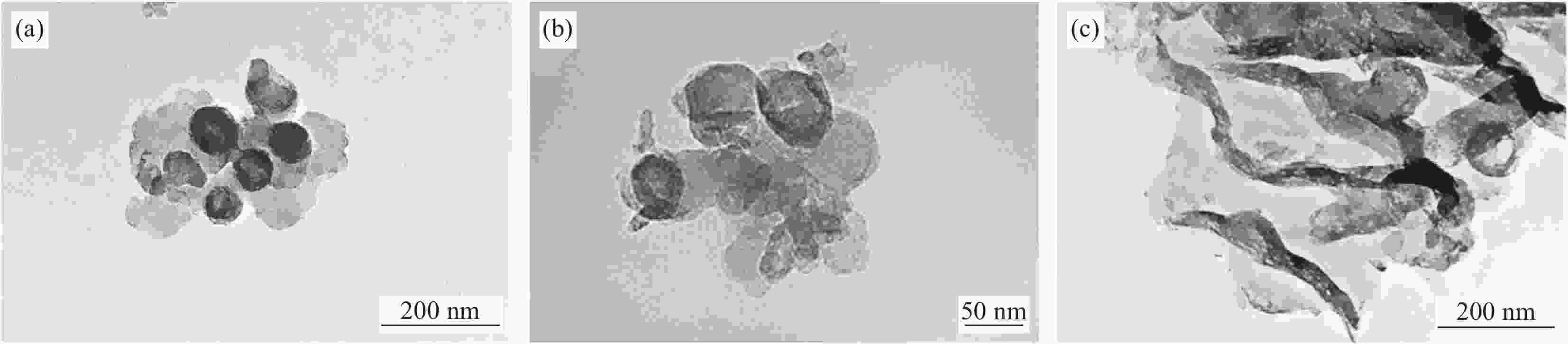
Improvement of the performance of photocatalytic degradation of acid orange Ⅱ by carbon nanospheres combined with g-C3N4
-
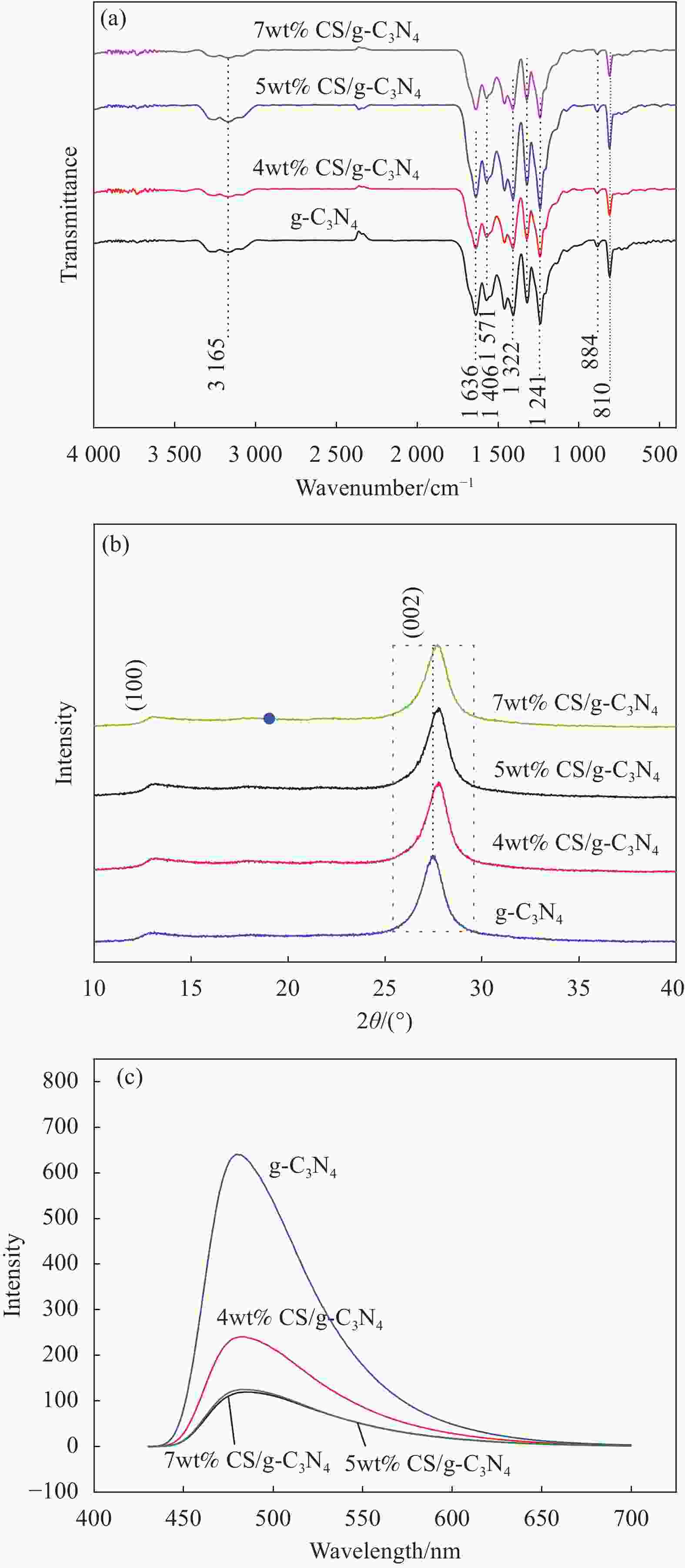
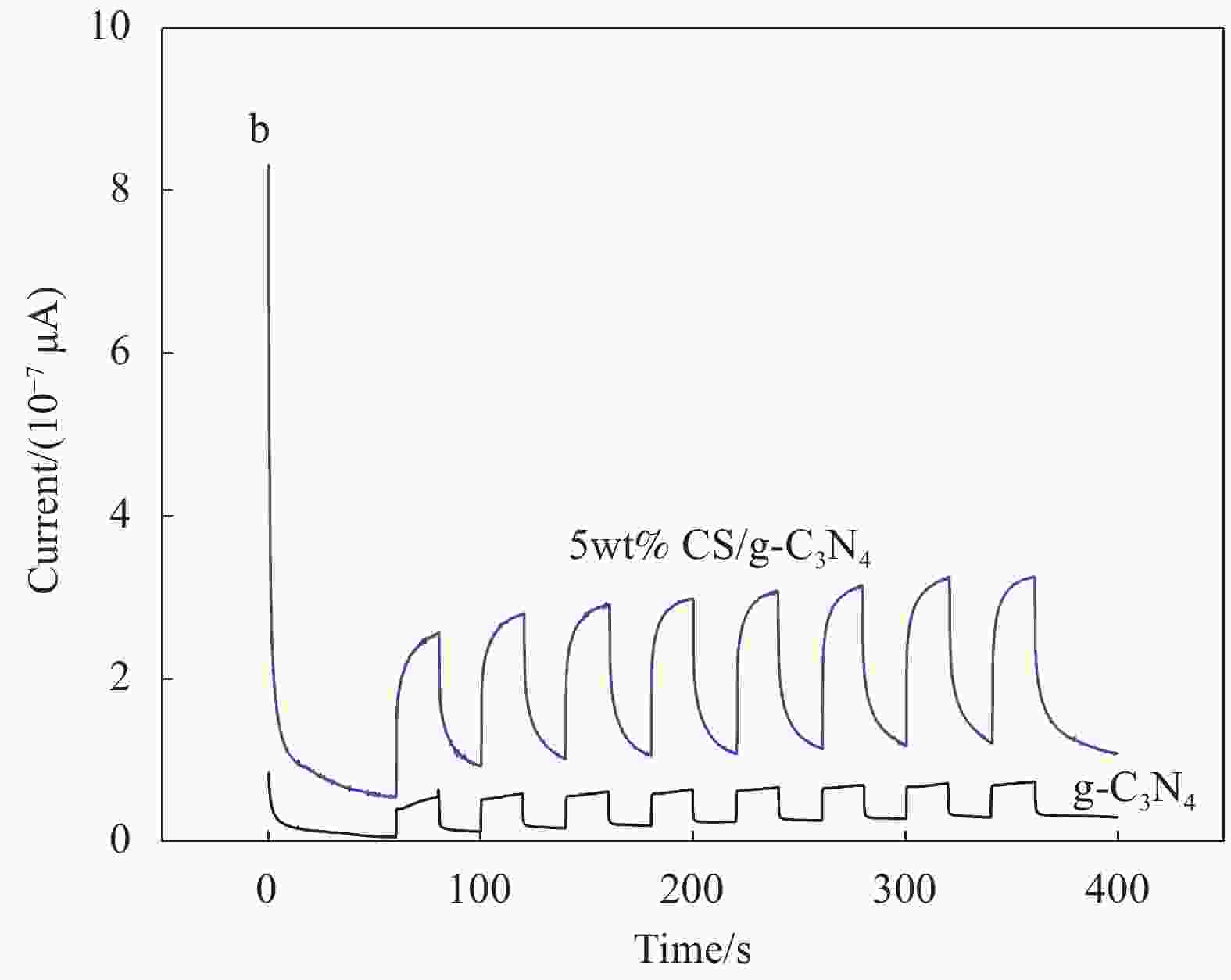
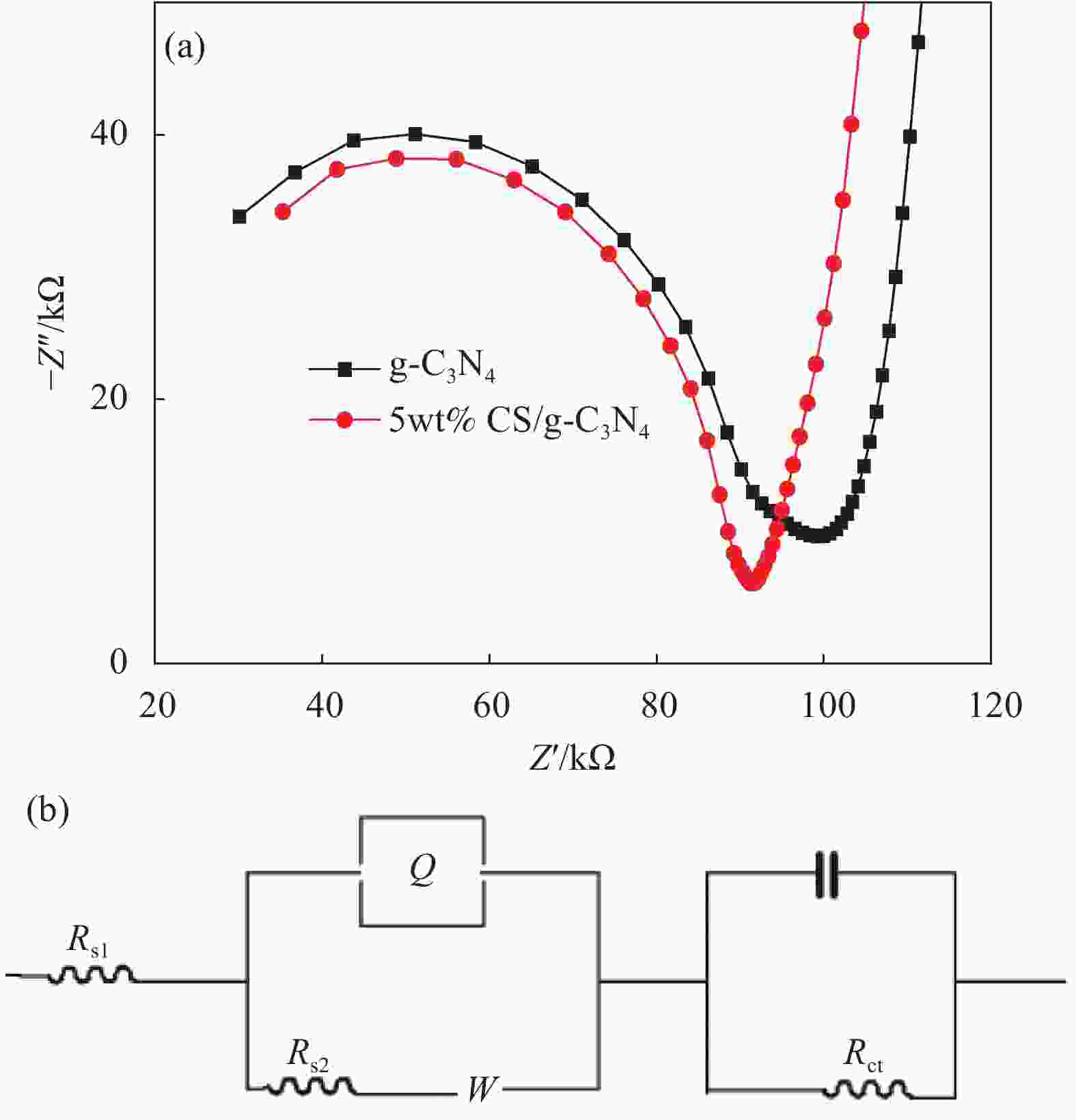
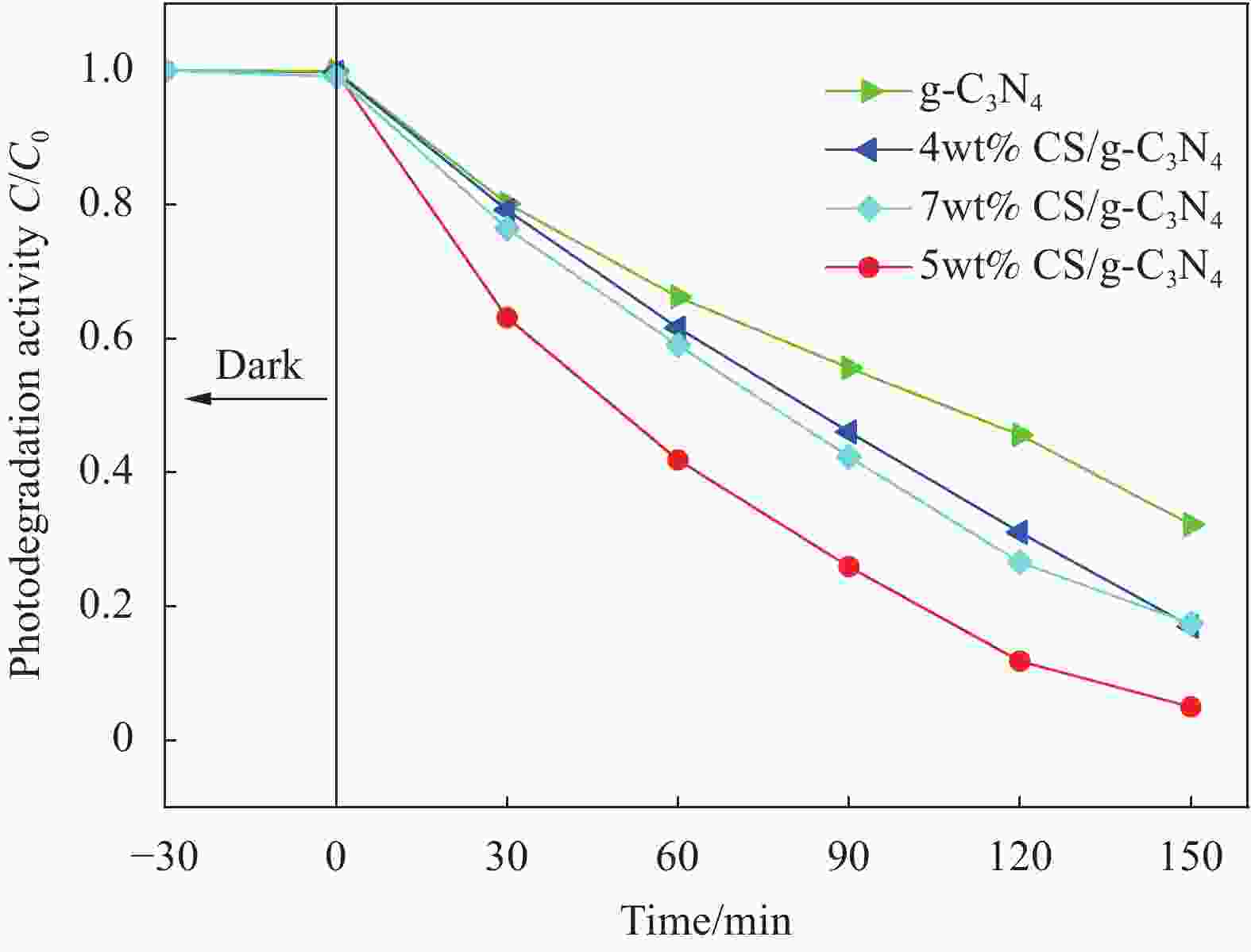
摘要: 基于g-C3N4构建的异质结光催化材料在降解有毒有害污染物方面体现出优良的效果。本研究通过水热法制备了一系列不同碳纳米球(Carbon nanospheres,CS)添加量的x-CS/g-C3N4 (x=4wt%、5wt%和7wt%)复合光催化剂,以氙灯光源模拟可见光,探究了x-CS/g-C3N4对酸性橙Ⅱ的光催化降解性能。结果表明:5wt% CS/g-C3N4的光催化活性最高,光催化反应150 min,酸性橙Ⅱ的降解率达到95%。表征结果表明,g-C3N4与CS具有类似的π-π共轭结构,易发生π-π堆积相互作用而有利于电子跃迁。二者复合后能有效增强g-C3N4对可见光的吸收效率,降低其表面/界面处的电荷转移电阻,显著增强载流子的传输能力。x-CS/g-C3N4可作为一种有效的可见光催化剂应用于有机染料降解,具有应用前景。Abstract: The heterojunction photocatalytic material constructed with g-C3N4 as the matrix shows excellent effects in degrading toxic and harmful pollutants. In this study, a series of x-CS/g-C3N4 (x=4wt%, 5wt% and 7wt%) composite photocatalysts with different addition amounts of carbon nanospheres (CS) were prepared by hydrothermal method, and the photocatalytic degradation performance of x-CS/g-C3N4 on acid orange II were explored when a xenon lamp was used as a visible light source. The results show that the photocatalytic activity of 5wt% CS/g-C3N4 is the highest, and the degradation rate of acid orange II reaches 95% when the photocatalytic reaction is 150 min. The characterization results show that g-C3N4 and CS have a similar π-π conjugate structure, and π-π stacking interaction is prone to occur, which is beneficial to electronic transition. The combination of g-C3N4 and CS can effectively enhance the absorption efficiency of g-C3N4 for visible light, reduce the charge transfer resistance at the surface/interface, and significantly enhance the transport capacity of carriers. x-CS/g-C3N4 can be used as an effective visible light catalyst for the degradation of organic dyes and has application prospects.
-
Key words:
- carbon nanospheres /
- g-C3N4 /
- composite photocatalyst /
- photocatalytic performance /
- acid orange II
-
表 1 g-C3N4和x-CS/g-C3N4催化剂的比表面积、孔容和孔径值
Table 1. Surface area, pore volume and pore size of g-C3N4 and x-CS/g-C3N4
Sample Specific surface
area/(m2·g−1)Pore
size/nmPore volume/
(cm3·g−1)g-C3N4 10.09 31.04 0.14 4wt% CS/g-C3N4 34.41 26.32 0.23 5wt% CS/g-C3N4 39.17 28.35 0.27 7wt% CS/g-C3N4 48.18 31.87 0.39 表 2 g-C3N4和x-CS/g-C3N4催化剂的吸收边和带隙能
Table 2. Absorption edge and bandgap energy of g-C3N4 and x-CS/g-C3N4 catalyst
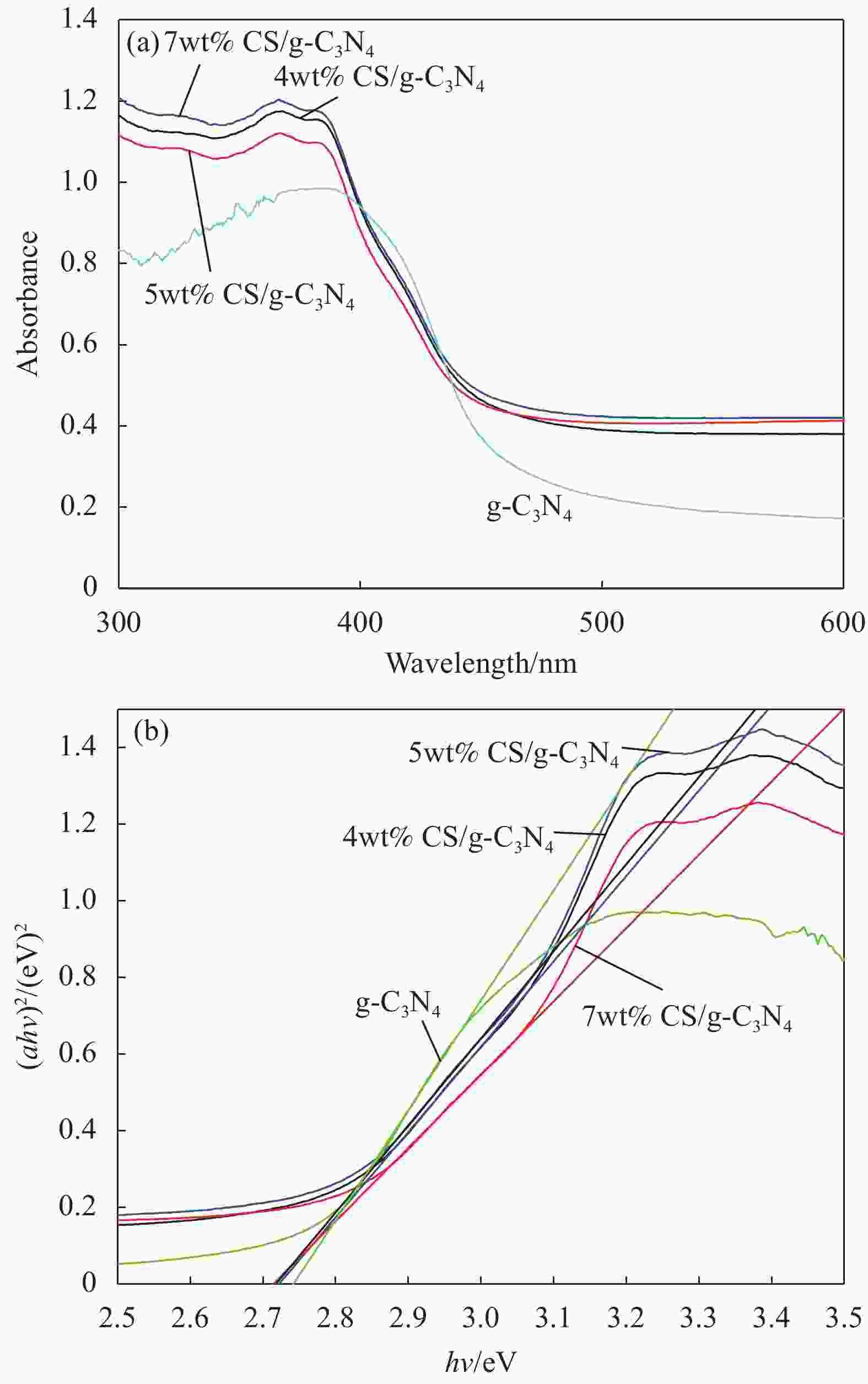
Sample λg/nm Eg/eV g-C3N4 452 2.74 4wt% CS/g-C3N4 455 2.72 5wt% CS/g-C3N4 457 2.71 7wt% CS/g-C3N4 457 2.71 Notes: λg—Absorption edge; Eg—Bandgap energy. 表 3 g-C3N4与不同碳量子点材料复合而成的光催化剂的光催化性能比较
Table 3. Comparison of the results for a number of CDs/g-C3N4-based nanocomposites
Photocatalyst Preparation method Degradation Light source Efficiency [Ref] 5wt% CS/g-C3N4 Hydrothermal Acid Orange II Xenon lamp (500 W) 95% in 150 min This study CQDs/g-C3N4 Precipitation RhB Xenon lamp (250 W) 95.2% in 210 min [32] g-C3N4/CDs/AgBr Precipitation RhB Xenon lamp (250 W) 96.0% in 40 min [33] g-C3N4/C-dots Hydrothermal MO Halide lamp (35 W) 92.0% in 180 min [34] Graphene/CQDs/g-C3N4 nanosheet Hydrothermal MO Xenon lamp (100 W) 91.1% in 240 min [35] SDAg-CQDs/ultrathin g-C3N4 Thermo-
polymerizationNaproxen Xenon lamp (350 W) 87.5% in 25 min [36] g-C3N4/CQDs Deposition RhB Xenon lamp (300 W) 100% in 210 min [37] g-C3N4/AgCl/CD Impregnation MB and RhB LED lamp (40 W) 100% in 75 min (MB) &
90% in 75 min (RhB)[38] CdS/CQDs/g-C3N4 Thermal
polymerizationMB, RhB, phenol Xenon lamp (300 W) 70, 95, 60% in 120 min (RhB, MB, phenol, respectively) [39] g-C3N4/Bi2WO6/NCQs In-situ calcination
and hydrothermalRhB and TC Xenon lamp (800 W) 95% in 45 min (RhB) & 80% in 60 min (TC) [40] Notes: CS—Carbon nanospheres; CQDs—Carbon quantum dots; CDs—Carbon dots, SDAg—Single atom-dispersed silver; NCQs—Nitrogen-doped carbon quantum dots; RhB—Rhodamine B; MO—Methyl orange; MB—Methylene blue; TC—Tetracycline. -
[1] 楚增勇, 原博, 颜廷楠. g-C3N4光催化性能的研究进展[J]. 无机材料学报, 2014, 29(8):785-794.CHU Z Y, YUAN B, YAN T N. Recent progress in photocatalysis of g-C3N4[J]. Acta Materiae Compositae Sinica,2014,29(8):785-794(in Chinese). [2] SCHWINGHAMMER K, MESCH M B, DUPPEL V, et al. Crystalline carbon nitride nanosheets for improved visible-light hydrogen evolution[J]. Journal of the American Chemical Society,2014,136(5):1730-1733. doi: 10.1021/ja411321s [3] NIU P, YANG Y Q, JIMMY C Y, et al. Switching the selectivity of the photoreduction reaction of carbon dioxide by controlling the band structure of a g-C3N4 photocatalyst[J]. Chemical Communications,2014,50(74):10837-10840. doi: 10.1039/C4CC03060E [4] TIAN J Q, LIU Q, GE C J, et al. Ultrathin graphitic carbon nitride nanosheets: A low-cost, green, and highly efficient electrocatalyst toward the reduction of hydrogen peroxide and its glucose biosensing application[J]. Nanoscale,2013,5(19):8921-8924. doi: 10.1039/c3nr02031b [5] YUAN Y P, XU W T, YIN L S, et al. Large impact of heating time on physical properties and photocatalytic H2 production of g-C3N4 nanosheets synthesized through urea polymerization in Ar atmosphere[J]. International Journal of Hydrogen Energy,2013,38(30):13159-13163. doi: 10.1016/j.ijhydene.2013.07.104 [6] HONG Y Z, LIU E L, SHI J Y, et al. A direct one-step synthesis of ultrathin g-C3N4 nanosheets from thiourea for boosting solar photocatalytic H2 evolution[J]. International Journal of Hydrogen Energy,2019,44(14):7194-7204. doi: 10.1016/j.ijhydene.2019.01.274 [7] DA S E S, MOURA N M M, COUTINHO A, et al. β-cyclodextrin as a precursor to holey C-doped g-C3N4 nanosheets for photocatalytic hydrogen generation[J]. ChemSusChem,2018,11(16):2681-2694. doi: 10.1002/cssc.201801003 [8] JIANG G, CAO J, CHEN M, et al. Photocatalytic NO oxidation on N-doped TiO2/g-C3N4 heterojunction: Enhanced efficiency, mechanism and reaction pathway[J]. Applied Surface Science,2018,458:77-85. doi: 10.1016/j.apsusc.2018.07.087 [9] ZHOU Y, ZHANG L, LIU J, et al. Brand new P-doped g-C3N4: Enhanced photocatalytic activity for H2 evolution and Rhodamine B degradation under visible light[J]. Journal of Materials Chemistry A,2015,3(7):3862-3867. doi: 10.1039/C4TA05292G [10] KONG W, ZHANG X, CHANG B, et al. Fabrication of B doped g-C3N4/TiO2 heterojunction for efficient photoelectrochemical water oxidation[J]. Electrochimica Acta,2018,282:767-774. doi: 10.1016/j.electacta.2018.06.090 [11] MUNOZ M J, FONTELLES O, FERRER M, et al. Disinfection capability of Ag/g-C3N4 composite photocatalysts under UV and visible light illumination[J]. Applied Catalysis B: Environmental,2016,183:86-95. doi: 10.1016/j.apcatb.2015.10.024 [12] PAWAR R C, KANG S, AHN S H, et al. Gold nanoparticle modified graphitic carbon nitride/multi-walled carbon nanotube (g-C3N4/CNTs/Au) hybrid photocatalysts for effective water splitting and degradation[J]. Rsc Advances,2015,5(31):24281-24292. doi: 10.1039/C4RA15560B [13] CAUX M, FINA F, IRVINE J T S, et al. Impact of the annealing temperature on Pt/g-C3N4 structure, activity and selectivity between photo degradation and water splitting[J]. Catalysis Today,2017,287:182-188. doi: 10.1016/j.cattod.2016.11.007 [14] ZHANG S, GU P, MA R, et al. Recent developments in fabrication and structure regulation of visible-light-driven g-C3N4-based photocatalysts towards water purification: a critical review[J]. Catalysis Today,2019,335:65-77. doi: 10.1016/j.cattod.2018.09.013 [15] AHMED A, NIAZI M B K, JAHAN Z, et al. In-vitro and in-vivo study of superabsorbent PVA/Starch/g-C3N4/Ag@TiO2 NPs hydrogel membranes for wound dressing[J]. European Polymer Journal,2020,130:109650. doi: 10.1016/j.eurpolymj.2020.109650 [16] NIKOKAVOURA A, TRAPALIS C. Graphene and g-C3N4 based photocatalysts for NOx removal: A review[J]. Applied Surface Science,2018,430:18-52. doi: 10.1016/j.apsusc.2017.08.192 [17] 王鹏, 李昭, 周颖梅, 等. 碳纳米管改性g-C3N4提升可见光催化降解性能[J]. 无机化学学报, 2019, 35(2):217-224.WANG P, LI Z, ZHOU Y M, et al. Synthesis of carbon nanotubes modified g-C3N4 photocatalysts for enhanced photocatalytic degradation activity[J]. Chinese Journal of Chemistry,2019,35(2):217-224(in Chinese). [18] TONG Z W, YANG D, SHI J F, et al. Three dimensional porous aerogel constructed by g-C3N4 and graphene oxide nanosheets with excellent visible light photocatalytic performance[J]. ACS Applied Materials & Interfaces,2015,7(46):25693-25701. [19] 刘翀, 刘丽来, 聂佳慧. 高活性碳球修饰g-C3N4的制备及光催化性能[J]. 高等学校化学学报, 2018, 39(7):1511-1517.LIU C, LIU L L, NIE J H. Synthesis of carbon ball modified g-C3N4 for improved photocatalytic activity[J]. Chemical Journal of Chinese Universities,2018,39(7):1511-1517(in Chinese). [20] YAN S C, LI Z S, ZOU Z G. Photodegradation performance of g-C3N4 fabricated by directly heating melamine[J]. Langmuir,2009,25(17):10397-10401. doi: 10.1021/la900923z [21] ZHAO W, GUO Y, WANG S M, et al. A novel ternary plasmonic photocatalyst: ultrathin g-C3N4 nanosheet hybrided by Ag/AgVO3 nanoribbons with enhanced visible-light photocatalytic performance[J]. Applied Catalysis B: Environmental,2015,165:335-343. doi: 10.1016/j.apcatb.2014.10.016 [22] GAO L, LI Y, Ren J, et al. Passivation of defect states in anatase TiO2 hollow spheres with Mg doping: Realizing efficient photocatalytic overall water splitting[J]. Applied Catalysis B: Environmental,2017,202:127-133. doi: 10.1016/j.apcatb.2016.09.018 [23] NIU P, LIU G, CHENG H M. Nitrogen vacancy-promoted photocatalytic activity of graphitic carbon nitride[J]. Journal of Physical Chemistry C,2012,116(20):11013-11018. doi: 10.1021/jp301026y [24] LI Y, ZHANG J, WANG Q, et al. Nitrogen-rich carbon nitride hollow vessels: synthesis, characterization, and their properties[J]. Journal of Physical Chemistry B,2010,114(29):9429-9434. doi: 10.1021/jp103729c [25] VINU A. Two-Dimensional hexagonally-ordered mesoporous carbon nitrides with tunable pore diameter, surface area and nitrogen content[J]. Advanced Functional Materials,2008,18(5):816-827. doi: 10.1002/adfm.200700783 [26] DANTE R C, MARTIN R P, CORREA G A, et al. Synthesis of graphitic carbon nitride by reaction of melamine and uric acid[J]. Materials Chemistry and Physics,2011,130(3):1094-1102. doi: 10.1016/j.matchemphys.2011.08.041 [27] LIU S, LI C, YU J, et al. Improved visible-light photocatalytic activity of porous carbon self-doped ZnO nanosheet-assembled flowers[J]. CrystEngComm,2011,13(7):2533-2541. doi: 10.1039/c0ce00295j [28] HAO Q, HAO S, NIU X, et al. Enhanced photochemical oxidation ability of carbon nitride by π-π stacking interactions with graphene[J]. Chinese Journal of Catalysis,2017,38(2):278-286. doi: 10.1016/S1872-2067(16)62561-5 [29] DONG G, ZHANG L. Porous structure dependent photoreactivity of graphitic carbon nitride under visible light[J]. Journal of Materials Chemistry,2012,22(3):1160-1166. doi: 10.1039/C1JM14312C [30] THOMAS A, FISCHER A, GOETTMANN F, et al. Graphitic carbon nitride materials: variation of structure and morphology and their use as metal-free catalysts[J]. Journal of Materials Chemistry,2008,18(41):4893-4908. doi: 10.1039/b800274f [31] 刘素芹, 王松, 戴高鹏, 等. 复合碳纳米管增强纳米Ag2CO3的可见光催化活性和稳定性[J]. 物理化学学报, 2014, 30(11):2121-2126. doi: 10.3866/PKU.WHXB201409191LIU S Q, WANG S, DAI G P, et al. Enhanced visible-light photocatalytic activity and stability of nano-sized Ag2CO3 combined with carbon nanotubes[J]. Acta Physico-Chimica Sinica,2014,30(11):2121-2126(in Chinese). doi: 10.3866/PKU.WHXB201409191 [32] HONG Y, MENG Y, ZHANG G, et al. Facile fabrication of stable metal-free CQDs/g-C3N4 heterojunctions with efficiently enhanced visible-light photocatalytic activity[J]. Separation and Purification Technology,2016,171:229-237. doi: 10.1016/j.seppur.2016.07.025 [33] MIAO X, JI Z, WU J, et al. g-C3N4/AgBr nanocomposite decorated with carbon dots as a highly efficient visible-light-driven photocatalyst[J]. Journal of Colloid and Interface Science,2017,502:24-32. doi: 10.1016/j.jcis.2017.04.087 [34] QIN T, YOU Z Y, WANG H, et al. Preparation and photocatalytic behavior of carbon-nanodots/graphitic carbon nitride composite photocatalyst[J]. Journal of the Electrochemical Society,2017,164:211-214. doi: 10.1149/2.1421704jes [35] HE H, HUANG L, ZHONG Z, et al. Constructing three-dimensional porous graphene-carbon quantum dots/g-C3N4 nanosheet aerogel metal-free photocatalyst with enhanced photocatalytic activity[J]. Applied Surface Science,2018,441:285-294. doi: 10.1016/j.apsusc.2018.01.298 [36] WANG F, WANG Y, FENG Y, et al. Novel ternary photocatalyst of single atom-dispersed silver and carbon quantum dots co-loaded with ultrathin g-C3N4 for broad spectrum photocatalytic degradation of naproxen[J]. Applied Catalysis B-Environmental,2018,221:510-520. doi: 10.1016/j.apcatb.2017.09.055 [37] SHI J, FENG S, CHEN T, et al. Effect of porous modification on the synthesis and photocatalytic activity of graphitic carbon nitride/carbon quantum dot nanocomposite[J]. Journal of Materials Science: Materials in Electronics,2018,29:17454-17462. doi: 10.1007/s10854-018-9845-y [38] MATHESWARAN P, THANGAVELU P, PALANIVEL B. Carbon dot sensitized integrative g-C3N4/AgCl hybrids: An synergetic interaction for enhanced visible light driven photocatalytic process[J]. Advanced Powder Technology,2019,30:1715-1723. doi: 10.1016/j.apt.2019.05.024 [39] FENG S, CHEN T, LIU Z, et al. Z-scheme CdS/CQDs/g-C3N4 composites with visible-near-infrared light response for efficient photocatalytic organic pollutant degradation[J]. Science of The Total Environment,2020,704:135404. doi: 10.1016/j.scitotenv.2019.135404 [40] JIA J, ZHANG X, JIANG C, et al. Visible-light-driven nitrogen-doped carbon quantum dots decorated g-C3N4/Bi2WO6 Z-scheme composite with enhanced photocatalytic activity and mechanism insight[J]. Journal of Alloys and Compounds,2020,835:155180. doi: 10.1016/j.jallcom.2020.155180 -






 下载:
下载: