO nanostructured thin films by varying angle of implantation[J]. RSC Advances,2018,8(12):6278-6287. doi: 10.1039/C7RA10615G [57] NEMIWAL M, ZHANG T C, KUMAR D. Recent progress in g-C3N4, TiO2 and ZnO based photocatalysts for dye degradation: Strategies to improve photocatalytic activity[J]. Science of The Total Environment,2021,767:144896. doi: 10.1016/j.scitotenv.2020.144896 [58] LIANG B, WANG D, JIANG H, et al. Recent advances in heteroatom doped graphitic carbon nitride (g-C3N4) and g-C3N4/metal oxide composite photocatalysts[J]. Current Organic Chemistry,2020,24(6):673-693. doi: 10.2174/1385272824666200309151648 [59] 于翔, 张雪寅, 李如洋, 等. TiO2/静电纺PAN基碳复合材料的制备及光催化性能[J]. 复合材料学报, 2020, 37(12):3177-3183.YU Xiang, ZHANG Xueyin, LI Ruyang, et al. Preparation and photocatalytic properties of TiO2/electrospinning PAN-based carbon composite material[J]. Acta Materiae Compositae Sinica,2020,37(12):3177-3183(in Chinese). [60] 胡海霞, 廖金龙, 朱曜峰, 等. 碳纤维负载NaTaO3光催化复合材料的制备及其性能[J]. 复合材料学报, 2019, 36(6):1501-1509.HU Haixia, LIAO Jinlong, ZHU Yaofeng, et al. Preparation and properties of NaTaO3 deposited on carbonfiber photocatalytic composites[J]. Acta Materiae Compositae Sinica,2019,36(6):1501-1509(in Chinese). [61] XIN S, MA B, LIU G, et al. Enhanced heterogeneous photo-Fenton-like degradation of tetracycline over CuFeO2/biochar catalyst through accelerating electron transfer under visible light[J]. Journal of Environmental Management,2021,285:112093. doi: 10.1016/j.jenvman.2021.112093 [62] LI J, HUI L, ZHANG W, et al. Scalable production of ultra small TiO2 nano crystal/activated carbon composites by atomic layer deposition for efficient removal of organic pollutants[J]. Advanced Powder Technology,2021,32(3):728-739. doi: 10.1016/j.apt.2021.01.013 [63] JIA J, WANG Y, XU M, et al. MOF-derived the direct Z-scheme g-C3N4/TiO2 with enhanced visible photocatalytic activity[J]. Journal of Sol-Gel Science and Technology,2020,93(1):123-130. doi: 10.1007/s10971-019-05172-3 [64] QAMAR M A, SHAHID S, JAVED M, et al. Designing of highly active g-C3N4 /Ni-ZnO photocatalyst nanocomposite for the disinfection and degradation of the organic dye under sunlight radiations[J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects,2021,614:126176. doi: 10.1016/j.colsurfa.2021.126176 [65] JAVED M, HUSSAIN S. Synthesis, characterization and photocatalytic applications of p (Aac) microgels and its composites of Ni doped ZnO nanorods[J]. Digest Journal of Nanomaterials and Biostructures,2020,15:217-230. [66] SHAHID I, MOHSIN J, ALI B, et al. Controlled synthesis of Ag-doped CuO nanoparticles as a core with poly(acrylic acid) microgel shell for efficient removal of methylene blue under visible light[J]. Journal of Materials Science: Materials in Electronics,2020,31:8423-8435. doi: 10.1007/s10854-020-03377-9 [67] QAMAR M A, SHAHID S, JAVED M. Synthesis of dynamic g-C3N4/Fe@ZnO nanocomposites for environmental remediation applications[J]. Ceramics International,2020,46:22171-22180. doi: 10.1016/j.ceramint.2020.05.294 [68] QAMAR M A, SHAHID S, JAVED M, et al. Fabricated novel g-C3N4/Mn doped ZnO nanocomposite as highly active photocatalyst for the disinfection of pathogens and degradation of the organic pollutants from wastewater under sunlight radiations[J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects,2020,611:125863. [69] 张隐, 黄慧玲, 魏留洋, 等. 生物质炭/ZnO复合材料的制备及其吸附-光催化性能[J]. 复合材料学报, 2019, 36(9):2187-2195.ZHANG Yin, HUANG Huiling, WEI Liuyang, et al. Preparation and adsorption-photocataly-sis properties of biochar/ZnO composites[J]. Acta Materiae Compositae Sinica,2019,36(9):2187-2195(in Chinese). -






 下载:
下载: