Preparation of nano core-shell PS-CHO@RGO composite microspheres by in-situ polymerization as a potassium hydrogen persulfate catalytic activator for methylene blue degradation
-
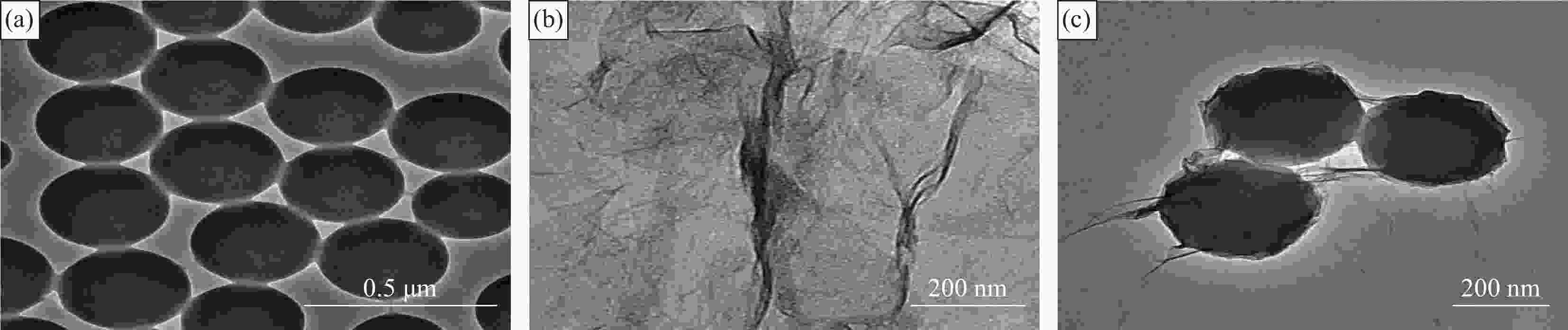
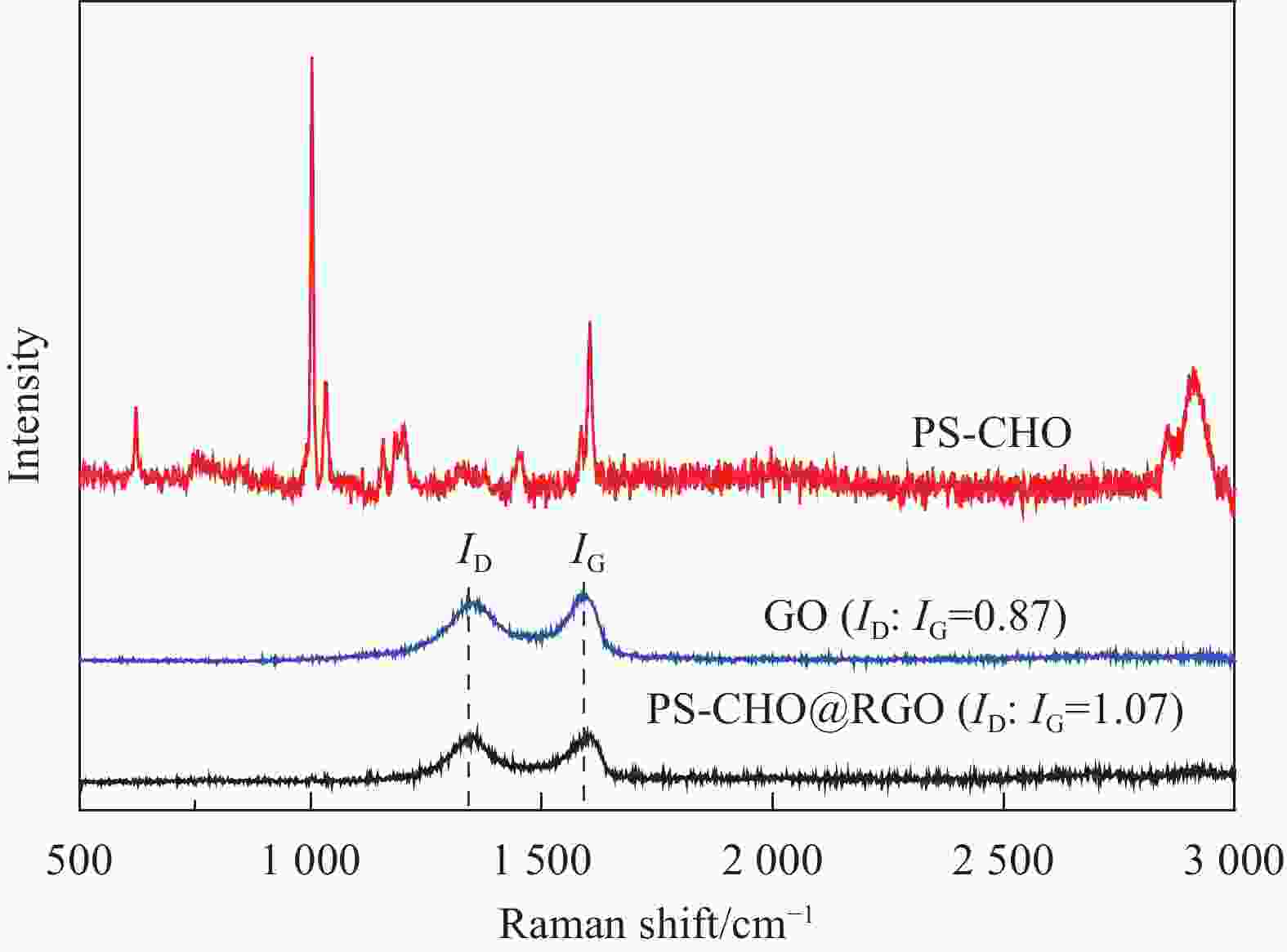
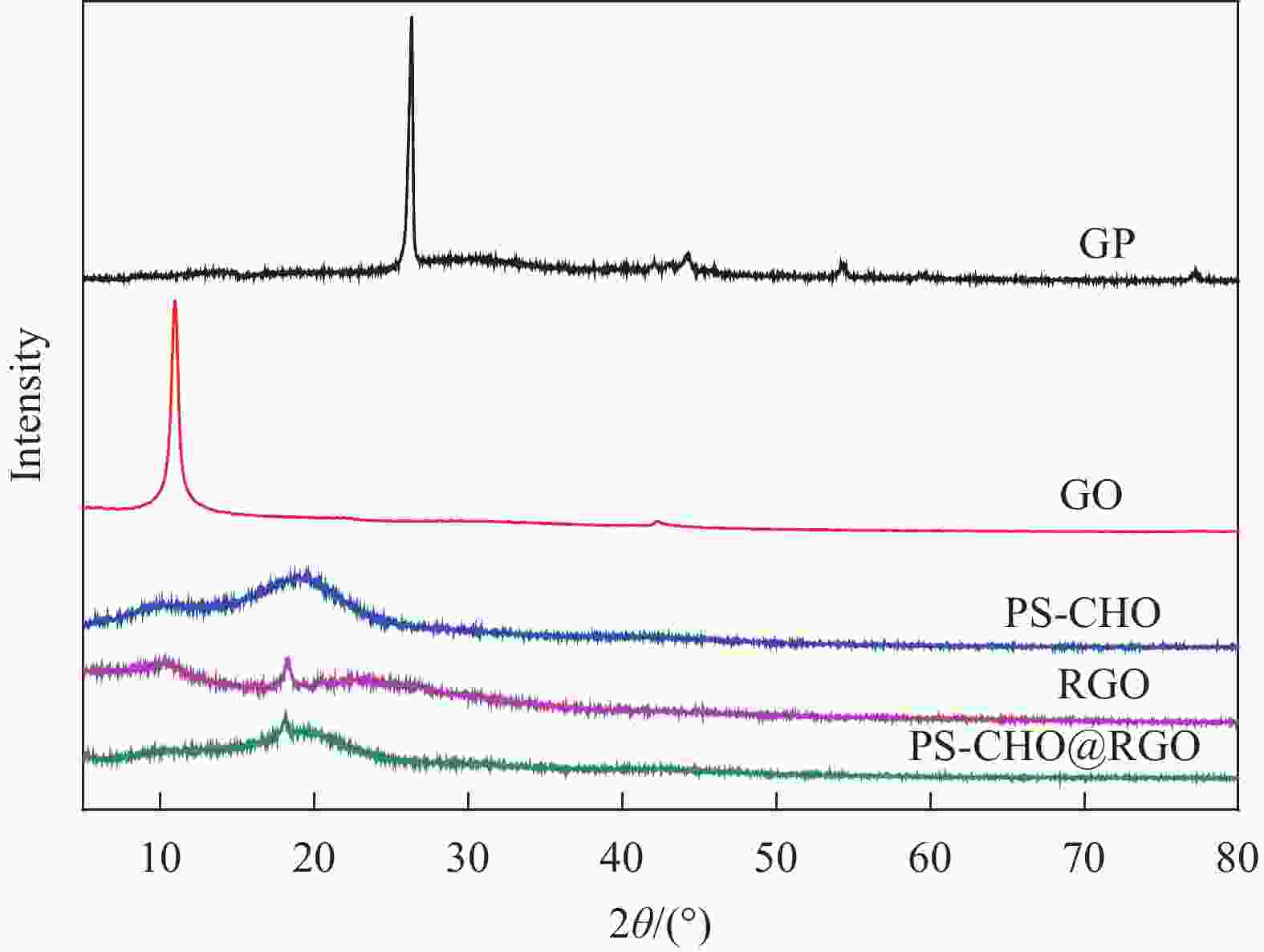
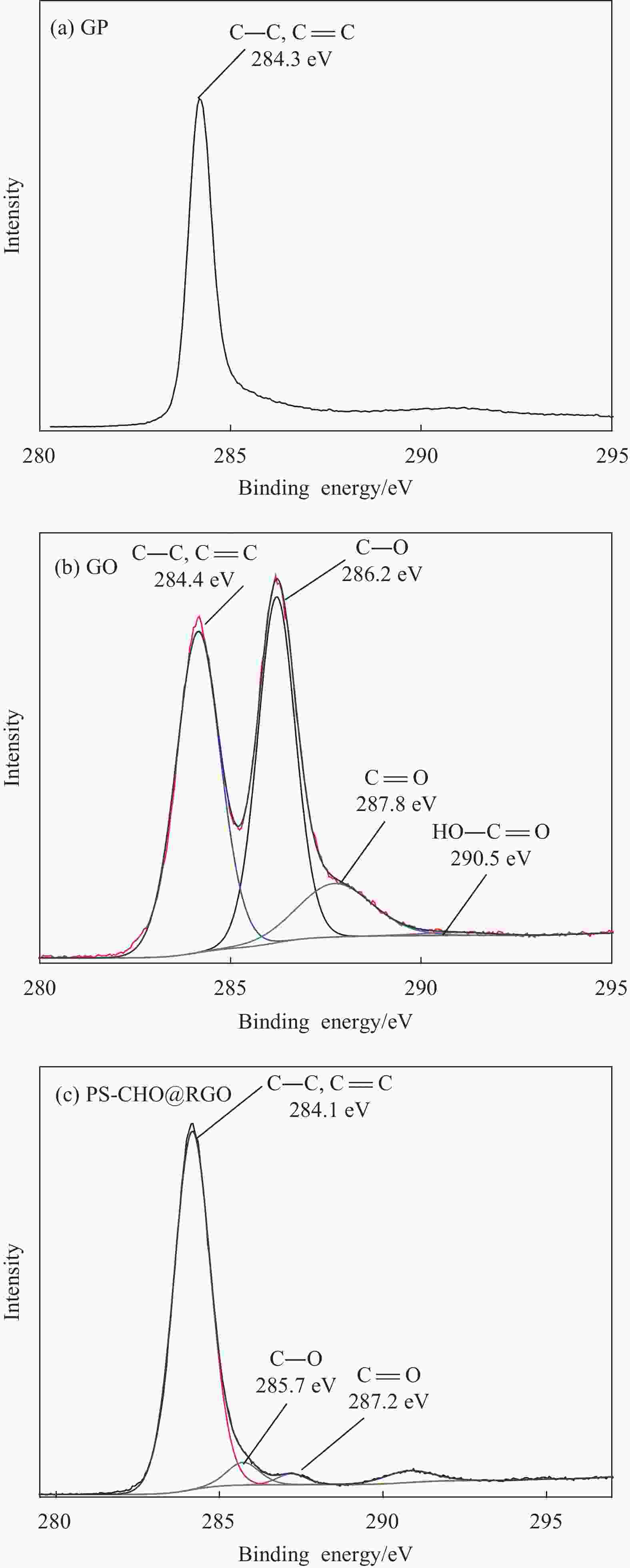
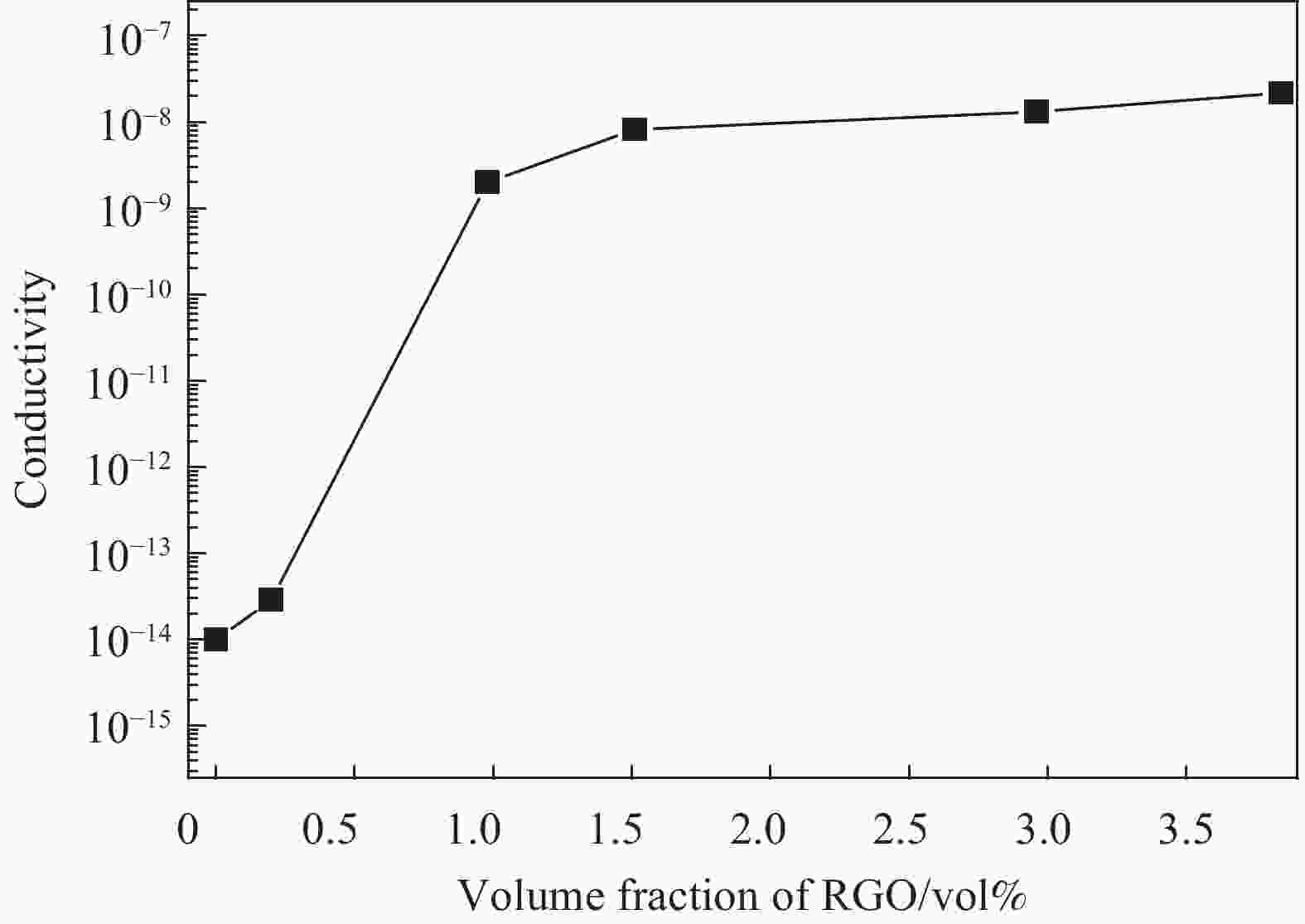
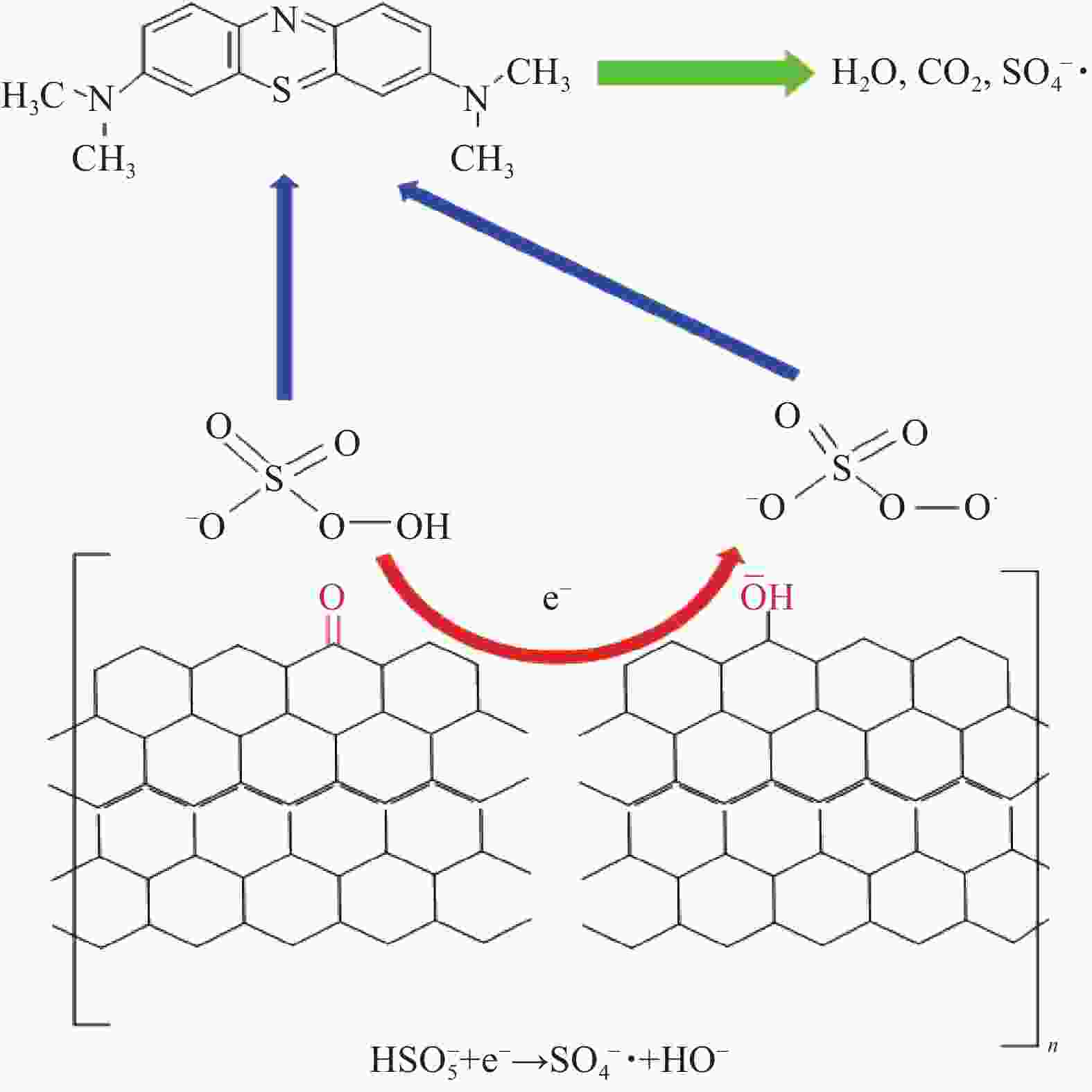
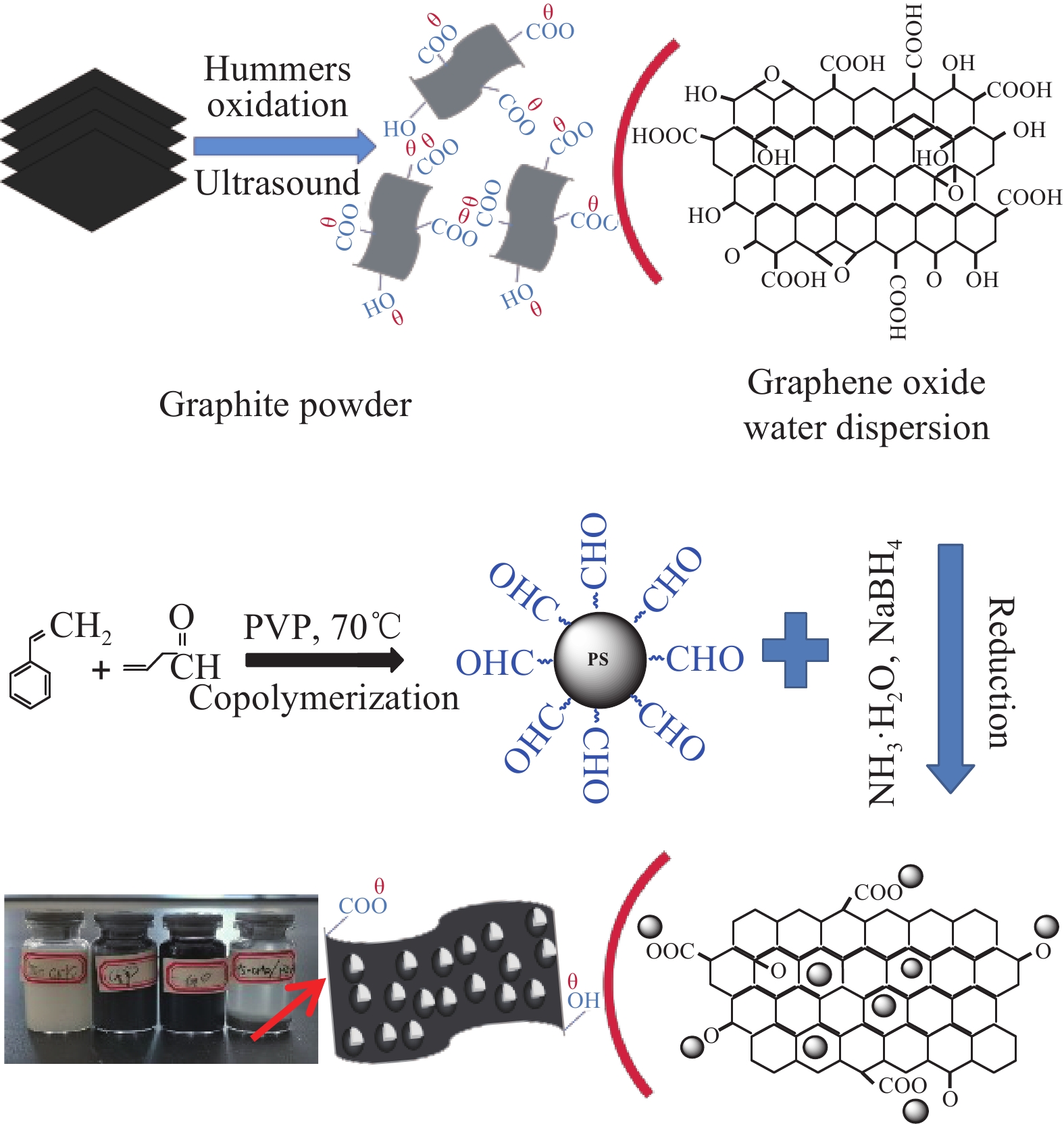
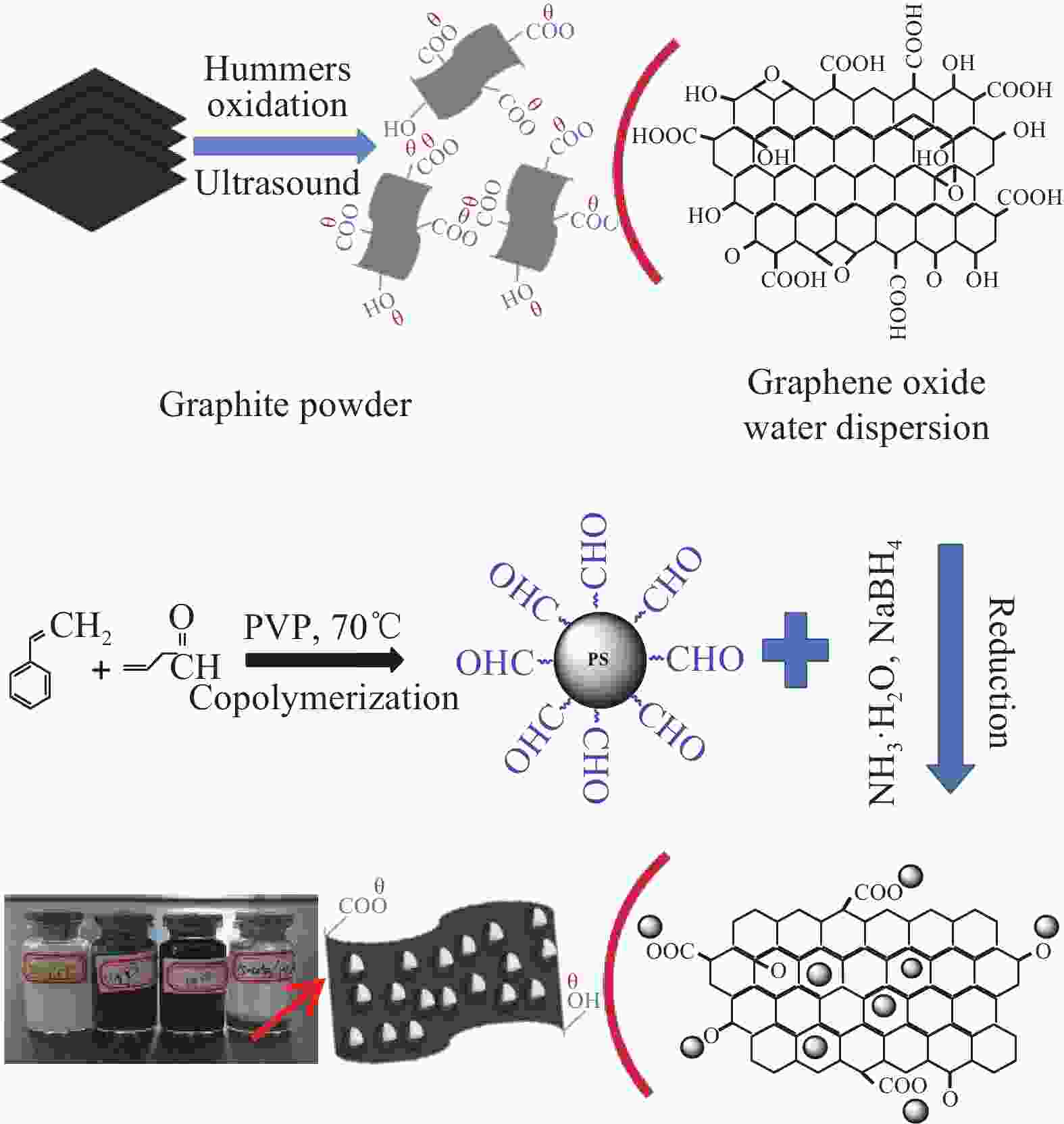
摘要: 还原氧化石墨烯(RGO)具有比表面积大、电子传输效率高、吸附速率快等优点,在处理油污、重金属离子、有机染料等领域均有应用,但由于自团聚而造成的分散性差等问题限制了其进一步应用。采用原位聚合法制备纳米核-壳型聚苯乙烯醛基微球(PS-CHO)@RGO复合微球。利用TEM、Raman、XRD、XPS及绝缘电阻测试仪对PS-CHO@RGO复合微球的形貌及理化性能进行表征。以亚甲基蓝(MB)为目标污染物,探究了PS-CHO@RGO复合微球在少量过硫酸氢钾(PMPS)存在下的氧化活性,并提出了降解机制。结果表明,RGO片层均匀包覆于PS-CHO微球表面,有效改善了分散性。制备所得PS-CHO@RGO复合微球的渗透阈值低,导电网络完善。降解实验中,PS-CHO@RGO复合微球可以激发PMPS生成硫酸根自由基(SO4−•),MB的氧化降解率显著提高,60 min内可达98%以上。PS-CHO@RGO复合微球同时表现出良好的稳定性,通过高速离心的方式实现循环利用。Abstract: The reduced graphene oxide (RGO) has been widely used in the treatment of oil, heavy metal ions, organic dyes and other fields due to the large specific surface area, high electron transport efficiency and fast adsorption rate. However, the poor dispersion caused by agglomeration limited the further research. In-situ polymerization was used to prepare nano core-shell polystyrene aldehyde microspheres (PS-CHO)@RGO composite microspheres. The morphology and physicochemical properties of the PS-CHO@RGO composite microspheres were characterized by TEM, Raman, XRD, XPS and insulation resistance tester. The methylene blue (MB) was selected as the target pollutant, the oxidation activity of PS-CHO@RGO composite microspheres in the presence of potassium hydrogen persulfate (PMPS) was investigated, and the degradation mechanism was proposed. The results indicate that RGO layer is uniformly coated on the surface of PS-CHO microspheres, which effectively improved the dispersion. The prepared PS-CHO@RGO composite microspheres are described as low penetration threshold and perfect conductive network. In the degradation experiment, PS-CHO@RGO composite microspheres can stimulate PMPS to generate sulfate radicals (SO4−•), over 98% of MB is catalytically degraded within 60 minutes. Meanwhile, the PS-CHO@RGO composite microspheres also show good stability and can be recycled by high-speed centrifugation.
-
Key words:
- composite microspheres /
- reduced graphene oxide (RGO) /
- core-shell type /
- methylene blue /
- catalytic /
- degradation
-
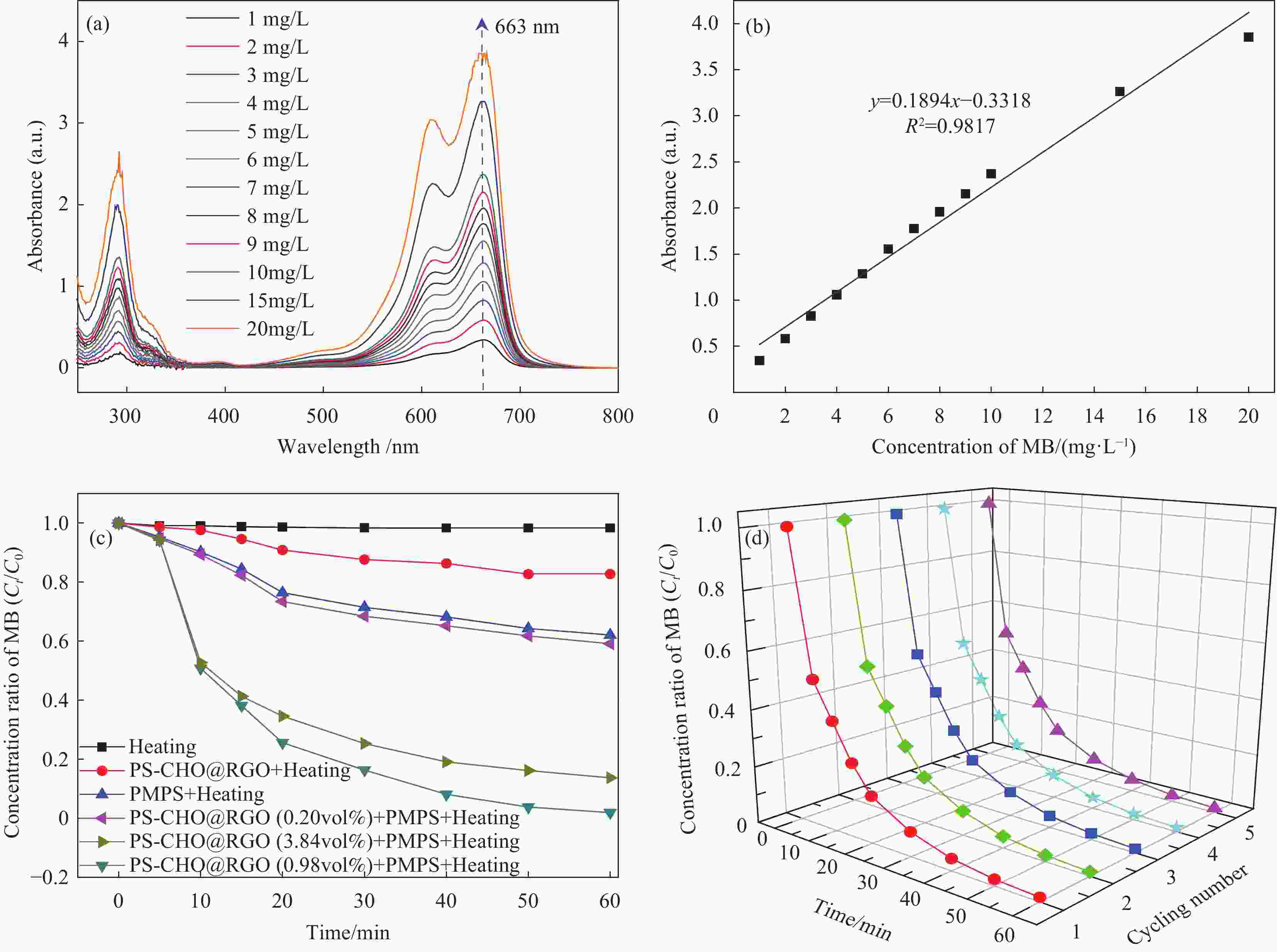
图 7 PS-CHO@RGO复合微球的催化活性: (a)不同浓度亚甲基蓝(MB)的吸光度; (b)吸光度与浓度线性方程;(c)不同反应条件下MB浓度的变化曲线; (d)循环催化曲线
Figure 7. Catalytic activities of PS-CHO@RGO composite microspheres: (a) Absorbance of different concentrations of methylene blue (MB); (b) Linear equation of absorbance and concentration of MB; (c) Concentration rate curves of MB under different reaction conditions; (d) Cyclic catalysis plots
PMPS—Potassium hydrogen persulfate
表 1 GP、GO和PS-CHO@RGO复合微球的化学元素含量
Table 1. Chemical element contents of GP, GO and PS-CHO@RGO composite microspheres
Sample C/at% O/at% C/O ratio GP 98.05 1.03 93.38 GO 60.23 39.67 1.51 PS-CHO@RGO 90.34 9.66 9.35 -
[1] WU J M, JING G J, LU X L, et al. The effect of sulfonated graphene on the rheological properties of cement paste[J]. Journal of Nanoscience and Nanotechnology,2020,20(12):7495-7505. doi: 10.1166/jnn.2020.18871 [2] LI D, KANER R B. Graphene-based materials[J]. Nature Nanotechnology,2008,3:101-105. doi: 10.1038/nnano.2007.451 [3] WEI X, MENG Z, RUIZ L, et al. Recoverable slippage mechanism in multilayer graphene leads to repeatable energy dissipation[J]. ACS Nano,2016,10(2):1820-1828. doi: 10.1021/acsnano.5b04939 [4] LI C, XUE Z, QIN J, et al. Synthesis of nickel hydroxide/delaminated-Ti3C2 MXene nanosheets as promising anode material for high performance lithium ion battery[J]. Journal of Alloys and Compounds,2020,842:155812. [5] ANSARI N, PAYAMI Z. Synthesis of magnetic graphene-Fe3O4 nanocomposites by electrochemical exfoliation method[J]. Journal of Nanostructures,2020,10(1):39-43. [6] CHI F, CHEN P, MAO C. Highly efficient photocatalytic disinfection of Escherichia coli by rose bengal-functionalized graphene oxide nanosheets[J]. Journal of Nanoscience and Nanotechnology,2020,20(12):7558-7568. doi: 10.1166/jnn.2020.18615 [7] RAMANATHAN T, ABDALA A A, STANKOVICH S, et al. Functionalized graphene sheets for polymer nanocomposites[J]. Nature Nanotechnology,2008,3:327-331. doi: 10.1038/nnano.2008.96 [8] SUN H, LIU S, ZHOU G, et al. Reduced graphene-oxide for catalytic oxidation of aqueous organic pollutants[J]. ACS Applied Materials & Interfaces,2012,4(10):5466-5471. [9] SHUKLA P, SUN H, WANG S, et al. Co-SBA-15 for heterogeneous oxidation of phenol with sulfate radical for wastewater treatment[J]. Catalysis Today,2011,175(1):380-385. doi: 10.1016/j.cattod.2011.03.005 [10] JAYANTHI S, LAVANYA T, DUTTA M, et al. Fabrication and characterization of graphene nanofibers by electrospinning technique and its electrochemical properties[J]. Journal of Nanoscience and Nanotechnology,2020,20(12):7659-7664. doi: 10.1166/jnn.2020.18625 [11] ANIPSITAKIS G P, STATHATOS E, DIONYSIOU D D. Heterogeneous activation of oxone using Co3O4[J]. The Journal of Physical Chemistry B,2005,109(27):13052-13055. doi: 10.1021/jp052166y [12] LING S K, WANG S, PENG Y. Oxidative degradation of dyes in water using Co2+/H2O2 and Co2+/peroxymonosulfate[J]. Journal of Hazardous Materials,2010,178(1-3):385-389. doi: 10.1016/j.jhazmat.2010.01.091 [13] BAI F, YANG X L, ZHAO Y Z, et al. Synthesis of core-shell microspheres with active hydroxyl groups by two-stage precipitation polymerization[J]. Polymer International, 2005, 54(1): 1 68-174. [14] OMI S, SAITO M, HASHIMOTO T, et al. Preparation of monodisperse polystyrene spheres incorporating polyimide prepolymer by dispersion polymerization in the presence of L-ascorbic acid[J]. Journal of Applied Polymer Science,1998,68(6):897-907. doi: 10.1002/(SICI)1097-4628(19980509)68:6<897::AID-APP4>3.0.CO;2-C [15] RIAHI K Z, SDIRI N, ENNIGROU D J, et al. Investigations on electrical conductivity and dielectric properties of graphene oxide nanosheets synthetized from modified Hummer’s method[J]. Journal of Molecular Structure,2020,1216:128304. [16] HOUAS A, LACHHEB H, KSIBI M, et al. Photocatalytic degradation pathway of methylene blue in water[J]. Applied Catalysis B: Environmental,2001,31(2):145-157. doi: 10.1016/S0926-3373(00)00276-9 [17] UMEBAYASHI T, YAMAKI T, TANAKA S, et al. Visible light-induced degradation of methylene blue on S-doped TiO2[J]. Chemistry Letters,2003,32(4):330-331. doi: 10.1246/cl.2003.330 [18] WU H, ZHAO W, HU H, et al. One-step in situ ball milling synthesis of polymer-functionalized graphene nanocomposites[J]. Journal of Materials Chemistry,2011,21(24):8626-8632. doi: 10.1039/c1jm10819k [19] LIU Y T, YANG J M, XIE X M, et al. Polystyrene-grafted graphene with improved solubility in organic solvents and its compatibility with polymers[J]. Materials Chemistry and Physics,2011,130(1-2):794-799. doi: 10.1016/j.matchemphys.2011.07.067 [20] FAN Z, WANG K, WEI T, et al. An environmentally friendly and efficient route for the reduction of graphene oxide by aluminum powder[J]. Carbon,2010,48(5):1686-1689. doi: 10.1016/j.carbon.2009.12.063 [21] ZHANG H, LIU S. Electrochemical biosensors based on nitrogen-doped reduced graphene oxide for the simultaneous detection of ascorbic acid, dopamine and uric acid[J]. Journal of Alloys and Compounds,2020,842:155873. [22] KWON O S, PARK S J, HONG J Y, et al. Flexible FET-type VEGF aptasensor based on nitrogen-doped graphene converted from conducting polymer[J]. ACS Nano,2012,6(2):1486-1493. doi: 10.1021/nn204395n [23] QI X Y, YAN D, JIANG Z G, et al. Enhanced electrical conductivity in polystyrene nanocomposites at ultra-low graphene content[J]. ACS Applied Materials & Interfaces,2011,8(3):3130-3133. [24] LUO Y, ZHAO P, YANG Q, et al. Fabrication of conductive elastic nanocomposites via framing intact interconnected graphene networks[J]. Composites Science and Technology,2014,100:143-151. doi: 10.1016/j.compscitech.2014.05.037 [25] JU S A, KIM K, KIM J H, et al. Graphene-wrapped hybrid spheres of electrical conductivity[J]. ACS Applied Materials & Interfaces,2011,3(8):2904-2911. [26] KAVITHA K, URADE A R, KAUR G, et al. Low-temperature chemical vapor deposition growth of graphene layers on copper substrate using camphor precursor[J]. Journal of Nanoscience and Nanotechnology,2020,20(12):7698-7704. doi: 10.1166/jnn.2020.18862 -





 下载:
下载: