Preparation of cellulose-sodium alginate-sepiolite porous bead and its application in adsorption of methylene blue
-
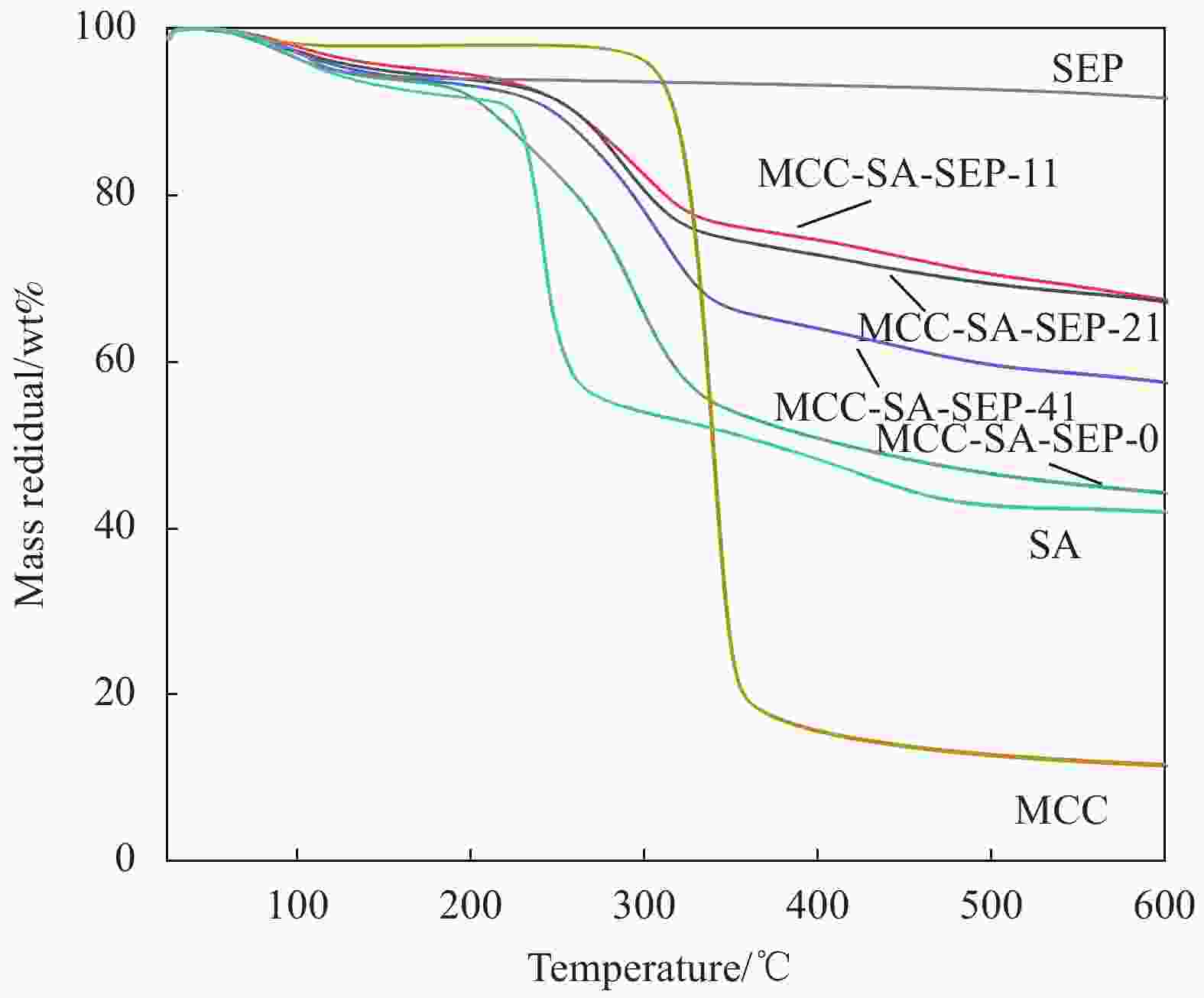
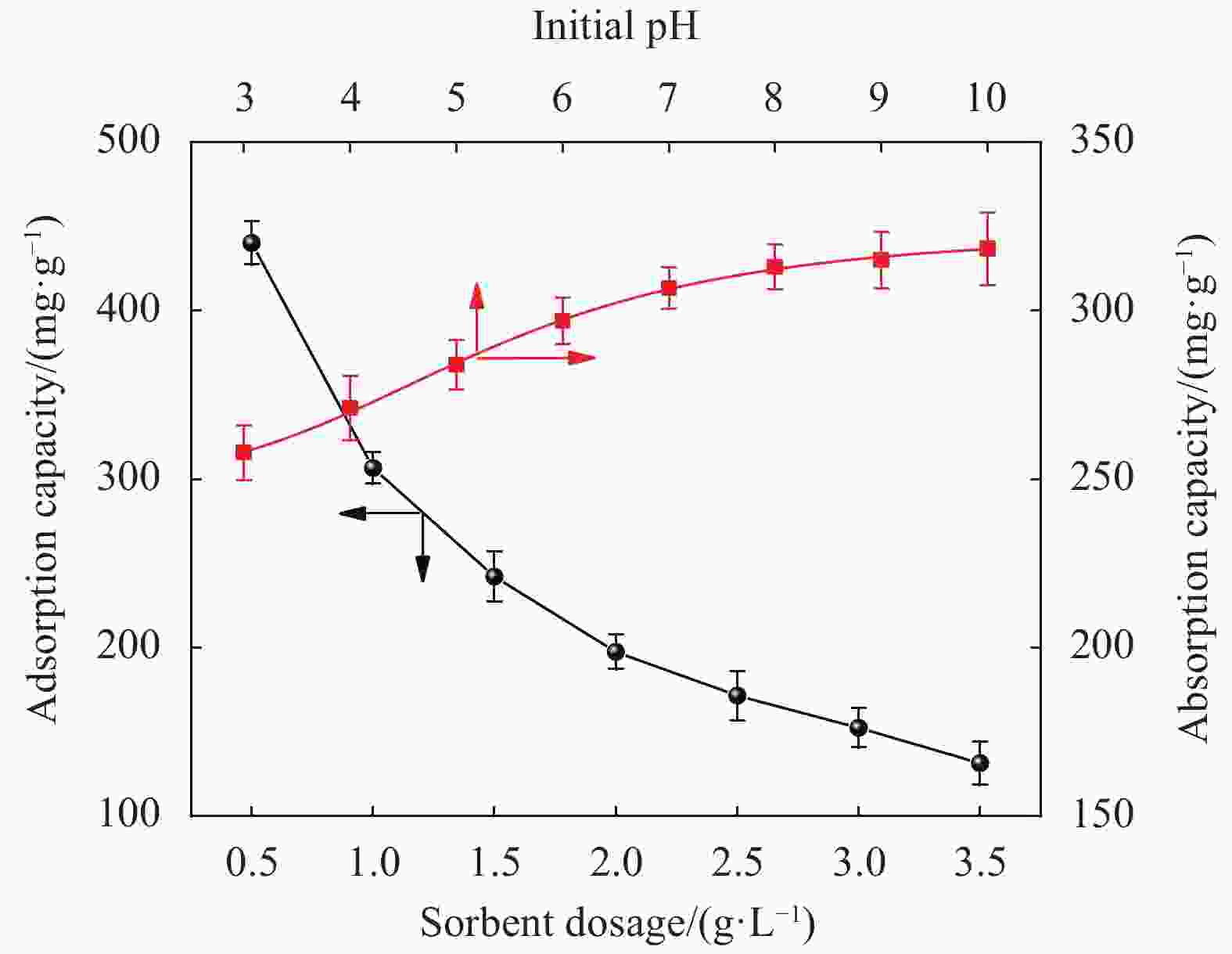
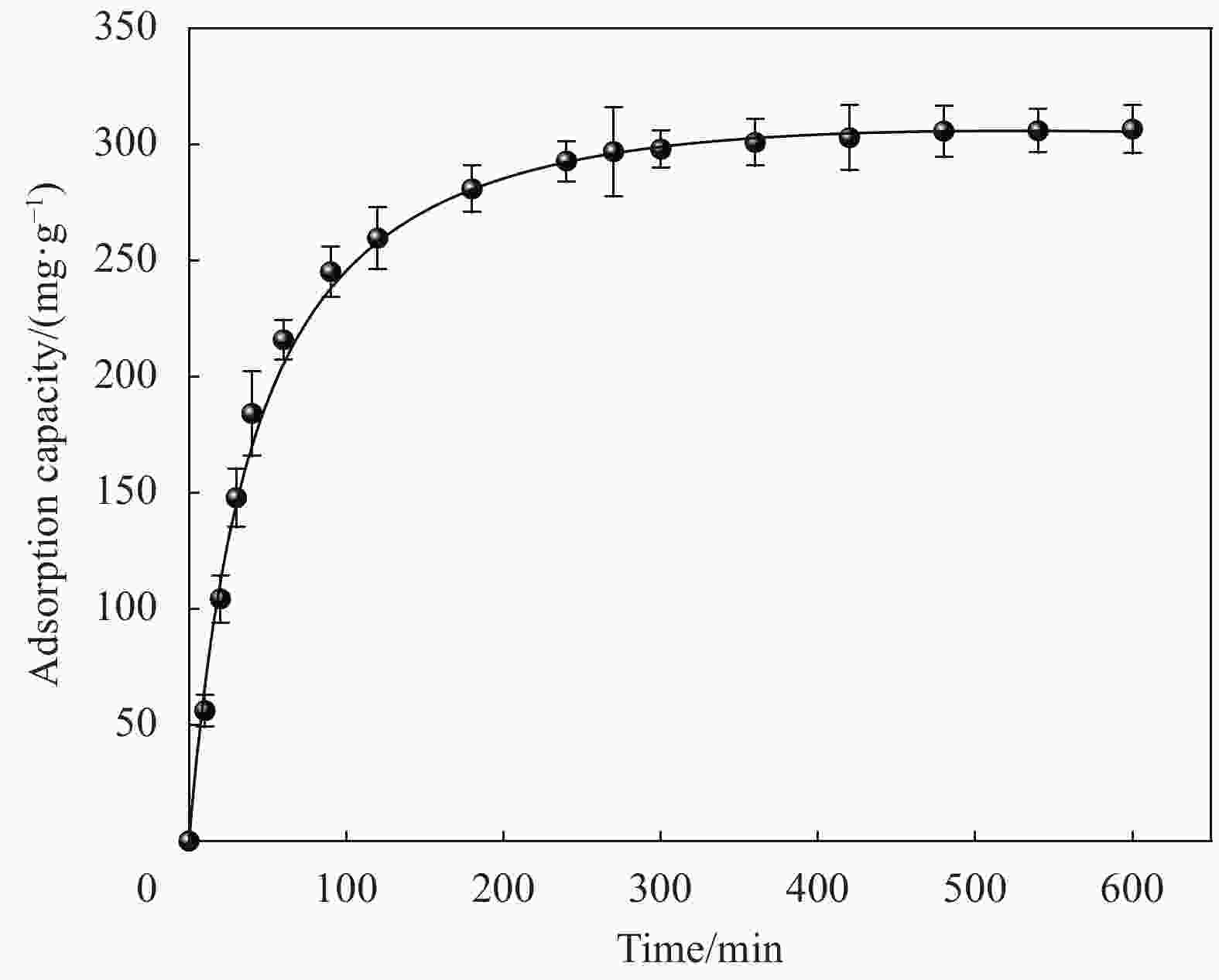
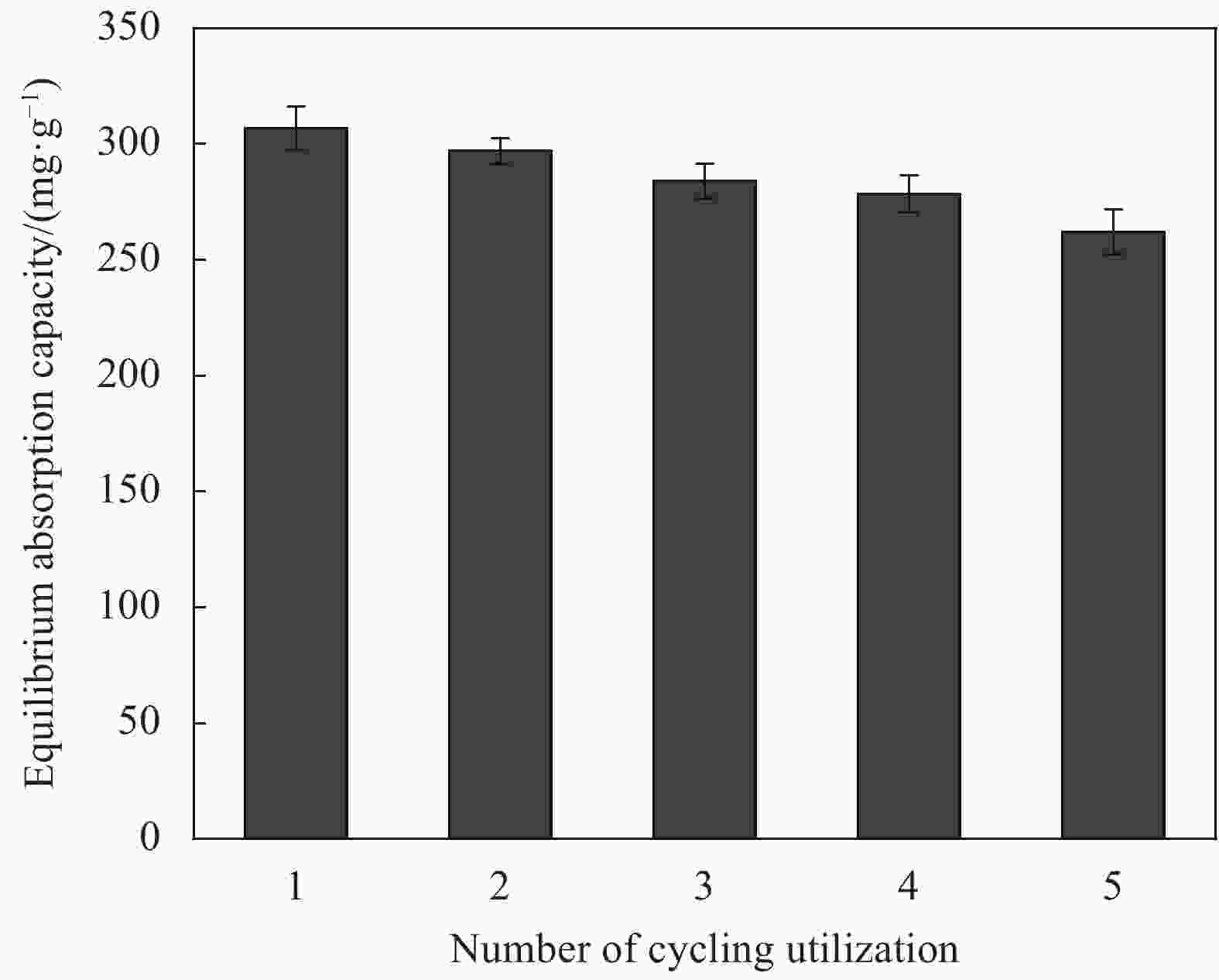
摘要: 以微晶纤维素(Microcrystalline cellulose,MCC)和海藻酸钠(Sodium alginate,SA)为网络框架,海泡石(Sepiolite,SEP)为功能单元,采用悬浮液滴法构建纤维素-海藻酸钠-海泡石(MCC-SA-SEP)双网络多孔复合微球。通过SEM和TG对复合微球结构和热稳定性能进行表征,并研究该微球对亚甲基蓝(Methylene blue,MB)水溶液的吸附性能。结果表明,MCC-SA-SEP复合微球呈现三维网络多孔结构,且随着SEP含量的增加热稳定性逐渐提高。吸附结果显示MCC-SA-SEP符合准二级动力学模型和Langmuir等温线,对MB的饱和吸附容量高达333.3 mg/g。经过五次再生循环后,对MB吸附能力仍能维持85.4%,表明该多孔复合微球可以作为一种高效可再生的有机-无机复合吸附剂用于染料废水处理。Abstract: Double network composite beads (MCC-SA-SEP) were synthesized by a floating droplet method, in which microcrystalline cellulose (MCC) and sodium alginate (SA) worked as the network frameworks, and sepiolite (SEP) was a functional component. The microstructure and thermal properties of the as-prepared MCC-SA-SEP beads were characterized by SEM and TG, respectively, and the adsorption performance for methylene blue (MB) aqueous solution was studied. The results present that the MCC-SA-SEP beads have three-dimensional porous structures, and the thermal stability increases gradually with the increasing of SEP contents. The adsorption process of MCC-SA-SEP follows the pseudo-second-order kinetic model and Langmuir isotherm, with the maximum adsorption capacity of 333.3 mg/g for MB. After five regeneration cycles, the adsorption capacity could still retain 85.4% of the initial adsorption amount, demonstrating a novel organic-inorganic hybrid adsorbent for dye waste water treatment.
-
Key words:
- microcrystalline cellulose /
- sodium alginate /
- sepiolite /
- beads /
- methylene blue /
- adsorption
-
表 1 不同SEP含量制备的微晶纤维素-海藻酸钠-海泡石(MCC-SA-SEP)复合微球
Table 1. Microcrystalline cellulose-sodium alginate-sepiolite (MCC-SA-SEP) composite beads prepared with various SEP contents
MCC-SA content/g SEP content/g Composite bead 2 0 MCC-SA-SEP-0 2 0.5 MCC-SA-SEP-41 2 1 MCC-SA-SEP-21 2 2 MCC-SA-SEP-11 表 2 MCC-SA-SEP-21多孔复合微球吸附MB的动力学模型拟合参数
Table 2. Parameters of kinetic adsorption models for MB onto MCC-SA-SEP-21 beads
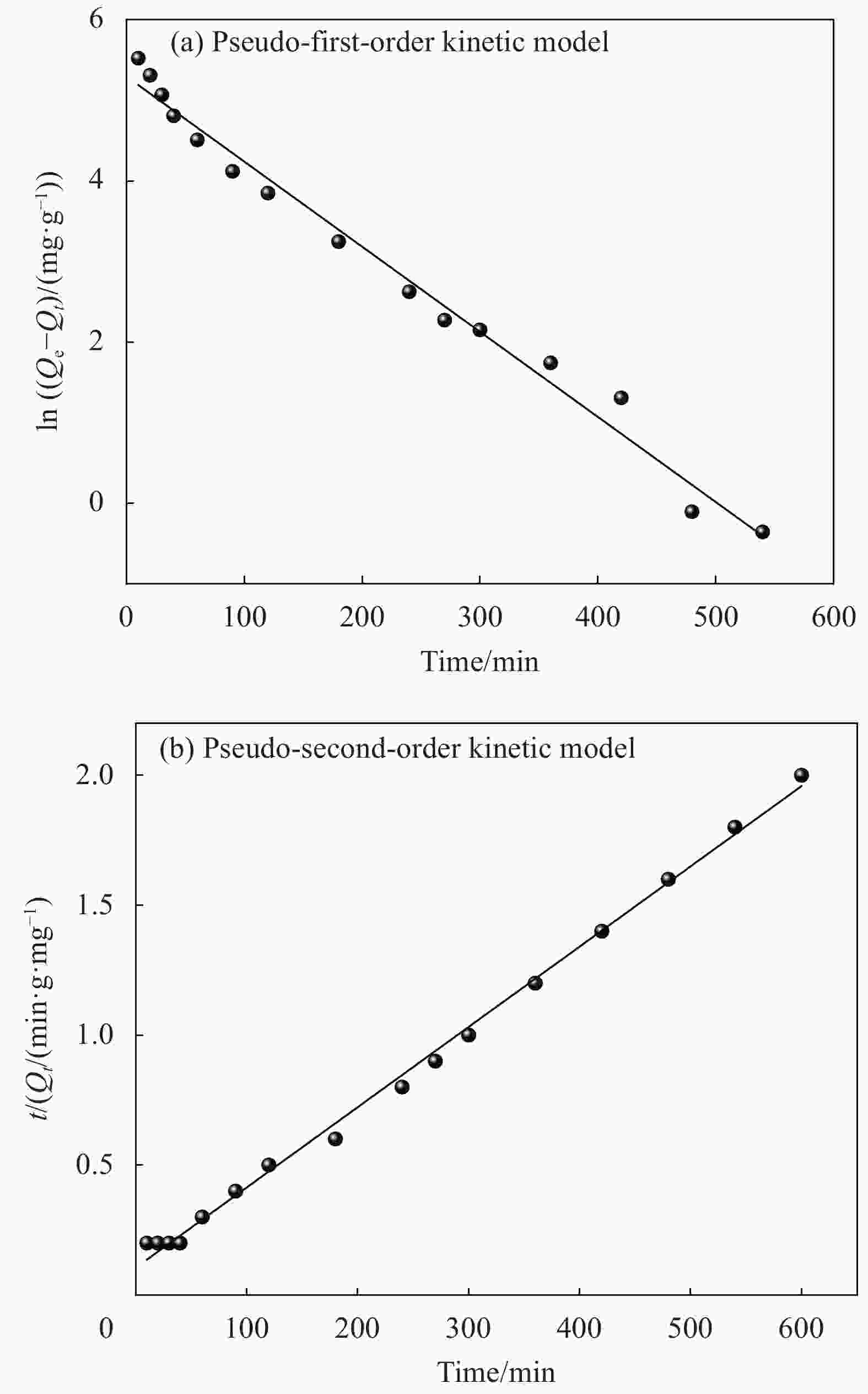
Adsorbate Qe(exp)/(mg·g−1) Pseudo-first-order model Pseudo-second-order model Q1e(cal)/(mg·g−1) k1/(min−1) R2 Q2e(cal)/(mg·g−1) k2/(g·mg−1·min−1) R2 MB 306.7 199.3 1.06×10−2 0.9855 322.6 9.1×10−5 0.9967 Notes: k1, k2—Pseudo-first-order kinetic and Pseudo-second-order kinetic constants, respectively; Qe(cal)—Calculation amount of MB removed per unit mass of adsorbent; Qe(exp)—Experimental amount of MB removed per unit mass of adsorbent. 表 3 MCC-SA-SEP-21复合微球对MB的吸附等温拟合结果
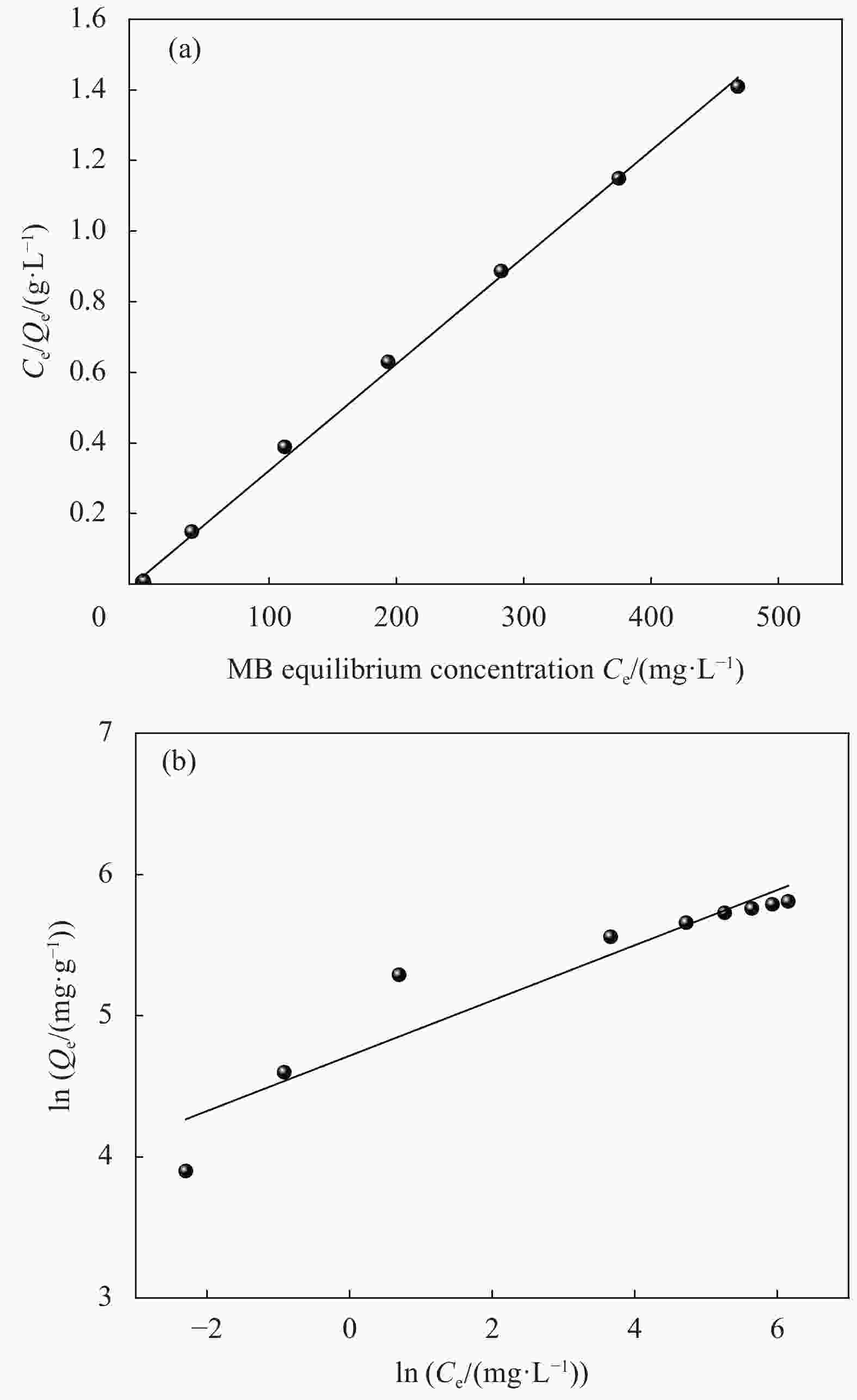
Table 3. Isothermal parameters for the adsorption of MB onto MCC-SA-SEP-21 beads
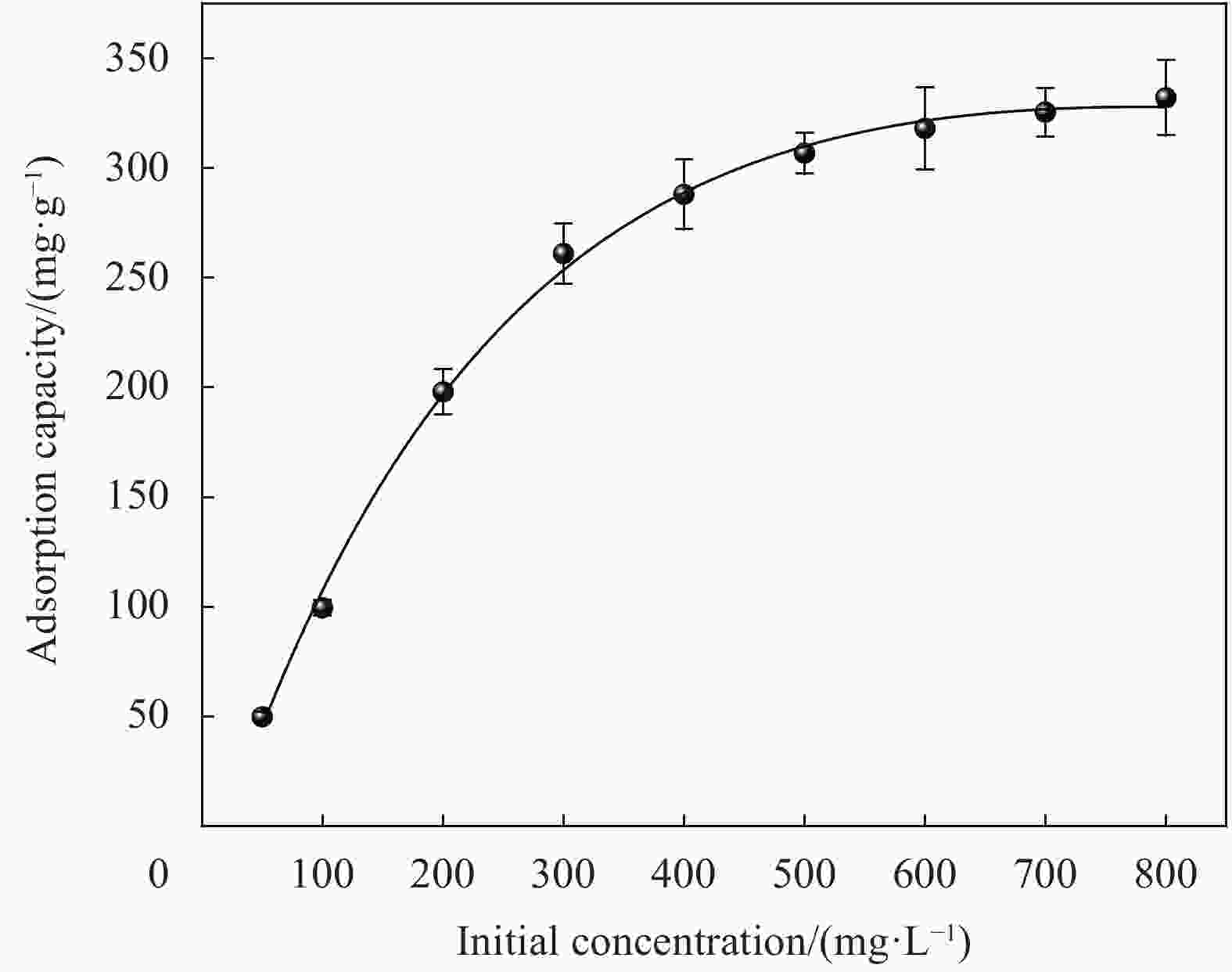
Adsorbate Langmuir Freundlich Qmax/(mg·g−1) kL/(L·mg−1) R2 kF/(L·mg−1) n R2 MB 333.3 0.147 0.9985 112 5 0.8949 Notes: Qmax—Langmuir adsorption maximum; kL—Langmuir coefficient of distribution of the adsorption; kF—Freundlich coefficient of distribution of the adsorption; n—Freundlich constants related to adsorption strength. 表 4 同类纤维素复合微球吸附剂对MB的吸附容量对比
Table 4. Adsorption capacity ratio of similar cellulose composite beads adsorbents to MB
Adsorbent Adsorption capacity/(mg·g−1) Reference SA/cellulose hydrogel beads 163.36 [26] Cellulose/diatomite composite aerogel beads 71.9424 [27] MCDBs 117.65 [14] CMC-AlG/GO hydrogels beads 78.5 [28] CNC-ALG 255.5 [29] CNC/MnO2/ALG beads 136.7 [30] MCC-SA-SEP 333.3 This work Notes: GO—Graphene oxide; MCDBs—Modified cellulose/diatomite beads; CMC—Carboxymethyl cellulose; CNC—Cellulose nanocrystal; ALG—Alginate. 表 5 MCC-SA-SEP-21复合微球对MB的吸附热力学参数
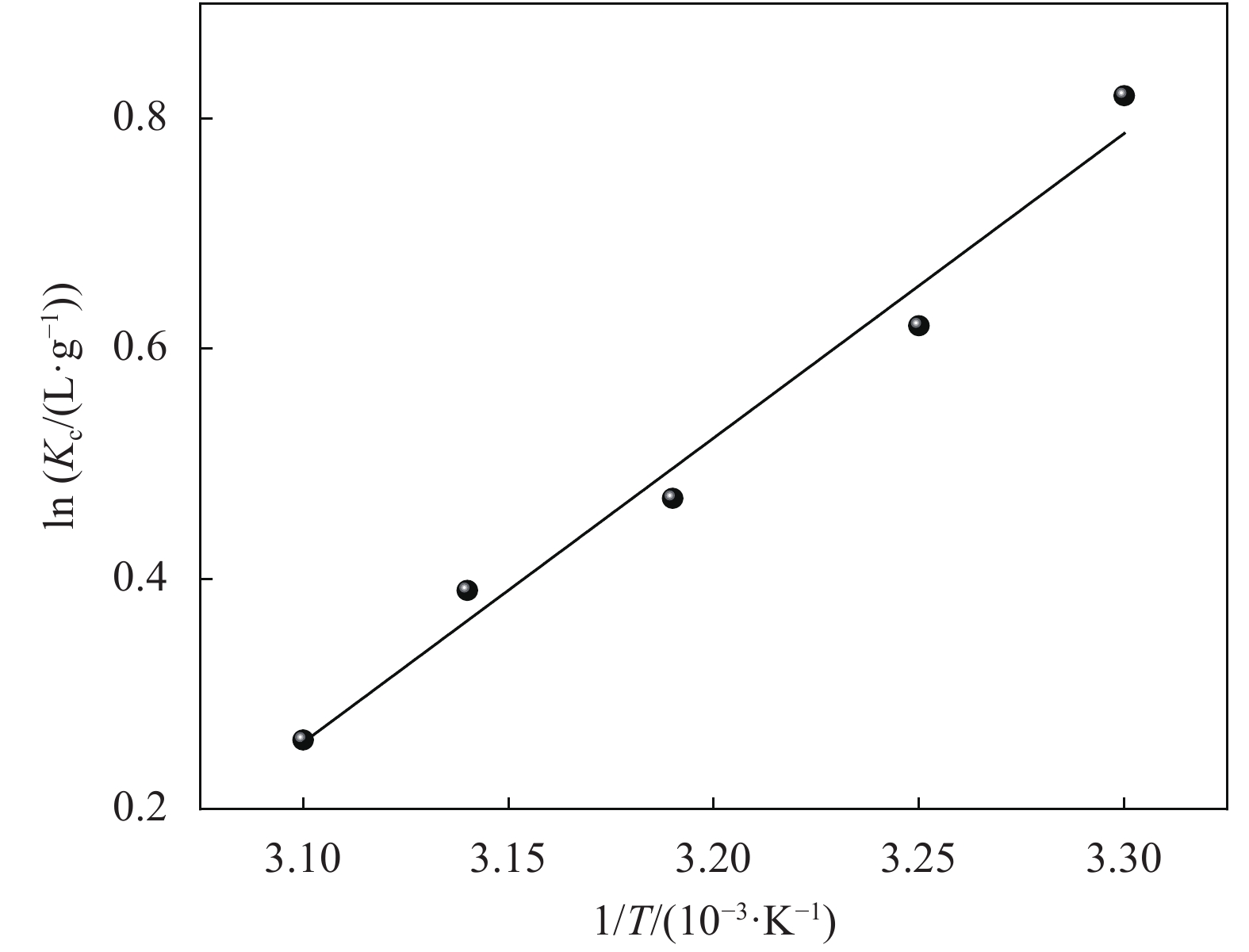
Table 5. Thermodynamic parameters for the adsorption of MB onto MCC-SA-SEP-21 beads
T/K ΔGo/(kJ·mol−1) ΔHo/(kJ·mol−1) ΔSo/(J·mol−1·K−1) 303 −2.1 −22 −66.1 308 −1.6 − − 313 −1.2 − − 318 −1 − − 323 −0.7 − − Notes: ΔGo—Gibbs free energy variation of the adsorption process; ΔHo—Enthalpy change of the adsorption process; ΔSo—Entropy change of the adsorption process. -
[1] DAI L, ZHU W, HE L, et al. Calcium-rich biochar from crab shell: An unexpected super adsorbent for dye removal[J]. Bioresource Technology,2018,267:510-516. doi: 10.1016/j.biortech.2018.07.090 [2] SONG W, GAO B, XU X, et al. Adsorption-desorption behavior of magnetic amine/Fe3O4 functionalized biopolymer resin towards anionic dyes from wastewater[J]. Bioresource Technology,2016,210:123-130. doi: 10.1016/j.biortech.2016.01.078 [3] HOLKAR C R, JADHAV A J, PINJARI D V, et al. A critical review on textile wastewater treatments: Possible approaches[J]. Journal of Environmental Management,2016,182:351-366. [4] GUPTA K C, KUMAR M N V. pH dependent hydrolysis and drug release behavior of chitosan/poly(ethylene glycol) polymer network microspheres[J]. Journal of Materials Science: Materials in Medicine,2001,12(9):753-759. doi: 10.1023/A:1017976014534 [5] PARK S, OH Y, YUN J, et al. Cellulose/biopolymer/Fe3O4 hydrogel microbeads for dye and protein adsorption[J]. Cellulose,2020,27(5):2757-2773. doi: 10.1007/s10570-020-02974-5 [6] 曾丹林, 陈诗渊, 张崎, 等. 纤维素制备微球材料的研究进展[J]. 材料导报, 2015, 29(17):68-72.ZENG Danlin, CHEN Shiyuan, ZHANG Qi, et al. Research progress of microspheres materials synthesized from cellulose[J]. Materials Reports,2015,29(17):68-72(in Chinese). [7] ROY D, SEMSARILAR M, GUTHRIE J T, et al. Cellulose modification by polymer grafting: A review[J]. Chemical Society Reviews,2009,38(7):2026-2064. [8] WAN Y, CHEN X, XIONG G, et al. Synthesis and characterization of three-dimensional porous graphene oxide/sodium alginate scaffolds with enhanced mechanical properties[J]. Materials Express,2014,4(5):429-434. doi: 10.1166/mex.2014.1188 [9] HU Z H, OMER A M, OUYANG X K, et al. Fabrication of carboxylated cellulose nanocrystal/sodium alginate hydrogel beads for adsorption of Pb(II) from aqueous solution[J]. International Journal of Biological Macromolecules,2018,108:149-157. doi: 10.1016/j.ijbiomac.2017.11.171 [10] 李延庆, 刘志明, 程小凯, 等. 海藻酸钠/纤维素复合微球的制备及性能表征[J]. 林产化学与工业, 2019, 39(2):67-72.LI Yanqing, LIU Zhiming, CHENG Xiaokai, et al. Preparation and performance characterization of sodium alginate/cellulose composite microspheres[J]. Chemistry and Industry of Forest Products,2019,39(2):67-72(in Chinese). [11] TARTAGLIONE G, TABUANI D, CAMINO G, et al. PP and PBT composites filled with sepiolite: Morphology and thermal behaviour[J]. Composites Science and Technology,2008,68(2):451-460. doi: 10.1016/j.compscitech.2007.06.023 [12] 张巍. 海泡石吸附混合污染物和气态污染物的研究进展[J]. 中国矿业, 2019, 28(2):126-132.ZHANG Wei. Research progress on sepiolite adsorption of mixed pollutants and gaseous pollutants[J]. China Mining Magazine,2019,28(2):126-132(in Chinese). [13] 杨欣洁. 新型磁性复合有机海泡石对双酚A的吸附试验研究[D]. 长沙: 湖南大学, 2015.YANG Xinjie. Adsorption of bisphenol A from aqueous solutions onto new magnetic composite organic sepiolite[D]. Changsha: Hunan University, 2015. [14] LI Y, XIAO H N, CHEN M D, et al. Absorbents based on maleic anhydride-modified cellulose fibers/diatomite for dye removal[J]. Journal of Materials Science,2014,46(19):6696-6704. [15] 李喜梅. 海藻酸盐纤维的热性能研究[D]. 青岛: 青岛大学, 2018.LI Ximei. Study on thermal properties of alginate fiber[D]. Qingdao: Qingdao University, 2018(in Chinese). [16] 宗鲁, 纪全, 谭利文, 等. 纤维素-钙纤维的制备及阻燃性能表征[J]. 高分子材料科学与工程, 2015, 31(2):176-180.ZONG Lu, JI Quan, TAN Liwen, et al. Preparation and flame retardant properties of cellulose-Ca fibers[J]. Polymer Materials Science & Engineering,2015,31(2):176-180(in Chinese). [17] ZHANG Y W, XU L, ZHAO L, et al. Radiation synthesis and Cr(VI) removal of cellulose microsphere adsorbent[J]. Carbohydrate Polymers,2012,88(3):931-938. doi: 10.1016/j.carbpol.2012.01.040 [18] 徐春霞, 降帅, 韩阜益, 等. 纤维素纳米纤丝气凝胶制备及其对亚甲基蓝的吸附性能[J]. 纺织学报, 2019, 40(10):20-25.XU Chunxia, JIANG Shuai, HAN Fuyi, et al. Preparation of cellulose nanofibrils aerogel and its adsorption of methylene blue[J]. Journal of Textile Research,2019,40(10):20-25(in Chinese). [19] LIU L, GAO Z Y, SU X P, et al. Adsorption removal of dyes from single and binary solutions using a cellulose-based bioadsorbent[J]. ACS Sustainable Chemistry & Engineering,2015,3(3):432-442. [20] TANZIFI M, YARAKI M T, KIADENHI A D, et al. Adsorption of amido black 10B from aqueous solution using polyaniline/SiO2 nanocomposite: Experimental investigtion and artificial neural network modeling[J]. Journal of Colloid and Interface Science,2018,510:246-261. doi: 10.1016/j.jcis.2017.09.055 [21] ZHOU Y, LIU X, XIANG Y, et al. Modification of biochar derived from sawdust and its application in removal of tetracycline and copper from aqueous solution: Adsorption mechanism and modelling[J]. Bioresource Technology,2017(245):266-273. [22] GULAY B, BEGUM A, YAKUP A. Adsorption kinetics and thermodynamic parameters of cationic dyes from aqueous solutions by using a new strong cation-exchange resin[J]. Chemical Engineering Journal,2009,1(52):339-346. [23] JIAO C L, TAO J, XIONG J Q, et al. In situ synthesis of MnO2-loaded biocomposite based on microcrystalline cellulose for Pb2+ removal from wastewater[J]. Cellulose,2017,24(6):2591-2604. doi: 10.1007/s10570-017-1271-4 [24] TAO J, XIONG J Q, JIAO C L, et al. Hybrid mesoporous silica based on hyperbranch-substrate nanonetwork as highly efficient adsorbent for water treatment[J]. ACS Sustainable Chemistry& Engineering,2016,4(1):60-68. [25] XU D, TAN X, CHEN C, et al. Removal of Pb(Ⅱ) from aqueous solution by oxidized multiwalled carbon nano-tubes[J]. Journal of Hazardous Materials,2008,154:407-416. doi: 10.1016/j.jhazmat.2007.10.059 [26] 吴鹏, 刘志明. 海藻酸钠/纤维素水凝胶球的制备与应用[J]. 功能材料, 2015, 46(10):10144-10147, 10152.WU Peng, LIU Zhiming. Preparation and application of sodium alginate/cellulose hydrogel beads[J]. Journal of Functional Materials,2015,46(10):10144-10147, 10152(in Chinese). [27] 王佳楠, 羿颖, 边勇军, 等. 纤维素/硅藻土复合气凝胶球的制备及其吸附性能的研究[J]. 纤维素科学与技术, 2019, 27(1):49-56.WANG Jianan, YI Ying, BIAN Yongjun, et al. Preparation of cellulose/diatomite composite aerogel beads and its adsorption performance[J]. Journal of Cellulose Science and Technology,2019,27(1):49-56(in Chinese). [28] ALLOUSS D, ESSAMLALI Y, AMADINE O, et al. Response surface methodology for optimization of methylene blue adsorption onto carboxymethyl cellulose-based hydrogel beads: Adsorption kinetics, isotherm, thermodynamics and reusability studies[J]. RSC Advances,2019,9(65):37858-37869. doi: 10.1039/C9RA06450H [29] MOHAMMED N, GRISHKEWICH N, WAEIJEN H A, et al. Continuous flow adsorption of methylene blue by cellulose nanocrystal-alginate hydrogel beads infixed bed columns[J]. Carbohydrate Polymers,2016,136:1194-1202. doi: 10.1016/j.carbpol.2015.09.099 [30] DIAO H L, ZHANG Z J, LIU Y X, et al. Facile fabrication of carboxylated cellulose nanocrystal-MnO(2)beads for high-efficiency removal of methylene blue[J]. Cellulose,2020,27(12):7053-7066. doi: 10.1007/s10570-020-03260-0 [31] KUMAR K V, KUMARAN A. Removal of methylene blue by mango seed kernel powder[J]. Biochemical Engineering Journal,2005,27(1):83-93. doi: 10.1016/j.bej.2005.08.004 [32] KUBILAY S, GÜRKAN R, SAVRAN A, et al. Removal of Cu(II), Zn(II) and Co(II) ions from aqueous solutions by adsorption onto natural bentonite[J]. Adsorption,2007,13(1):41-51. doi: 10.1007/s10450-007-9003-y [33] STANCIU M C, NICHIFOR M. Adsorption of anionic dyes on a cationic amphiphilic dextran hydrogel: Equilibrium, kinetic, and thermodynamic studies[J]. Colloid and Polymer Science,2019,297(1):45-57. doi: 10.1007/s00396-018-4439-z [34] JIANG Y F, SUN H, YVES U J, et al. Impact of biochar produced from post-harvest residue on the adsorption behavior of diesel oil on loess soil[J]. Environmental Geochemistry and Health,2016,38(1):243-253. doi: 10.1007/s10653-015-9712-1 -






 下载:
下载: