Preparation of ZIF-67/waste cotton cellulose composite aerogels and the removal performance on dyes
-
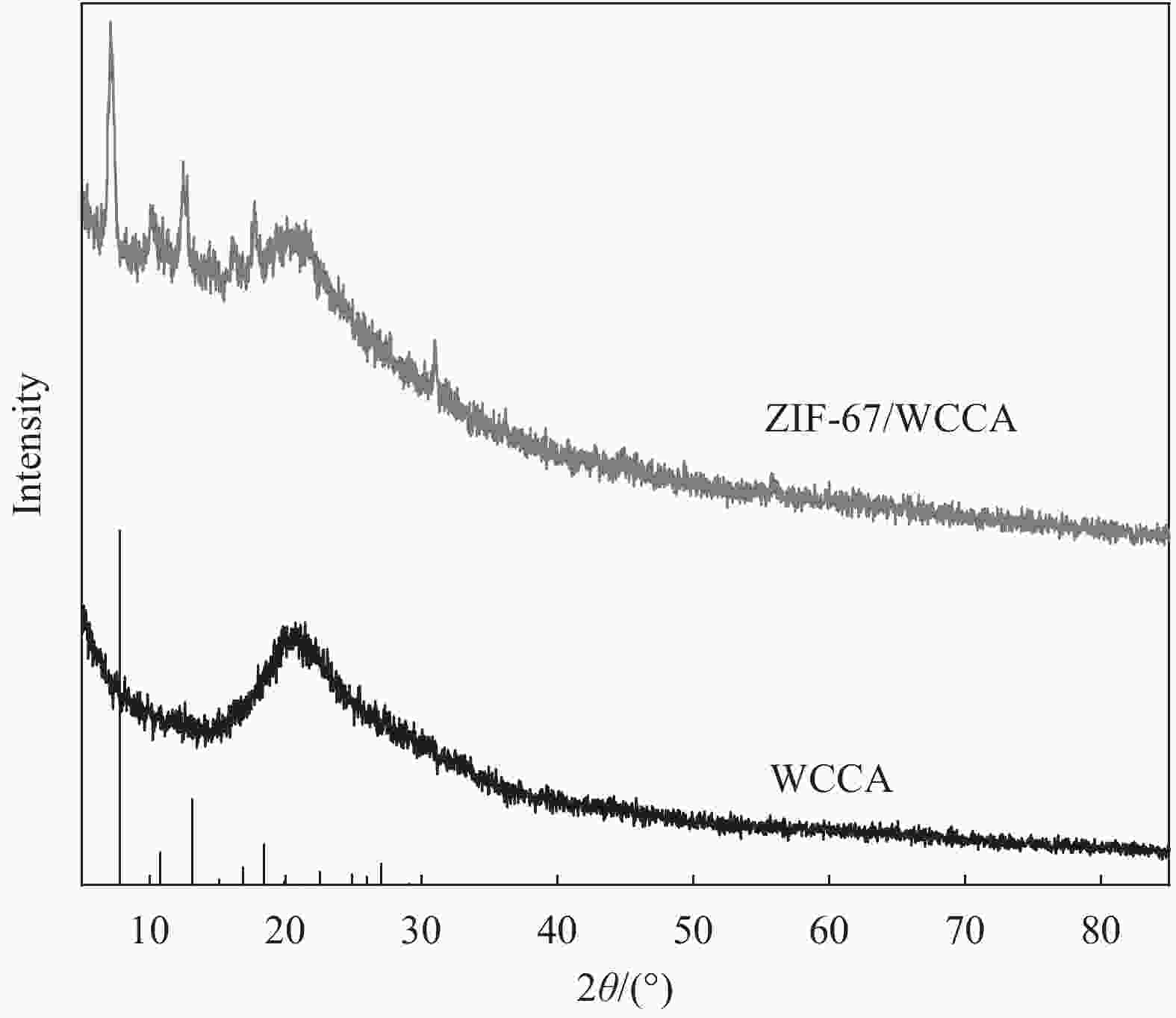
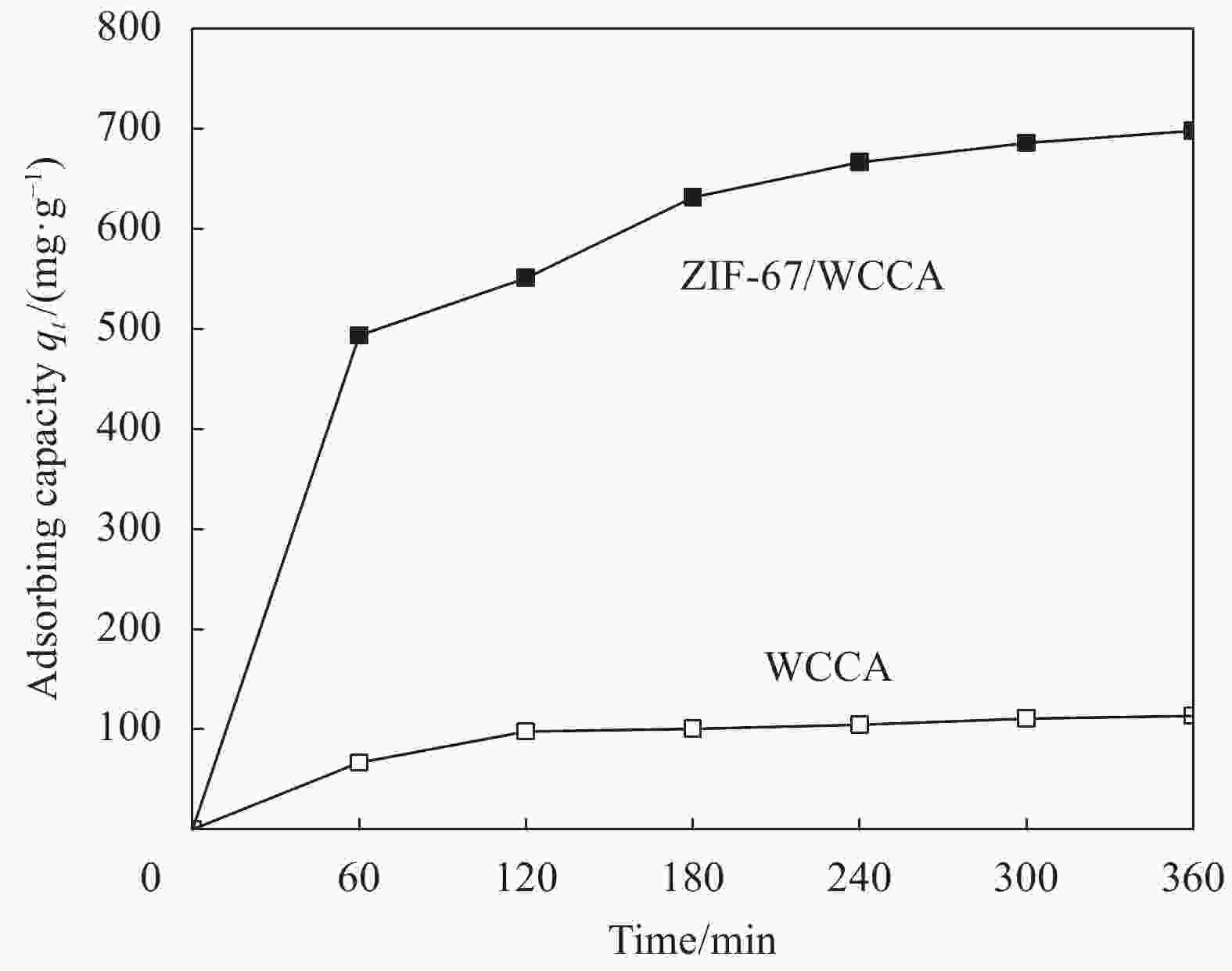
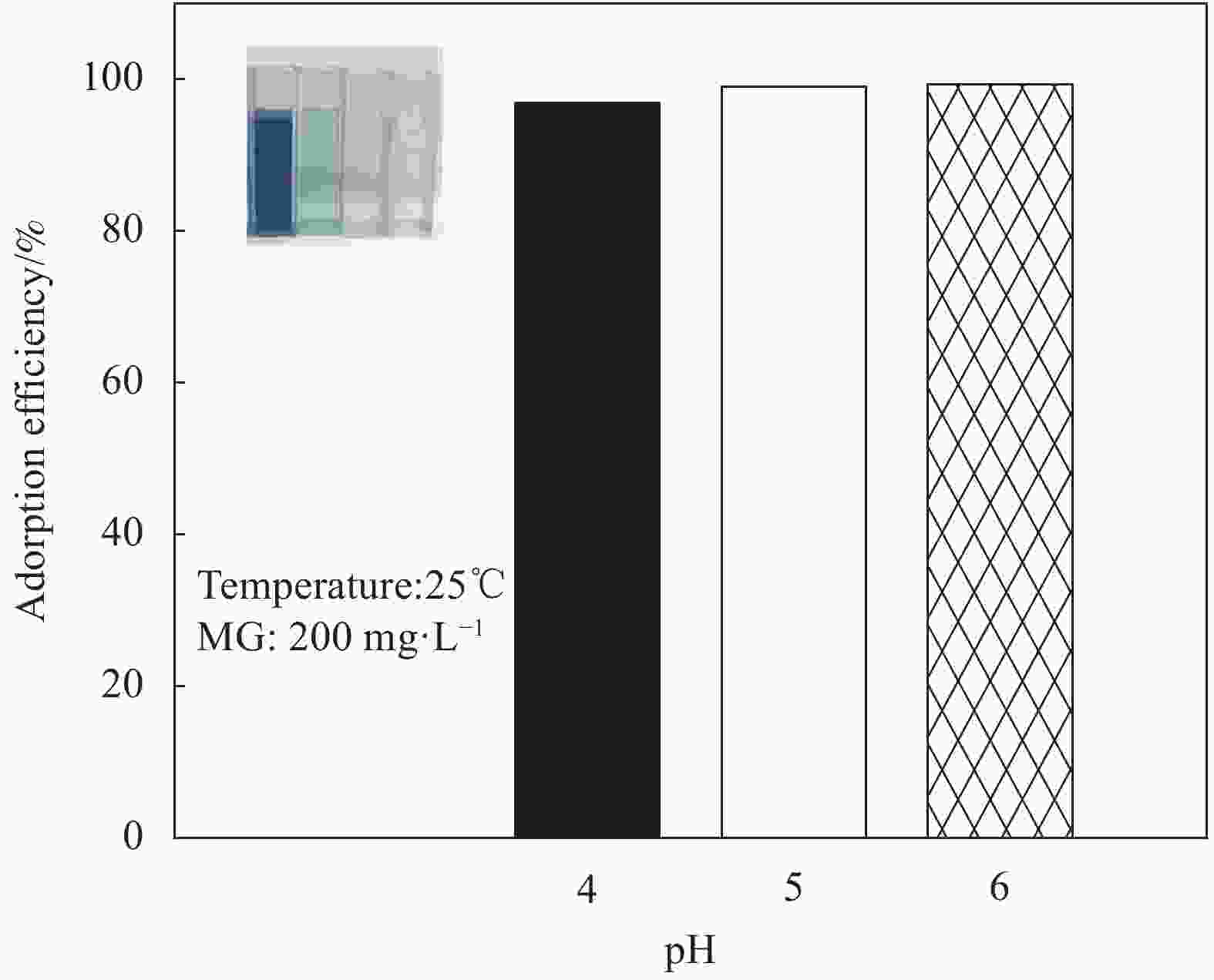
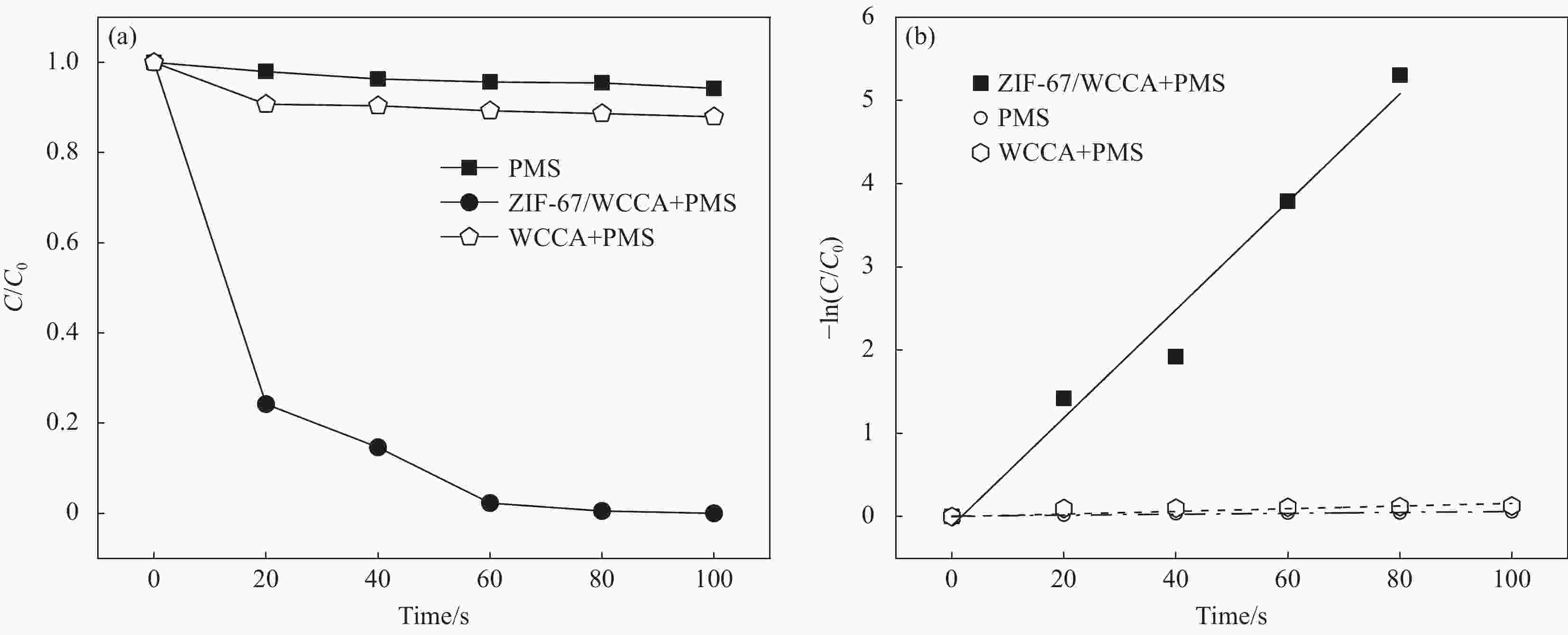
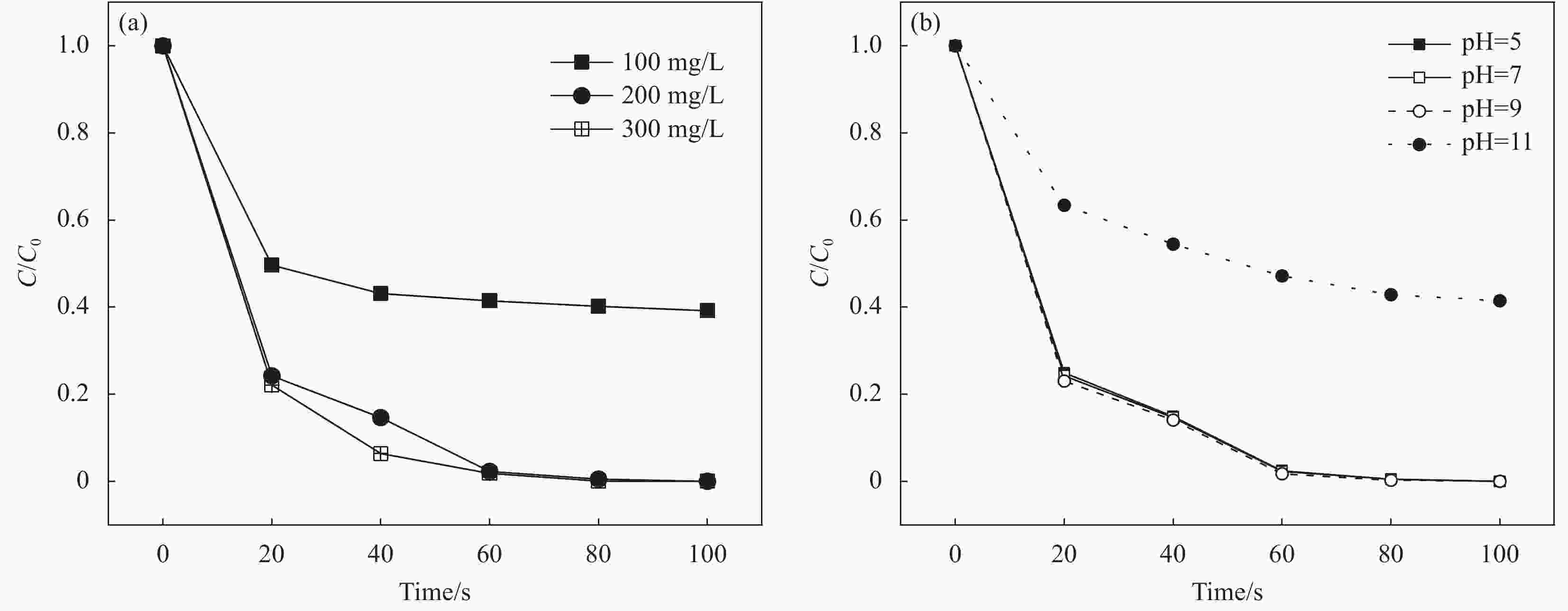
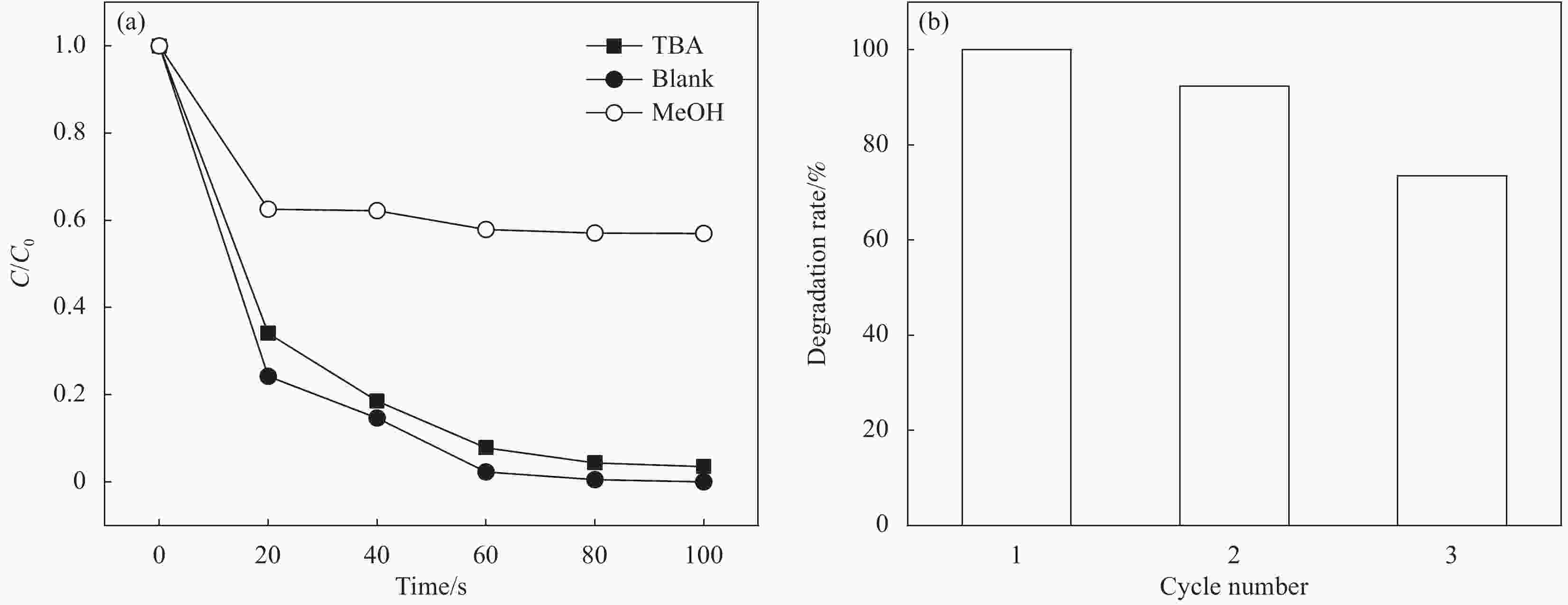
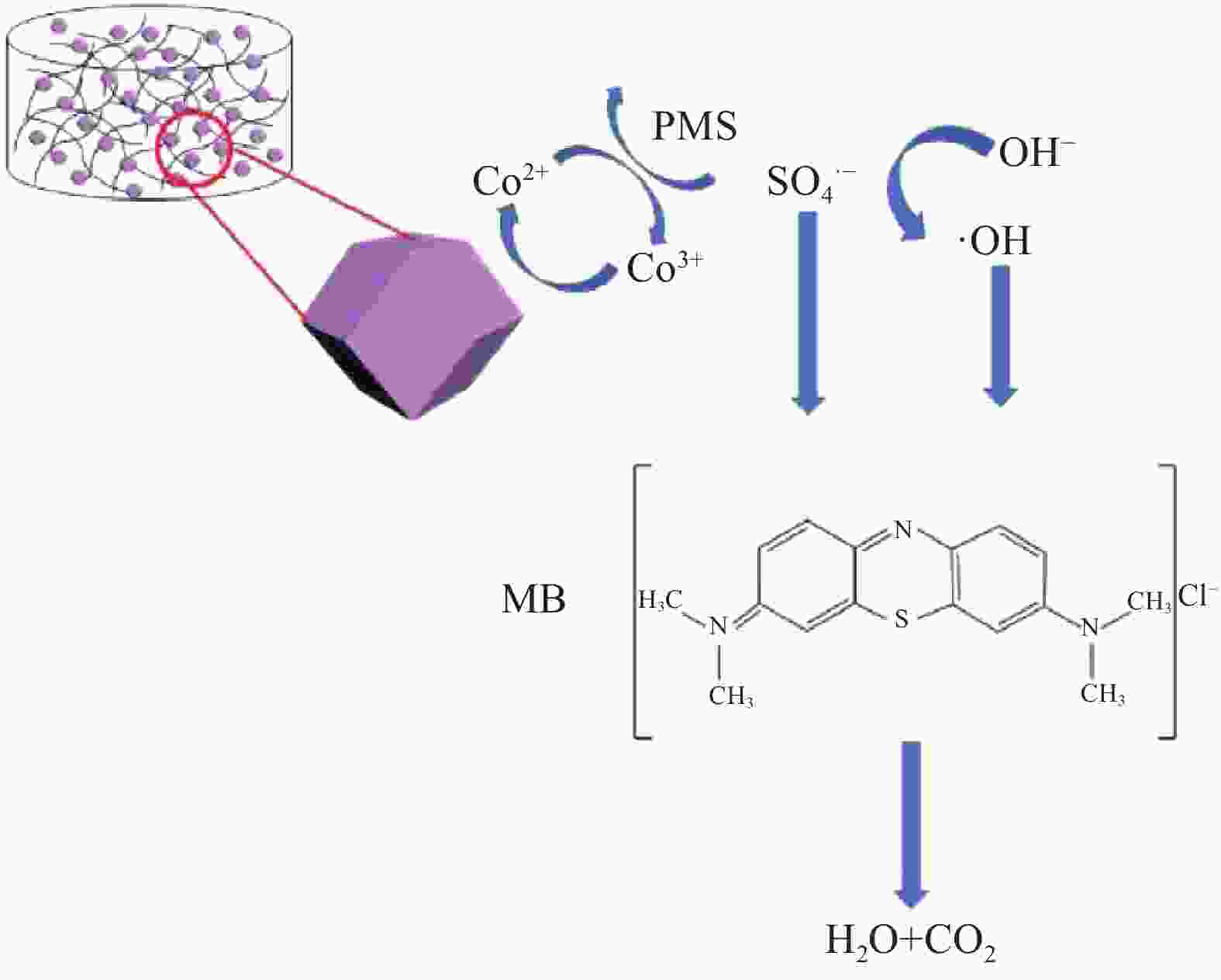
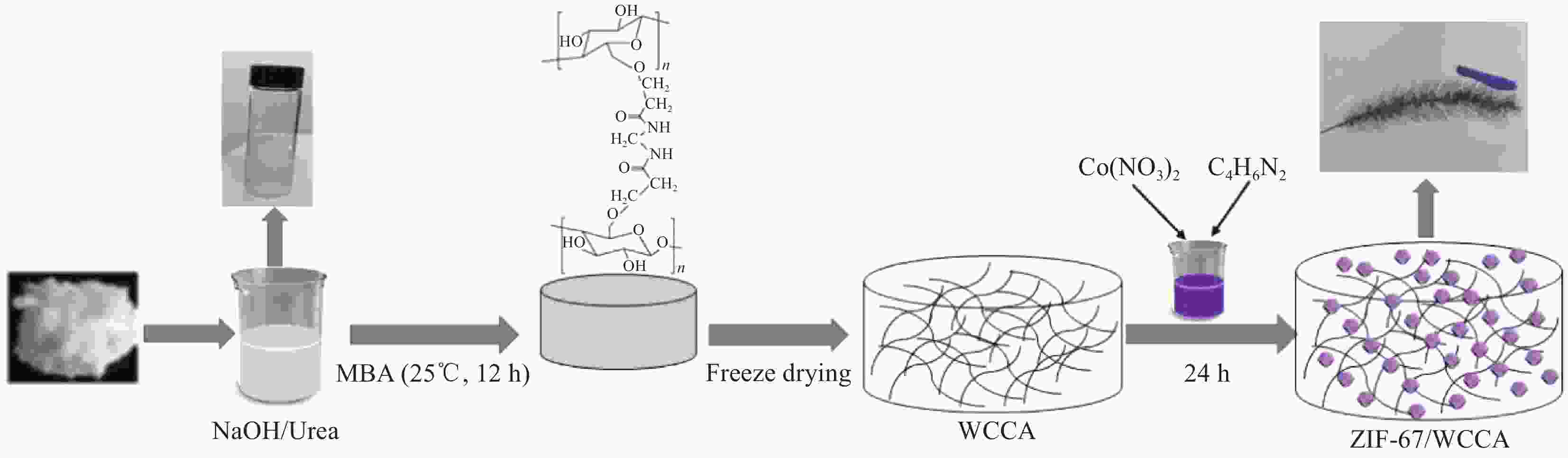
摘要: 为高效去除污水中的有机染料,以废旧棉织物为纤维素原材料,碱/尿素为溶解体系,N, N'-亚甲基双丙烯酰胺(MBA)作为交联剂,通过冷冻干燥技术制备了废棉纤维素气凝胶(WCCA),在其表面原位生成钴基沸石咪唑酯有机框架(ZIF-67),制备了ZIF-67/WCCA,用于吸附孔雀石绿(MG)和催化过硫酸氢钾(PMS)降解亚甲基蓝(MB)。借助SEM和XRD对ZIF-67/WCCA的结构和成分进行表征,并探讨了影响其染料吸附和催化降解性能的条件。结果表明,具有菱形十二面体结构的ZIF-67负载于WCCA网络框架上;在室温下,对MG吸附量达到1474.01 mg·g−1;在100 s左右催化PMS对MB的降解率可达100%。ZIF-67/WCCA可应用于污水中染料吸附和催化降解。Abstract: In order to efficiently remove organic dyes from waste water, ZIF-67/waste cotton cellulose composite aerogels (ZIF-67/WCCA) were prepared by freeze drying technology using waste cotton fabrics as cellulose raw materials, alkali/urea as dissolution system, N, N’-Methylenediacrylamide (MBA) as cross-linking agent, and cobalt-based zeolite imidazolate metal-organic framework (ZIF-67) nanoparticles were grown in situ on the surface of WCCA. ZIF-67/WCCA were prepared and used to adsorb malachite green (MG) and degrade methylene blue (MB) with peroxymonosulfate (PMS) activation. The structure and composition of the ZIF-67/WCCA were characterized by scanning electron microscopy and X-ray diffraction. The conditions affecting the adsorption and degradation properties of the dyes were also discussed. The results show that ZIF-67 nanoparticles of dodecahedron are loaded on the network framework of cellulose aerogels from waste cotton. The adsorption capacity of MG can reach 1474.01 mg·g−1 at room temperature, and the degradation rate of MB at 100 s can reach 100% by using ZIF-67/WCCA. ZIF-67/WCCA can be applied in fields of dye adsorption and catalytic degradation in sewage.
-
Key words:
- waste cotton /
- cellulose aerogel /
- ZIF-67 /
- dye /
- adsorption /
- catalytic degradation
-
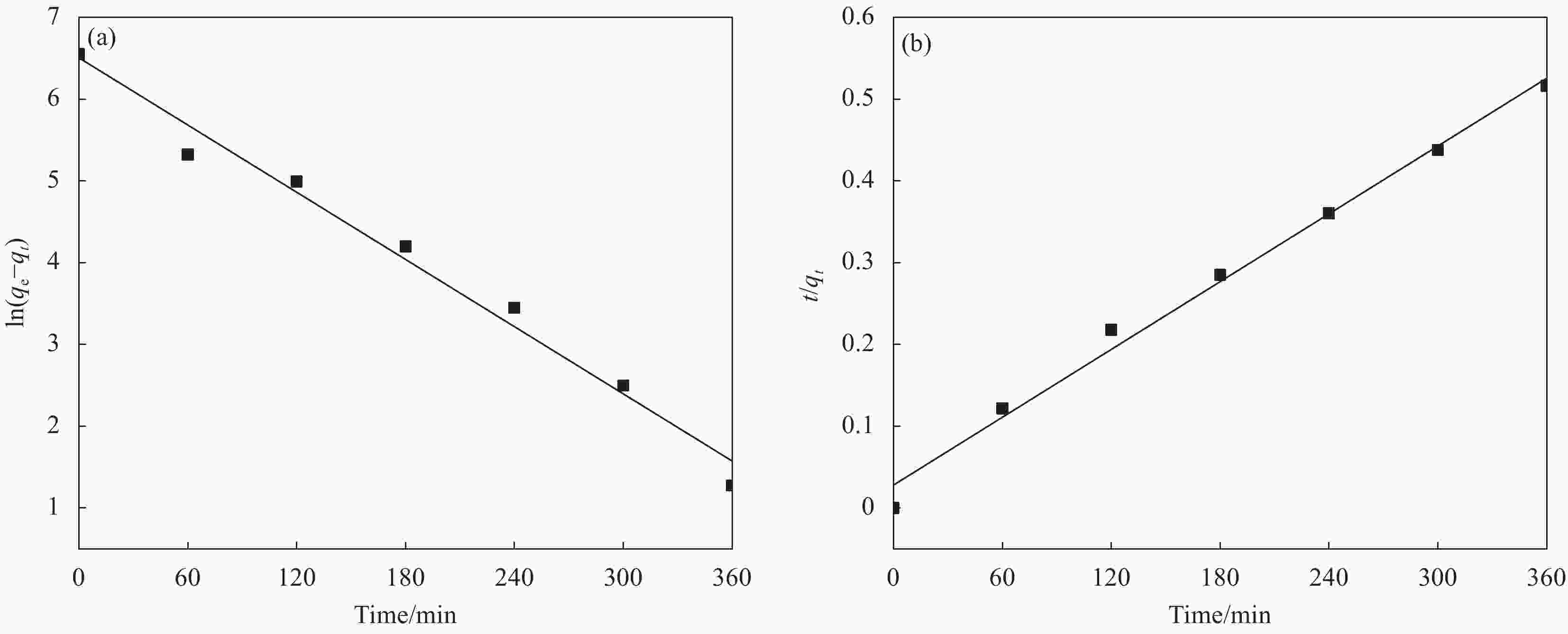
图 5 ZIF-67/WCCA的动力学拟合曲线:(a) 准一级动力学模型;(b) 准二级动力学模型
Figure 5. Fitted adsorption kinetic plots of ZIF-67/WCCA: (a) Pseudo-first-order kinetic model; (b) Pseudo-second-order kinetic model
qe—Adsorption capacity at adsorption equilibrium; qt—Adsorption capacity of aerogels to dyes after a certain adsorption time t
表 1 ZIF-67/WCCA吸附亚甲基蓝(MG)的动力学参数
Table 1. Adsorption kinetic parameters of methylene blue (MG) on ZIF-67/WCCA
Pseudo-first-order Pseudo-second-order k1/min−1 R12 k2/(min·g·mg−1) R22 0.0229 0.983 0.0000678 0.991 Notes: k1—Pseudo-first-order adsorption rate constants; R12—
Correlation coefficient of pseudo-first-order kinetics model; k2—Pseudo-second-order adsorption rate constants; R22—
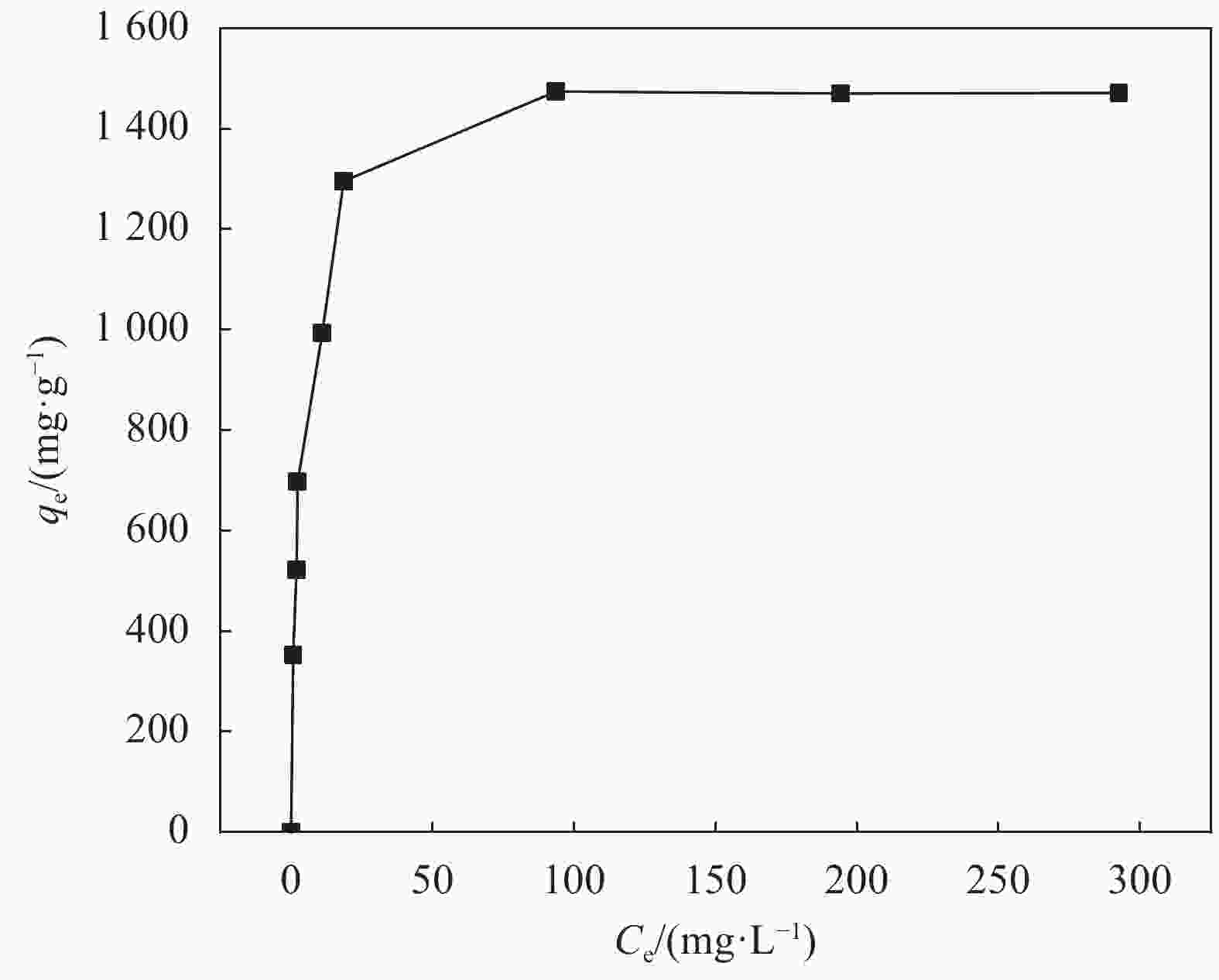
Correlation coefficient of pseudo-second-order kinetics model.表 2 ZIF-67/WCCA吸附MG的等温线模型参数
Table 2. Parameters of adsorption isotherms model for MG on ZIF-67/WCCA
Langumir Freundlich Ce/qe=1/qmKL+Ce/qm lnqe=(1/n)lnCe+lnKF qe exp/(mg·g−1) qm/(mg·g−1) KL RL2 KF RF2 1474.01 1498.35 3.209 0.999 492.8 0.876 Notes: qe exp—Equilibrium adsorption capacities of the dyes; qm exp—Maximum adsorption capacities of the dyes; KL—Langmuir equilibrium adsorption constants; KF—Freundlich equilibrium adsorption constants; RL2—Correlation coefficient of Langumir model; RF2—Correlation coefficient of Freundlich model. -
[1] MATE C J, MISHRA S. Synthesis of borax cross-linked Jhingan gum hydrogel for remediation of Remazol Brilliant Blue R (RBBR) dye from water: Adsorption isotherm, kinetic, thermodynamic and biodegradation studies[J]. International Journal of Biological Macromolecules,2020,151:677-690. doi: 10.1016/j.ijbiomac.2020.02.192 [2] DAI C Y, ZHANG M H, GUO X W, et al. Mesoporous composite Ni-C-N/SA for selective adsorption of methylene blue from water[J]. Chemical Engineering Journal,2021,407:127181. doi: 10.1016/j.cej.2020.127181 [3] 李婷婷, 李瑞雪, 马政, 等. 纤维素-海藻酸钠-海泡石多孔微球的制备及其对亚甲基蓝吸附性能[J]. 复合材料学报, 2021, 38(12): 4273-4281.LI Tingting, LI Ruixue, MA Zheng, et al. Preparation of cellulose-sodium alginate-sepiolite porous bead and its application in adsorption of methylene blue[J]. Acta Materiae Compositae Sinica, 2021, 38(12): 4273-4281(in Chinese). [4] YU C Q, WEN M, LI S H, et al. Elbaite catalyze peroxymonosulfate for advanced oxidation of organic pollutants: Hydroxyl groups induced generation of reactive oxygen species[J]. Journal of Hazardous Materials,2020,398:122932. doi: 10.1016/j.jhazmat.2020.122932 [5] PANG Y, KONG L, CHEN D, et al. Facilely synthesized cobalt doped hydroxyapatite as hydroxyl promoted peroxymonosulfate activator for degradation of Rhodamine B[J]. Journal of Hazardous Materials,2020,384:121447. doi: 10.1016/j.jhazmat.2019.121447 [6] XIAO Z Y, LI Y, FAN L, et al. Degradation of organic dyes by peroxymonosulfate activated with water-stable iron-based metal organic frameworks[J]. Journal of Colloid and Interface Science,2021,589:298-307. doi: 10.1016/j.jcis.2020.12.123 [7] GHORAI K, PANDA A, BHATTACHARJEE M, et al. Facile synthesis of CuCr2O4/CeO2 nanocomposite: A new Fenton like catalyst with domestic LED light assisted improved photocatalytic activity for the degradation of RhB, MB and MO dyes[J]. Applied Surface Science,2021,536:147604. doi: 10.1016/j.apsusc.2020.147604 [8] DU Z F, CUI C, ZHANG S H, et al. Visible-light-driven photocatalytic degradation of Rhodamine B using Bi2WO6/GO deposited on polyethylene terephthalate fabric[J]. Journal of Leather Science and Engineering,2020,2(1):16. doi: 10.1186/s42825-020-00029-w [9] LI X, WAN J Q, MAY W, et al. Study on cobalt-phosphate (Co-Pi) modified BiVO4/Cu2O photoanode to significantly inhibit photochemical corrosion and improve the photoelectrochemical performance[J]. Chemical Engineering Journal,2021,404:127054. doi: 10.1016/j.cej.2020.127054 [10] HE X H, CHEN T T, JIANG T Y, et al. Preparation and adsorption properties of magnetic hydrophobic cellulose aerogels based on refined fibers[J]. Carbohydrate Polymers,2021,260:117790. doi: 10.1016/j.carbpol.2021.117790 [11] SONG Y S, PENG Y S, LONG N V, et al. Multifunctional self-assembly 3D Ag/g-C3N4/RGO aerogel as highly efficient adsorbent and photocatalyst for R6G removal from wastewater[J]. Applied Surface Science,2021,542:148584. doi: 10.1016/j.apsusc.2020.148584 [12] MAHMOODI N M, MOKHTARI-SHOURIJEH Z, LANGARI S, et al. Seifpanahi-Shabani, silica aerogel/polyacrylonitrile/polyvinylidene fluoride nanofiber and its ability for treatment of colored wastewater[J]. Journal of Molecular Structure,2021,1227:129418. doi: 10.1016/j.molstruc.2020.129418 [13] YANG L, ZHAN Y F, GONG Y J, et al. Development of eco-friendly CO2-responsive cellulose nanofibril aerogels as “green” adsorbents for anionic dyes removal[J]. Journal of Hazardous Materials,2021,405:124194. doi: 10.1016/j.jhazmat.2020.124194 [14] QIN Q, GUO R H, REN E H, et al. Waste cotton fabric/zinc borate composite aerogel with excellent flame retardancy[J]. ACS Sustainable Chemistry & Engineering,2020,8:10335-10344. [15] QIN Q, GUO R H, REN E H, et al. Waste cotton fiber/Bi2WO6 composite film for dye removal[J]. Cellulose,2019,26:3909-3922. doi: 10.1007/s10570-019-02345-9 [16] ÜTEBAY B, ÇELIK P, ÇAY A. Effects of cotton textile waste properties on recycled fibre quality[J]. Journal of Cleaner Production,2019,222:29-35. doi: 10.1016/j.jclepro.2019.03.033 [17] ERKARTAL M, ERKILIC U, TAM B, et al. From 2-methylimidazole to 1, 2, 3-triazole: A topological transformation of ZIF-8 and ZIF-67 by post-synthetic modification[J]. Chemical Communications (Camb),2017,53:2028-2031. doi: 10.1039/C6CC08746A [18] LI N, CHEN G Y, ZHAO J H, et al. Self-cleaning PDA/ZIF-67@PP membrane for dye wastewater remediation with peroxymonosulfate and visible light activation[J] Journal of Membrane Science, 2019, 591: 117341. [19] 刘兴静, 孙赟, 林亚玲等. 天然纤维表面化学处理性能研究[J]. 化工新型材料, 2012, 40(5):51-53. doi: 10.3969/j.issn.1006-3536.2012.05.018LIU Xingjing, SUN Yun, LIN Yaling, et al. Properties of natural fibersurface after chemical treatment[J]. New chemical materials,2012,40(5):51-53(in Chinese). doi: 10.3969/j.issn.1006-3536.2012.05.018 [20] SONG W Q, ZHU M, ZHU Y F, et al. Zeolitic imidazolate framework-67 functionalized cellulose hybrid aerogel: An environmentally friendly candidate for dye removal[J]. cellulose,2020,27:2161-2172. doi: 10.1007/s10570-019-02883-2 [21] GUO W W, WANG X, ZHANG P, et al. Nano-fibrillated cellulose-hydroxyapatite based composite foams with excellent fire resistance[J]. Carbohydrate Polymers,2018,195:71-78. doi: 10.1016/j.carbpol.2018.04.063 [22] LIN K Y, CHANG H A. Ultra-high adsorption capacity of zeolitic imidazole framework-67 (ZIF-67) for removal of malachite green from water[J] Chemosphere, 2015, 139: 624-631. [23] XIANG C, WANG C, GUO R H, et al. Synthesis of carboxymethyl cellulose-reduced graphene oxide aerogel for efficient removal of organic liquids and dyes[J]. Journal of Materials Science,2018,54:1872-1883. [24] WU Y H, REN W J, LI Y W, et al. Zeolitic Imidazolate Framework-67@Cellulose aerogel for rapid and efficient degradation of organic pollutants[J]. Journal of Solid State Chemistry,2020,291:121621. doi: 10.1016/j.jssc.2020.121621 [25] WANG S Z, LIU H Y, WANG J L. Nitrogen, sulfur and oxygen co-doped carbon-armored Co/Co9S8 rods (Co/Co9S8@N—S—O—C) as efficient activator of peroxymonosulfate for sulfamethoxazole degradation[J]. Journal of Hazardous Materials,2020,387:121669. doi: 10.1016/j.jhazmat.2019.121669 -





 下载:
下载: