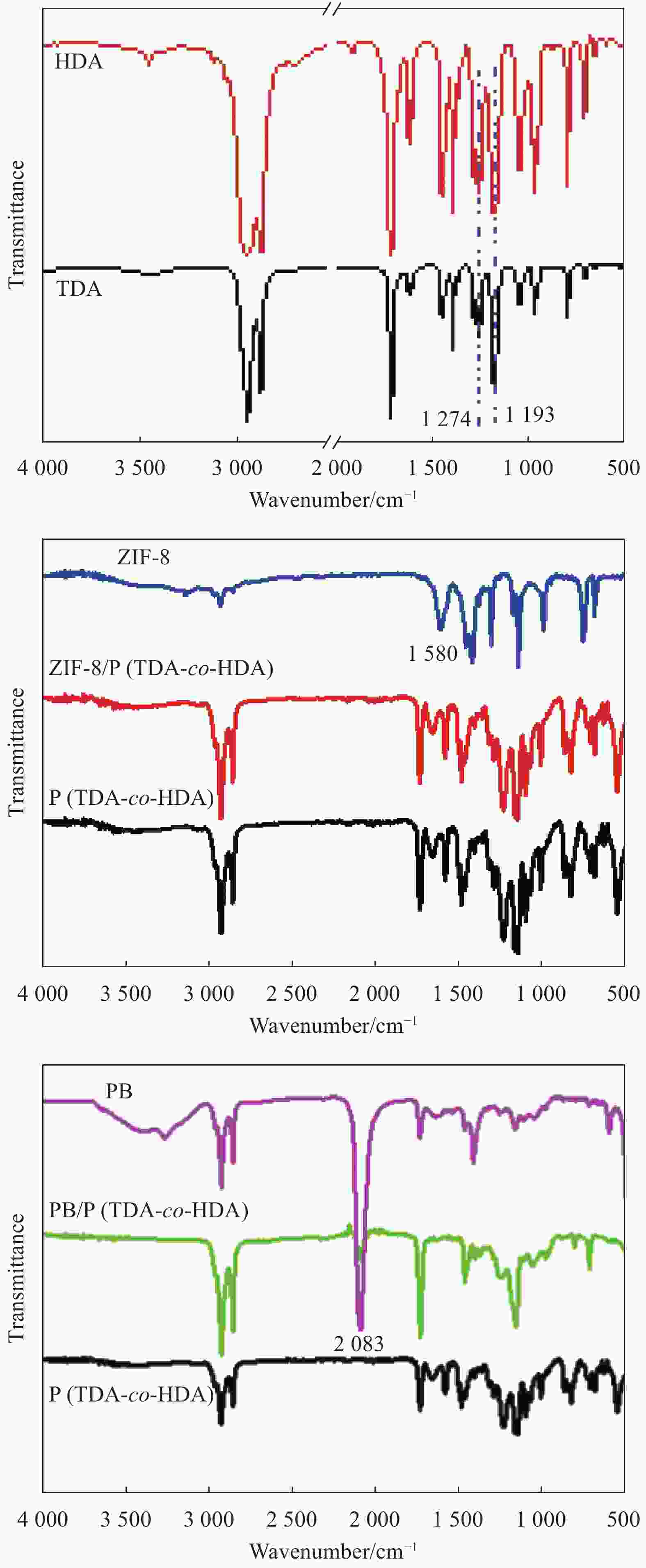
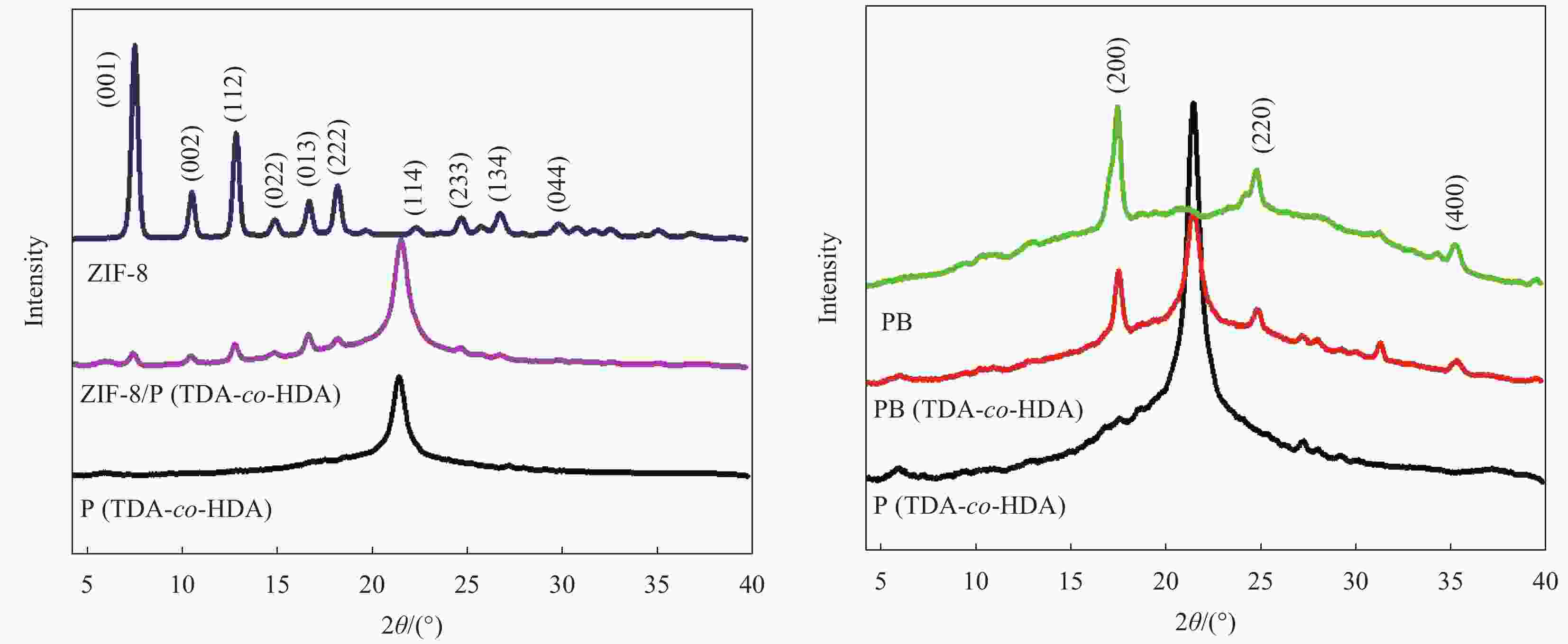
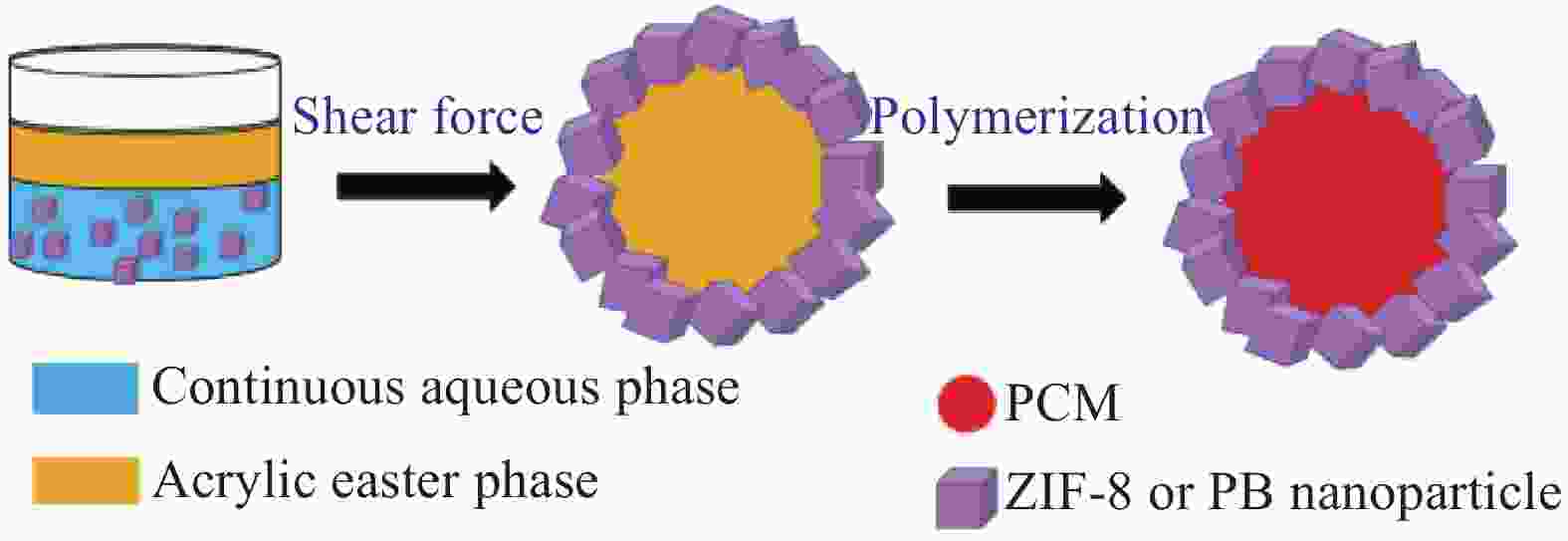
Fabrication and characterization of shape-stabilized phase change materials of ZIF-8/P(tetradecyl acrylate-co-hexadecyl acrylate) and prussian blue/ (tetradecyl acrylate-co-hexadecyl acrylate)
-
摘要: 形状稳定的相变材料(SPCMs)是绿色的可重复使用的储能材料。由于丙烯酸正烷基酯共聚物的熔融温度可通过控制侧链长度来调节,因此可以得到适宜的相变温度。用一种基于金属-有机骨架纳米粒子稳定悬浮聚合法制备出了功能MOFs/聚合物复合材料—ZIF-8/丙烯酸十四-十六酯共聚物(ZIF-8/P(TDA-co-HDA))与PB/丙烯酸十四-十六酯共聚物(PB/P(TDA-co-HDA)),通过使用此技术,可以将功能性纳米颗粒固定在聚合物表面上,ZIF-8和PB起到形状稳定的作用。ZIF-8/P(TDA-co-HDA)的吸放热温度分别为37.5℃和8.4℃,相变焓值为63 J/g,PB/P(TDA-co-HDA)在39.1℃吸热,10.1℃放热,相变焓值为68 J/g。该种材料在60℃时保持其形状没有任何泄漏,这远高于P(TDA-co-HDA)的熔融温度。在1000个热循环后,ZIF-8/P(TDA-co-HDA)和PB/P(TDA-co-HDA)仍表现出良好的结晶行为和热可靠性。制备的新型形状稳定相变材料在热能存储应用中具有潜在的用途。Abstract: A novel strategy based on Metal-organic frameworks nanoparticles (MOFs NPs)-stabilized suspension polymerization has been achieved for the fabrication of multifunctional ZIF-8/P(TDA-co-HDA) and PB/P(TDA-co-HDA) composites. By using this technique, functional nanoparticles can be immobilized on the surface of polymer. In this paper, ZIF-8 and PB nanoparticles (NPs) were used as stabilizer for the suspension polymerization in water and ZIF-8/P(TDA-co-HDA) and PB/P(TDA-co-HDA) composites are successfully synthesized. ZIF-8/P(TDA-co-HDA) and PB/P(TDA-co-HDA) absorbed heat at 37.5℃, 39.1℃ and released it at 8.4℃, 10.1℃ with a heat storage capacity of 63 J/g, 68 J/g, respectively. The material retains its shape without any leakage at 60℃, which is much higher than that of the melting temperature of P(TDA-co-HDA). The ZIF-8/P(TDA-co-HDA) and PB/P(TDA-co-HDA) composites exhibit good crystallization behaviors and excellent thermal reliabilities after 1000 thermal cycles. The thermal properties of the ZIF-8/P(TDA-co-HDA) and PB/P(TDA-co-HDA) composites were also investigated. The novel shape-stabilized PCMs fabricated in this study have potential uses in thermal energy storage applications.
-
表 1 P(TDA-co-HDA)、ZIF-8/P(TDA-co-HDA)和PB/P(TDA-co-HDA)的相变特性
Table 1. Thermal properties of P(TDA-co-HDA), ZIF-8/P(TDA-co-HDA) and PB/P(TDA-co-HDA)
Sample ΔHm/(J·g−1) Tmo/℃ Tmp/℃ ΔHc/(J·g−1) Tco/℃ Tcp/℃ T5wt%/℃ P(TDA-co−HDA) 70 30.9 38.2 −70 19.9 9.1 290.8 ZIF-8/P(TDA-co-HDA) 62 27.9 37.5 −63 19.6 8.4 227.6 PB/P(TDA-co−HDA 68 31.9 39.1 −68 19.7 10.1 254.8 Notes: Tmo—Onset melting temperature on the DSC heating curve; Tmp—Peak melting temperature on the DSC heating curve; ΔHm—Melting enthalpy; Tco—Onset crystallizing temperature on the crystallization curve; Tcp—Peak crystallizing temperature on the crystallization curve; ΔHc—Crystallizing enthalpy; T5wt%—Temperature at mass loss of 5wt%. 表 2 热循环处理前后ZIF-8/P(TDA-co-HDA)和PB/P(TDA-co-HDA)的DSC数据
Table 2. DSC datas of ZIF-8/P(TDA-co-HDA) and PB/P(TDA-co-HDA) before and after thermal cycling treatments
Sample ΔHm/(J·g−1) Tmo/℃ Tmp/℃ ΔHc/(J·g−1) Tco/℃ Tcp/℃ ZIF-8/P(TDA-co-HDA) 1 cycle 62 27.9 37.5 −63 19.6 8.4 1000 cycles 60 27.6 37.4 −61 19.5 8.5 PB/P(TDA-co-HDA) 1 cycle 68 31.9 39.1 −68 19.7 10.1 1000 cycles 67 32.1 39.2 −66 19.9 9.9 -
[1] AYDM A A, OKUTAN H. High-chain fatty acid esters of myristyl alcohol with even carbon number: novel organic phase change materials for thermal energy storage-1[J]. Solar Energy Materials & Solar Cells,2011,95(10):2752-2762. [2] WANG C Y, FENG L L, XIN G B, et al. Graphene oxide stabilized polyethylene glycol for heat storage[J]. Physical Chemistry Chemical Physics PCCP,2012,14(38):13233-13238. doi: 10.1039/c2cp41988b [3] LI D, WANG J P, WANG Y N, et al. Effect of N-isopropylacrylamide on the preparation and properties of microencapsulated phase change materials[J]. Energy,2016,106:221-230. doi: 10.1016/j.energy.2016.03.035 [4] ZHANG Z L, ZHANG X X, SHI H F, et al. Thermo-regulated sheath/core submicron fiber with poly(diethylene glycol hexadecyl ether acrylate) as a core[J]. Textile Research Journal,2015,86(5):493-501. [5] LI W, ZHANG R, JIANG N, et al. Composite macrocapsule of phase change materials/expanded graphite for thermal energy storage[J]. Energy,2013,57:607-614. doi: 10.1016/j.energy.2013.05.007 [6] PEI D F, CHEN S, LI W, et al. Poly(mono/diethylene glycol n-tetradecyl ether vinyl ether)s with various molecular weights as phase change materials[J]. Polymers,2018,10(2):197-215. doi: 10.3390/polym10020197 [7] BEHZADI S, FARID M M. Long term thermal stability of organic PCMs[J]. Applied Energy,2014,; 122:11-16. [8] Li W, GENG X Y, HUANG R, et al. Microencapsulated comb-like polymeric solid-solid phase change materials via in-situ polymerization[J]. Polymers,2018,10(2):172-178. doi: 10.3390/polym10020172 [9] AMIN M, PUTRA N, KOSASIH E A, et al. Thermal properties of beeswax/graphene phase change material as energy storage for building applications[J]. Applied Thermal Engineering,2017,112:273-280. doi: 10.1016/j.applthermaleng.2016.10.085 [10] MEMON S A. Phase change materials integrated in building walls: A state of the art review[J]. Renewable & Sustainable Energy Reviews,2014,31:870-906. [11] 宋晓庆, 姜猛进, 叶光斗, 等. 石蜡/聚乙烯醇相变储能纤维的制备与表征[J]. 复合材料学报, 2008, 25(1):17-22. doi: 10.3321/j.issn:1000-3851.2008.01.003SONG X Q, JIANG M J, YE G D, et al. Preparation and characterization of paraffin/polyvinyl alcohol phase change fibers for energy storage[J]. Acta Materiae Compositae Sinica,2008,25(1):17-22(in Chinese). doi: 10.3321/j.issn:1000-3851.2008.01.003 [12] CHEN C, WANG L, HUANG Y. Electro spun phase change fibers based on polyethylene glycol/cellulose acetate blends[J]. Applied Energy,2011,88:3133-3139. doi: 10.1016/j.apenergy.2011.02.026 [13] CAO R R, LI X, CHEN S, et al. Fabrication and characterization of novel shape-stabilized synergistic phase change materials based on PHDA/GO composites[J]. Energy,2017,138:157-166. doi: 10.1016/j.energy.2017.07.049 [14] CAO R R, LIU H H, CHEN S, et al. Fabrication and properties of graphene oxide-grafted-poly(hexadecyl acrylate) as a solid-solid phase change material[J]. Composites Science and Technology,2017,149:262-268. doi: 10.1016/j.compscitech.2017.06.019 [15] O"LEARY K A, PAUL D R. Physical properties of poly(n-alkyl acrylate) copolymers. Part 2. Crystalline/non-crystalline combinations[J]. Polymer,2006,47:1245-1258. doi: 10.1016/j.polymer.2005.12.006 [16] 李树芹, 陈赛, 王学晨, 等. 梳状相变聚合物为芯材的复合调温纤维的制备及热性能[J]. 复合材料学报, 2018, 35(1):8-15.LI S Q, CHEN S, WANG X C, et al. Fabrication and properties of wet-spun thermo-regulating fibers with a novel comb-like phase change polymer core[J]. Acta Materiae Compositae Sinica,2018,35(1):8-15(in Chinese). [17] BANERJEE R, PHAN A, WANG B, et al. High-throughput synthesis of Zeolitic Imidazolate frameworks and application to CO2 capture[J]. Science,2008,319(5865):939-943. doi: 10.1126/science.1152516 [18] YUAN S, FENG L, WANG K, et al. Stable metal-organic frameworks: Design, synthesis, and applications[J]. Advanced Materials,2018,30(37):1704303-1704338. doi: 10.1002/adma.201704303 [19] BANGERJEE R, FURUKAWA H, BRITT D, et al. Control of pore size and functionality in isoreticular zeolitic imidazolate frameworks and their carbon dioxide selective capture properties[J]. Journal of the American Chemical Society,2009,131(11):3875-3877. doi: 10.1021/ja809459e [20] EDDAOUDI M, KIM J, ROSI N, et al. Systematic design of pore size and functionality in isoreticular MOFs and their application in methane storage[J]. Science,2002,295:469-472. doi: 10.1126/science.1067208 [21] YAGHI O M, O"KEEFFE M, OCKWING N W, et al. Reticular synthesis and the design of new materials[J]. Nature,2003,423(6941):705-714. doi: 10.1038/nature01650 [22] CHEN S, YU Y, CAO R R, et al. Fabrication and characterization of novel shape-stabilized phase change materials based on P(TDA-co-HDA)/GO composites[J]. Polymers,2019,11(7):1113-1125. doi: 10.3390/polym11071113 -





 下载:
下载: