Synthesis and visible light photocatalytic properties of Bi2MoO6/WO3 composite photocatalysts
-
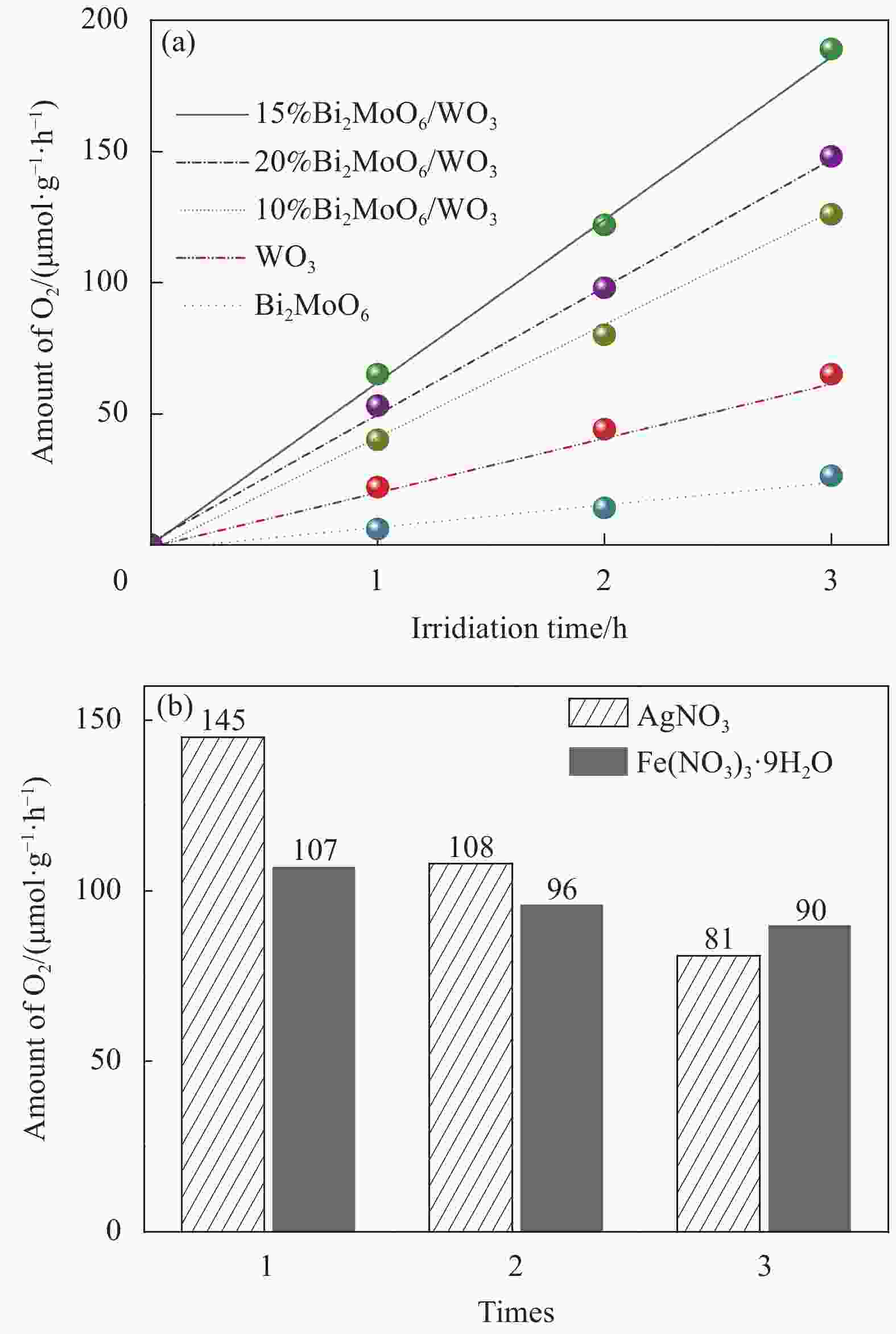
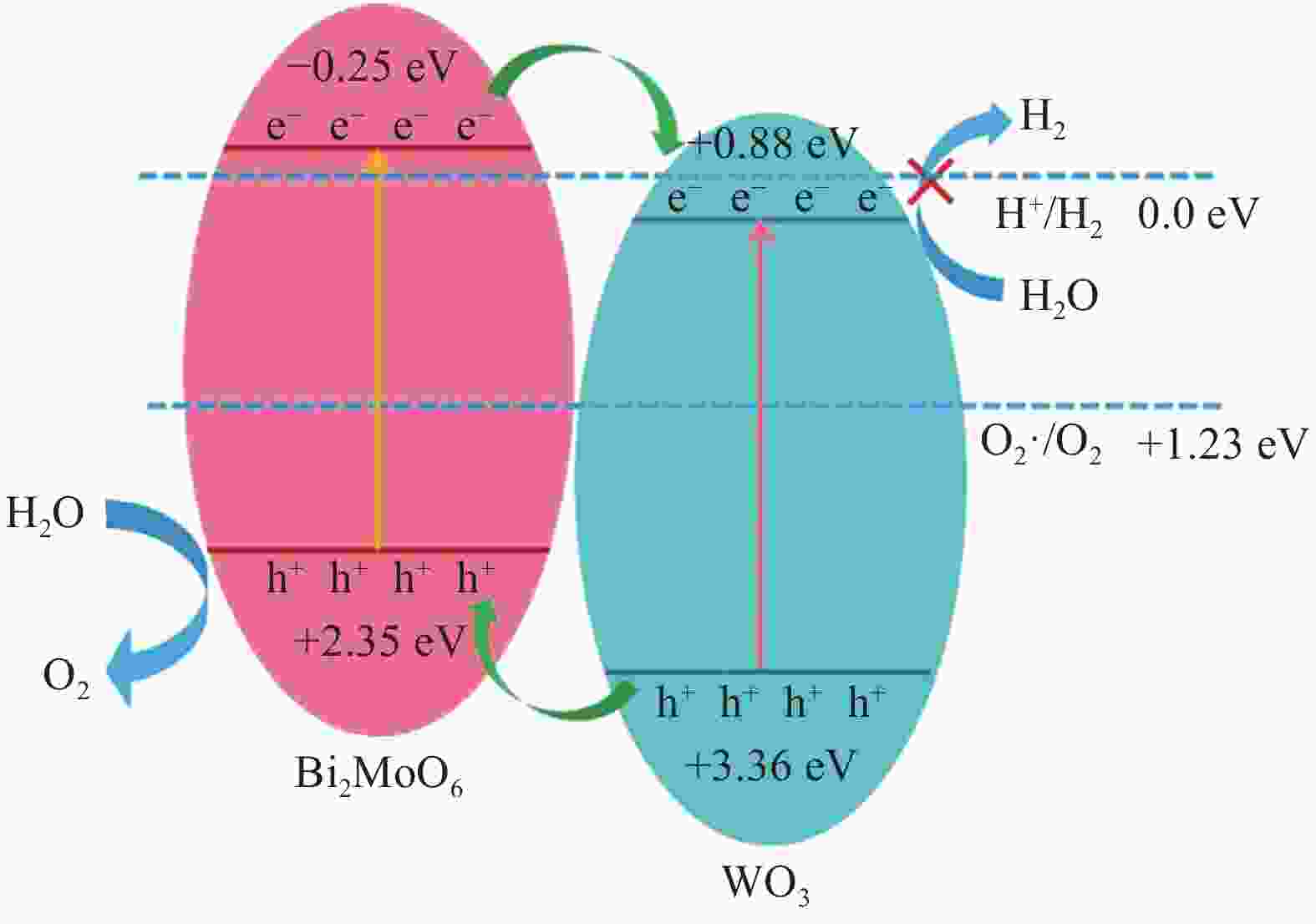
摘要: 采用水热法成功制备了不同Bi2MoO6含量的Bi2MoO6/WO3复合光催化剂,利用XRD、SEM、UV-Vis、EIS和PL对样品进行了微观结构、形貌、光吸收特性、光谱响应和光电流的测试与表征,并考察了Bi2MoO6/WO3复合材料光催化分解水制氧的活性。结果表明,Bi2MoO6/WO3复合样品的光催化活性明显高于纯WO3和Bi2MoO6样品。在模拟太阳光照射下,15%Bi2MoO6/WO3复合催化剂的光催化产氧效率是纯WO3的产氧效率的2.3倍,以Fe(NO3)3·9H2O为牺牲剂时,复合催化剂的产氧效率可达到107 μmol/(g·h),且具有良好的循环稳定性。分析发现Bi2MoO6/WO3复合样品中Bi2MoO6颗粒与WO3纳米棒的异质结结构提高了光生载流子传输和转移效率,减少了光生电子-空穴对的复合几率,因此有助于增强光催化活性。Abstract: Bi2MoO6/WO3 composite photocatalysts with various Bi2MoO6 amounts were successfully synthesized by hydrothermal method. The microstructure, morphologies, optical absorption properties and spectral response of Bi2MoO6/WO3 composites were measured and characterized by XRD, SEM, UV-Vis, EIS and PL. Moreover, the photocatalytic activities of the samples were further investigated. The results show that the photocatalytic activity of the 15%Bi2MoO6/WO3 composite is obviously higher than that of the pure WO3 and Bi2MoO6: the photocatalytic oxygen production efficiency of the Bi2MoO6/WO3 is 2.3 times that of the pure WO3. The oxygen production efficiency of composite catalyst obtained 107 μmol/(g·h) after introducing Fe(NO3)3·9H2O assacrificial agent and exhibited good cycling stability. It is inferred that the Bi2MoO6 nanoparticle-WO3 nanorod heterojunction structure improves the transport and transfer efficiency of photo-generated carriers, reduces the recombination probability of electron-hole pairs, which is helpful to enhance the photocatalytic activity.
-
Key words:
- Bi2MoO6 /
- WO3 /
- nanorods /
- composite photocatalyst /
- photocatalytic oxygen production
-
图 4 模拟太阳光照射下WO3、Bi2MoO6及不同复合比例Bi2MoO6/WO3样品的光催化产氧性能 (a) 和牺牲剂对15%Bi2MoO6/WO3样品产氧性能的影响 (b)
Figure 4. Performance of photocatalytic oxygen production for WO3, Bi2MoO6 and Bi2MoO6/WO3 composites with different contents of Bi2MoO6 (a) and effect of sacrificial agents on the oxygen production performance of 15%Bi2MoO6/WO3 (b)
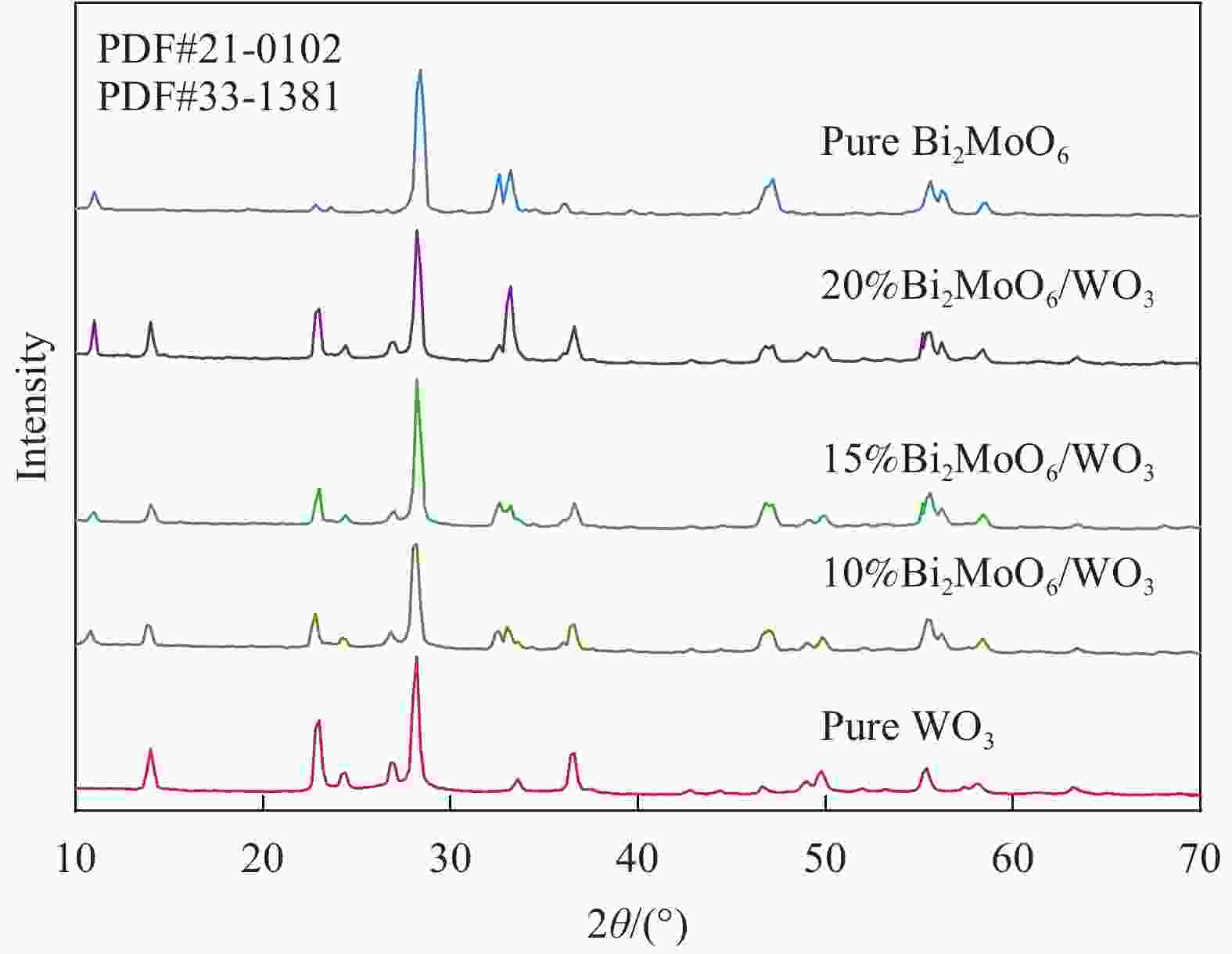
表 1 Bi2MoO6/WO3的命名
Table 1. Naming of Bi2MoO6/WO3
Sample Mass ratio of Bi2MoO6:WO3/% 5%Bi2MoO6/WO3 5 10%Bi2MoO6/WO3 10 15%Bi2MoO6/WO3 15 20%Bi2MoO6/WO3 20 表 2 WO3和Bi2MoO6的能带位置计算结果
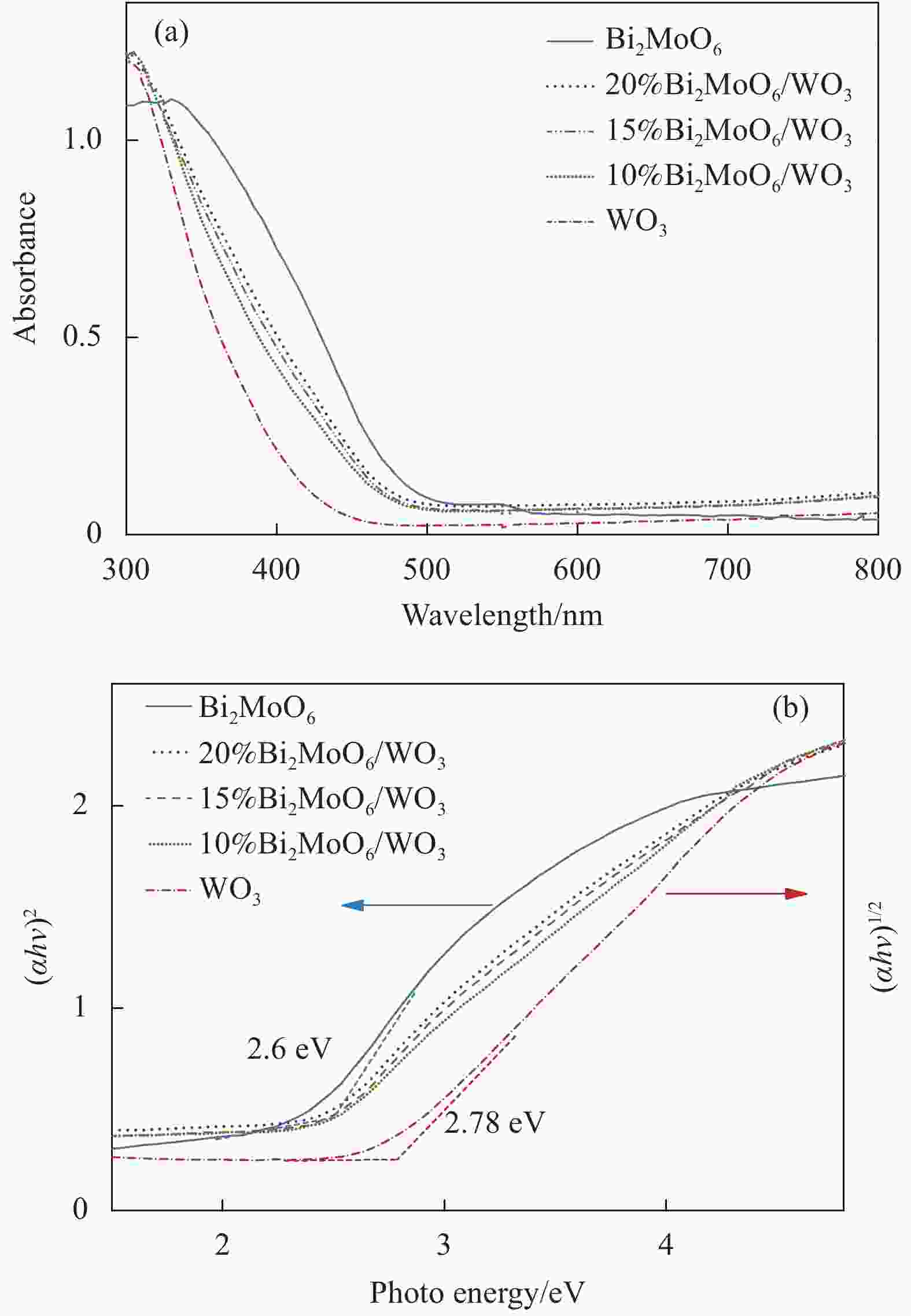
Table 2. Calculated band position values of Bi2MoO6 and WO3
Sample X/eV Ee/eV ECB /eV EVB/eV Eg/eV WO3 6.57 4.5 +0.88 +3.46 2.78 Bi2MoO6 5.55 4.5 −0.25 +2.35 2.60 Notes: X—Electronegativity of the semiconductor; Ee—Energy of free electrons on the hydrogen scale; ECB—Conduct band edge potential; EVB—Valence band-edge potential; Eg—Band gap energy. -
[1] GHOLAMI P, KHATAEE A, BHATNAGAR A, et al. Synthesis of N-doped magnetic WO3-x@mesoporous carbon using a diatom template and plasma modification: Visible-light-driven photocatalytic activities[J]. ACS Applied Materials Interfaces,2021,13(11):13072-13086. doi: 10.1021/acsami.0c21076 [2] 刘成宝, 唐飞, 朱晨, 等. WO3-Ag/石墨相 C3N4 Z型复合光催化剂的合成及其光催化性能[J]. 复合材料学报, 2021, 38(1):209-220.LIU C B, TANG F, ZHU C, et al. Preparation and photocatalytic properties of WO3-Ag/graphitic C3N4 Z-scheme composite photocatalyst[J]. Journal of Composite Materials,2021,38(1):209-220(in Chinese). [3] WANG F, VALENTIN C, PACCHIONI G. Doping of WO3 for photocatalytic water splitting: Hints from density functional theory[J]. Journal of Physical Chemistry C,2012,116(16):8901-8909. doi: 10.1021/jp300867j [4] LI S, WANG J, XIA Y, et al. Boosted electron-transfer by coupling Ag and Z-scheme heterostructures in CdSe-Ag-WO3-Ag for excellent photocatalytic H2 evolution with simultaneous degradation[J]. Chemical Engineering Journal,2021,417:129298. doi: 10.1016/j.cej.2021.129298 [5] ZHAO Y, ZHANG Y, YE Z, et al. CdS-dots decorated WO3 nanorods photocatalyst with highly ordered interfacial structure for enhanced photocatalytic activities via Z-scheme mechanism[J]. International Journal of Hydrogen Energy,2021,46:9040-9051. doi: 10.1016/j.ijhydene.2021.01.023 [6] ZHOU Q, SONG Y, LI N, et al. Direct dual Z-scheme Bi2WO6/GQDs/WO3 inverse opals for enhanced photocatalytic activities under visible light[J]. ACS Sustainable Chemistry & Engineering,2020,21:7921-7927. [7] WANG X, SUN M, MURUGANAN T, et al. Electrochemically self-doped WO3/TiO2 nanotubes for photocatalytic degradation of volatile organic compounds[J]. Applied Catalysis B: Environmental,2020,260:118205. doi: 10.1016/j.apcatb.2019.118205 [8] LING Y, DAI Y. Direct Z-scheme hierarchical WO3/BiOBr with enhanced photocatalytic degradation performance under visible light[J]. Applied Surface Science,2020,509:145201. doi: 10.1016/j.apsusc.2019.145201 [9] ZHANG B, LI J, GAO Y, et al. To boost photocatalytic activity in selective oxidation of alcohols on ultrathin Bi2MoO6 nanoplates with Pt nanoparticles as cocatalyst[J]. Journal of Catalysis,2017,345:96-103. doi: 10.1016/j.jcat.2016.11.023 [10] YANG X, XIANG Y, QU Y, et al. Novel in situ fabrication of conjugated microporous poly(benzothiadiazole)-Bi2MoO6 Z-scheme heterojunction with enhanced visible light photocatalytic activity[J]. Journal of Catalysis,2017,345:319-328. doi: 10.1016/j.jcat.2016.11.014 [11] MA T, WU J, MI Y, et al. Novel Z-Scheme g-C3N4/C@ Bi2MoO6 composite with enhanced visible-light photocatalytic activity for β-naphthol degradation[J]. Separation and Purification Technology,2017,183:54-65. doi: 10.1016/j.seppur.2017.04.005 [12] GOVINDARAJ T, MAHENDRAN C, MARNADU R, et al. The remarkably enhanced visible-light-photocatalytic activity of hydrothermally synthesized WO3 nanorods: An effect of Gd doping[J]. Ceramics International,2021,47:4267-4278. doi: 10.1016/j.ceramint.2020.10.004 [13] JI Z, WU J, JIA T, et al. In-situ growth of TiO2 phase junction nanorods with Ti3+ and oxygen vacancies to enhance photocatalytic activity[J]. Materials Research Bulletin,2021,140:111291. doi: 10.1016/j.materresbull.2021.111291 [14] HUANG S, LONG Y, RUAN S, et al. Enhanced photocatalytic CO2 reduction in defect-engineered Z-scheme WO3-x/g-C3N4 heterostructures[J]. ACS Omega,2019,13:15593-15599. [15] MA H, HE Y, LI X, et al. In situ loading of MoO3 clusters on ultrathin Bi2MoO6 nanosheets for synergistically enhanced photocatalytic NO abatement[J]. Applied Catalysis B: Environmental,2021,292:120159. doi: 10.1016/j.apcatb.2021.120159 [16] AMANO F, ISHINAGA E, YAMAKAT A. Effect of particle size on the photocatalytic activity of WO3 particles for water oxidation[J]. The Journal of Physical Chemistry C,2013,117(44):22584-22590. doi: 10.1021/jp408446u [17] LI S, WANG C, CHEN X, et al. Photocatalytic degradation of antibiotics using a novel Ag/Ag2S/ Bi2MoO6 plasmonic p-n heterojunction photocatalyst: mineralization activity, degradation pathways and boosted charge separation mechanism[J]. Chemical Engineering Journal,2021,415:128991. doi: 10.1016/j.cej.2021.128991 [18] KOHANTORABI M, MOUSSAVI G, MOHAMMADI S, et al. Photocatalytic activation of peroxymonosulfate (PMS) by novel mesoporous Ag/ZnO@NiFe2O4 nanorods, inducing radical-mediated acetaminophen degradation under UVA irradiation[J]. Chemosphere,2021,277:130271. doi: 10.1016/j.chemosphere.2021.130271 [19] QIN Y, LU J, MENG F, et al. Rationally constructing of a novel 2D/2D WO3/Pt/g-C3N4 Schottky-Ohmic junction towards efficient visible-light-driven photocatalytic hydrogen evolution and mechanism insight[J]. Journal of Colloid and Interface Science,2021,586:576-587. doi: 10.1016/j.jcis.2020.10.123 [20] XUE C, ZHANG P, SHAO G, et al. Effective promotion of spacial charge separation in direct Z-scheme WO3/CdS/WS2 tandem heterojunction with enhanced visible-light-driven photocatalytic H2 evolution[J]. Chemical Engineering Journal,2020,398:125602. doi: 10.1016/j.cej.2020.125602 [21] JIA J, JIANG C, ZHANG X, et al. Urea-modified carbon quantum dots as electron mediator decorated g-C3N4/WO3 with enhanced visible-light photocatalytic activity and mechanism insight[J]. Applied Surface Science,2019,45:143524. [22] TIAN J, HAO P, WEI N, et al. 3D Bi2MoO6 nanosheet/TiO2 nanobelt heterostructure: Enhanced photocatalytic activities and photoelectochemistry performance[J]. ACS Catalysis,2015,5:4530-4536. doi: 10.1021/acscatal.5b00560 -






 下载:
下载: