Synthesis of chitosan functionalized magnetic graphene oxide composite and adsorption on methyl orange
-
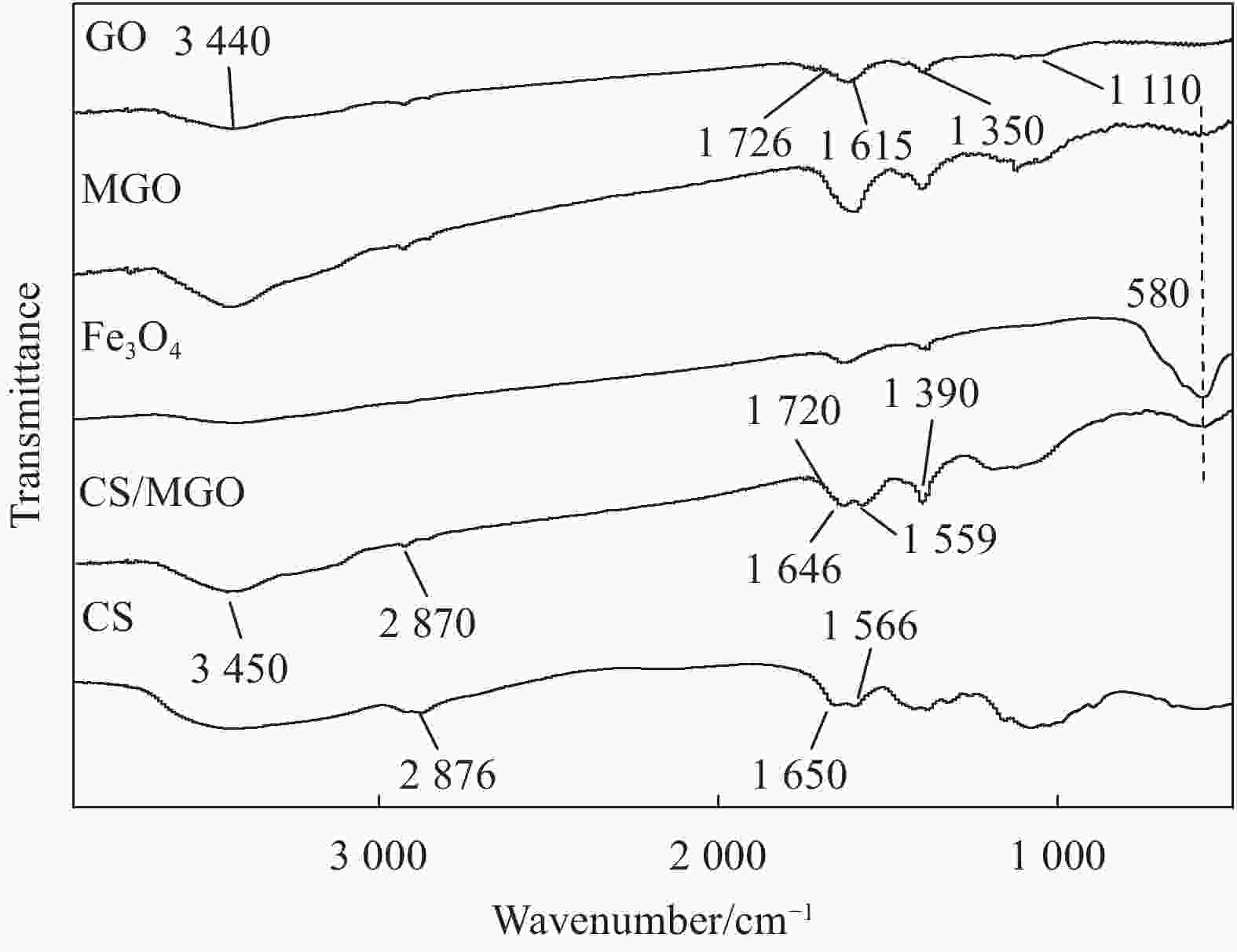
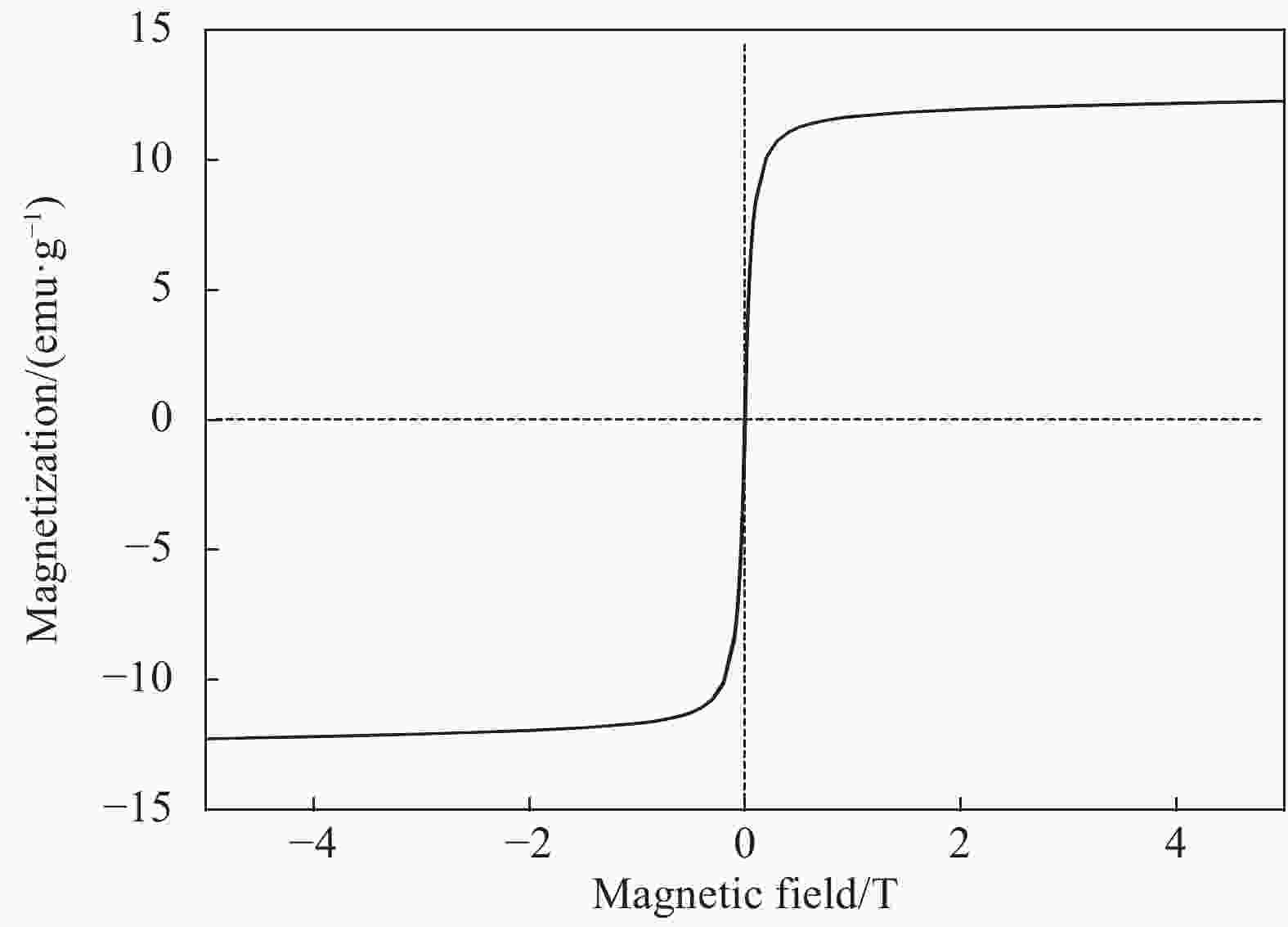
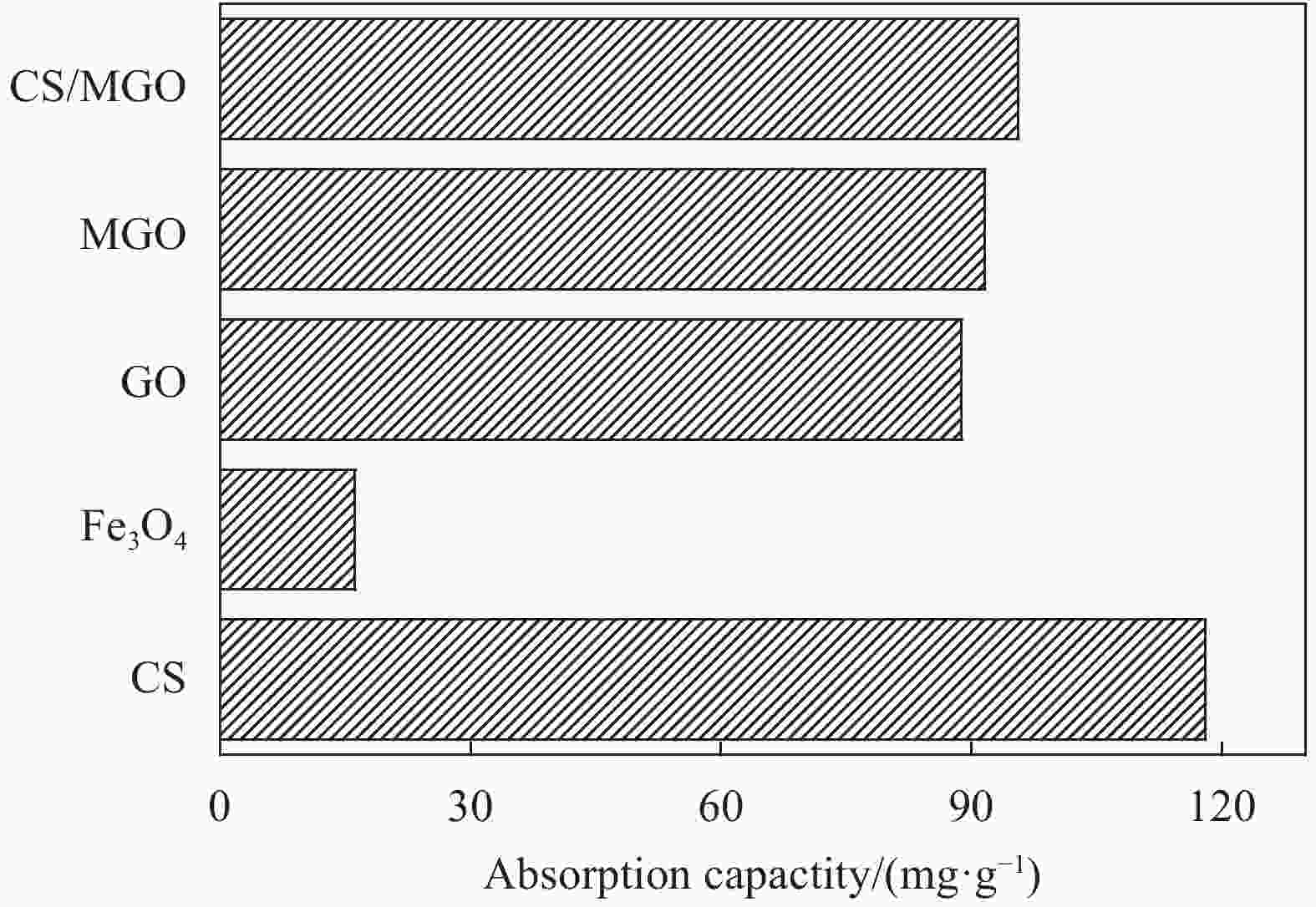
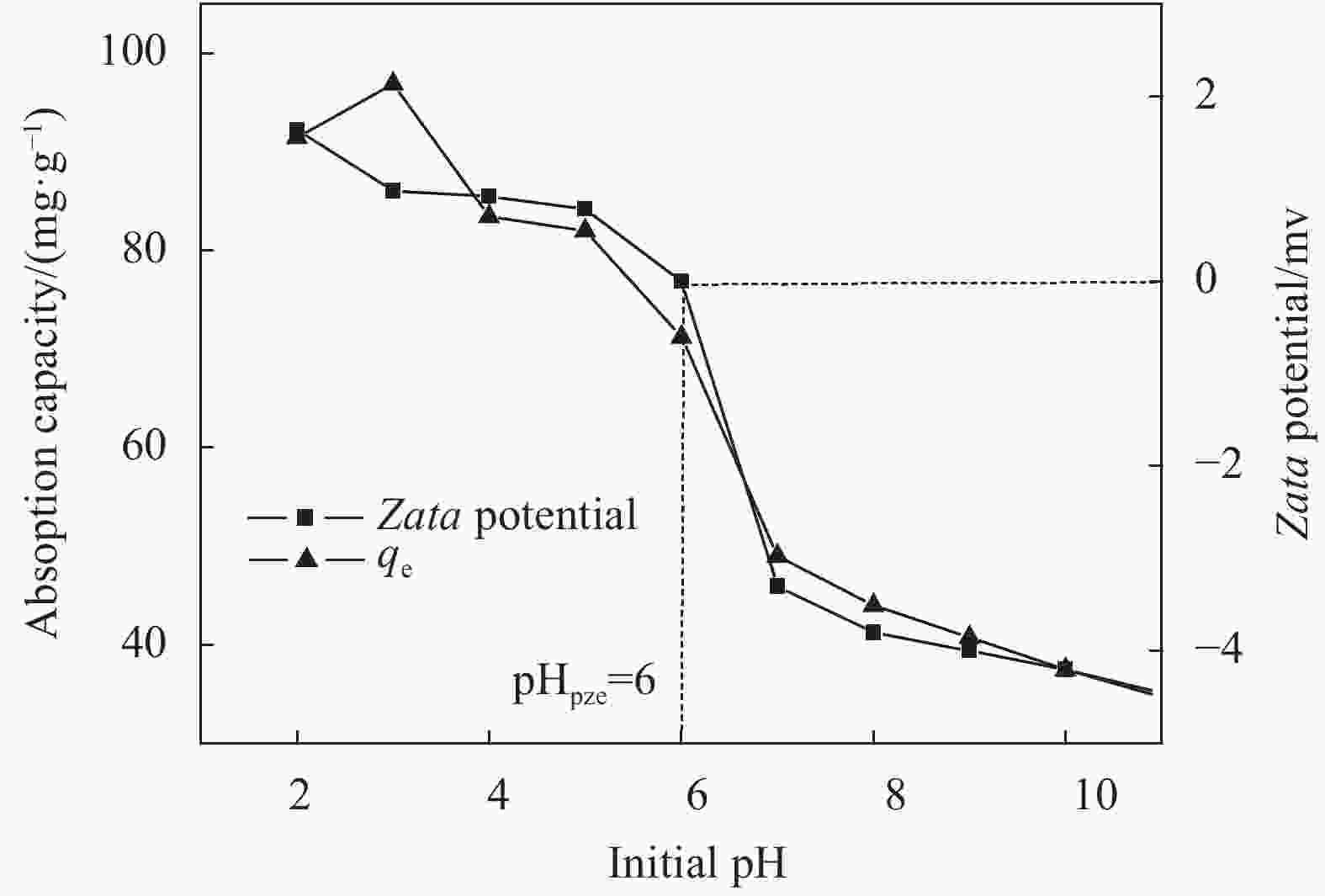
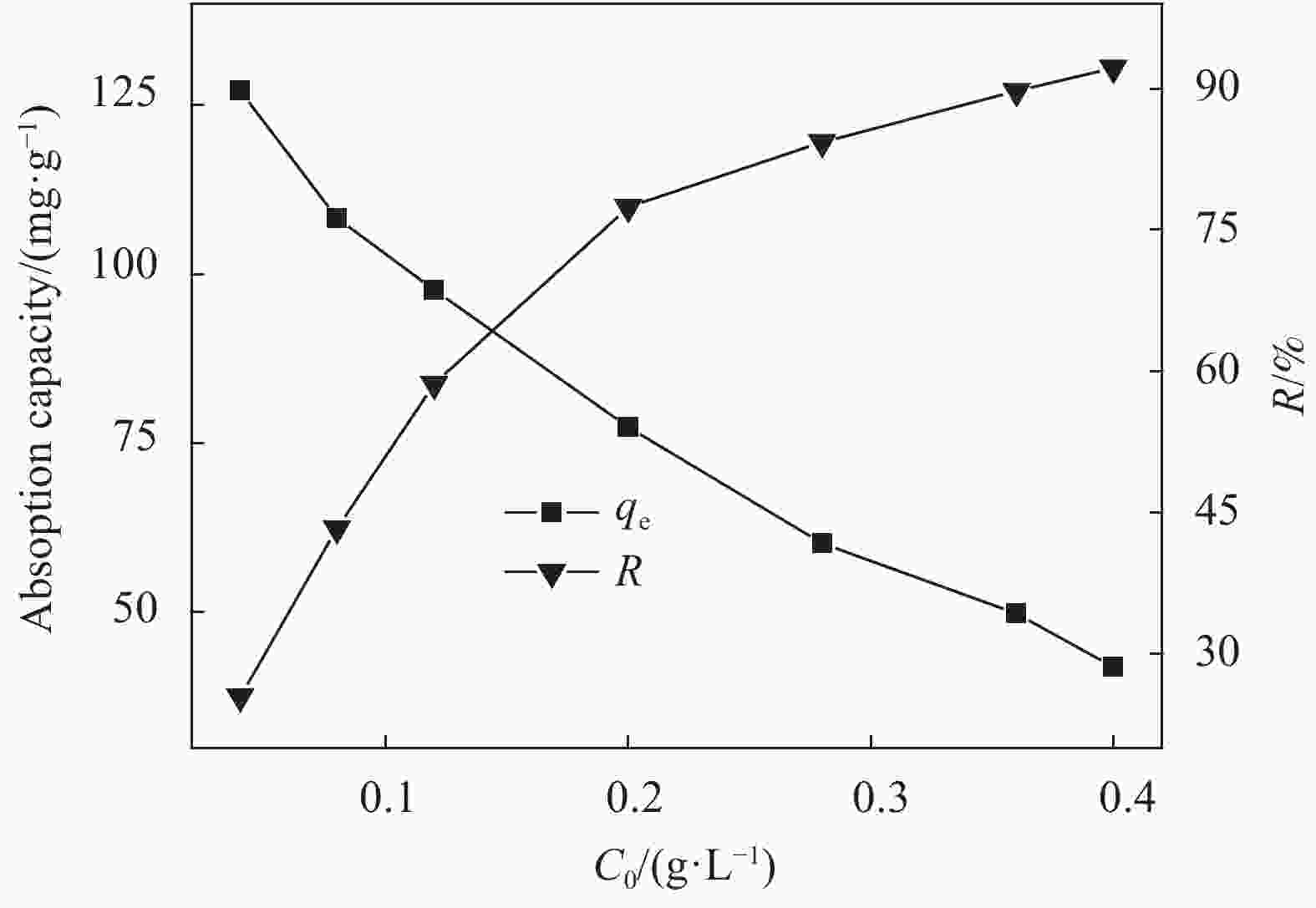
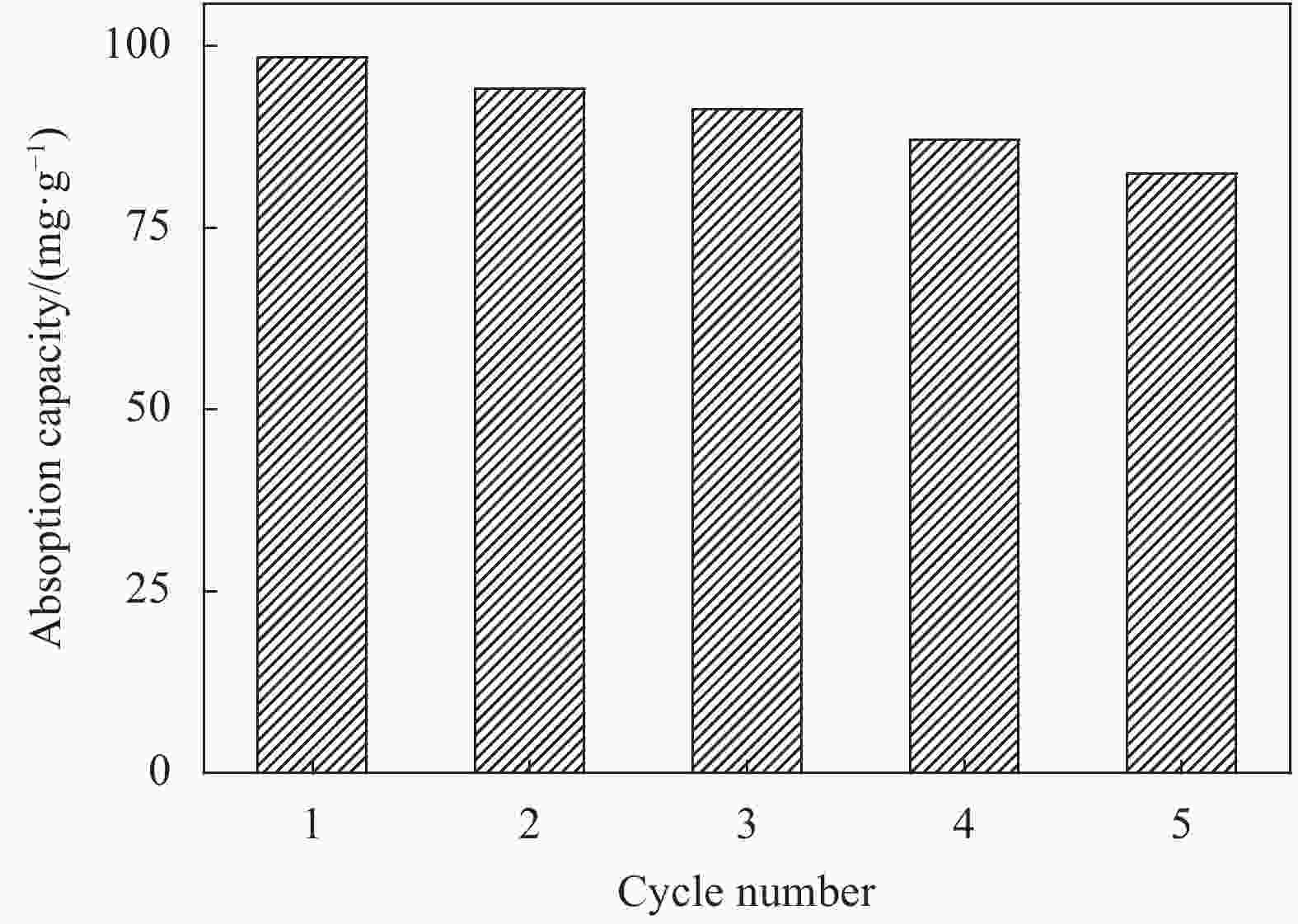
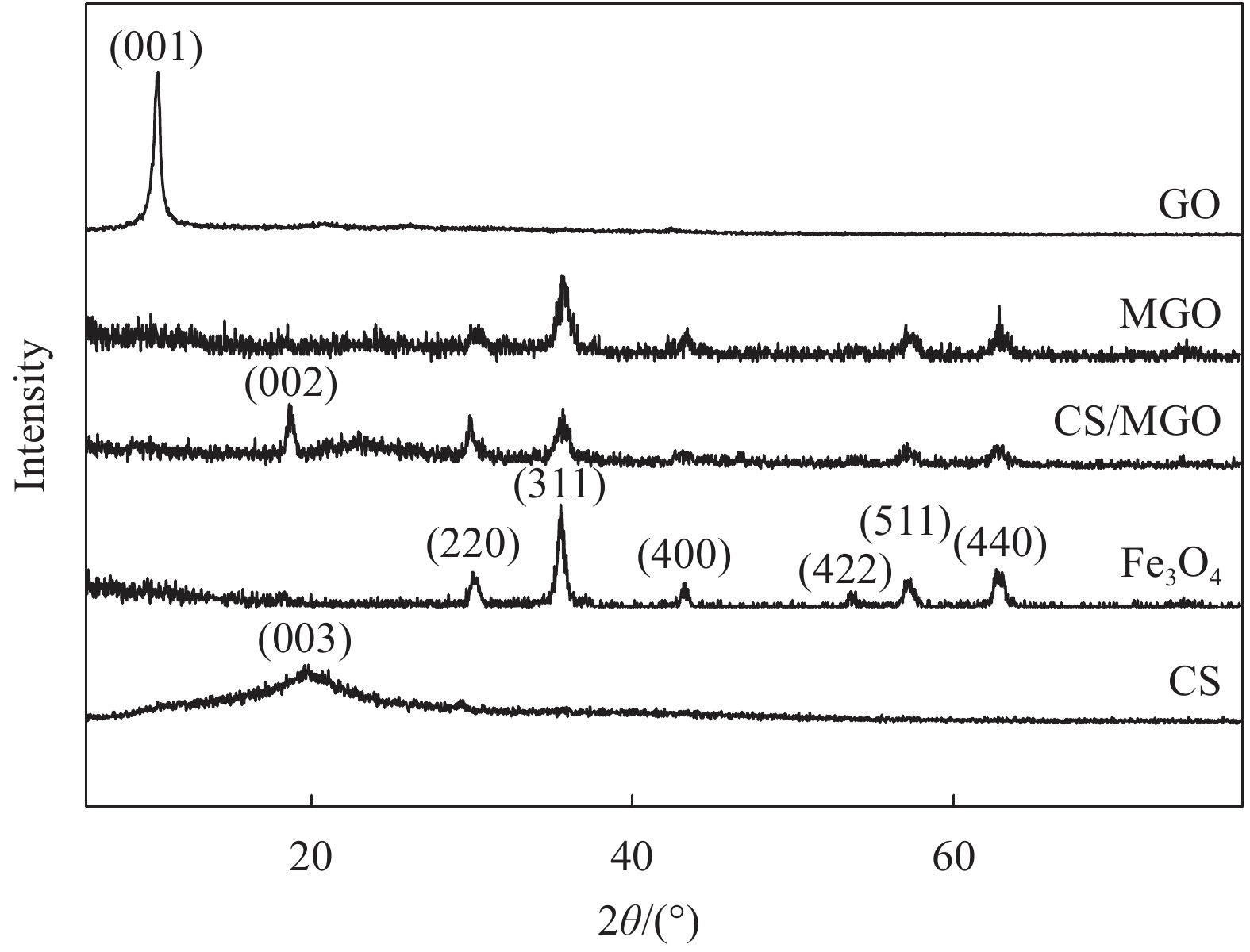
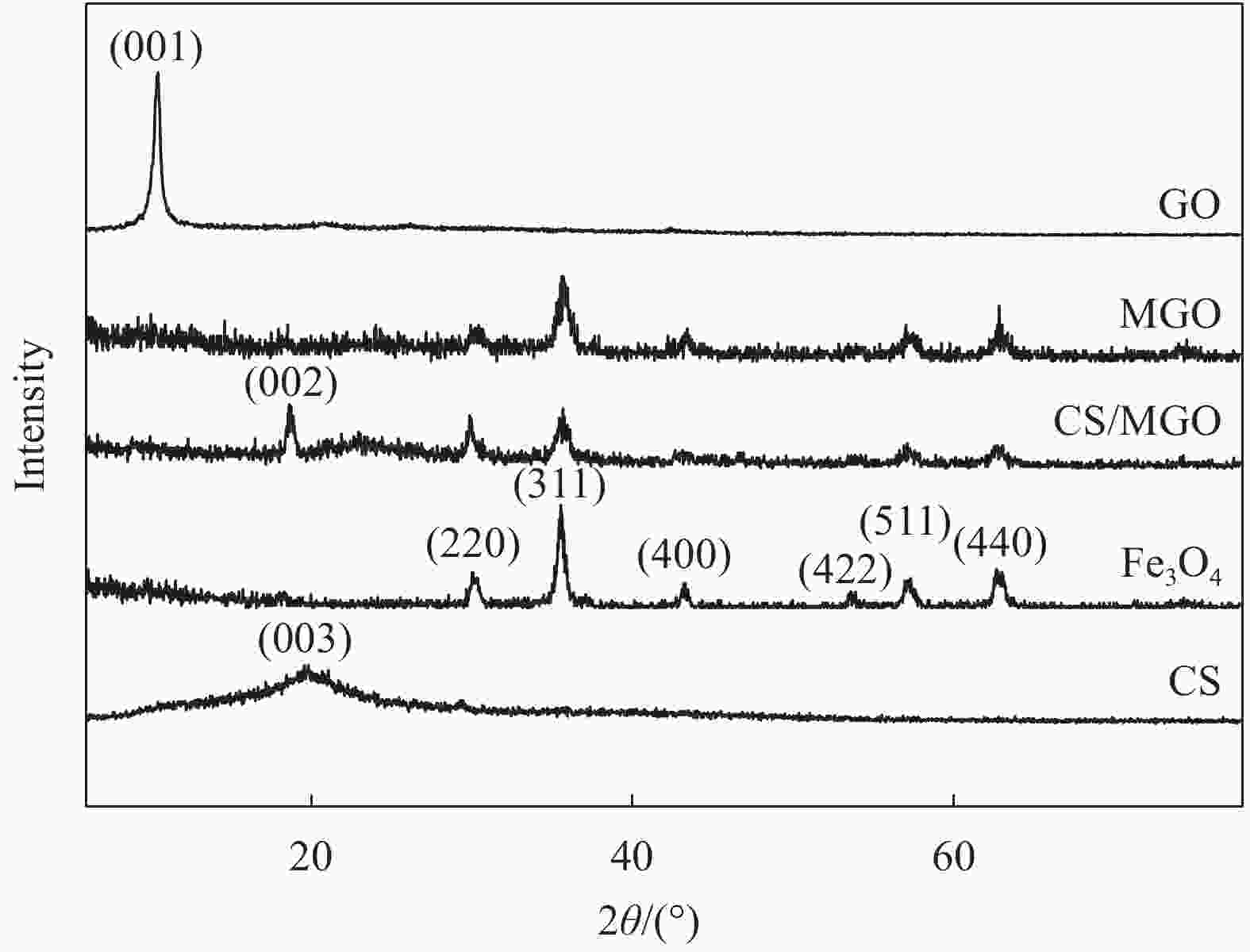
摘要: 用改进Hummers方法和水热法制备壳聚糖功能化磁性氧化石墨烯(CS/MGO)复合材料,通过SEM、FTIR、XRD、BET和振动样品磁强计(VSM)对材料结构和性能进行表征和测试,并对水中甲基橙(Methyl orange,MO)吸附研究。结果表明:氧化石墨烯(Graphene oxide,GO)与壳聚糖(Chitosan,CS)成功键合,热稳定性好,被壳聚糖修饰后比表面积为36.873 m2·g−1,磁性粒子均匀分布在GO表面,磁性响应明显。考察pH值、MO初始浓度、CS/MGO复合材料添加量及再生性能对MO去除率的影响,结果表明:在pH=3、MO初使浓度为20 mg·L−1、吸附材料为0.12 g·L−1时,210 min后达到吸附平衡,经5次循环后为初使吸附容量的83.7%。吸附过程符合拟二级动力学模型,吸附等温线符合Langmuir模型,在298.15、303.15、308.15 K温度下最大吸附量分别为129.96、138.94、145.03 mg·g−1;吸附热力学表明,吸附过程为吸热、熵增自发的吸附过程。Abstract: The chitosan/magnetic graphene oxide (CS/MGO) composites were synthesized by the modified Hummers and hydrothermal methods and applied as an adsorbent for the removal of methyl orange (MO). CS/MGO composite was characterized by SEM, XRD, BET, FTIR and a vibrating sample magnetometer (VSM). Results show that Fe3O4 nanoparticles mainly exist on the surface of graphene oxide and chitosan (CS) composite with less aggregation and a good magnetic response. In addition, the thermal stability is good, and the specific surface area of CS/MGO is 36.873 m2·g−1. CS/MGO composite could be easily separated by magnetic separation and demonstrates good stability and reusability. The effects of pH, initial concentration of MO, CS/MGO composite amount and regeneration on the removal of MO were systematically investigated. The results reveal that the initial MO concentration of 20 mg·L−1, CS/MGO composite amount of 0.12 g·L−1, and pH=3 lead to the adsorption equilibrium after 210 min. CS/MGO composite maintains 83.7% of its maximum MO adsorption capacity after five consecutive cycles. The adsorption process conforms to the pseudo-second-order kinetic model, and the adsorption isotherms conform to the Langmuir model. The maximum adsorption amounts at 298.15, 303.15 and 308.15 K are 129.96, 138.94 and 145.03 mg·g−1, respectively. The adsorption thermodynamics indicate that the adsorption process is endothermic; entropy increases the spontaneous adsorption process.
-
Key words:
- hydrothermal method /
- chitosan /
- grapheme oxide /
- composites /
- adsorption /
- methyl orange
-
表 1 CS/MGO复合材料吸附MO动力学拟合参数(T=298.15 K)
Table 1. Kinetic parameters of adsorption of MO onto CS/MGO composite (T=298.15 K)
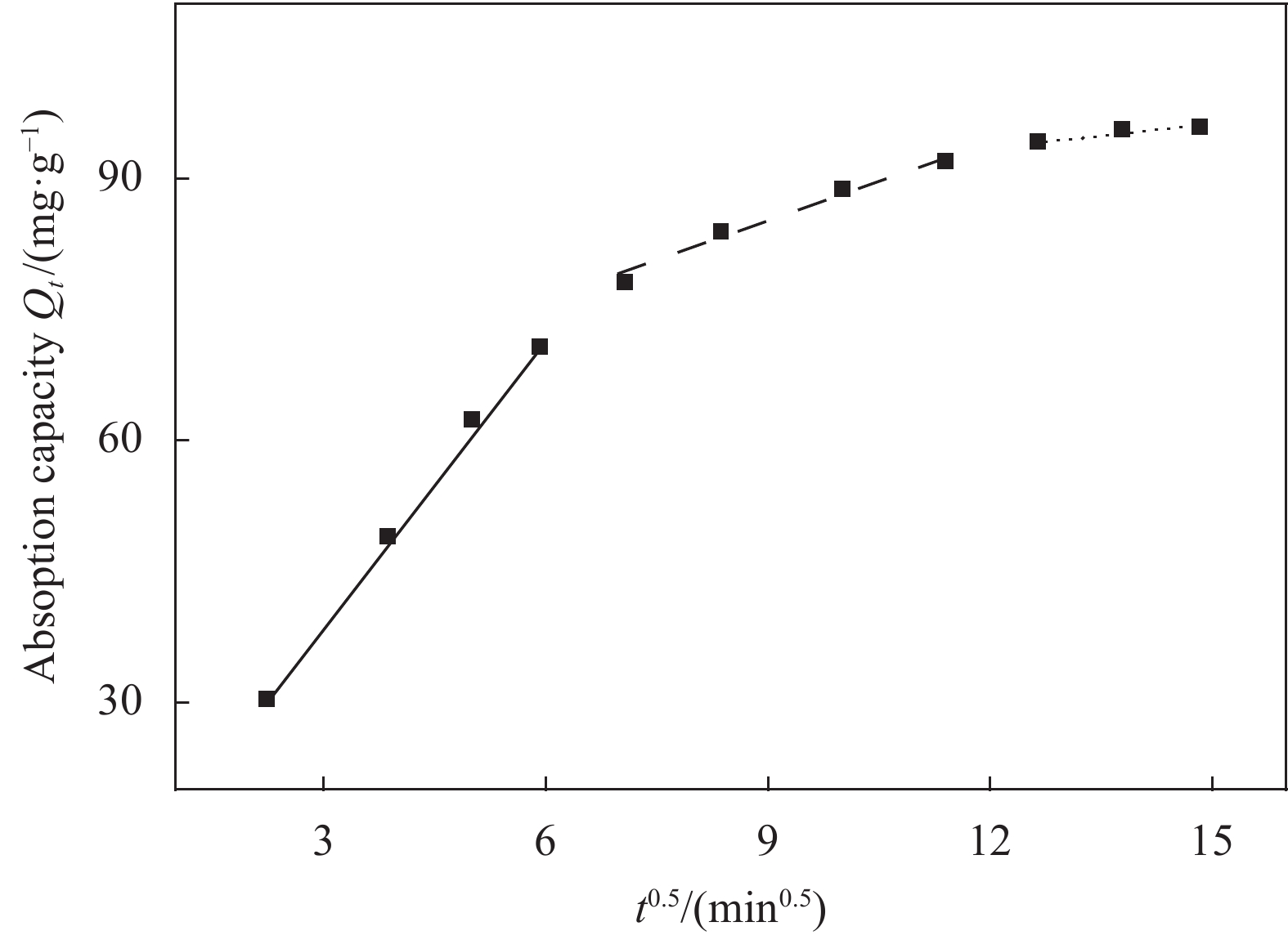
Model Parameter Value Pesudo-first-order Qe(cal)/(mg·g−1) 91.922 K1/10−2min−1 4.656 R2 0.9449 Pesudo-second-order Qe(cal)/(mg·g−1) 102.399 K2/(10−4g·mg−1·min−1) 6.437 R2 0.9921 Intra-particle diffusion Kp/(mg·g−1·min−0.5) 4.724 C 35.5936 R2 0.8409 Notes: K1, K2, Kp—Psudo-first-order kinetic constant and Psudo-second-order kinetic constant and Intra-particle diffusion rate constant, respectively; Qe(cal)—Calculation amount of MO removed per unit mass of adsorbent; C—Adsorption constant. 表 2 CS/MGO对MO吸附等温模型拟合结果
Table 2. Isotherm parameters for the adsorption of CS/MGO composite to MO
Temperature/K Langmuir Freundlich Qm/(mg·g−1) KL/(L·mg−1) R2 n/(mg·g−1) KF/(mg·g−1) R2 298.15 129.96 0.228 0.996 1.993 29.865 0.972 303.15 138.94 0.247 0.996 2.051 34.479 0.984 308.15 145.03 0.263 0.996 2.156 38.205 0.983 Notes: Qm—Langmuir adsorption maximum; KL—Langmuir coefficient of distribution of the adsorption; KF—Freundlich coefficient of distribution of the adsorption; n—Freundlich isotherm constant. 表 3 CS/MGO复合材料吸附MO热力学参数
Table 3. Thermodynamic parameters for the adsorption of MO onto CS/MGO composite
T/K ∆Gθ/(kJ·mol−1) ∆Hθ/(kJ·mol−1) ∆Sθ/(kJ·K−1·mol−1) 298.15 −7.92 16.60 82.34 303.15 −8.39 308.15 −8.74 Notes: ∆Gθ—Gibbs free energy variation of the adsorption process; ∆Hθ—Enthalpy change of the adsorption process; ∆Sθ—Entropy change of theadsorption process. -
[1] SUBBAIAHM V, KIM D S. Adsorption of methyl orange from aqueous solution by aminated pumpkin seed powder: Kinetics, isotherms, and thermodynamic studies[J]. Ecotoxicology and Environmental Safety,2016,128:109-117. doi: 10.1016/j.ecoenv.2016.02.016 [2] FU L, BAI Y N, LU Y Z, et al. Degradation of organic pollutants by anaerobic methane-oxidizing microorganisms using methyl orange as example[J]. Journal of Hazardous Materials,2019,364:264-271. doi: 10.1016/j.jhazmat.2018.10.036 [3] GAO M, WANG Z, YANG C, et al. Novel magnetic graphene oxide decorated with persimmon tannins for efficient adsorption of malachite green from aqueous solutions[J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects,2019,566:48-57. [4] 毕玉玺, 凌辉, 唐振平, 等. 磁性介孔TiO2/氧化石墨烯复合材料的制备及其对U(Ⅵ)的吸附[J]. 复合材料学报, 2019, 36(9):2176-2186.BI Y X, LING H, TANG Z P, et al. Preparation of magnetic mesoporous TiO2/graphene oxide composites and their adsorption for U(Ⅵ)[J]. Acta Materiae Compositae Sinica,2019,36(9):2176-2186(in Chinese). [5] CHENG Z, LIAO J, HE B, et al. One-step fabrication of graphene oxide enhanced magnetic composite gel for highly efficient dye adsorption and catalysis[J]. ACS Sustainable Chemistry & Engineering,2015,3(7):1677-1685. [6] OTHMANN H, ALIAS N H, SHAHRUDDIN M. Z, et al. Adsorption kinetics of methylene blue dyes onto magnetic grapheneoxide[J]. Journal of Environmental Chemical Engineering,2018,6(2):2803-2811. doi: 10.1016/j.jece.2018.04.024 [7] LIU S, HUANG B, CHAI L, et al. Enhancement of As (V) ad-sorption from aqueous solution by a magnetic chitosan/biochar composite[J]. RSC Advances,2017,7(18):10891-10900. doi: 10.1039/C6RA27341F [8] LE T T N, LE V T, DAO M U, et al. Preparation of magnetic graphene oxide/chitosan composite beads for effective removal of heavy metals and dyes from aqueous solutions[J]. Chemical Engineering Communications,2019,206(10):1337-1352. doi: 10.1080/00986445.2018.1558215 [9] XU, L, HUANG Y A, ZHU Q J, et al. Chitosan in molecularly-imprinted polymers: Current and future prospects[J]. International Journal of Molecular Sciences,2015,16(8):18328-18347. doi: 10.3390/ijms160818328 [10] SUN L, YUAN Z, GONG W, et al. The mechanism study of trace Cr (VI) removal from water using Fe0nanorods modified with chitosan in porous anodic alumina[J]. Applied Surface Science,2015,328:606-613. doi: 10.1016/j.apsusc.2014.12.094 [11] 郜玉楠, 周历涛, 王静, 等. 壳聚糖/沸石分子筛复合吸附颗粒的制备与性能[J]. 复合材料学报, 2019, 36(3):701-707.GAO Y N, ZHOU L T, WANG J, et al. Preparation and performances of chitosan/zeolite molecular sieve composite adsorbed particles[J]. Acta Materiae Compositae Sinica,2019,36(3):701-707(in Chinese). [12] FAN L, LUO C, SUN M, et al. Preparation of novel magnetic chitosan/graphene oxide composite as effective adsorbents toward methylene blue[J]. Bioresource Technology,2012,114:703-706. doi: 10.1016/j.biortech.2012.02.067 [13] 丁杰, 沙焕伟, 赵双阳, 等. 磁性氧化石墨烯/壳聚糖制备及其对磺胺嘧啶吸附性能研究[J]. 环境科学学报, 2016, 36(10):3691-3700.DING J, SHA H W, ZHAO S Y, et al. Synthesis of graphene oxide/magnetite chitosan composite and adsorption performance for sulfadiazine[J]. Acta Scientiae Circumstantiae,2016,36(10):3691-3700(in Chinese). [14] 李林波, 马键. 氧化石墨烯/改性磁性壳聚糖复合材料对Cu2+的吸附研究[J]. 化工新型材料, 2019, 47(8):261-264.LI L B, MA J. Study on adsorption of Cu2+by GO/modified magnetic chitosan composite[J]. New Chemical Materials,2019,47(8):261-264(in Chinese). [15] MARCANO D C, KOSYNKIN D V, BERLIN J M, et al. Improved synthesis of graphene oxide[J]. ACS Nano,2010,4(8):4806-4814. doi: 10.1021/nn1006368 [16] CUI L M, WANG Y G, GAO L, et al. EDTA functionalized magnetic graphene oxide for removal of Pb(II), Hg(II) and Cu(II) in water treatment: Adsorption mechanism and separation property[J]. Chemical Engineering Journal,2015,281:1-10. doi: 10.1016/j.cej.2015.06.043 [17] DUANH, LI L, WANG X, et al. β-Cyclodextrin/chitosan-magnetic graphene oxide-surface molecularly imprinted polymer nanocomplex coupled with chemiluminescence biosensing of bovine serum albumin[J]. RSC Advances,2015,5(84):68397-68403. doi: 10.1039/C5RA11061K [18] SUTAR D S, NARAYANAM P K, SINGH G, et al. Spectroscopic studies of large sheets of graphene oxide and reduced graphene oxide monolayers prepared by Langmuir-Blodgett technique[J]. Thin Solid Films,2012,520(18):5991-5996. doi: 10.1016/j.tsf.2012.05.018 [19] 常会, 范文娟, 曾成华, 等. 氨基功能化磁性氧化石墨烯吸附亚甲基蓝的性能探讨[J]. 冶金分析, 2019, 39(8):52-60.CHANG H, FAN W J, ZENG C H, et al. Discussion on adsorption property of amino-functionalized magnetic graphene oxide to methylene blue[J]. Metallurgical Analysis,2019,39(8):52-60(in Chinese). [20] 龚新怀, 辛梅华, 李明春, 等. 磁性响应茶渣制备及其对水溶液中亚甲基蓝的吸附[J]. 化工进展, 2019, 38(2):1113-1121.GONG X H, XIN M H, LI M C, et al. Preparationof magnetically responsive tea waste andit's adsorption of methylene blue from aqueous[J]. Chemical Industry and Engineering Progress,2019,38(2):1113-1121(in Chinese). [21] ZHANG, Y, WEN, G, FAN, S, et al. Partially reduced and nitrogen-doped graphene oxides with phenylethylamine for high-performance supercapacitors[J]. Journal of Materials Science,2018,53(16):11715-11727. doi: 10.1007/s10853-018-2471-5 [22] 于长江, 王苗, 董心雨, 等. 海藻酸钙@Fe3O4/生物碳磁性复合材料的制备及其对Co(Ⅱ)的吸附性能和机制[J]. 复合材料学报, 2018, 35(6):1549-1557.YU C J, WANG M, DONG X Y, et al. Preparationand characterization of calcium alginate@Fe3O4/biochar magnetic microsphereand its adso[J]. Acta Materiae Compositae Sinica,2018,35(6):1549-1557(in Chinese). [23] 姚时, 张鸣帅, 李林璇, 等. 茶渣负载纳米四氧化三铁复合材料制备及其对亚甲基蓝的吸附机理[J]. 环境化学, 2018, 37(1):96-107. doi: 10.7524/j.issn.0254-6108.2017050401YAO S, ZHANG M S, LI L X, et al. Preparationof tea waste-nano Fe3O4 composite and its removal mechanism of methylene blue from aqueous solution[J]. Environmental Cheistry,2018,37(1):96-107(in Chinese). doi: 10.7524/j.issn.0254-6108.2017050401 [24] 朱脉勇, 陈齐, 童文杰, 等. 四氧化三铁纳米材料的制备与应用[J]. 化学进展, 2017, 29(11):1366-1394.ZHU M Y, CHEN Q, TONG W J, et al. Preparation and application of Fe3O4 nanomaterials[J]. Progress in Chemistry,2017,29(11):1366-1394(in Chinese). [25] 史月月, 单锐, 袁浩然. 改性稻壳生物炭对水溶液中甲基橙的吸附效果与机制[J]. 环境科学, 2019, 40(6):2783-2792.SHI Y Y, SHAN R, YUAN H R. Effect and mechanisms of methyl orange removal from aqueous solutions by modified riceshell biochar[J]. Environmental Science,2019,40(6):2783-2792(in Chinese). [26] 常春, 刘天琪, 王瑀婷, 等. 水热法制备玉米叶基生物炭对亚甲基蓝的吸附性能研究[J]. 环境科学学报, 2017, 37(7):2680-2690.CHANG C, LIU T Q, WANG Y T, etal. Hydrothermal preparation of maize leaf based biochar and its adsorption performance for methylene blue[J]. Acta Scientiae Circumstantiae,2017,37(7):2680-2690(in Chinese). [27] 吴艳, 罗汉金, 王侯. 改性木屑对水中刚果红的吸附性能研究[J]. 环境科学学报, 2014, 34(7):1680-1688.WU Y, LUO H J, WANG H. Adsorption properties of modified sawdust for conge red removal from wastewater[J]. Acta Scientiae Circumstantiae,2014,34(7):1680-1688(in Chinese). [28] 布林朝克, 郭婷, 张邦文, 等. 部分还原氧化石墨烯-Fe3O4对水中Mn(Ⅱ)的快速去除[J]. 高等学校化学学报, 2017, 38(2):217-224. doi: 10.7503/cjcu20160664BULIN C K, GUO T, ZHANG B W, et al. Fast removal of aqueous Mn(II) using partially reduced graphene oxide-Fe3O4[J]. Chemical Journal of Chinese Universities,2017,38(2):217-224(in Chinese). doi: 10.7503/cjcu20160664 -





 下载:
下载: