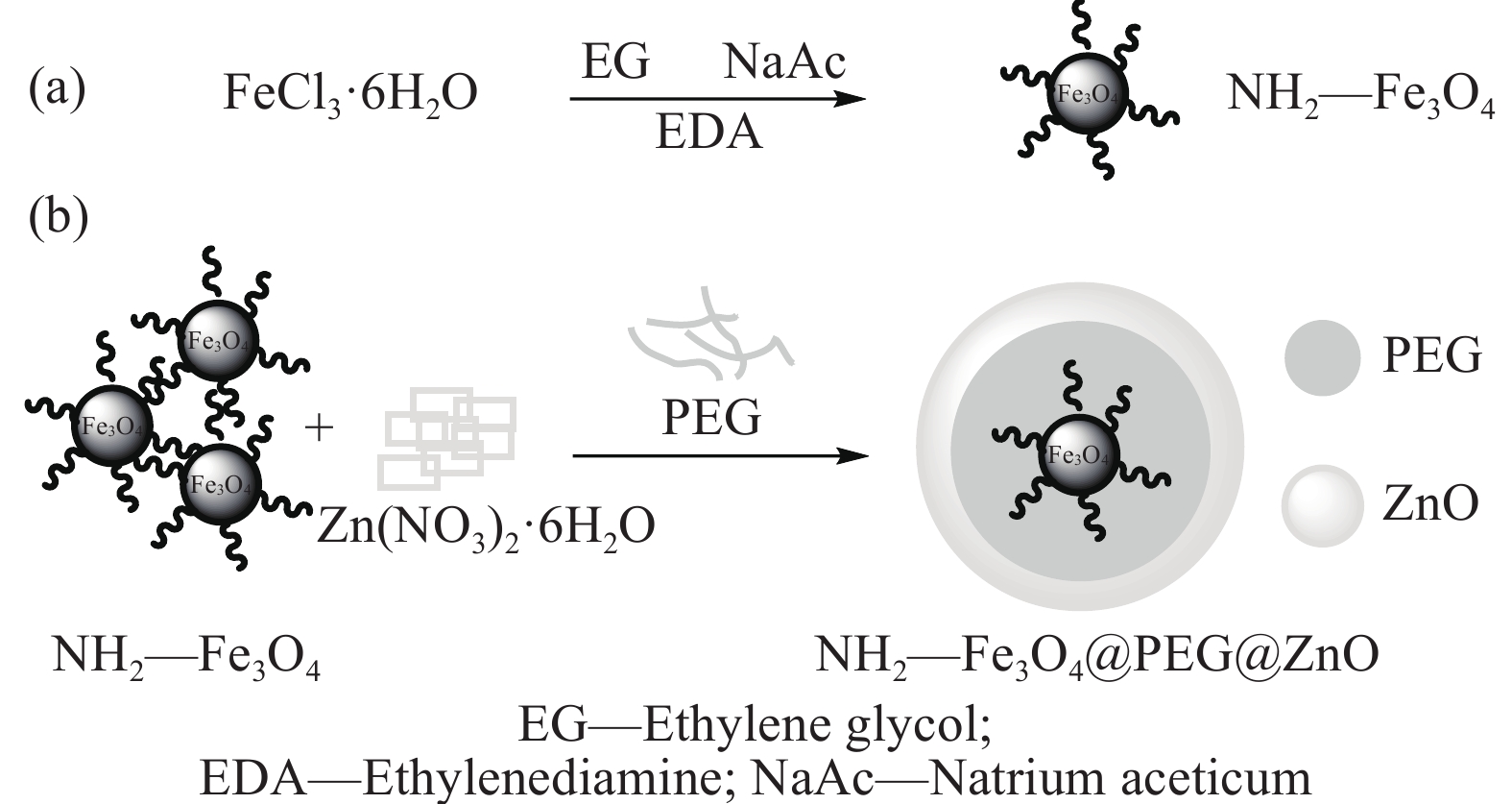
Preparation and photocatalytic properties of NH2—Fe3O4@polyethylene glycol@ZnO nanospheres
-
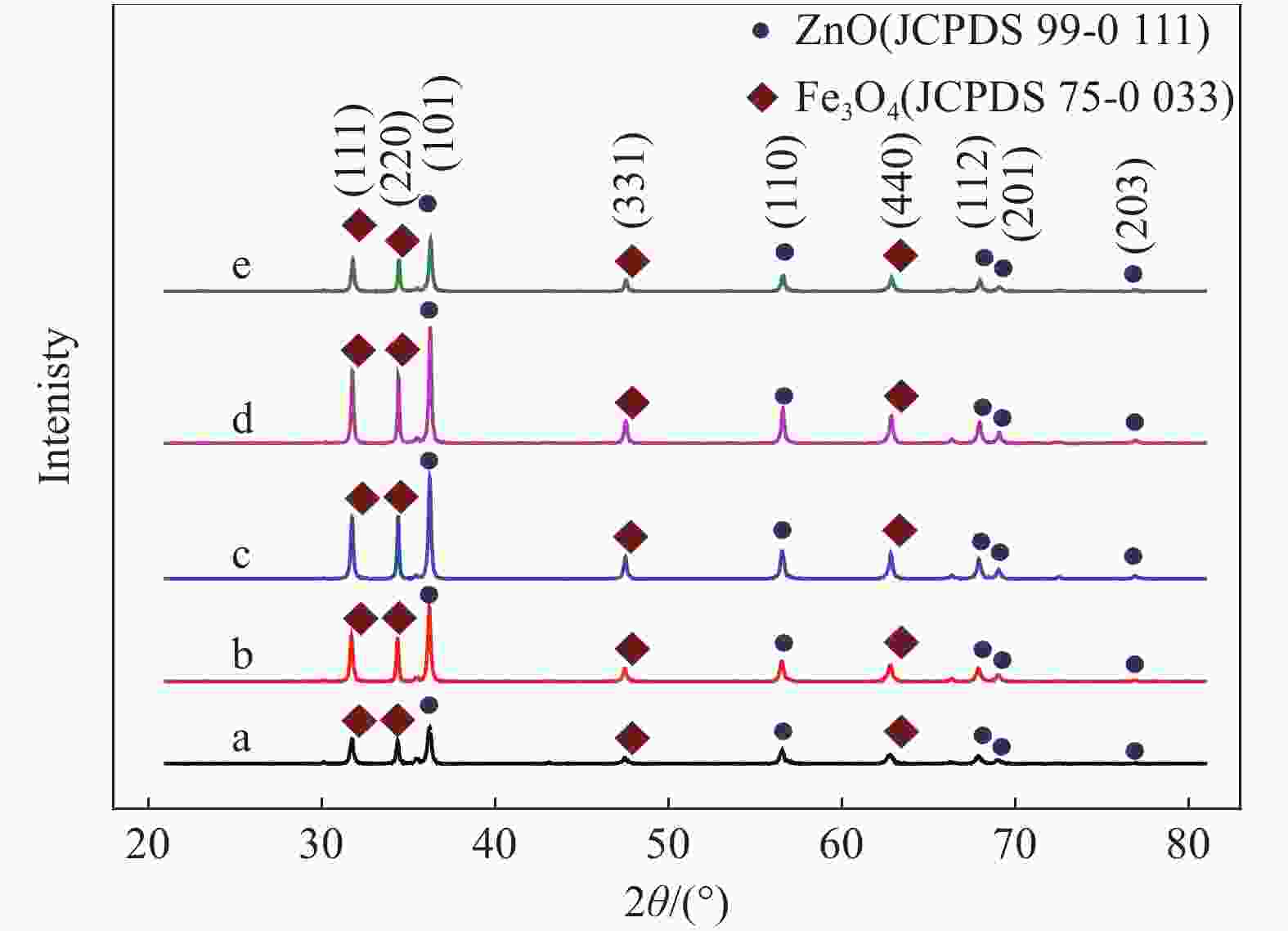
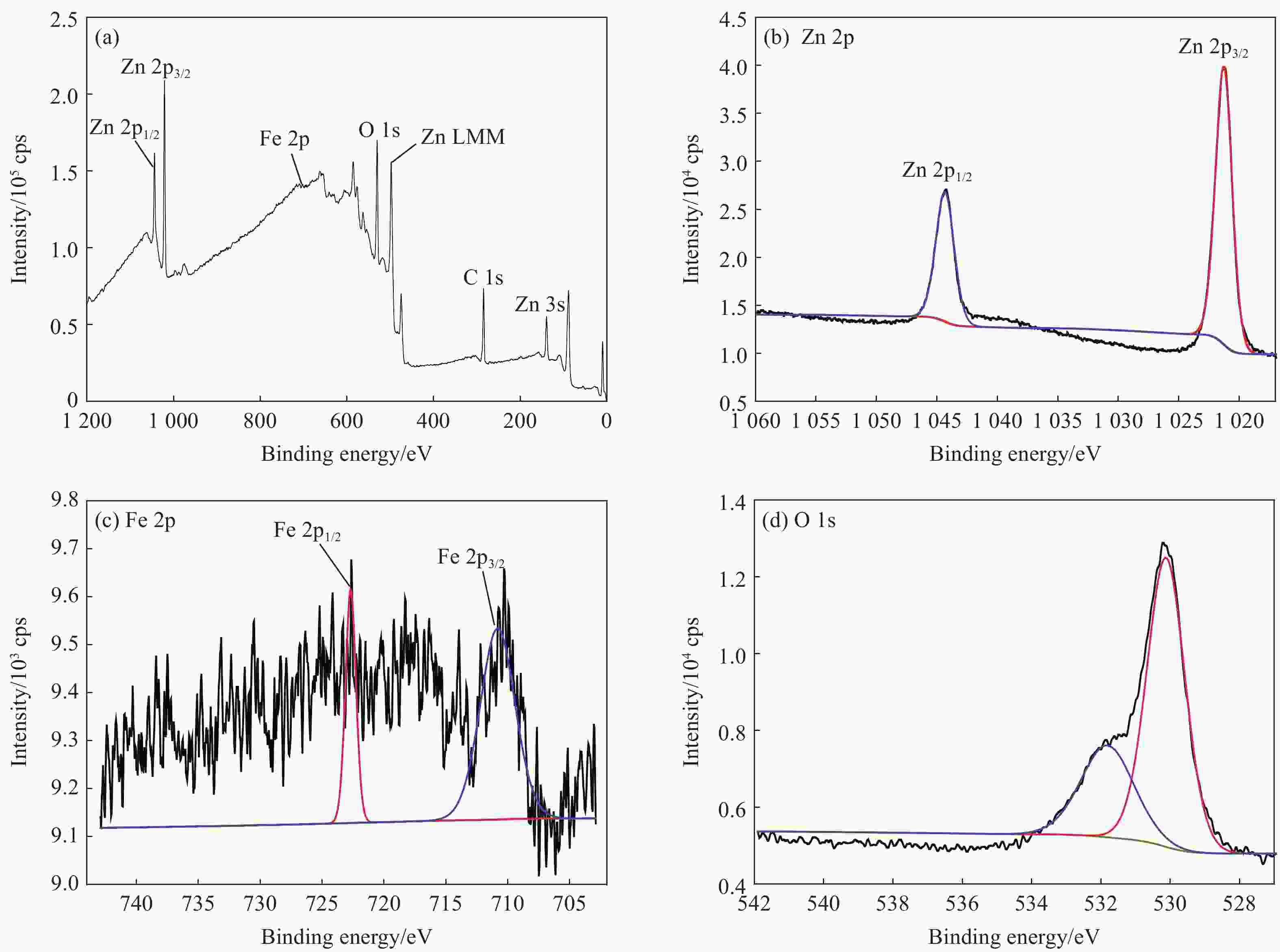
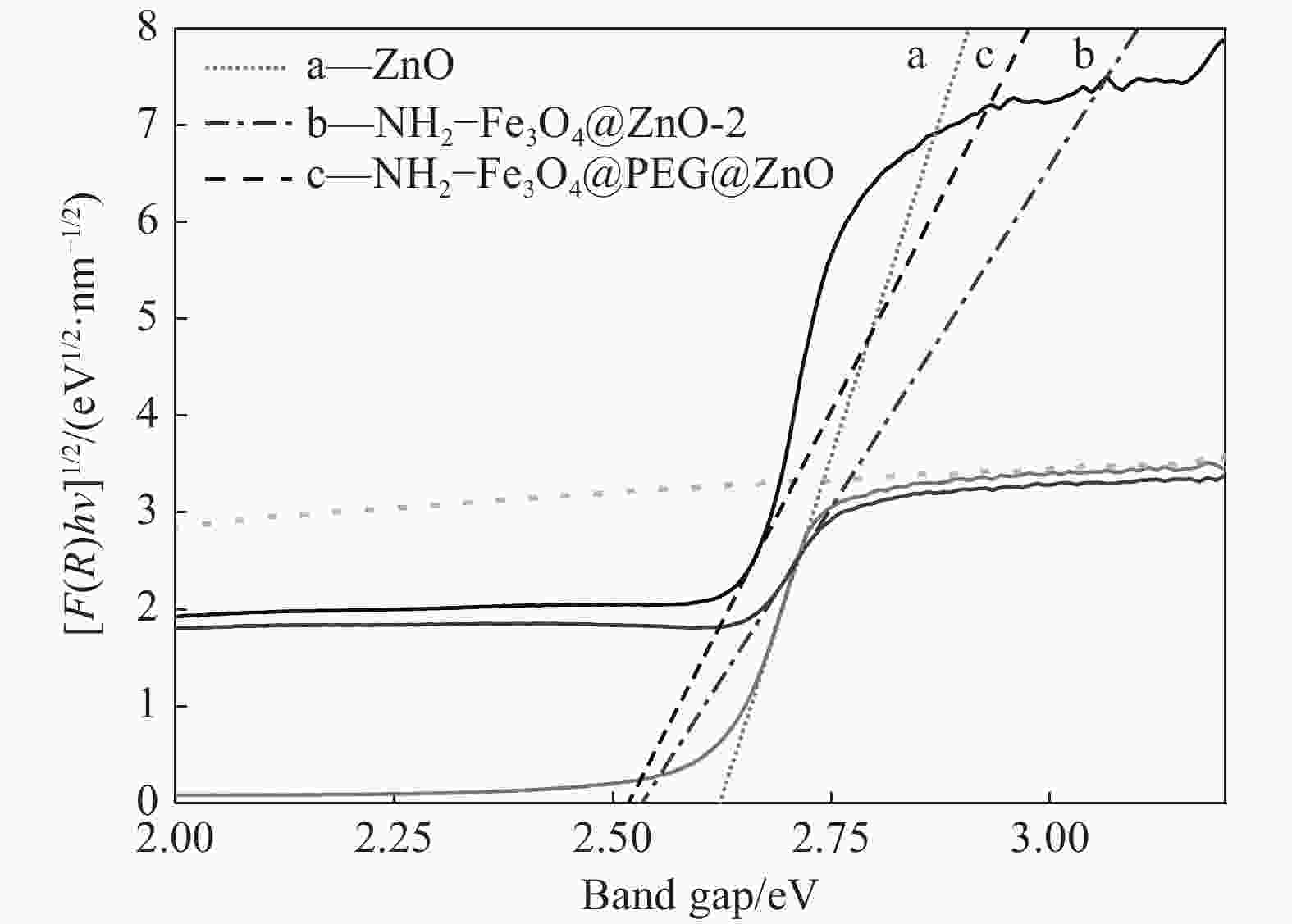
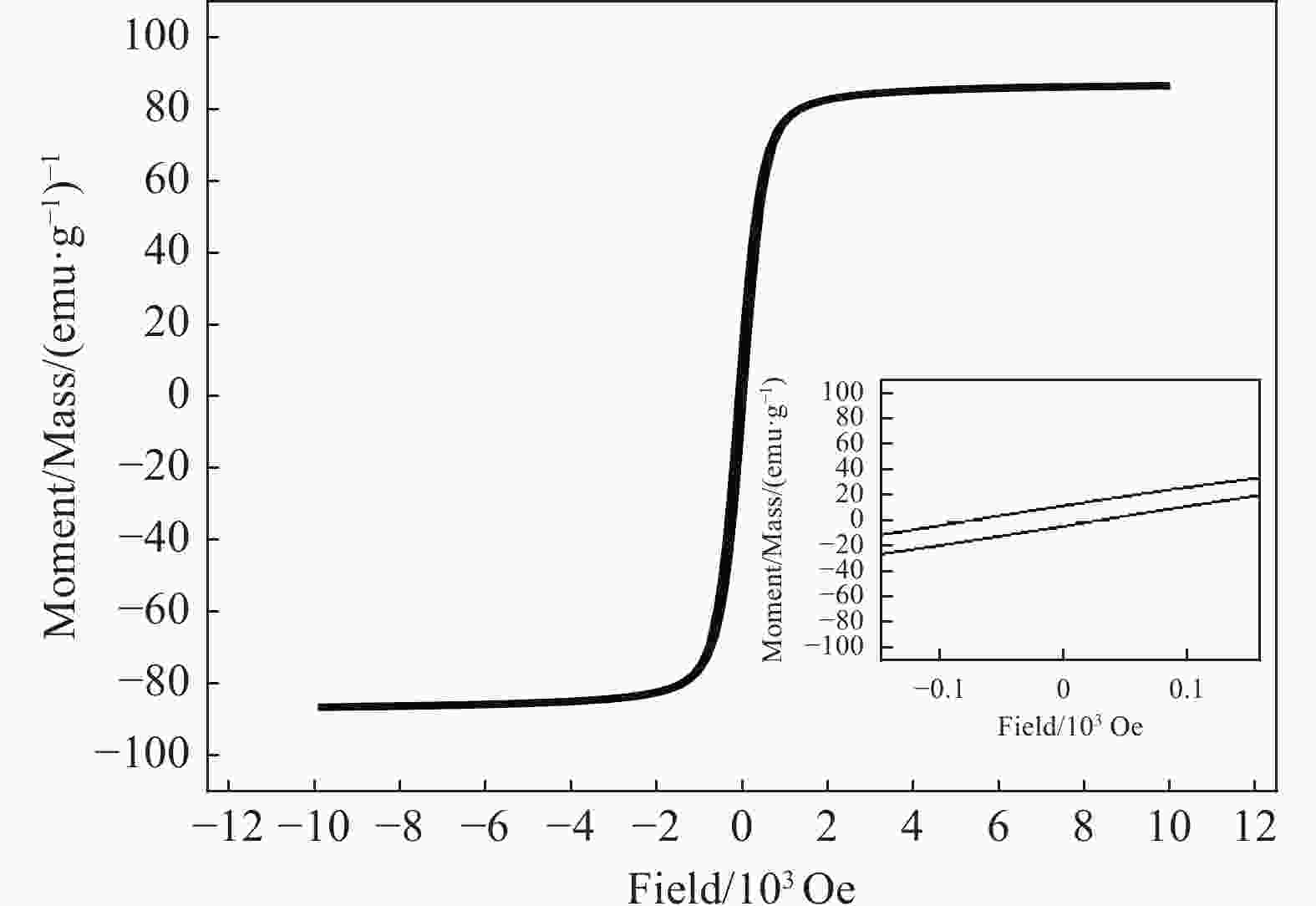
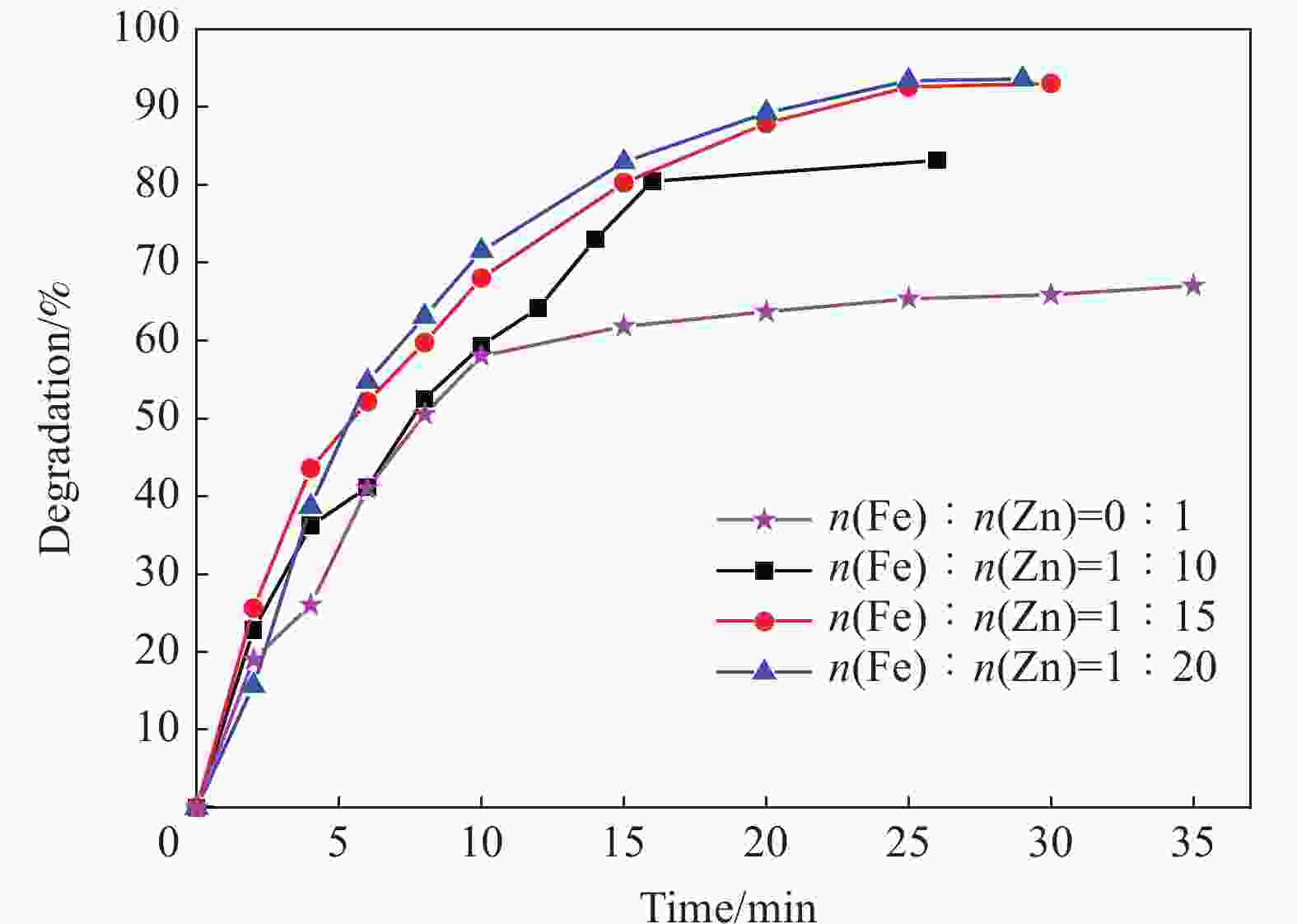
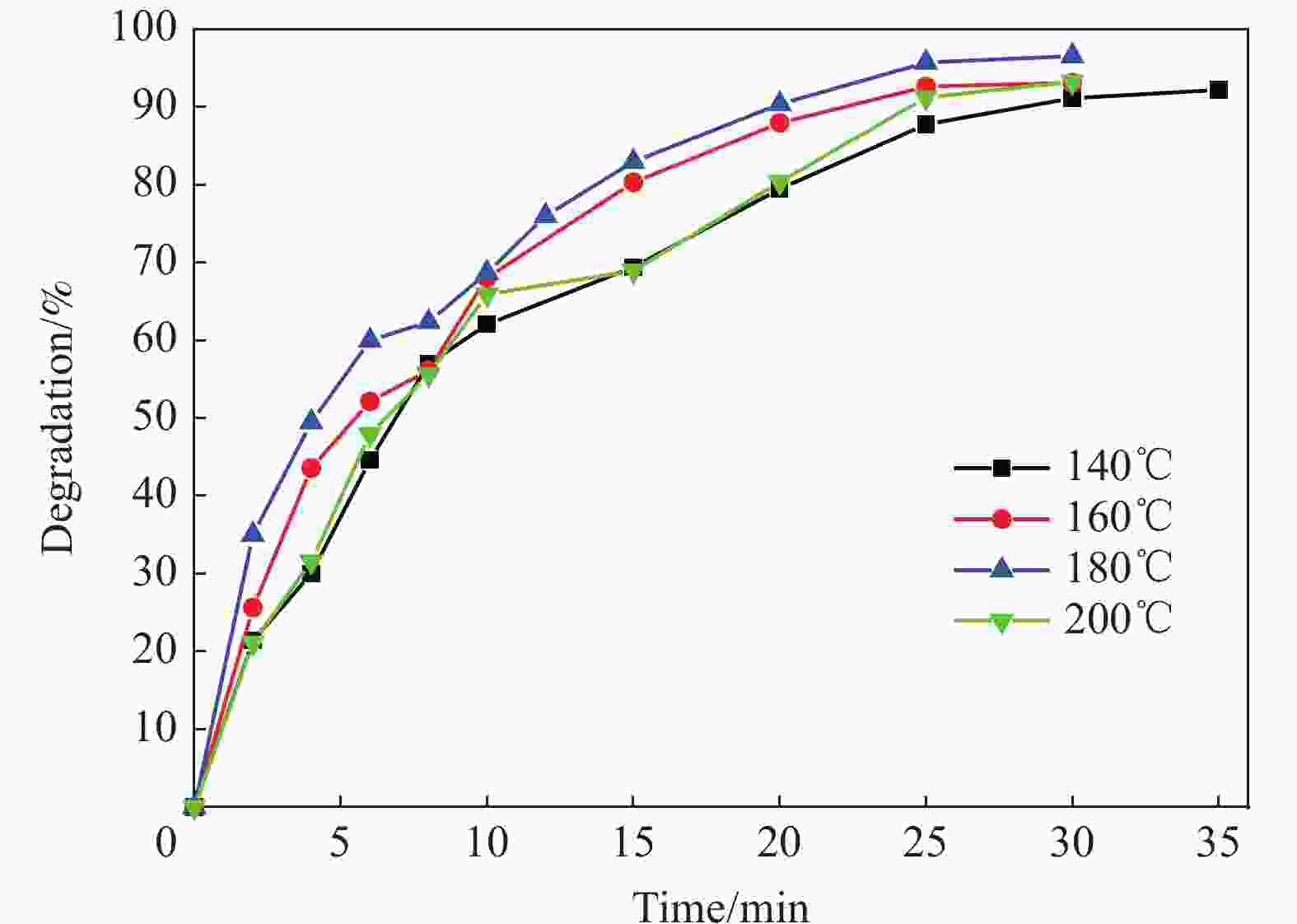
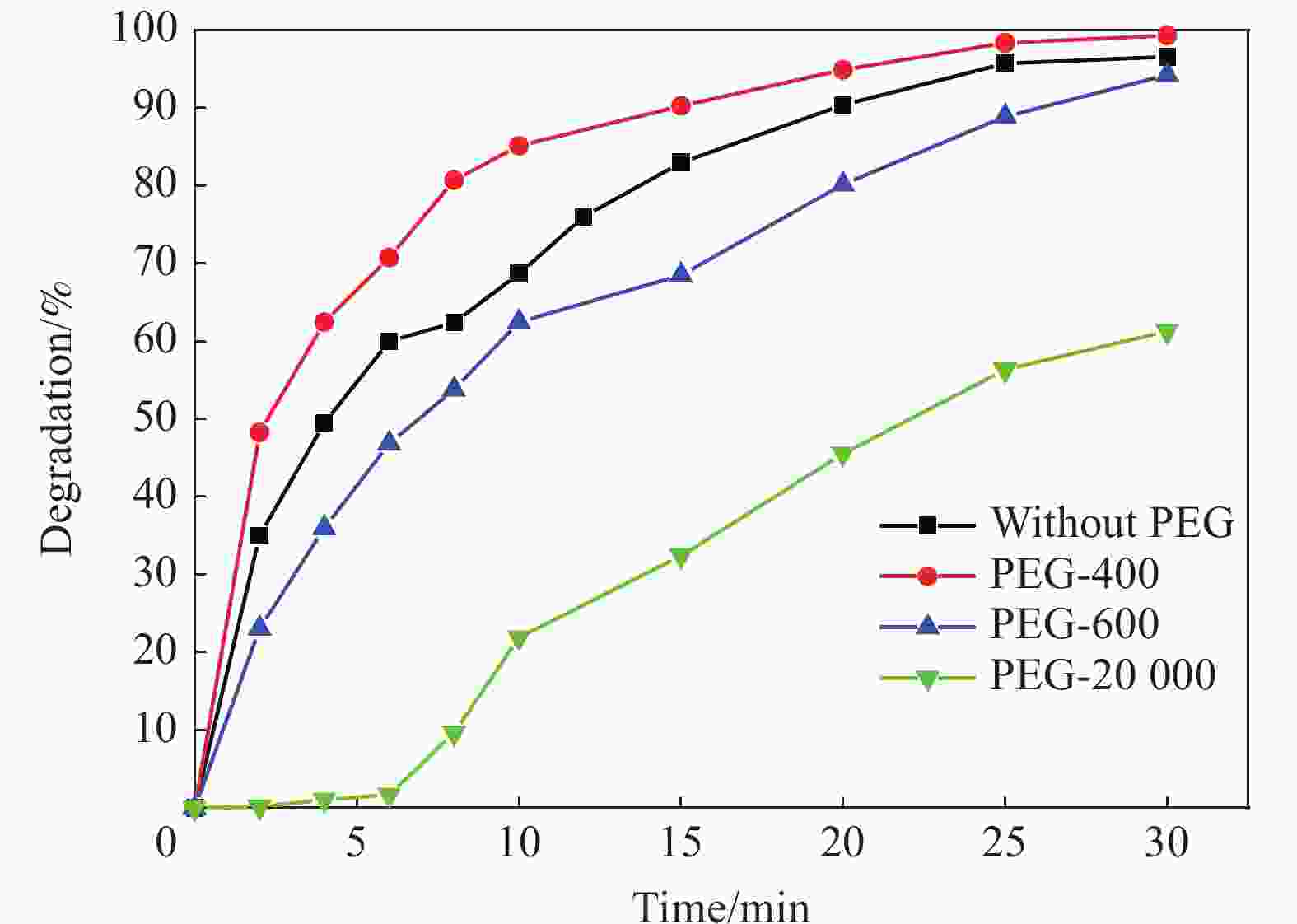
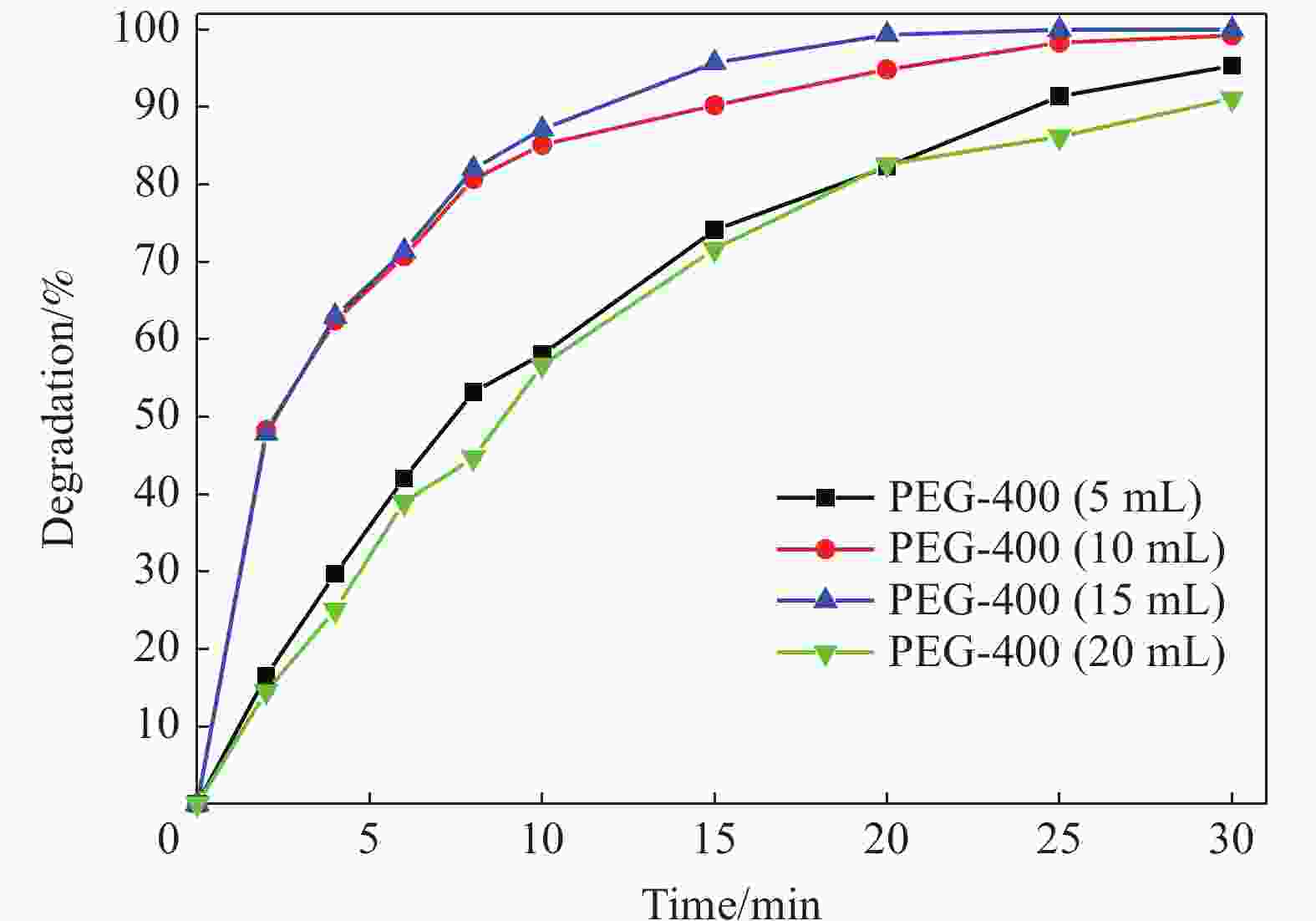
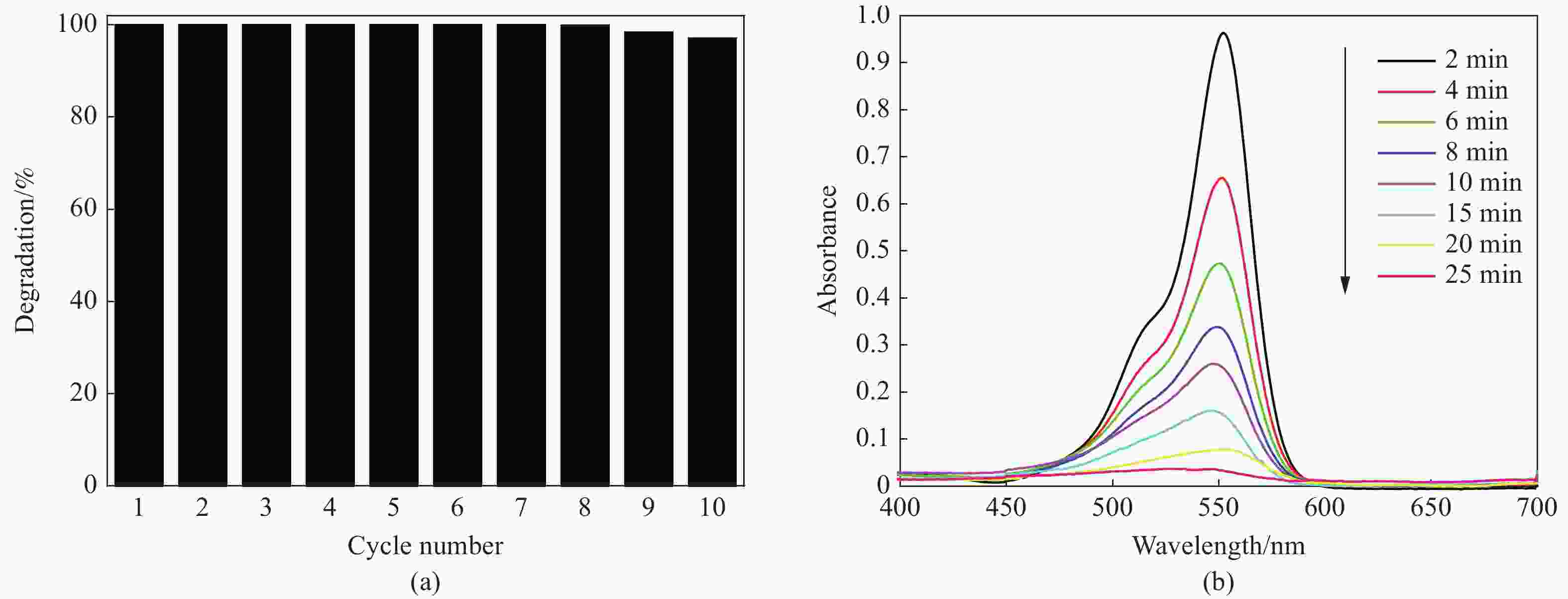
摘要: 首先合成氨基功能化Fe3O4(NH2—Fe3O4),并以NH2—Fe3O4为磁核,六水合硝酸锌(Zn(NO3)2·6H2O)为锌源,在表面活性剂聚乙二醇(PEG,PEG-400)辅助下通过水热法制备PEG修饰的ZnO(NH2—Fe3O4@PEG@ZnO)磁性复合材料。利用XRD、SEM、TEM、XPS、紫外-可见-近红外分光光度计、比表面吸附仪(BET)、振动样品磁强计(VSM)等对NH2—Fe3O4@PEG@ZnO复合材料组成、形貌、磁性能等进行表征。并进一步以罗丹明B(RhB)染料为模拟污染物,对NH2−Fe3O4@PEG@ZnO复合材料的光催化降解性能进行研究,采用单因素法探究Fe与Zn的原子比(n(Fe)∶n(Zn))、合成温度、表面活性剂种类及用量对NH2—Fe3O4@PEG@ZnO复合材料光催化降解性能的影响。结果表明,n(Fe)∶n(Zn)=1∶15、水热合成温度为180℃制备的NH2—Fe3O4@ZnO复合材料具有良好的光降解性能,0.0500 g NH2—Fe3O4@ZnO复合材料在紫外光照射20 min内对50 mL RhB(1.0×10−5 mol·L−1)溶液降解率为90.36%。而相同条件制备的NH2—Fe3O4@PEG@ZnO复合材料呈微球状,比表面积为11.43 m2·g−1,禁带宽度为2.51 eV,对RhB的光催化降解率可提高至99.36%,循环使用10次后,其对RhB的光催化降解率仍可达96.48%,PEG-400对NH2—Fe3O4@ZnO复合材料的光催化活性具有较大的协同效应。Abstract: The amino functionalized Fe3O4(NH2—Fe3O4) had been successfully prepared as magnetic core. With zinc nitrate hexahydrate (Zn(NO3)2·6H2O) as zinc source and polyethylene glycol(PEG, PEG-400) as surfactant, the magnetic composite based on zinc oxide and PEG-400-modified (NH2—Fe3O4@PEG@ZnO) composite was synthesized by hydrothermal process. The composition, morphology and magnetic properties of the NH2—Fe3O4@PEG@ZnO composite were characterized by a series of techniques including XRD, SEM, TEM, XPS, UV-VIS-NIR spectroscopy, specific surface area analyzer(BET), vibrating sample magnetometer(VSM) and so on. The photocatalytic performance of NH2—Fe3O4@PEG@ZnO composite under ultraviolet light was investigated by using the degradation of Rhodamine B (RhB) dye as a simulated pollutant. The influencing factors such as the atomic ratio of Fe to Zn(n(Fe)∶n(Zn)), reaction temperature, types and dosages of surfactant on photocatalytic degradation performance were investigated by one-factor method. The results show that NH2—Fe3O4@ZnO composite has good photocatalytic activity which is synthesized by n(Fe)∶n(Zn) is 1∶15 and the hydrothermal temperature is 180℃. The degradation rate of 50 mL RhB(1.0×10−5 mol·L−1) is 90.36% when use 0.0500 g NH2—Fe3O4@ZnO composite as a catalyst under ultraviolet light within 20 min. The specific surface area of NH2—Fe3O4@PEG@ZnO composite is 11.43 m2·g−1, the forbidden band width is about 2.51 eV and degradation rate of RhB is up to 99.36%. After 10 times of recycling, the photocatalytic degradation rate of RhB can still reach 96.48%. The combination of NH2−Fe3O4@ZnO composite and PEG-400 has a synergistic effect on improving the photocatalytic activity.
-
Key words:
- magnetic composites /
- photocatalytic degradation /
- nano ZnO /
- amino functionalized /
- Rhodamine B
-
表 1 NH2—Fe3O4@ZnO和NH2—Fe3O4@PEG@ZnO复合材料配比
Table 1. Component contents of NH2—Fe3O4@ZnO and NH2—Fe3O4@PEG@ZnO composite
Sample n(Fe)∶n(Zn) NH2—Fe3O4/g Zn(NO3)2·6H2O/g PEG-400/mL NH2—Fe3O4@ZnO-1 1∶10 0.0773 2.970 − NH2—Fe3O4@ZnO-2 1∶15 0.0773 4.455 − NH2—Fe3O4@ZnO-3 1∶20 0.0773 5.940 − NH2—Fe3O4@PEG@ZnO 1∶15 0.0773 4.455 15 Note: n(Fe) : n(Zn)—Atomic ratio of Fe to Zn. 表 2 NH2—Fe3O4@ZnO-2和NH2—Fe3O4@PEG@ZnO复合材料比表面积
Table 2. BET surface areas of NH2—Fe3O4@ZnO-2 and NH2—Fe3O4@PEG@ZnO composites
Catalyst BET surface area/(m2·g−1) NH2—Fe3O4@ZnO-2 0.0756 NH2—Fe3O4@PEG@ZnO 11.43 表 3 NH2—Fe3O4@PEG@ZnO复合材料光催化降解RhB拟一级动力学方程及参数
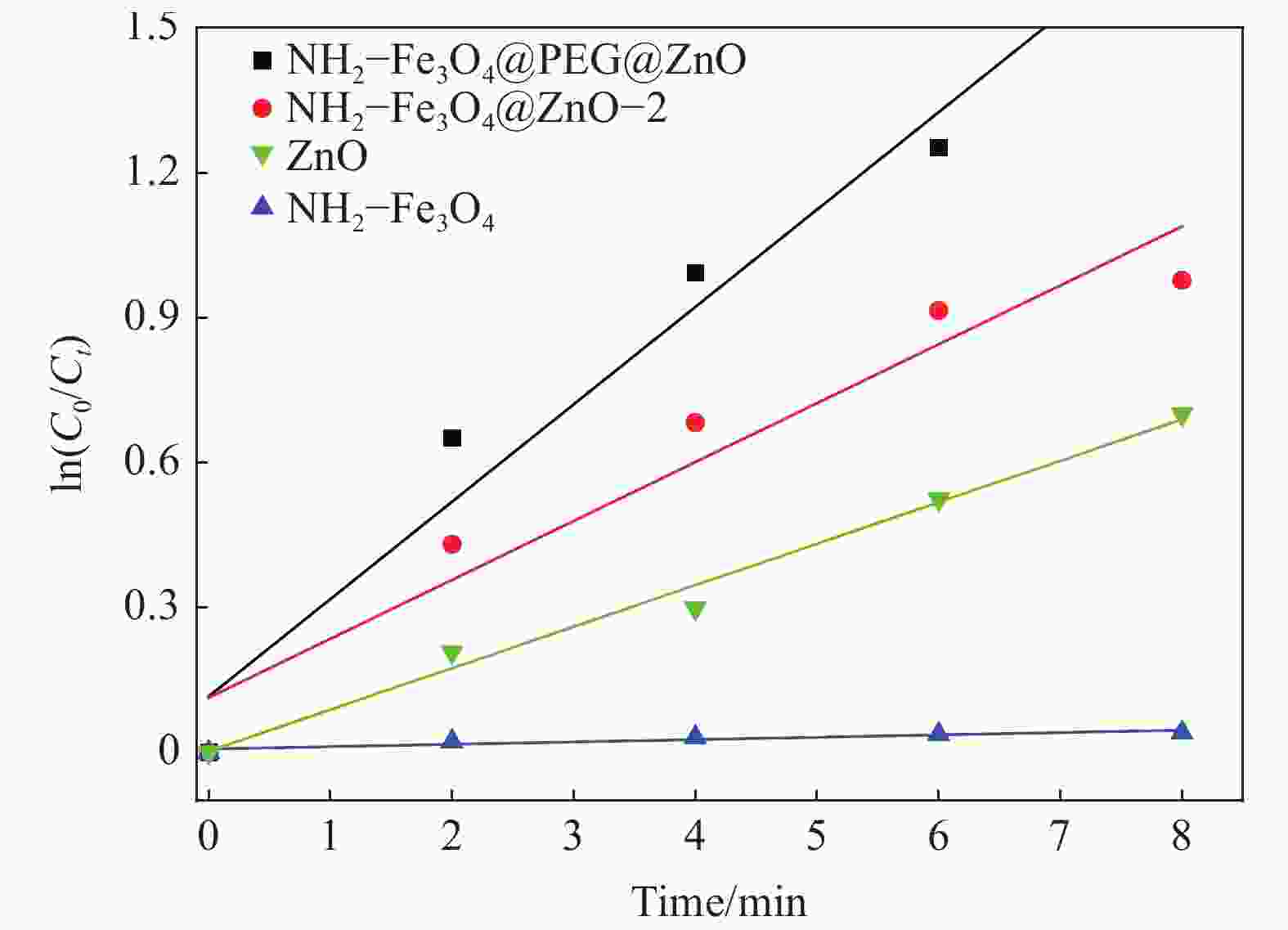
Table 3. Pseudo first-order kinetic equations and parameters of NH2—Fe3O4@PEG@ZnO composite degrading RhB
Composite Kinetic equations R2 K/min−1 NH2—Fe3O4 Y=0.0049x+0.0073 0.8288 0.0049 ZnO Y=0.0859x+0.0028 0.9841 0.0859 NH2—Fe3O4 @ZnO-2 Y=0.1220x+0.1135 0.9115 0.1220 NH2—Fe3O4@PEG@ZnO Y=0.2016x+0.1160 0.9669 0.2016 Notes: R2—Correlation coefficient; K—Apparent rate constant. -
[1] CHEN J, CHAO F, MU X, et al. ZnIn2S4/UiO-66-(SH)2 composites as efficient visible-light photocatalyst for RhB degradation[J]. Inorganic Chemistry Communications,2019,102:25-29. doi: 10.1016/j.inoche.2019.02.008 [2] REDDY P A K, RDEEY P V L, KWON E, et al. Recent advances in photocatalytic treatment of pollutants in aqueous media[J]. Environment International,2016,91:94-103. doi: 10.1016/j.envint.2016.02.012 [3] TONG H, OUYANG S, BI Y, et al. Nano-photocatalytic materials: Possibilities and challenges[J]. Advanced Materials,2012,24(2):229-251. doi: 10.1002/adma.201102752 [4] CHEN X, SU T, CHEN Y, et al. D-ribose as a contributor to glycated haemoglobin[J]. EBioMedicine,2017,25:143-153. doi: 10.1016/j.ebiom.2017.10.001 [5] MISHRA M, CHUN D. α-Fe2O3 as a photocatalytic material: A review[J]. Applied Catalysis A: General,2015,498:126-141. doi: 10.1016/j.apcata.2015.03.023 [6] CHEN D, HUANG F, REN G, et al. ZnS nano-architectures: Photocatalysis, deactivation and regeneration[J]. Nanoscale,2010,2(10):2062-2064. doi: 10.1039/c0nr00171f [7] GHAFURI H, DEHGHANI M, RASHIDIZADEH A, et al. Synthesis and characterization of magnetic nanocomposite Fe3O4@TiO2/Ag, Cu and investigation of photocatalytic activity by degradation of rhodamine B (RhB) under visible light irradiation[J]. Optik,2019,179:646-653. doi: 10.1016/j.ijleo.2018.10.180 [8] SIWIŃSKA-STEFAŃSKA K, KUBIAK A, PIASECKI A, et al. Hydrothermal synthesis of multifunctional TiO2-ZnO oxide systems with desired antibacterial and photocatalytic properties[J]. Applied Surface Science,2019,463:791-801. doi: 10.1016/j.apsusc.2018.08.256 [9] LIU Y, ZHANG Q, XU M, et al. Novel and efficient synthesis of Ag-ZnO nanoparticles for the sunlight-induced photocatalytic degradation[J]. Applied Surface Science,2019,476:632-640. doi: 10.1016/j.apsusc.2019.01.137 [10] ATLA S B, LIN W R, CHIEN T C, et al. Fabrication of Fe3O4/ZnO magnetite core shell and its application in photocatalysis using sunlight[J]. Materials Chemistry and Physics,2018,216:380-386. doi: 10.1016/j.matchemphys.2018.06.020 [11] WANG J, YANG J, LI X, et al. Preparation and photocatalytic properties of magnetically reusable Fe3O4@ZnO core/shell nanoparticles[J]. Physica E: Low-dimensional Systems and Nanostructures,2016,75:66-71. doi: 10.1016/j.physe.2015.08.040 [12] 胡亚一, 陈嘉磊, 刘琦, 等. 杂多酸离子液体负载氨基化Fe3O4磁性复合材料的制备及其超声辅助催化脱硫性能[J]. 复合材料学报, 2020, 37(3):650-661.HU Y Y, CHEN J L, LIU Q, et al. Preparation of heteropoly acid ionic liquids supported amino-functionalized Fe3O4 magnetic composite and its catalytic property for ultrasound-assisted desulfurization[J]. Acta Materiae Compositae Sinica,2020,37(3):650-661(in Chinese). [13] 王燕, 叶思, 吕珊珊, 等. 磁性Fe3O4@离子印迹聚(MMA-HPMA-DVB)复合材料的合成及其对水中Ni(Ⅱ)选择性吸附[J]. 复合材料学报, 2017, 34(12):2846-2855.WANG Y, YE S, LV S S, et al. Preparation of ion imprinted Fe3O4@IIP(MMA- HPMA-DVB) magnetic composite and its s-elective adsorption to Ni(Ⅱ)[J]. Acta Materiae Compositae Sinica,2017,34(12):2846-2855(in Chinese). [14] LIU K, QIN Y, MUHAMMAD Y, et al. Effect of Fe3O4 content and microwave reaction time on the properties of Fe3O4/ZnO magnetic nanoparticles[J]. Journal of Alloys and Compounds,2019,781:790-799. doi: 10.1016/j.jallcom.2018.12.085 [15] HABIBI-YANGJEH A, SHEKOFTEH-GOHARI M. Synthesis of magnetically recoverable visible-light-induced photocatalysts by combination of Fe3O4/ZnO with BiOI and polyaniline[J]. Progress in Natural Science: Materials International,2019,29(2):145-155. [16] ZHANG L, DONG B, XU L, et al. Three-dimensional ordered ZnO-Fe3O4 inverse opal gas sensor toward trace concentration acetone detection[J]. Sensors and Actuators B: Chemical,2017,252:367-374. doi: 10.1016/j.snb.2017.05.167 [17] ZENG J, LI Z, PENG H, et al. Core-shell Sm2O3@ZnO nano-heterostructure for the visible light driven photocatalytic performance[J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects,2019,560:244-251. [18] ZAINURI N Z, HAIROM N H H, SIDIK D A B, et al. Palm oil mill secondary effluent (POMSE) treatment via photocatalysis process in presence of ZnO-PEG nanoparticles[J]. Journal of Water Process Engineering,2018,26:10-16. doi: 10.1016/j.jwpe.2018.08.009 [19] 龙泽清. Fe掺纳米ZnO及Fe3O4/ZnO的制备及光催化性能研究[D]. 太原: 中北大学, 2016.LONG Z Q. A research on the preparation and photocatalytic performance of Fe-doped ZnO and Fe3O4/ZnO[D]. Taiyuan: North University of China, 2016(in Chinese). [20] DIKICI T, DEMIRCI S. Influence of thermal oxidation temperature on the microstructure and photoelectrochemical properties of ZnO nanostructures fabricated on the zinc scraps[J]. Journal of Alloys and Compounds,2019,779:752-761. doi: 10.1016/j.jallcom.2018.11.241 [21] SARANYA A, DEVASENA T, SIVARAM H, et al. Role of hexamine in ZnO morphologies at different growth temperature with potential application in dye sensitized solar cell[J]. Materials Science in Semiconductor Processing,2019,92:108-115. doi: 10.1016/j.mssp.2018.03.028 [22] HOLI A M, ZAINAL Z, AYAL A K, et al. Ag2S/ZnO nanorods composite photoelectrode prepared by hydrothermal method based on influence of growth temperature[J]. Optik,2019,184:473-479. [23] VIDYASAGAR C C, ARTHOBA NAIK Y. Surfactant (PEG 400) effects on crystallinity of ZnO nanoparticles[J]. Arabian of Chemistry,2016,9(4):507-510. doi: 10.1016/j.arabjc.2012.08.002 [24] 张丽, 阎建辉, 周民杰, 等. 高比表面空心球状ZnO/ZnAI2O4复合光催化剂制备及活性[J]. 无机化学学报, 2012, 28(9):1827-1834.ZHANG L, YAN J H, ZHOU M J, et al. Preparation and photocatalytic property of hollow sphere-like ZnO/ZnAI2O4 composite photocatalysts with high specific surface area[J]. Chinese Journal of Inorganic Chemistry,2012,28(9):1827-1834(in Chinese). [25] ZHENG W, DING R, YAN X, et al. PEG induced tunable morphology and band gap of ZnO[J]. Materials Letters,2017,201:85-88. doi: 10.1016/j.matlet.2017.04.133 [26] DEHGHAN S, KAKAVANDI B, KALANT-ARY R R. Heterogeneous sonocatalytic degradation of amoxicillin using ZnO@Fe3O4 magnetic nanocomposite: Influential factors, reusability and mechanisms[J]. Journal of Molecular Liquids,2018,264:98-109. doi: 10.1016/j.molliq.2018.05.020 [27] LIU Y, LIU G, XU J, et al. Distraction osteogenesis in the dog mandible under 60-Gy irradiation[J]. Oral Surgery, Oral Medicine, Oral Pathology and Oral Radiology,2012,113(2):183-187. doi: 10.1016/j.tripleo.2011.01.041 [28] CHEN Y F, TAN L L, LIU J M, et al. Calix[4]arene based dye-sensitized Pt@UiO-66-NH2 metal-organic framework for efficient visible-light photocatalytic hydrogen production[J]. Applied Catalysis B: Environmental,2017,206:426-433. doi: 10.1016/j.apcatb.2017.01.040 -





 下载:
下载: