Research progress of graphene oxide composite coatings in metal corrosion protection
-
摘要:
氧化石墨烯(GO)作为石墨烯的衍生物具有优异的综合性能,在金属的防腐蚀领域中表现出了巨大的应用潜力。GO不仅具有石墨烯的二维层状结构,还含有羟基、羰基、羧基和环氧基团等官能团可作为活性位点与其他物质进行共价/非共价性功能化改性,因此GO常被用作填料来增强涂层的综合性能。本文以GO复合涂层为中心,简要地介绍了其理化性质,以当前世界金属腐蚀的情况和腐蚀类型为切入点,针对一些常用的腐蚀防护方法进行了讨论。综述了近年来国内外关于GO与有机物和无机物的复合涂层在金属腐蚀与防护领域的研究进展并对复合涂层的防腐机制进行了简述;最后,总结了目前研究工作中存在的关键科学难题与挑战,对涂层的研究方向与应用前景进行了展望。
Abstract:As a derivative of graphene, graphene oxide (GO) has excellent comprehensive performance, showing great application potential in metal corrosion and protection. GO has not only a two-dimensional layered structure but also contains hydroxyl, carbonyl, carboxyl, epoxy groups and other functional groups that can be used as active sites with other substances for covalent/non-covalent functionalization modification, so GO is often used as a filler to enhance the comprehensive performance of coatings. This paper reviews the recent research progress on GO composite coating in metal corrosion and protection. The first part summarizes the morphology of metal corrosion and the corrosion protection methods, explains the primary forms of metal corrosion, and discusses the current main metal corrosion protection methods. The second part introduces the physicochemical properties of graphene and its derivatives. In this paper, GO is the main focus. It has tremendous application potential in metal corrosion protective coatings due to its sheet structure, excellent dispersion, and abundant oxygen-containing functional groups on the surface, which are the active point of the reaction and easy modification. The third part introduces the current GO composite coating, which compares the corrosion resistance of traditional coatings, GO organic/inorganic unit composite coatings and GO multi-composite coatings. It is found that the performance of GO multi-component composite coating is far better than that of GO organic/inorganic unit composite coating and traditional coating. This is due to the excellent dispersion of GO, which enables it to fill the pores that occur during the curing and film-forming process of organic polymer materials and improve the corrosion resistance of the coating. The photogenic cathodic protection mechanism and current situation of GO anti-corrosion coating are introduced, and due to the green environmental protection and excellent anti-corrosion effect, it is regarded as the future development trend of GO composite coating. The fourth part summarizes the key scientific issues and challenges of GO composite anti-corrosion coating in the current research. It looks forward to the research direction and application prospect of GO composite anti-corrosion coating.
-
碳纤维增强热塑性树脂基复合材料因其良好的力学性能在车辆船舶、航空航天、汽车制造等众多领域得到广泛应用[1-6]。其中,大连理工大学蹇锡高院士团队研发的杂萘联苯聚芳醚砜酮(PPBESK)相比较同类型产品具有耐高温、易溶解、易加工等优点[7],其作为树脂基体有较好前景。在复合材料中,基体为连续相,其对复合材料的热量传递具有决定作用。然而,树脂基体无规结构使其热散射严重,导致其导热系数较差,限制了其在电子封装、快速散热领域中的使用。因此,提高碳纤维(CF)增强PPBESK复合材料的导热系数(λ)具有重要意义。
研究者采用两种设计方向来提高树脂基复合材料的导热系数(λ)。一是合成本征高导热树脂基体,但树脂是由相互纠缠的分子链所构成,其结构包含了复杂的支链和侧链。目前关于分子链结构与树脂导热能力之间的具体联系规律尚未明确[8]。因此,直接从结构上改善树脂本征导热系数的难度较高。二是引入高导热填料如石墨烯[9],氮化硼[10],氮化铝[11],碳化硅[12]和碳纳米管[13]。徐任信[14]等人利用浇注法合成了CF/AlN/EP复合材料,有效提升了其导热性能。钱欣[15]等人则选用石墨作为增强材料,与酚醛树脂结合制备了新型树脂基复合材料,并发现随着石墨含量的提升,复合材料的导热率也呈现上升趋势。顾军渭[16]等人则选用不同种类的碳化硅与环氧树脂为原料,通过浇铸成型制备了SiC/环氧树脂基复合材料,实验结果表明,纳米级SiC含量的增加能显著提高复合材料的导热率。
碳纳米管,具有高导热系数和大比表面积,掺入聚合物基体后,有望改善复合材料的导热性能和力学性能[17, 18]。其结构可以形象地描述为石墨烯片层的卷曲与焊接,这种结构赋予了碳纳米管与石墨烯相似的优异导通性能,其热导率可在
2500 -6000 W·m−1·K−1之间[19, 20]。此外,碳纳米管的高度sp2杂化结构赋予其高模量和高强度的特点。其抗拉强度高达200 GPa,是钢材的百倍之多;同时,其弹性模量也达到了钢材的5倍左右。凭借其独特的结构和卓越的物理性能,少量加入就有望合成具有优异力学性能和导热性能的结构/功能一体化复合材料。然而,对碳纳米管改善树脂基体力学性能和导热性的系统性研究较少。为了提高CF增强PPBESK复合材料导热及力学性能,笔者将碳纳米管(CNTs)引入CF/PPBESK复合材料,研究了CNTs对CF/PPBESK复合材料导热系数和力学性能的影响,并阐明了其导热及力学性能增强机制。为制备导热结构/功能一体化复合材料提供了理论依据和实验研究。1. 实验材料及方法
1.1 主要原材料
PPBESK (密度1.33 g/cm3,特性黏度0.48 dl/g,拉伸强度87 MPa)由大连高聚物新材料有限公司制造;
碳纳米管(纯度95%,直径10 nm)江苏先锋纳米材料科技有限公司提供。
DMAc(纯度99%)天津博迪化工有限公司生产;
碳纤维(直径7 μm,密度1.8 g/cm3,拉伸强度为4.9 GPa,拉伸模量为230 GPa)由日本东丽公司生产。
1.2 仪器与设备
扫描电子显微镜(SEM):FLexSEM1000,SU8200,日本日立株式会社;
电子光学显微镜:Axiocam 208 color,蔡司集团;
电子万能试验机:Instron 5869,英国英斯特朗公司;
动态机械热分析(DMA)仪:TA Q800型,美国TA公司。
红外热成像仪:UTi260 A,中国优利德科技股份有限公司
1.3 测试与表征
采用日本日立株式会社FLexSEM1000,SU8200扫描电镜和蔡司集团Axiocam 208 color光学显微镜观察复合材料的显微组织和形貌。在SEM观察之前,采用溅射工艺在样品上喷金,获得清晰稳定的图像。
根据ASTM D790-10[21]标准,使用50 KN Instron 5869试验机测量复合材料的弯曲强度。试件尺寸为80 mm × 12.5 mm × 2 mm。施加弯曲力的十字头速度为2 mm·min−1。
根据ASTM D695 M[22]标准,使用50 KN Instron 5869试验机测量复合材料的抗压强度。试件尺寸为75 mm × 10 mm × 2 mm。施加力的十字头速度为1 mm·min−1。
根据ASTM D3039[23]标准,使用50 KN Instron 5869试验机测量复合材料的拉伸强度。试件尺寸为250 mm × 15 mm × 1 mm。施加力的十字头速度为2 mm·min−1。
根据ISO14130[21]标准,使用50 KN Instron 5869试验机测量复合材料的层间剪切强度(ILSS)。试件尺寸为20 mm × 10 mm × 2 mm。施加剪切力时,横头速度为1 mm·min−1。根据式ILSS=4F3bh计算ILSS值。
其中F为断裂时的最大压缩载荷(N), b为试件宽度(mm), h为试件厚度(mm)。
动态力学分析(DMA)测量在空气气氛中等速升温至300℃,使用美国TA公司TA Q800型装置,频率为1 Hz。加热速率为3℃/min。试验程序为单悬臂模型。试件尺寸为35 mm × 6 mm × 2 mm。利用LFA 467微闪烁激光器进行了空气中透层热扩散系数的测量。根据式λ=αρCp计算含/不含碳纳米管的CF/PPBESK层压板的垂直导热系数λ其中α为热扩散系数,ρ为试样的体积密度,Cp为比热容。
将中国优利德科技股份有限公司UTi260 A手持红外热成像通过支架固定,将样品置于150℃加热台加热,每隔1 s拍摄一张热成像图。
1.4 CNTs@CF/PPBESK复合材料的制备
本研究采用溶液浸渍法制备了CNTs@CF/PPBESK复合材料。具体步骤如下:首先将纤维缠绕在230 mm × 150 mm的铁框上,然后将碳纳米管溶解在DMAc中,再将该溶液超声震荡6 h,之后将PPBESK溶解在上述溶液中,然后将缠绕在铁框上的CF束用CNTs@PPBESK/DMAc溶液浸泡得到预浸料。将预浸料置于烘箱中蒸发DMAc溶剂(100℃/4 h, 120℃/2 h, 150℃/2 h, 180℃/2 h, 220℃/2 h),然后用热压机在330℃下以5 MPa的压力对15层预浸料进行压缩,得到2 mm厚的CNTs@CF/PPBESK复合材料。使用纯预浸料以相同的方法制备原始CF/PPBESK复合材料。整个制备过程如图所示:
2. 结果与讨论
2.1 导热性能
所有试件的垂直导热系数测量数据已在图2中详细展示。经过CNTs改性的CF增强复合材料的导热系数均显著超过了未改性的层压板。随着CNTs含量的递增,复合层压板的导热系数展现出一种先增后减的趋势。具体来说,当CNTs含量达到10 wt%时,复合材料的导热系数达到峰值,为1.016 W/(m·K),相较于纯CF增强复合材料,其提升幅度高达72%。然而,随着CNTs含量的进一步增加,导热系数却开始逐渐下降。这一现象的主要原因在于,随着CNTs含量的增加,CNTs在树脂基体中有效分散的难度增大,造成了CNTs和树脂基体的接触面积反而减小,进而导致了整体导热率的降低[24]。此外,此外,从图2中还可以观察到CNTs对热扩散系数的影响。改性后的CF增强复合材料的热扩散系数同样显著高于未改性的层压板,并且其随CNTs含量的变化趋势与导热系数的变化趋势基本一致。
此外,为了更清楚地说明CNTs与CF对复合材料导热性能的作用以及之间的导热协效性,对PPBESK、10 wt%CNTs/PPBESK、10 wt%CNTs@CF/PPBESK复合材料进行了导热性能测试。从图3中可以看出,PPBESK与10 wt%CNTs/PPBESK导热率较差,而10 wt%CNTs@CF/PPBESK复合材料的导热率几乎是10 wt%CNTs/PPBESK复合材料的两倍,这充分证明了CNTs与·CF对复合材料导热率的协效型。
通过红外热成像技术模拟了在实际应用过程中复合材料的热传导能力。从图4中可以了解到纯CF/PPBESK复合材料与CNTs@CF/PPBESK (4wt%)复合材料的温度变化有明显不同。经过CNTs改性的复合材料导热速率有明显改善。进一步证明了CNTs的引入对纯CF/PPBESK复合材料的导热率有积极作用。
在纯CF/PPBESK复合材料中,存在较大的富树脂区,CF被树脂分隔开,两相间巨大的声子散射使其导热性能差。因此,虽然CF具有良好的导热性,但CF/PPBESK复合材料由于富树脂区导热性不足而表现出较差的导热性。如图5所示,在引入CNTs后,复合材料中相邻的CF被CNTs连接,形成导热网路。热量通过CNTs与CF形成的导热网络高效传递,有效地降低了富树脂区域的声子散射,从而增强了导热性。
2.2 弯曲性能
CNTs的引入同样会对复合材料的力学性能产生显著影响。为了深入探究这一影响,我们进行了针对性的弯曲试验。观察图6中的数据,可以发现,纯CF/PPBESK复合材料的弯曲强度为
1328 MPa随着CNTs含量的增加,CNTs@CF/PPBESK复合材料的弯曲性能起初呈现出提升的趋势。当CNTs含量达到4 wt%时,弯曲强度达到了最大值,即1695 MPa,相较于纯CF/PPBESK复合材料,提升了28%。然而,随着CNTs含量的进一步增加,弯曲强度开始逐渐下降。当CNTs含量达到15 wt%时,弯曲强度降至最低,为1233 MPa,这主要归因于CNTs的团聚。为了进一步揭示CNTs对弯曲性能的具体影响,利用扫描电镜对弯曲试验后的样品断裂面进行了深入观察。从图7(a)中,可以清晰地看到纯CF/PPBESK复合材料的断裂面上存在着较多的孔洞。这些孔洞表明,在弯曲断裂的过程中,CF在PPBESK基体中被强力拉出,形成了明显的缺陷。而在添加了CNTs后,复合材料的断裂面展现出了显著的变化。观察图7(b),可以发现,在含有CNTs@CF/PPBESK(4 wt%)复合材料中,纤维被拉出的现象明显减少。这表明CNTs的加入有效地增强了纤维与基体之间的界面附着力。在局部放大图7(c)中,可以更加清晰地观察到CNTs的存在。它们紧密地分布在纤维和基体之间,形成了一个坚固的网络结构。在纯CF/PPBESK复合材料中,由于纤维上的表面活性基团数量较少,导致纤维与基体之间的界面附着力较弱,容易产生应力集中和裂纹扩展,进而引发界面的破坏[25, 26]。而引入CNTs后,情况得到了显著的改善。CNTs以其高比表面积和优良的力学性能[27],大幅增加了树脂基体与纤维增强体之间的应力传输能力,从而提升了复合材料的力学性能。此外,一部分CNTs可能会迁移至CF表面,增加了纤维与树脂间的接触面积,改善了CF与树脂之间的结合强度。这两方面的共同作用使得CNTs显著提升了CF/PPBESK复合材料的弯曲性能。
2.3 压缩性能
为了探究CNTs含量对CF/PPBESK复合材料压缩性能的影响,进行了压缩实验。与弯曲强度趋势类似,随着CNTs浓度的增加,其变化趋势并非单调上升,而是呈现出先升高后降低的态势。具体来说,在加入CNTs之前,纯CF/PPBESK复合材料的压缩强度为729 MPa。当向复合材料中加入至4 wt%的CNTs时,其压缩强度提升至
1001 MPa,相比纯CF/PPBESK复合材料,这一提升幅度高达37%。然而,当继续增加CNTs的含量时,情况开始发生变化。随着CNTs质量分数达到15 wt%,复合材料的压缩强度最终降至629 MPa,甚至低于纯CF/PPBESK复合材料的压缩强度。这可能是因为随着CNTs含量的增加,其在复合材料中的分散变得困难,并形成了团聚,这导致复合材料内部形成了大量缺陷,从而在受到外力作用时,更容易发生破坏。观察图9压缩样件断裂面可以得出:纯CF/PPBESK复合材料在受到压力而发生断裂时,明显出现了分层破坏。这主要是由于碳纤维与树脂之间的界面结合较为薄弱,树脂与基体间存在微小的裂纹。当受到外部应力的作用时,这些微裂纹便会沿着界面逐渐扩展,最终导致复合材料的分层破坏。当加入4 wt%的CNTs后,情况发生了显著的变化。复合材料的破坏形式由分层失效转变为了纤维与基体的共同断裂(45°剪切破坏)。CNTs的存在增强了树脂与碳纤维之间的应力传输能力,使得外力能够更好地由基体传递至纤维,而不是沿着界面处扩散。另一方面,碳纳米管能够起到偏转裂纹的作用,这消耗了大量的裂纹扩展能,从而使得其能够承受更大的力,具有更高的压缩强度。
2.4 复合材料层间剪切强度和断裂形态
从图10 CNTs含量对层间剪切强度的影响中可以看出,加入CNTs后会对CF/PPBESK复合材料的剪切强度造成影响。低填充量的CNTs可以提高CF/PPBESK复合材料的剪切强度,当CNTs的质量分数为4 wt%时,剪切模量达到了70 MPa,相比纯CF/PPBESK复合材料提升了14%。随着CNTs填充量的增加,剪切强度会下降。
从图11(a),(b)SEM图中可以看出,纯CF/PPBESK复合材料破坏后CF表面相对光滑,CF表面几乎没有树脂,而从11(c)中则可以看出,CF表面粗糙,可以清楚观察到纤维表面附有大量的树脂碎片。此外,如图11(d)的放大图像中可清晰的看出CNTs的存在。在模压过程中,一部分CNTs能够扩散到CF表面,增加其与树脂间的接触面积,从而改善了其与树脂间的机械互锁能力。此外,CNTs能够起到偏转裂纹的作用,一定程度上阻止了层间裂纹的扩展,增强了界面剪切强度。
2.5 拉伸性能
CNTs加入到CF/PPBESK复合材料体系中会对基体与CF的界面造成较大的影响,进而也会影响复合材料的拉伸强度,为了深入探究这一影响,进行了拉伸试验。从图5中了解到,纯CF/PPBESK的拉伸强度为
1450 MPa,当CNTs含量达到4 wt%时,CNTs@CF/PPBESK复合材料拉伸强度最高,达到了1696 MPa。之后随CNTs含量的增加,CNTs@CF/PPBESK复合材料的拉伸强度降低,当CNTs含量为15 wt%时,复合材料的拉伸强度最低,为1240 MPa。这归因于CNTs的团聚。通过观察失效拉伸样条,纯CF/PPBESK复合材料中CF与PPBESK之间结合不紧密,而CNTs@CF/PPBESK复合材料由于CNTs的引入改善了树脂与CF的机械锁合作用,界面性能有了明显改善,从而提高了复合材料的拉伸强度。
2.6 复合材料动态热力学分析
对纯CF/PPBESK复合材料与CNTs@CF/PPBESK进行了动态热力学分析,如图11所示,纯CF/PPBESK复合材料的储能模量为14 GPa,而CNTs@CF/PPBESK(4 wt%)复合材料的储能模量为22 GPa,明显可以看出,相较于改性前,改性后的复合材料Tg也有升高。这是因为CNTs比表面积大,并且具有良好的力学性能,与树脂基体结合后,可以将其看作复合材料的增强相。其次,由于CNTs的存在,限制了分子链的移动。在这两种因素协同作用下,CNTs@CF/PPBESK复合材料的刚性得到增强。
3. 结 论
(1)引入碳纳米管(CNTs)后,碳纤维(CF)/聚芳醚砜酮(PPBESK)复合材料的导热性能和力学性能均得到了提升。
(2)通过扫描电镜观察证明了引入CNTs后复合材料形成了导热网路。
(3)通过动态热力学分析证明CNTs的引入提高了CF/PPBESK复合材料的Tg和储能模量。
(4)本研究有助于进一步推进结构/功能一体化复合材料的商用步伐,为社会创造巨大收益。
-
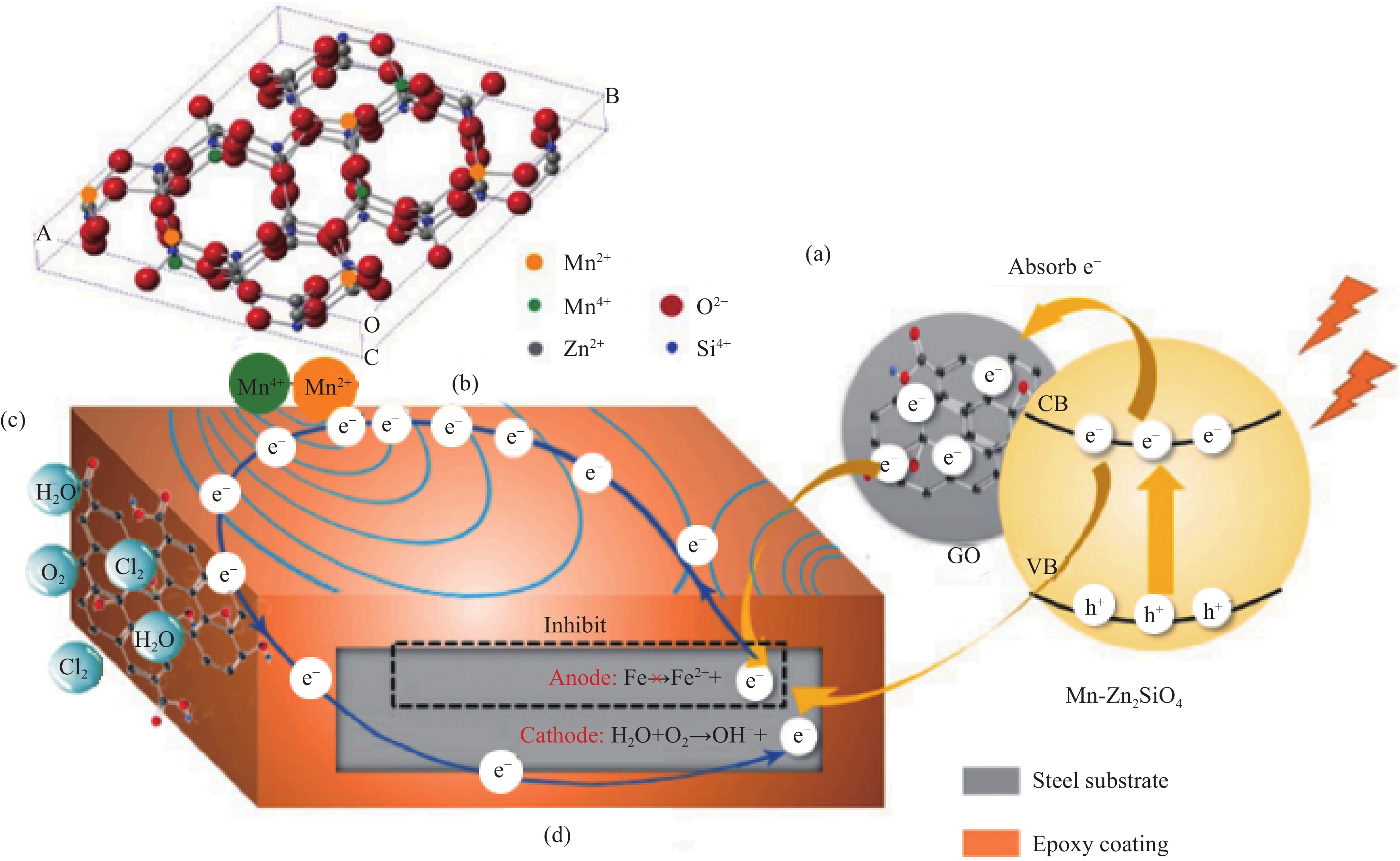
图 5 GO/Mn-Zn2SiO4材料的腐蚀防护机制图[43]:(a)光生阴极效应;(b)无机锰离子磁感应效应;(c)二维GO屏蔽效应;(d)阴极二次保护效应
Figure 5. Corrosion protection mechanism of GO/Mn-Zn2SiO4 material[43]: (a) Photogenerated cathode effect; (b) Inorganic manganese ion magnetic induction effect; (c) Two-dimensional GO shielding effect; (d) Cathode secondary protection effect
VB—Valence band; CB—Conduction band
表 1 GO作为涂层填料相对于其他常见无机填料的优缺点
Table 1 Advantages and disadvantages of GO as a coating filler over other common inorganic fillers
Type Character Advantages Disadvantages GO 1. High specific surface area, good mechanical properties, excellent barrier and shielding properties;
2. Excellent response reactivity, thermal and chemical stabilities;
3. Oxygen-containing functional group can serve as active sites for reactions; Hydrophilic groups on the surface are more easily modified by polymers or alkali metal oxides;
4. "Maze effect" can increase the diffusion path of the corrosion factor in the coating, and has a high resistance to permeability.1. Easy to agglomerate, dispersibility and stability are reduced after agglomeration;
2. Electrical conductivity, prone to galvanic coupling corrosion at locations of coating defects.Nano-ZnO 1. High melting point;
2. Good oxidation and corrosion resistance.1. High surface activity, easy to agglomerate and lose the special effect of nanoparticles after agglomeration;
2. Hydrophilic and oleophobic, poor dispersibility and stability in organic media;
3. Weak bonding with the substrate, poor interfacial compatibility, easy to produce voids, micro-cracks and other interfacial defects.Nano-Al2O3 1. High strength, thermal conductivity and wear resistance;
2. Excellent electrical insulation;
3. Stable physical and chemical properties.1. Poor compatibility with the substrate, poor dispersion, easy to agglomerate;
2. Functionalisation of the surface may lead to a reduction in filler size, resulting in defects on the surface;
3. Different shapes and sizes also have an effect on the corrosion resistance of the coating.Micro/Nano-
SiO21. High hardness, high mechanical strength;
2. Excellent thermal and chemical stability;
3. Low density, small particle size, large specific
surface area;
4. Colorless, odorless and pollution-friendly;
5. Good corrosion resistance.1. Fine particles and high hydrophilicity, easy to agglomerate;
2. The coating is prone to cracking during curing and reducing the corrosion resistance of the coating. -
[1] BALANDIN A A, GHOSH S, BAO W Z, et al. Superior thermal conductivity of single-layer graphene[J]. Nano Letters, 2008, 8(3): 902-907. DOI: 10.1021/nl0731872
[2] GUO S J, DONG S J. Graphene nanosheet: Synthesis, molecular engineering, thin film, hybrids, and energy and analytical applications[J]. Chemical Society Reviews, 2011, 40(5): 2644-2672. DOI: 10.1039/c0cs00079e
[3] LIANG X, LI X J, TANG Y, et al. Hyperbranched epoxy resin-grafted graphene oxide for efficient and all-purpose epoxy resin modification[J]. Journal of Colloid and Interface Science, 2022, 611: 105-117. DOI: 10.1016/j.jcis.2021.12.068
[4] WANG J Q, ZHANG P, LIANG B, et al. Graphene oxide as an effective barrier on a porous nanofibrous membrane for water treatment[J]. ACS Applied Materials & Interfaces, 2016, 8(9): 6211-6218. DOI: 10.1021/acsami.5b12723
[5] CHEN J H, LI W G, ZHAO Y T, et al. Application of graphene in anti-corrosive and anti-fouling coating[J]. Surface Technology, 2019, 48(6): 89-97.
[6] ZHANG Y J, SHAO Y W, LIU X L, et al. A study on corrosion protection of different polyaniline coatings for mild steel[J]. Progress in Organic Coatings, 2017, 111: 240-247. DOI: 10.1016/j.porgcoat.2017.06.015
[7] CHENG L, LIU C, HAN D, et al. Effect of graphene on corrosion resistance of waterborne inorganic zinc-rich coatings[J]. Journal of Alloys and Compounds, 2019, 774: 255-264. DOI: 10.1016/j.jallcom.2018.09.315
[8] LE H S, NGUYEN D L, JUKKA S. Enhanced mechanical and thermal properties of polyurethane functionalized graphene oxide composites by in situ polymerization[J]. Plastics, Rubber and Composites, 2019, 48(10): 466-476. DOI: 10.1080/14658011.2019.1664820
[9] SALAVAGIONE H J, MARTÍNEZ G, ELLIS G. Recent advances in the covalent modification of graphene with polymers[J]. Macromolecular Rapid Communications, 2011, 32(22): 1771-1789. DOI: 10.1002/marc.201100527
[10] SUN Y B, YANG S B, CHEN Y, et al. Adsorption and desorption of U(VI) on functionalized graphene oxides: A combined experimental and theoretical study[J]. Environmental Science & Technology, 2015, 49(7): 4255-4262.
[11] ASALDOUST S, RAMEZANZADEH B. Synthesis and characterization of a high-quality nanocontainer based on benzimidazole-zinc phosphate (ZP-BIM) tailored graphene oxides; A facile approach to fabricating a smart self-healing anti-corrosion system[J]. Journal of Colloid and Interface Science, 2020, 564: 230-244.
[12] LI Y F, LIU J L, DENG J S, et al. Fabrication of graphene oxide reinforced plasma sprayed Al2O3 coatings[J]. Ceramics International, 2023, 49: 1667-1677. DOI: 10.1016/j.ceramint.2022.09.129
[13] HAMID Z A, GOMAA M H, EL-HOUT S I, et al. α-Fe2O3/Fe3O4@GO nanosheets boost the functionality of Ni-P thin film deposited by electroless method[J]. Surface & Coatings Technology, 2023, 456: 129288.
[14] LALA S R F, SRIVASTAVA C. Correlation between texture, grain boundary constitution and corrosion behaviour of copper-chromium coatings containing graphene oxide[J]. Metallurgical and Materials Transactions A, 2023, 54: 634-645. DOI: 10.1007/s11661-022-06908-7
[15] BAGDE A, SHRIRAME S, BHANVASE B A, et al. Investigation on simultaneous enhancement in mechanical and anticorrosion performance of graphene oxide-zinc phosphate nanocomposite-based coatings[J]. Diamond & Related Materials, 2024, 144: 110960.
[16] 李智, 刘崇宇, 葛毓立, 等. 氧化石墨烯对纳米金属陶瓷复合镀层组织性能影响[J]. 表面技术, 2023, 52(10): 394-401. LI Zhi, LIU Chongyu, GE Yuli, et al. Effect of graphene oxide on microstructure and properties of nano-cermet composite coatings[J]. Surface Technology, 2023, 52(10): 394-401(in Chinese).
[17] ZHANG Y H. Application of water-soluble polymer inhibitor in metal corrosion protection: Progress and challenges[J]. Frontiers in Energy Research, 2022, 10: 3389.
[18] VAZIRINASAB E, JAFARI R, MOMEN G. Application of superhydrophobic coatings as a corrosion barrier: A review[J]. Surface & Coatings Technology, 2018, 341: 40-56.
[19] ZHANG F Y, LIU W Q, LIANG L Y, et al. Applications of hydrophobic α,ω-bis(amino)-terminated polydimethylsiloxane-graphene oxide in enhancement of anti-corrosion ability of waterborne polyurethane[J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2020, 600: 124981. DOI: 10.1016/j.colsurfa.2020.124981
[20] WANG M J, WANG A, ZHAO W J, et al. The ion diffusion-directed self-assembled graphene oxide coating and its synergistic lubrication mechanism against environmental moisture[J]. Tribology International, 2024, 191: 109182. DOI: 10.1016/j.triboint.2023.109182
[21] GUO R T, LI W, WANG X Y, et al. Antimicrobial corrosion study of the epoxy coating with the graphene oxide supported Schiff base quaternary ammonium salt additives[J]. Materials Today Communications, 2023, 35: 105517. DOI: 10.1016/j.mtcomm.2023.105517
[22] 李凤英, 鞠鹏飞, 陈磊, 等. 聚苯胺原位聚合改性氧化石墨烯制备复合涂层及其耐腐蚀性能研究[J]. 表面技术, 2021, 50(11): 287-296. LI Fengying, JU Pengfei, CHEN Lei, et al. Preparation and corrosion resistance of polyaniline/modified graphene oxide composite coating[J]. Surface Technology, 2021, 50(11): 287-296(in Chinese).
[23] ZHU X T, GAO Z S, LI F C, et al. Superlyophobic graphene oxide/polydopamine coating under liquid system for liquid/liquid separation, dye removal, and anti-corrosion[J]. Carbon, 2022, 190: 329-336. DOI: 10.1016/j.carbon.2022.01.018
[24] GAUR M S, RAGHAV R K, SAGAR R, et al. Investigation of anticorrosion properties of epoxy GO nanocomposites spin coated aluminum alloy 7075[J]. Polymers and Polymer Composites, 2022, 30: 1-10.
[25] DU X Q, LIU Y W, CHEN D C, et al. Co-electrodeposition of silane and graphene oxide on copper to enhance the corrosion protection performance[J]. Surface & Coatings Technology, 2022, 436: 128279.
[26] PANG W T, JIANG H, WANG S, et al. Graphene oxides enhanced polyurethane based composite coating with long term corrosion resistance and self-healing property[J]. European Polymer Journal, 2024, 207: 112825. DOI: 10.1016/j.eurpolymj.2024.112825
[27] 郭洪飞, 赵增祺, 朝宝, 等. 2-6二氨基吡啶改性氧化石墨烯复合涂层的制备及防腐性能[J]. 中国表面工程, 2022, 35(2): 126-139. GUO Hongfei, ZHAO Zengqi, CHAO Bao, et al. Preparation and anticorrosion performance of 2-6 diaminopyridine modified graphene oxide composite coating[J]. China Surface Engineering, 2022, 35(2): 126-139(in Chinese).
[28] XUE B, YU M, LIU J, et al. Corrosion protection of AA2024-T3 by sol-gel film modified with graphene oxide[J]. Journal of Alloys and Compounds, 2017, 725: 84-95.
[29] PARHIZKAR N, RAMEZANZADEH B, SHAHRABI T. Corrosion protection and adhesion properties of the epoxy coating applied on the steel substrate pre-treated by a sol-gel based silane coating filled with amino and isocyanate silane functionalized graphene oxide nanosheets[J]. Applied Surface Science, 2018, 439: 45-59. DOI: 10.1016/j.apsusc.2017.12.240
[30] 楠顶, 李鑫, 徐宇, 等. 硅烷偶联剂改性氧化石墨烯增强环氧树脂复合涂料的防腐性能[J]. 化学工业与工程, 2023, 40(6): 130-135. NAN Ding, LI Xin, XU Yu, et al. Epoxy resin composite coating with silane coupling agent modified graphene oxide generating effective corrosion protection[J]. Chemical Industry and Engineering, 2023, 40(6): 130-135(in Chinese).
[31] LI M M, ZHOU D N, ZHAO B F, et al. Simultaneously improved corrosion/wear resistances of epoxy coating on Mg alloy via the coupled hybridization of GO and nano-SiO2[J]. Diamond and Related Materials, 2022, 128: 109224. DOI: 10.1016/j.diamond.2022.109224
[32] PRASETYA N B A, PUTRI M R, NGADIWIYANA, et al. Polyeugenol-gum arabic/graphene oxide composite coating for high performance anticorrosion material[J]. Case Studies in Chemical and Environmental Engineering, 2024, 9: 100658. DOI: 10.1016/j.cscee.2024.100658
[33] DADKHAH S, GHARIEH A. UV-cured acrylated PANI/graphene oxide nanocomposite coating with superior anticorrosive protection and self-healing abilities[J]. Progress in Organic Coatings, 2024, 189: 108346. DOI: 10.1016/j.porgcoat.2024.108346
[34] HUANG Y X, YANG S H, HU Y, et al. Construction of anticorrosive coatings with emergency response closure by introducing functionalized graphene oxide[J]. Chemical Engineering Journal, 2024, 487: 150539. DOI: 10.1016/j.cej.2024.150539
[35] XIE C, ZHANG P, XUE M S, et al. Long-lasting anti-corrosion of superhydrophobic coating by synergistic modification of graphene oxide with polydopamine and cerium oxide[J]. Construction and Building Materials, 2024, 418: 135283. DOI: 10.1016/j.conbuildmat.2024.135283
[36] WANG H H, YE M Y, WU J H, et al. Development of amine functionalized graphene oxide/fluorinated polyurethane topcoat with integrated anti-corrosion, anti-aging, and anti-bacterial performance[J]. Progress in Organic Coatings, 2024, 189: 108337. DOI: 10.1016/j.porgcoat.2024.108337
[37] XU C A, CHU Z Z, LI X C, et al. Vanillin and organosilicon functionalized graphene oxide modified ester resin composite coatings with excellent anti-corrosion properties[J]. Progress in Organic Coatings, 2023, 183: 107804. DOI: 10.1016/j.porgcoat.2023.107804
[38] SHU S S, WU L, LIU J M, et al. Synthesis and corrosion resistance of silane coupling agent modified graphene oxide/waterborne polyurethane[C]//Proceedings of 2019 5th International Conference on Applied Materials and Manufacturing Technology (ICAMMT 2019). Singapore: IOP Publishing Ltd., 2019, 631: 022058.
[39] WU H, CHENG L, LIU C B, et al. Engineering the interface in graphene oxide/epoxy composites using bio-based epoxy-graphene oxide nanomaterial to achieve superior anticorrosion performance[J]. Journal of Colloid and Interface Science, 2021, 587: 755-766. DOI: 10.1016/j.jcis.2020.11.035
[40] XU C A, LIANG W Z, NI C L, et al. Silicone and nano-diamond modified graphene oxide anticorrosive coating[J]. Surface & Coatings Technology, 2024, 479: 130584.
[41] ZHU J K, LI H, YANG Z Y, et al. CaIn2S4 nanosheets and SnO2 nanoflowers co-sensitized TiO2 nanotubes photoanode for continuous and efficient photocathodic protection of Q235 carbon steel[J]. Journal of Alloys and Compounds, 2024, 970: 172570. DOI: 10.1016/j.jallcom.2023.172570
[42] 陈凡伟, 刘斌, 蹇冬辉, 等. 光生阴极保护技术的研究进展及其存在的问题[J]. 材料工程, 2021, 49(12): 83-90. DOI: 10.11868/j.issn.1001-4381.2021.000469 CHEN Fanwei, LIU Bin, JIAN Donghui, et al. Research progress and existing problems of photocathodic protection technology[J]. Journal of Materials Engineering, 2021, 49(12): 83-90(in Chinese). DOI: 10.11868/j.issn.1001-4381.2021.000469
[43] HUANG W Q, CHAI Z L, ZHAO S R, et al. Design synthesis and excellent anti-corrosion property of GO/Mn-Zn2SiO4 composite materials[J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2023, 656: 130281. DOI: 10.1016/j.colsurfa.2022.130281
[44] 陈廷廷, 李波, 张小龙, 等. GO/PANI改性环氧树脂@TiO2/CdS涂层的光生阴极保护性能[J]. 微纳电子技术, 2023, 60(8): 1211-1223. CHEN Tingting, LI Bo, ZHANG Xiaolong, et al. Photogenerated cathodic protection performance of GO/PANI modified epoxy resin@TiO2/CdS coating[J]. Micronanoelectronic Technology, 2023, 60(8): 1211-1223(in Chinese).
[45] 李波, 何锦航, 余思伍, 等. TiO2/GO/EP复合涂层的光电化学防腐蚀性能研究[J]. 涂料工业, 2023, 53(1): 22-29, 36. DOI: 10.12020/j.issn.0253-4312.2022-194 LI Bo, HE Jinhang, YU Siwu, et al. Study on photoelectrochemical corrosion protection performance of TiO2/GO/EP composite coating[J]. Paint & Coatings Industry, 2023, 53(1): 22-29, 36(in Chinese). DOI: 10.12020/j.issn.0253-4312.2022-194
[46] MA L W, WANG J K, ZHAO F T, et al. Plasmon-mediated photothermal and superhydrophobic TiN-PTFE film for anti-icing/deicing applications[J]. Composites Science and Technology, 2019, 181: 107696. DOI: 10.1016/j.compscitech.2019.107696
[47] QIAN B, ZHENG Z L, MICHAILIDS M, et al. Mussel-inspired self-healing coatings based on polydopamine-coated nanocontainers for corrosion protection[J]. ACS Applied Materials & Interfaces, 2019, 10(11): 10283-10291.
[48] MARINETTI S, VAVILOV V. IR thermographic detection and characterization of hidden corrosion in metals: General analysis[J]. Corrosion Science, 2010, 52(3): 865-872. DOI: 10.1016/j.corsci.2009.11.005
[49] KHALILI P, CAWLEY P. The choice of ultrasonic inspection method for the detection of corrosion at inaccessible locations[J]. NDT & E International, 2018, 99: 80-92.
[50] SILVA M Z, GUOYON R, LEPOUTRE F. Hidden corrosion detection in aircraft aluminum structures using laser ultrasonics and wavelet transform signal analysis[J]. Ultrasonics, 2003, 414(4): 301-305.
[51] DU G, LI J, WANG W K, et al. Detection and characterization of stress-corrosion cracking on 304 stainless steel by electrochemical noise and acoustic emission techniques[J]. Corrosion Science, 2011, 53(9): 2918-2926. DOI: 10.1016/j.corsci.2011.05.030
[52] TSANGOURI E, AGGELIS D G, TITTELBOOM K V, et al. Detecting the activation of a self-healing mechanism in concrete by acoustic emission and digital image correlation[J]. The Scientific World Journal, 2013, 2013: 424560. DOI: 10.1155/2013/424560
[53] BUCHHEIT R G, GUAN H, MAHAJANAM S, et al. Active corrosion protection and corrosion sensing in chromate-free organic coatings[J]. Progress in Organic Coatings, 2003, 74(3-4): 174-182.
[54] ZHELUDKEVICH M L, TEDIM J, FERREIRA M G S. "Smart" coatings for active corrosion protection based on multi-functional micro and nanocontainers[J]. Electrochimica Acta, 2012, 82: 314-323. DOI: 10.1016/j.electacta.2012.04.095
[55] FUGOLIN A P P, FERRACANE J L, PFEIFER C S. "Fatigue-crack propagation behavior in microcapsule-containing self-healing polymeric networks"[J]. Materials & Design, 2022, 223: 111142.
[56] EXBRAYAT L, SALALUK S, UEBEL M, et al. Nanosensors for monitoring early stages of metallic corrosion[J]. ACS Applied Energy Materials, 2019, 2(2): 812-818.
[57] HU M H, PEIL S, XING Y W, et al. Monitoring crack appearance and healing in coatings with damage self-reporting nanocapsules[J]. Materials Horizons, 2018, 5: 51-58.
[58] MAIA F, TEDIM J, BASTOS A C, et al. Nanocontainer-based corrosion sensing coating[J]. Nanotechnology, 2013, 24(41): 415502. DOI: 10.1088/0957-4484/24/41/415502
[59] ZHENG X, WANG Q, LI Y, et al. Microcapsule-based visualization smart sensors for damage detection: Principles and applications[J]. Advanced Materials Technologies, 2019, 52: 1900832.
[60] LIU J G, HUANG W R, ZHANG K L, et al. Early warning and self-repair properties of o-phenanthroline modified graphene oxide anti-corrosion coating[J]. Progress in Organic Coatings, 2024, 189: 108274. DOI: 10.1016/j.porgcoat.2024.108274
[61] LI Y F, NING C Y. Latest research progress of marine microbiological corrosion and bio-fouling, and new approaches of marine anti-corrosion and anti-fouling[J]. Bioactive Materials, 2019, 4: 189-195. DOI: 10.1016/j.bioactmat.2019.04.003
[62] MELCHERS R E. Microbiological and abiotic processes in modelling longer-term marine corrosion of steel[J]. Bioelectrochemistry, 2014, 97: 89-96. DOI: 10.1016/j.bioelechem.2013.07.002
[63] LI Z C, LIU P, CHEN S W, et al. Bioinspired marine antifouling coatings: Antifouling mechanisms, design strategies and application feasibility studies[J]. European Polymer Journal, 2023, 190: 111997. DOI: 10.1016/j.eurpolymj.2023.111997
[64] LEONARDI A K, OBER C K. Polymer-based marine antifouling and fouling release surfaces: Strategies for synthesis and modification[J]. Annual Review of Chemical and Biomolecular Engineering, 2019, 10: 241-264. DOI: 10.1146/annurev-chembioeng-060718-030401
[65] XIE Q Y, PAN J S, MA C F, et al. Dynamic surface antifouling: Mechanism and systems[J]. Soft Matter, 2019, 15(6): 1087-1107.
[66] BEYER J, SONG Y, TOLLEFSEN K E, et al. The ecotoxicology of marine tributyltin (TBT) hotspots: A review[J]. Marine Environmental Research, 2022, 179: 105689. DOI: 10.1016/j.marenvres.2022.105689
[67] HU P, XIE Q Y, MA C F, et al. Silicone-based fouling-release coatings for marine antifouling[J]. Langmuir, 2022, 36(9): 2170-2183.
[68] LI C, YANG J J, HE W J, et al. A review on fabrication and application of tunable hybrid micro-nano array surfaces[J]. Advanced Materials Interfaces, 2023, 10(6): 2202160.
[69] ZHENG N, JIA B, LIU J, et al. Multi-strategy combined bionic anti-fouling coating for long-term and stable protection against marine biofouling[J]. Journal of Materials Science and Technology, 2025, 210: 265-277.
-
目的
氧化石墨烯(GO)作为石墨烯的衍生物具有高比表面积、良好的机械性能、阻隔屏蔽性、反应活性、热稳定性及化学稳定性等优异的综合性能,在金属防腐蚀领域中表现出了巨大的应用潜力。本文综述了近年来国内外关于GO复合涂层在金属腐蚀与防护领域的研究进展,同时也对GO复合涂层未来发展的趋势进行了展望。
方法本文以GO复合涂层为中心,分别从GO-无机物复合涂层、GO-有机物复合涂层、GO多元复合涂层三个方面展开,将GO作为涂层填料相对于其他常见无机填料的优缺点进行了对比,同时对国内外现有GO复合涂层在制备方法、掺杂成分、GO掺杂浓度、涂层耐蚀性提高等方面取得的进展进行了归纳总结。
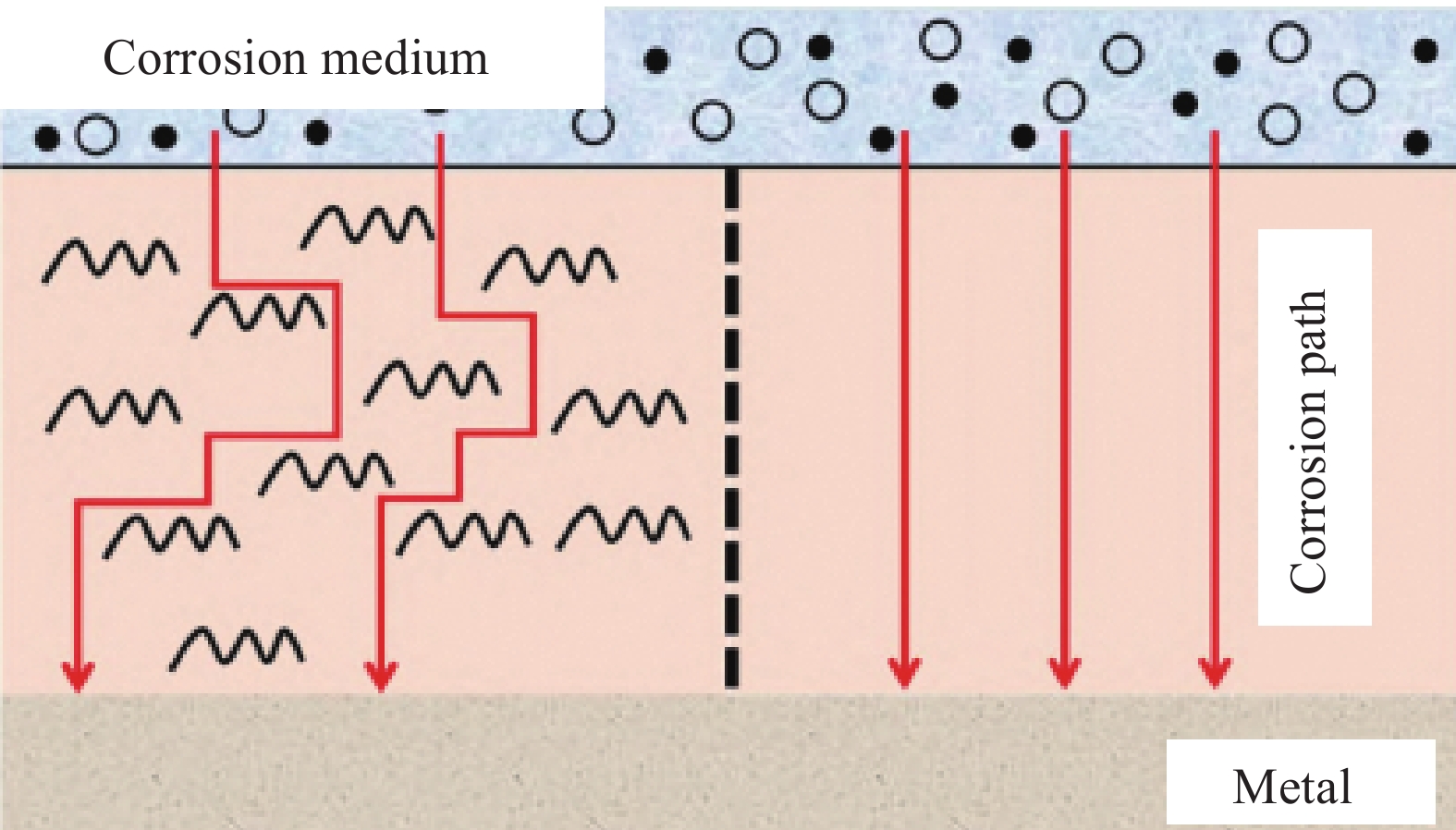
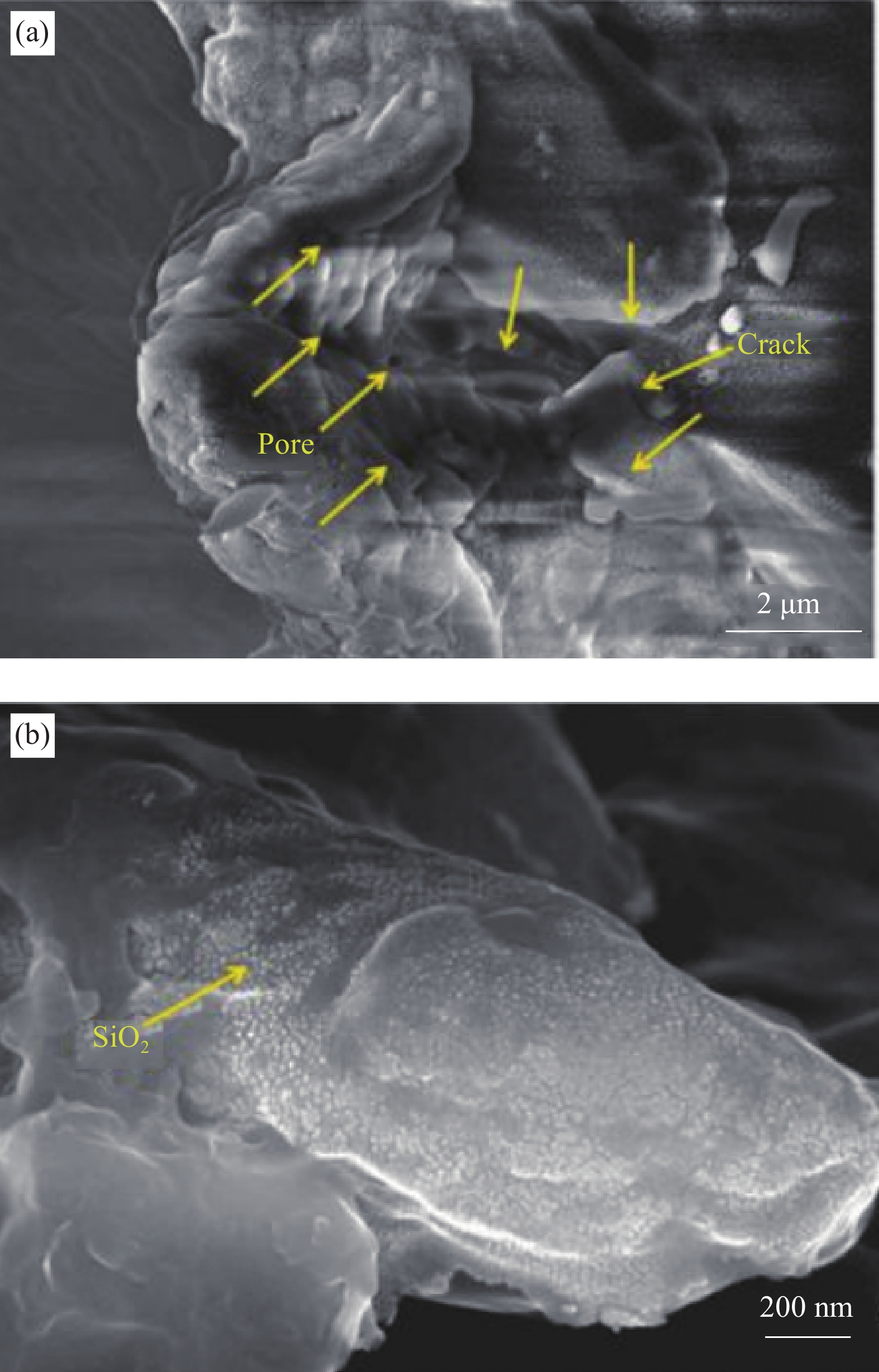
结果GO掺杂在不同的复合涂层中的作用机理、涂层优势及不足之处各不相同。(1)GO与无机物的复合机理主要是通过分子间氢键及静电力的作用,GO在其中起到的是物理阻隔增加腐蚀介质渗透路径及细化无机粒子晶粒的作用。但GO表面的含氧官能团能够提供大量的反应结合位点这一优势不能被充分的利用,而且由于范德华力及p-p键的相互作用,GO作为填料在无机物中很容易团聚,导致分散性和稳定性都降低,“迷宫效应”无法充分发挥,这些都导致涂层的耐蚀性受到一定限制。(2)相较于无机物,GO与有机物复合时,更能发挥出其自身的优异性能。GO的分散性使它能够填补有机高分子材料固化和成膜过程中出现的孔隙;GO的含氧官能团与有机物能够通过反应形成共价键,对有机物的化学性能和力学性能均有所改变,同时保持着GO原有的物理阻隔性能这一优点。但仍有一些不足之处存在其中。首先,GO易团聚依然是GO-有机物复合涂层耐蚀性提高的关键制约因素之一。其次,在涂层制备过程中,有机溶剂的挥发及不合理的固化方式都会导致传统的聚合物涂层在固化后,不仅会有微孔、微裂纹等缺陷的产生,而且残余的亲水基团或表面活性剂易形成极性孔道。这些缺陷及孔道会为腐蚀介质的入侵提供便捷途径,加速腐蚀介质的吸收和渗透,导致涂层的耐蚀性变差。最后,GO具有一定的导电性,在涂层缺陷的位置容易发生局部电偶腐蚀。(3)GO多元复合涂层不仅使GO的分散性有一定提高,而且其自身的可结合反应位点的利用率也大大提高。关于GO复合涂层的最新研究聚焦在以下几个方面:(1)改善GO团聚性;(2)GO光阴极保护;(3)自预警自修复GO复合涂层。
结论针对目前GO复合涂层存在的问题,未来的重点研究方向应集中在:(1)开发在耐高、低温领域可应用的GO复合涂层;(2)探索新的改性方法对复合涂层中的GO绝缘处理;(3)减少GO智能复合涂层自修复时间,提高自修复效率。





 下载:
下载: