Research progress on biomass and biomass-related hemostatic materials
-
摘要:
伤口的快速止血和愈合对于解决意外事故造成的出血具有重要意义,相关止血材料的开发和应用一直备受关注。以角蛋白、丝素蛋白、胶原蛋白为代表的蛋白类和以纤维素、壳聚糖、海藻酸为代表的多糖类等生物质材料,因其无毒性、低抗原性、良好的生物相容性、生物可降解性等优点在止血领域展现了前所未有的应用价值。基于此,本文对生物质止血材料的设计、制备及止血应用的最新研究进展进行了全面综述,并对其发展前景做了展望,以期为新型高效止血材料的开发和实际应用提供思路。
Abstract:The rapid hemostasis and healing of wounds after trauma hold great significance to solve the bleeding caused by accidents. Therefore, the development and application of related hemostatic materials have attracted much attention. Biomass-derived materials such as proteins including keratin, silk fibroin, collagen and polysaccharides including cellulose, chitosan, and alginic acid are favored by researchers because of their non-toxicity, low antigenicity, good biocompatibility, and biodegradability, and have shown unprecedented application value in the field of hemostasis. In this paper, the latest research results on the design, the preparation and the application of biomass hemostatic materials are comprehensively reviewed, and their development prospects are prospected, which will provide ideas for the development and practical application of new high-efficiency hemostatic materials.
-
Keywords:
- biomass materials /
- protein /
- polysaccharide /
- hemostasis properties /
- wound hemostasis
-
四环素广泛应用在医疗救治、农业生产、畜牧业管理以及水产养殖等多个关键领域[1-4]。然而,四环素所拥有的高稳定化学性质,使其在生物体内吸收和代谢效率低下,造成大量抗生素残留物质在自然环境中积聚,传统的处理技术往往难以达到优异的去除效果。光催化技术降解有机污染物技术因其能够实现高效转化、绿色环保、运行稳定等一系列优越特性,受到了科研界的广泛关注与推崇,被认为是未来解决环境污染问题最具潜力的技术路径之一[5-7]。
晶态红磷(cRP)作为一种廉价、易得且低毒的半导体材料,其结构特性赋予了它良好的光催化性能和可见光响应在光催化领域展现出了独特的优势[6, 8-10]。在光的照射下,当光子的能量大于禁带间隙时,红磷价带上的光生电子实现跃迁,到导带上产生光生空穴,实现电荷分离。随后,红磷表面的光生电子被捕获并生成具有超强还原性的•O−2。尽管红磷具有实际应用的潜力,但其催化活性受限于导电性差、光生电子空穴复合较快、活性位点少等不足。通过改进制备方法、优化光催化剂的结构以及与其他材料进行复合等方式,有望进一步提升红磷的光催化性能。如Liu等[11]通过超声剥离将晶态红磷从块体转化成了纤维状结构,提高了红磷的接触面积。Shen等[12]通过在制备时加入Si片的方法控制合成不同类型的晶态红磷,优化了晶态红磷的结构。
构建异质结是将光催化剂与相匹配的其他材料结合,是优化光催化性能的一种常用且有效的方法[13-14]。通过此方法可以得到具有更多活性位点和自由基产生的光催化剂[15],可以提高红磷光催化剂的氧化能力和还原能力,使其能够更有效地降解污染物。在构建异质结时,需要选择与红磷带隙结构相匹配的材料,以确保两者之间能够形成紧密的结合,并实现有效的电子传输和分离。
金属有机骨架(Metal-organic frameworks,MOFs)作为一种新兴的微介孔杂化材料,以其独特且丰富的结构特征,在光催化领域成为了研究热点。在MOFs材料中MIL[15-17]系列、IRMOF[18-21]系列、ZIF[18-21]系列、UiO[22-28]系列和PCN系列等,都以其独特的结构和性能在各个领域发挥着重要作用。在MOFs中,铁基MOFs (MIL家族)以其孔隙率大、比表面积大、成本效益高和环境友好等特性,在光催化领域引起了广泛关注。然而, MOFs材料在水热条件下的稳定性较差,这在一定程度上限制了其在光催化领域的应用。如2020年,Lei等[15]采用简单的低温溶剂热法制备了MIL-101(Fe)与非金属红磷的Z型异质结复合材料,表现出了不错的光催化性能。
本文结合晶态红磷纳米带(cRP NRs)和MIL-101-NH2两者的优点,通过构造异质结的方法将其进行复合,来提高光催化性能以及材料稳定性,以进一步实现对有机污染物的高效降解。首先制备出晶态红磷纳米带,随后再以2-氨基对苯二甲酸为配体制备MIL-101-NH2铁基MOF材料。然后将cRP NRs与MIL-101-NH2通过溶剂热法制备出复合材料。为了确定该复合材料的成功合成,本文通过SEM、TEM和XRD等表征来对复合材料进行了验证,并对其形貌结构进行了深入分析。然后在光催化降解四环素中的实验中确定了复合材料的最佳比例和其优秀的降解效果。此外,本文还通过XPS表征分析电子在复合材料中的转移情况,通过固体紫外吸收光谱和Mott-Schottky曲线得到复合材料的禁带宽度和价带导带信息,确定了复合材料形成的异质结类型。通过上述工作,可以充分地证明cRP NRs/MIL-101-NH2这种新型复合材料在光催化降解中优秀的性能和出色的稳定性,并为之后制备催化效果能加优异的复合材料提供了更多的选择。
1. 实验部分
1.1 原材料
商业红磷(Red phosphorus,RP)、I2 (Iodine)、FeCl3·6 H2O、2-氨基对苯二甲酸(2-aminoterephthalic acid)、硝酸银(Silver nitrate,AgNO3)、氢氟酸(Hydrofluoric acid,HF)、甲基橙(Methyl orange,MO)和四环素(Tetracycline,TC)均从上海阿拉丁生化科技股份有限公司采购。所使用的商业硅片来自浙江立晶,为p-Si (100)型。
1.2 表征仪器
采用美国赛默飞FEI Inspect F50扫描电子显微镜进行形貌测试。该仪器配备了EDAX超辛烷值检测器,在测试过程中,设置了扫描范围为500 nm至10 μm。采用了美国赛默飞FEI Talos F200S透射电镜仪器进行分析。在测试过程中,设定了扫描范围为5 nm至1 μm。选取需要测试的样品,随后将其加入无水乙醇中,利用超声波将溶液中的样品充分分散。分散完成后,将溶液中的样品转移到超薄铜网上。经过调校后,对样品进行形貌记录。包括样品的晶格排列、界面形态以及元素分布等关键信息。
X’Pert PRO衍射仪采用了日本Rigaku Ultima IV。使用Cu 靶,Kα辐射波长为λ=0.15406 nm,以40 kV作为工作电压和30 mA的电流作为工作电流,让衍射仪在5°~50°的扫描角范围内,以1°/min的扫描速度去对样品进行扫描。
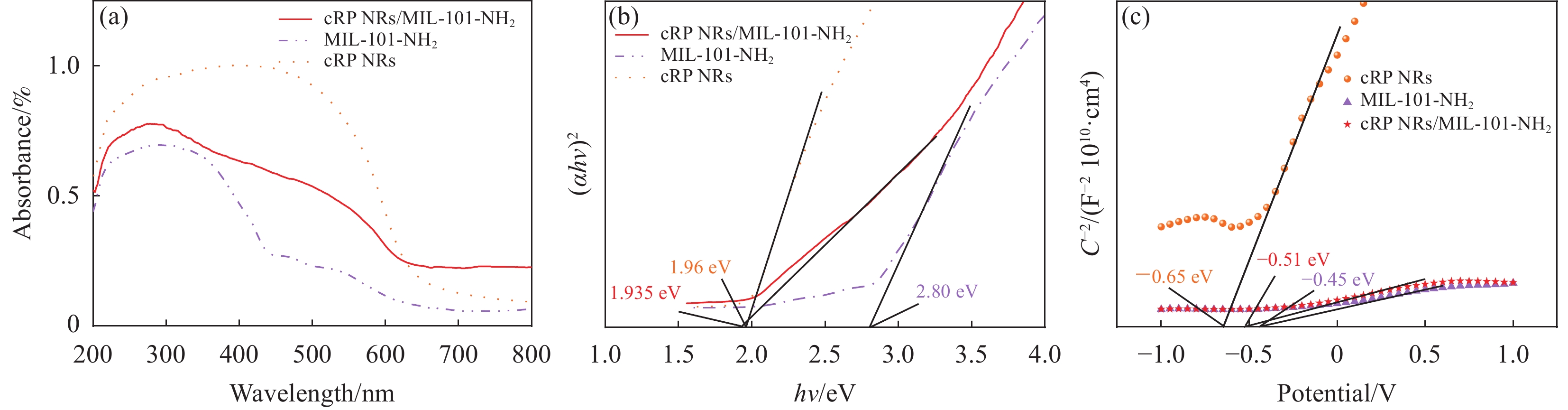
使用UV-3600 (Shimadzu,日本)测试样品的吸光度,测试范围为200~800 nm,先在样品槽中加入适量的硫酸钡粉末作为背景测试基线后,再将待测样品制成薄片或溶解后进行吸光度测试。并通过下式得到Tauc's图,通过绘制曲线斜率最大处切线与x轴相交所得的横坐标可以得到各个材料的禁带宽度。
(αhv)1n=K(hv−Eg) (1) 其中:α为吸光度;v为频率;h为普朗克常数6.63×10−34 J·s;K为常数;Eg为禁带宽度。因为红磷为直接带隙的半导体,所以n=1/2。
本次实验中使用的X射线光电子能谱检测仪是美国赛默飞的K-ALPHA仪器,测量的表层深度为10 nm。最终的数据都经过C1s 峰值的284.8 eV来进行校准。
采用上海辰华CHI 660E电化学工作站,对样品进行光电性能表征。选用了0.5 mol/L的Na2SO4溶液作为电解液,以铂网作为辅助电极,Ag/AgCl电极为参比电极,对样品进行瞬态光电流测试和Mott-Schottky曲线测试。
1.3 cRP NRs的制备
首先在石英管中加入600 mg商用红磷和40 mg的I2,将经过刻蚀干燥后的硅晶片放置于石英管中并开始抽取真空,在压力表显示达到−0.03 MPa的真空压力后,封装石英管。
随后,将封装完毕的石英管放入马弗炉(MF-1200C-M,安徽贝意克设备技术有限公司)中,设定升温速率控制在5℃/min,从室温逐渐升温至560℃,并在此温度下稳定保持20 h,以确保红磷与硅晶片之间的充分反应。之后,再次以5℃/min的降温速率控制冷却过程,直至炉温降至室温。
待石英管冷却后将其敲碎,从硅片将红磷刮取收集。接着,将刮下的cRP放入研钵中,进行30 min的充分研磨。随后,利用乙醇对研磨后的红磷进行彻底洗涤,以去除杂质。经过真空泵的抽滤后,将红磷放置在电鼓风干燥箱(DHG-9070A,上海一恒科学仪器有限公司)中,设置温度为40℃,干燥2 h,以去除残余的乙醇并确保红磷的干燥状态。
接着称取300 mg的cRP,并将其均匀分散在150 mL的无水乙醇中,以确保红磷颗粒在溶剂中的充分分散。随后,使用了超声波细胞粉碎机(Scientz-1500F,中国宁波新芝生物科技有限公司),并设置了特定的操作参数:功率为40%,脉冲周期为6 s开启,8 s关闭。超声处理完成后,首先将悬浊液转移至离心管中,并以
3000 r/min的转速离心10 min。这一步骤的目的是初步分离出较大颗粒的固体物质,留下含有纤维状红磷的液相。接着,将液相进行第二次离心,转速提高至11000 r/min,再次离心10 min,以进一步去除残留的清液和细小颗粒。最后,将材料放入冷冻干燥机(SCIENTZ-10N,中国宁波新芝生物科技有限公司)中,以40℃的温度干燥24 h,以此得到cRP NRs。1.4 MIL-101-NH2的制备
采用溶剂热法制备了MIL-101-NH2。首先将FeCl3·6H2O (10 mmoL)和2-氨基对苯二甲酸(12.5 mmoL)同时溶解于50 mL DMF中。随后,将上述两种溶液在磁力搅拌下混合均匀,并转移到100 mL特氟龙内衬高压釜(KH100,西安常仪仪器设备有限公司)中。在110℃反应20 h。反应结束后,
11000 r/min离心5 min收集所得颗粒,再用DMF和乙醇洗涤3次,放入真空干燥箱在70℃下真空干燥12 h,得到目标MIL-101-NH2。1.5 复合材料的制备
采用溶剂热法制备了cRP NRs/MIL-101-NH2。分别称取50 mg的cRP NRs和为cRP NRs质量50%、75% 、100% 和125%的MIL-101-NH2放入4个反应釜中(4个反应釜中cRP NRs和MIL-101-NH2的质量比例为2∶1、4∶3、1∶1和4∶5),并加入50 mL 无水乙醇。随后将上述两种材料在磁力搅拌下混合均匀,在110℃反应20 h。反应结束后,
11000 r/min离心5 min收集所得颗粒,再用乙醇洗涤3次,放入真空干燥箱在60℃下真空干燥12 h,得到4种比例复合的cRP NRs/MIL-101-NH2。1.6 光催化实验
本文选取四环素(TC)作为研究对象进行了光催化降解实验。在反应管中加入了10 mg的催化剂,随后加入了50 mL的TC (50 mg/L)水溶液。在开始光催化反应之前,首先设置了30 min的暗反应阶段,以便让催化剂与污染物之间建立起稳定的吸附平衡。暗反应阶段结束后,打开500 W氙灯照射120 min。在整个实验期间每隔15 min便采集4 mL的样品,并通过离心分离得到上清液。最后,利用紫外-可见光分光光度计(UV-1800PC,上海美普达仪器有限公司)在357 nm处测定上清液的吸光度。通过测量该波长的吸光度,能够准确地计算出TC在水溶液中的降解情况。
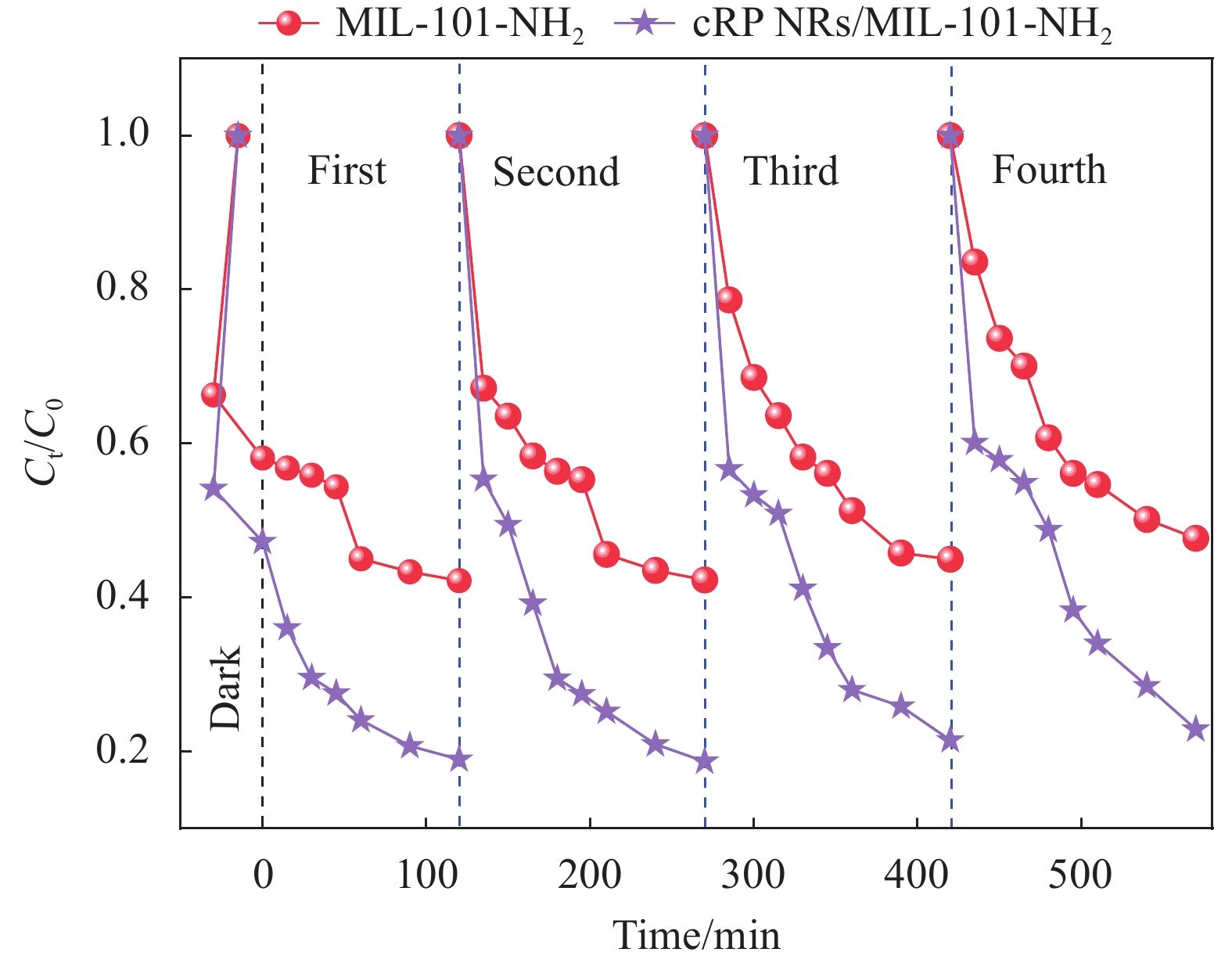
为了评估催化剂的稳定性,进行了循环实验,该实验与降解实验的操作步骤基本一致。收集离心后的催化剂。使用真空泵对催化剂进行抽滤,以去除残留的水分和溶液。将抽滤后的催化剂放入鼓风干燥箱中,在60℃下干燥30 min,以去除残余的水分。将干燥好的催化剂重新放入新的TC溶液中,并重复上述实验步骤。如此反复进行4次。
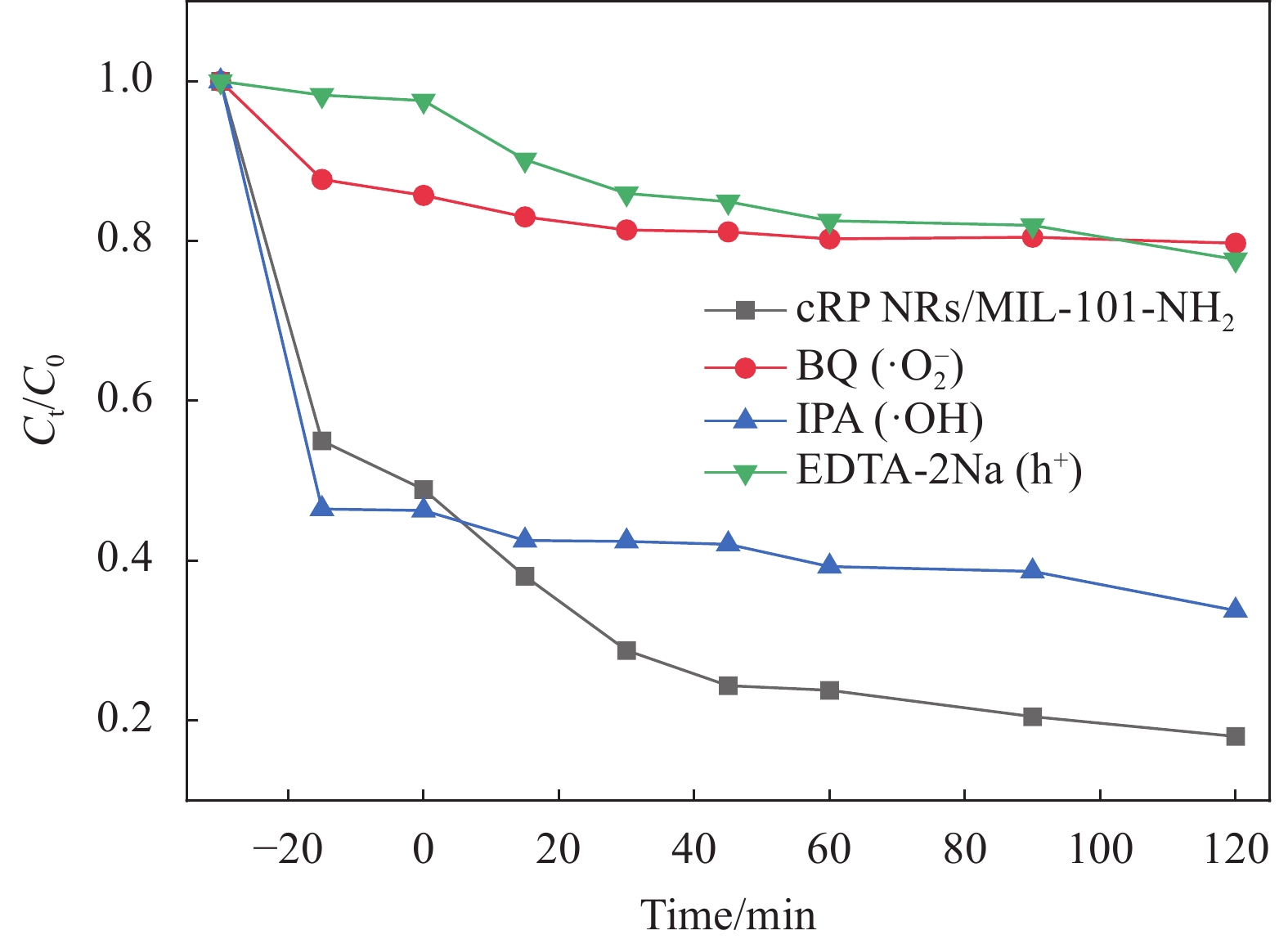
实验设计将催化剂加入50 mg/L的TC的溶液中,并引入不同的添加剂以观察它们对催化过程的影响,同时设置不添加捕获剂的实验作为对照试验,从而分析出本研究制备的催化剂的催化机制。分别向溶液中加入苯醌(BQ)、异丙醇(IPA)和乙二胺四乙酸钠(EDTA-2Na)。每种添加剂的加入量根据其抑制特定反应步骤的能力来确定,通过预实验来确定最佳添加量。将采集的样品立即离心,取上清液进行后续分析。例如,如果BQ显著抑制了催化反应,这可能表明超氧自由基(•O−2)在催化过程中起到了关键作用;如果IPA或EDTA-2Na对反应有明显影响,则可能涉及羟基自由基(•OH)或空穴(h+)在催化过程中的作用。
根据有机溶液在特定光长下的吸光度,可以得到样品在不同时间的浓度,根据降解效率的计算公式可以得到该时间的降解效率。根据Lambert-Beer law,可得下式:
Φ=C0−CtC0×100%=A0−AtA0×100% (2) 其中:Φ为降解效率;C0为初始浓度;Ct为t时刻溶液浓度;A0为初始溶液吸光度;At为t时刻溶液吸光度。可以计算出在某个时刻t的溶液浓度,并根据所得浓度做出降解效率曲线。
2. 结果与讨论
2.1 样品的形貌与结构表征
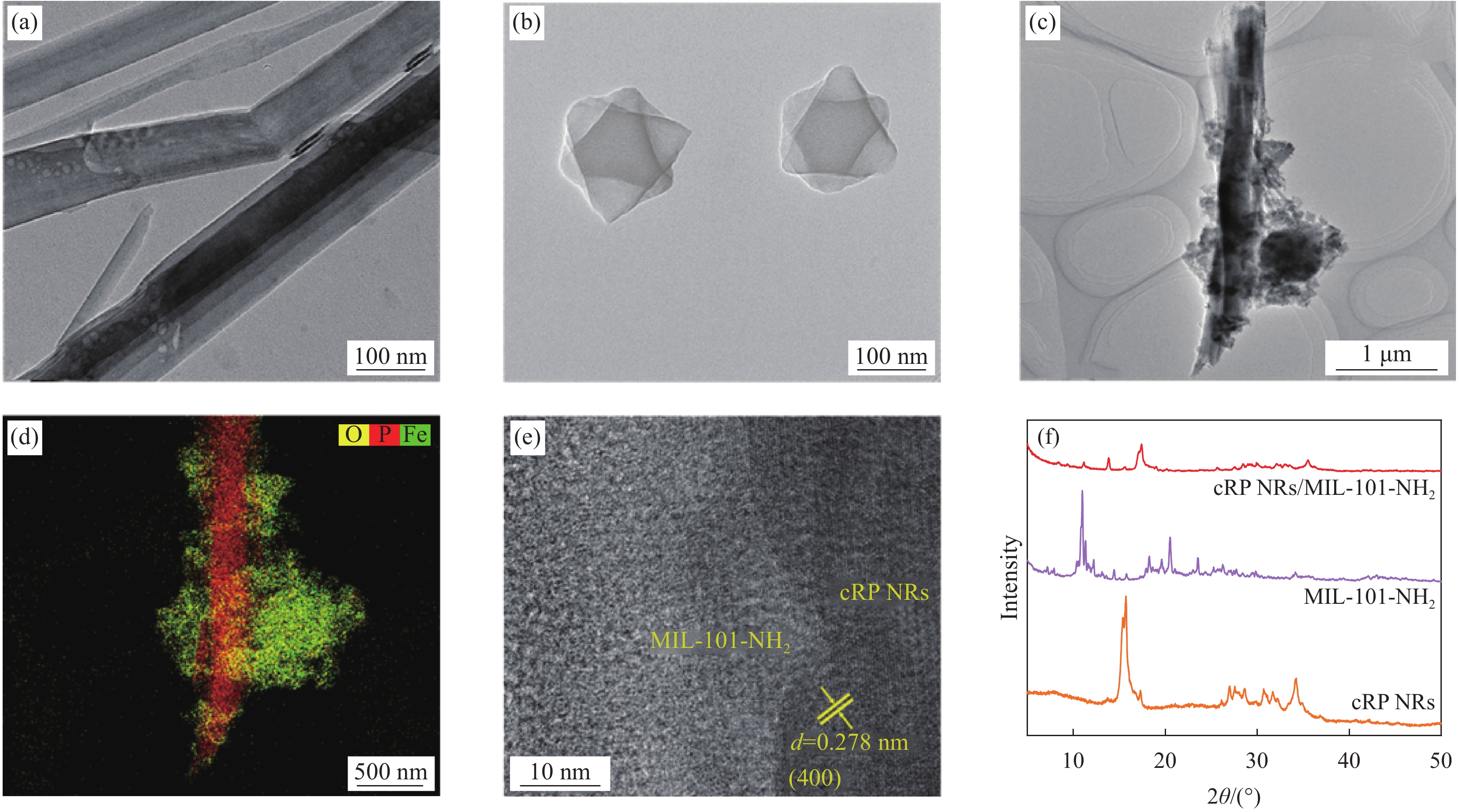
从图1中的SEM图像可以观察到cRP NRs整体为纤维状结构。MIL-101-NH2铁基金属框架催化剂为较为规则的多面体的晶粒。这些晶粒的形状较为规则,但是大小分布并不均匀。材料表面存在一些细小的孔隙。这些孔隙MOFs是材料本身的特性之一,其存在对于材料的相容性和应用性能有着重要影响。通过观察图1(c),可以清晰地观察到MIL-101-NH2分布于纤维状红磷及红磷纳米带的各个角落,并且紧紧贴合在纳米带的表面。
为了进一步深入分析材料的复合状态,采用了TEM透射电镜对MIL-101-NH2与cRP NRs复合后的形态进行了表征。在图2(b)中,MIL-101-NH2所展现的形态与之前在SEM扫描电镜下的观察结果保持了一致性。同时也可以更加清晰地看到MIL-101-NH2与cRP NRs之间紧密的贴合。在图2(e)中,可以清晰地观察到cRP NRs所特有的0.278 nm的(400)晶面的晶格条纹,呈现出了其独特的晶体结构[11-12]。而与之相对,MIL-101-NH2则呈现出无晶格条纹的状态,展示出其不同于cRP的另一种物质特性。在高分辨率透射电镜的元素分布扫描中,对P、O、Fe 3种元素进行了详尽的元素分布面谱分析。结果显示,Fe元素在复合材料中将红磷包裹,这种分布模式进一步证实了铁基MOF与红磷之间的紧密结合。而O元素的存在则暗示了复合材料中可能存在的氧化情况。通过这一系列的TEM观测和分析,进一步证实了新型cRP NRs/MIL-101-NH2的成功合成。
![]() 图 2 ((a)~(c)) cRP NRs、MIL-101-NH2和cRP NRs/MIL-101-NH2的TEM图像;(d) cRP NRs/MIL-101-NH2的元素分布;(e) cRP NRs/MIL-101-NH2的晶格条纹图像;(f) cRP NRs、MIL-101-NH2和 cRP NRs/MIL-101-NH2的XRD图谱Figure 2. ((a)-(c)) TEM images of cRP NRs, MIL-101-NH2 and cRP NRs/MIL-101-NH2; (d) Elemental distribution of cRP NRs/MIL-101-NH2; (e) Lattice fringe images of cRP NRs/MIL-101-NH2; (f) XRD patterns of cRP NRs, MIL-101-NH2 and cRP NRs/MIL-101-NH2d—Diameter
图 2 ((a)~(c)) cRP NRs、MIL-101-NH2和cRP NRs/MIL-101-NH2的TEM图像;(d) cRP NRs/MIL-101-NH2的元素分布;(e) cRP NRs/MIL-101-NH2的晶格条纹图像;(f) cRP NRs、MIL-101-NH2和 cRP NRs/MIL-101-NH2的XRD图谱Figure 2. ((a)-(c)) TEM images of cRP NRs, MIL-101-NH2 and cRP NRs/MIL-101-NH2; (d) Elemental distribution of cRP NRs/MIL-101-NH2; (e) Lattice fringe images of cRP NRs/MIL-101-NH2; (f) XRD patterns of cRP NRs, MIL-101-NH2 and cRP NRs/MIL-101-NH2d—Diameter为了探究合成样品的结晶完整度和合成情况我们进行了XRD测试。从图2(f)中可以清楚的看到, cRP NRs与MIL-101-NH2制备出的新型复合材料分别对比两种原材料的XRD图,新型复合材料的主要特征峰与cRP和cRP NRs在角度为15°和16°处红磷的(001)和(200)晶面的特征峰虽然发生了些许偏移但是其峰的形状基本一致[11]。而MIL-101-NH2的峰同样出现,但是峰的高度大幅减小,证明了复合材料的成功合成,也说明了在新型复合材料中红磷占据了主体地位。
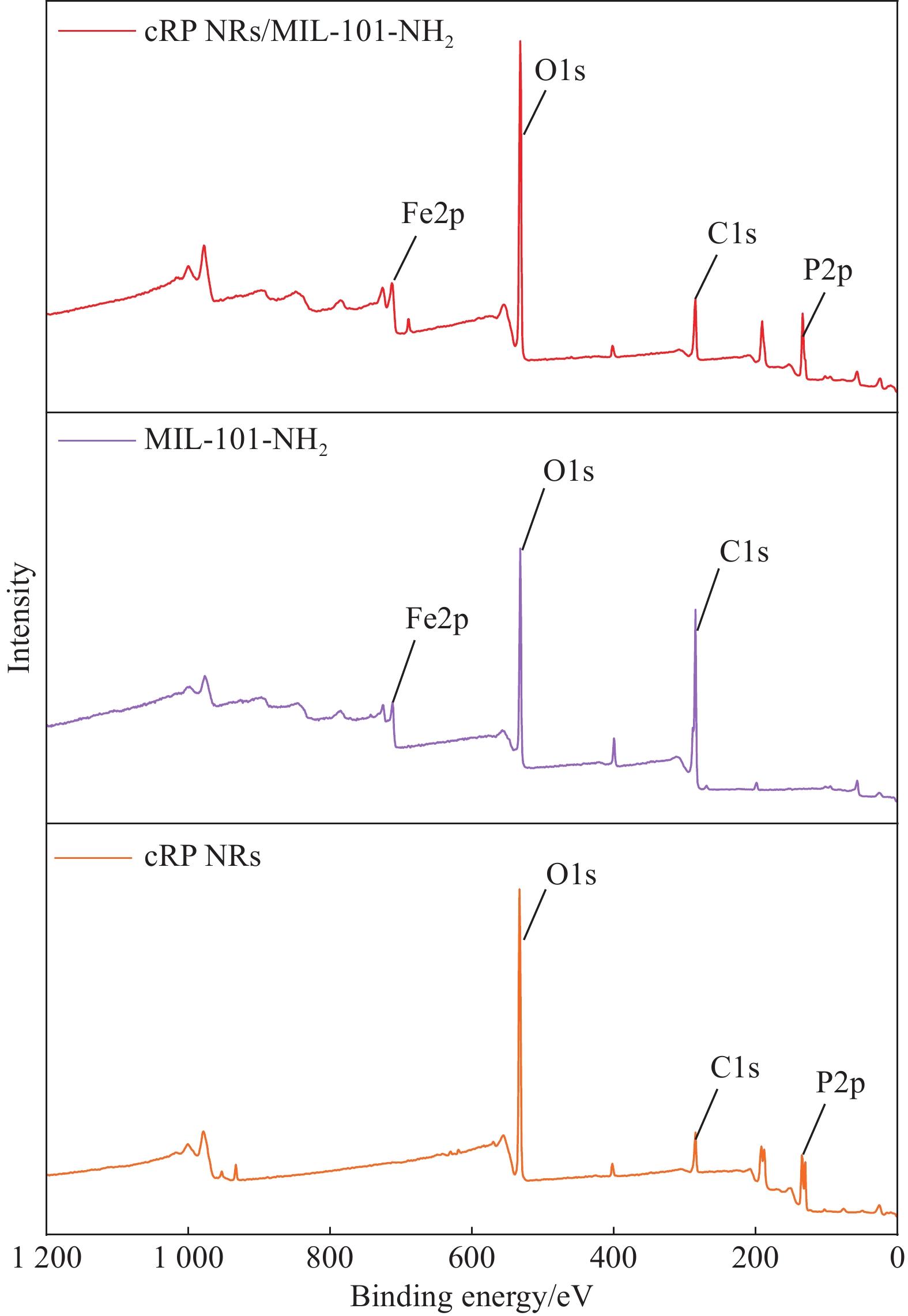
图3为cRP NRs、MIL-101-NH2和 MIL-101-NH2的XPS全谱图。从图中观察到,cRP NRs/MIL-101-NH2的XPS全元素谱中显示铁、氧、碳、磷几种元素共存,出现了在cRP NRs和MIL-101-NH2上没有的峰,证明了cRP NRs与MIL-101-NH2的成功结合。为了明确cRP NRs/MIL-101-NH2复合材料中的元素价态和表面电子转移情况,如图4中进一步对4种元素进行了XPS分析。
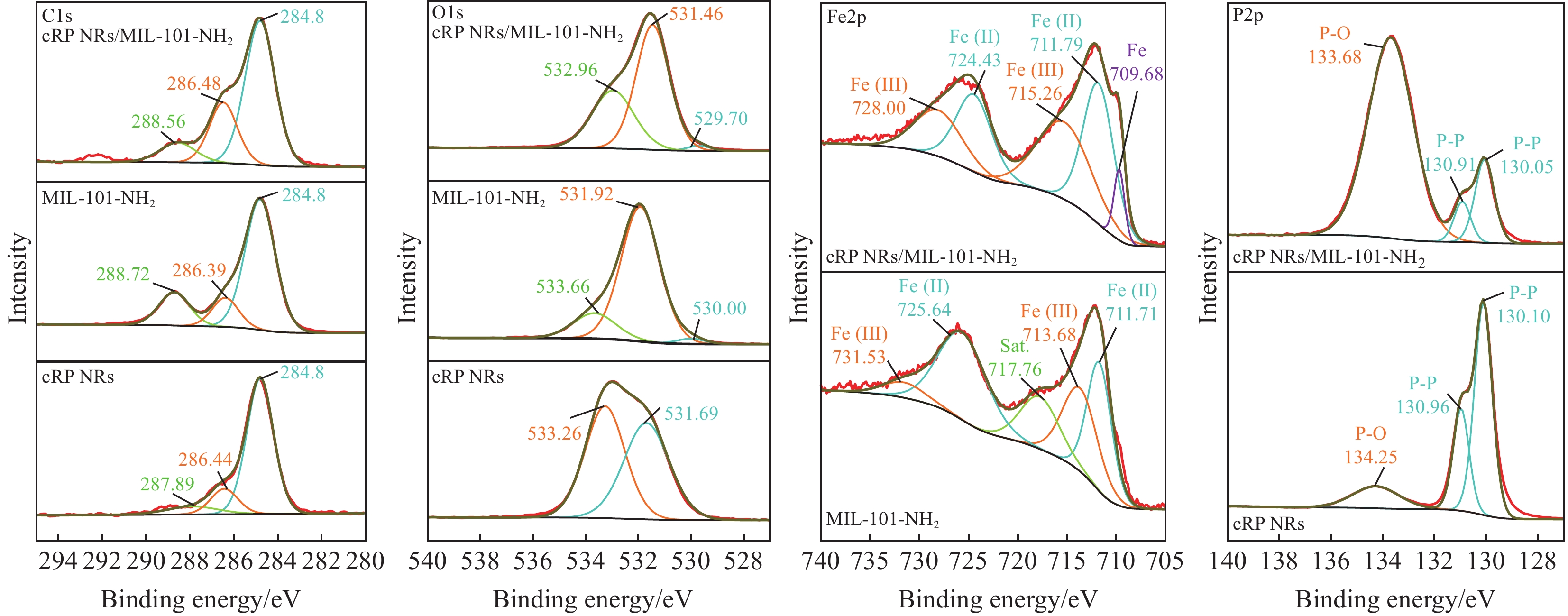
cRP NRs/MIL-101-NH2在图4的C元素谱中的C1s在284.8 eV处有一个强烈的峰为C—C峰,在286.44 eV、286.39 eV 和286.48 eV处分别对应着cRP NRs、MIL-101-NH2材料和复合材料的C—O峰,287.89 eV、288.72 eV和288.56 eV 对应着O—C=O的峰[29-30]。在O1s光谱中,可以看到,位于530.00 eV和529.70 eV对应的是Fe—O键,在cRP NRs的氧元素光谱中并未有对应的峰,而复合后则出现说明了MIL-101-NH2和cRP NRs的成功复合,而后面出现在533.18 eV和532.84 eV处的峰则代表了有机配体中的O和氧空位上吸附的H2O[30]。
样品中氧空位的形成是由于溶剂热过程中溶剂分子的蒸发,样品中的Fe2+必须通过除去晶格氧来平衡电荷。这种电荷平衡的趋势导致晶格氧附近的电子密度增加,从而削弱了复合材料中的Fe—O键。在P2p光谱中有3个特征峰,130.1 eV和130.96 eV处的峰分配给P2p1/2和P2p3/2代表P—P键,134.25 eV处的峰与五氧化二磷(P2O5)的典型波段相对应。在P2p光谱中对应的cRP NRs/MIL-101-NH2复合材料中,P—P键的峰的位置变成了130.05 eV和130.91 eV,其位置也发生了右移,说明复合材料中P元素因结合能的降低,存在电子密度增加情况。再观察Fe2p对应的Fe元素光谱,在MIL-101-NH2中711.71 eV和725.64 eV代表了二价Fe2+ ,在713.68 eV和731.53 eV代表着三价Fe3+,717.76 eV处为卫星峰[13]。而复合材料在711.79 eV处所对应的Fe2+的峰发生了左移,说明复合材料中存在因Fe元素结合能的上升,而导致电子密度下降的情况。
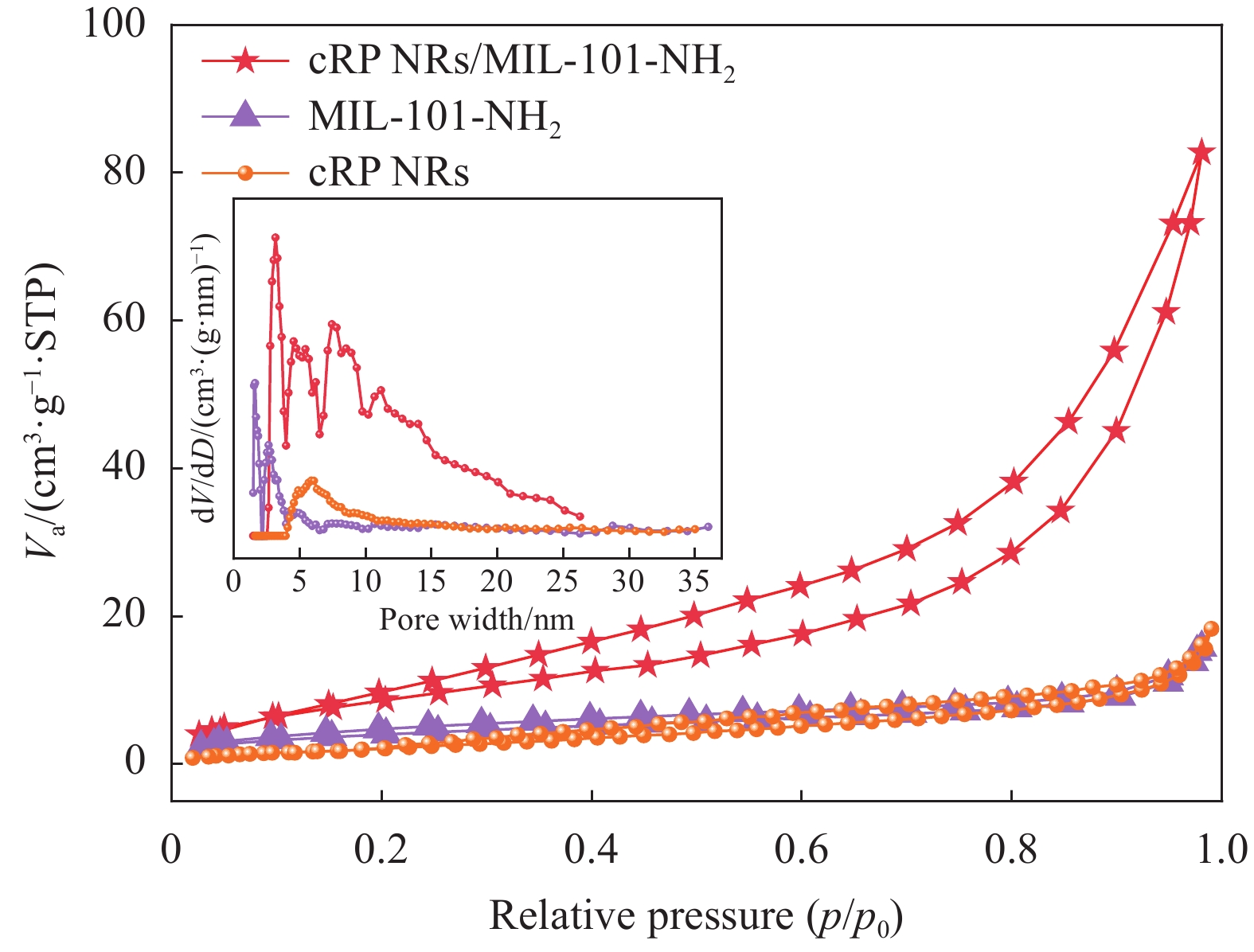
为了进一步明确cRP NRs负载对复合材料比表面积的影响,对样品进行了N2吸附-解吸测定。在图5中,cRP NRs/MIL-101-NH2等温线为与微孔结构相关的I型性质曲线。
比表面积的排序如下:cRP NRs/MIL-101-NH2(33.547 m2/g) > MIL-101-NH2(14.644 m2/g) > cRP NRs(11.065 m2/g),可以发现复合材料的比表面积高于复合前材料的比表面积。由此可以推断,复合材料对污染物具有更好的吸附和降解效果。
2.2 样品的光学性质
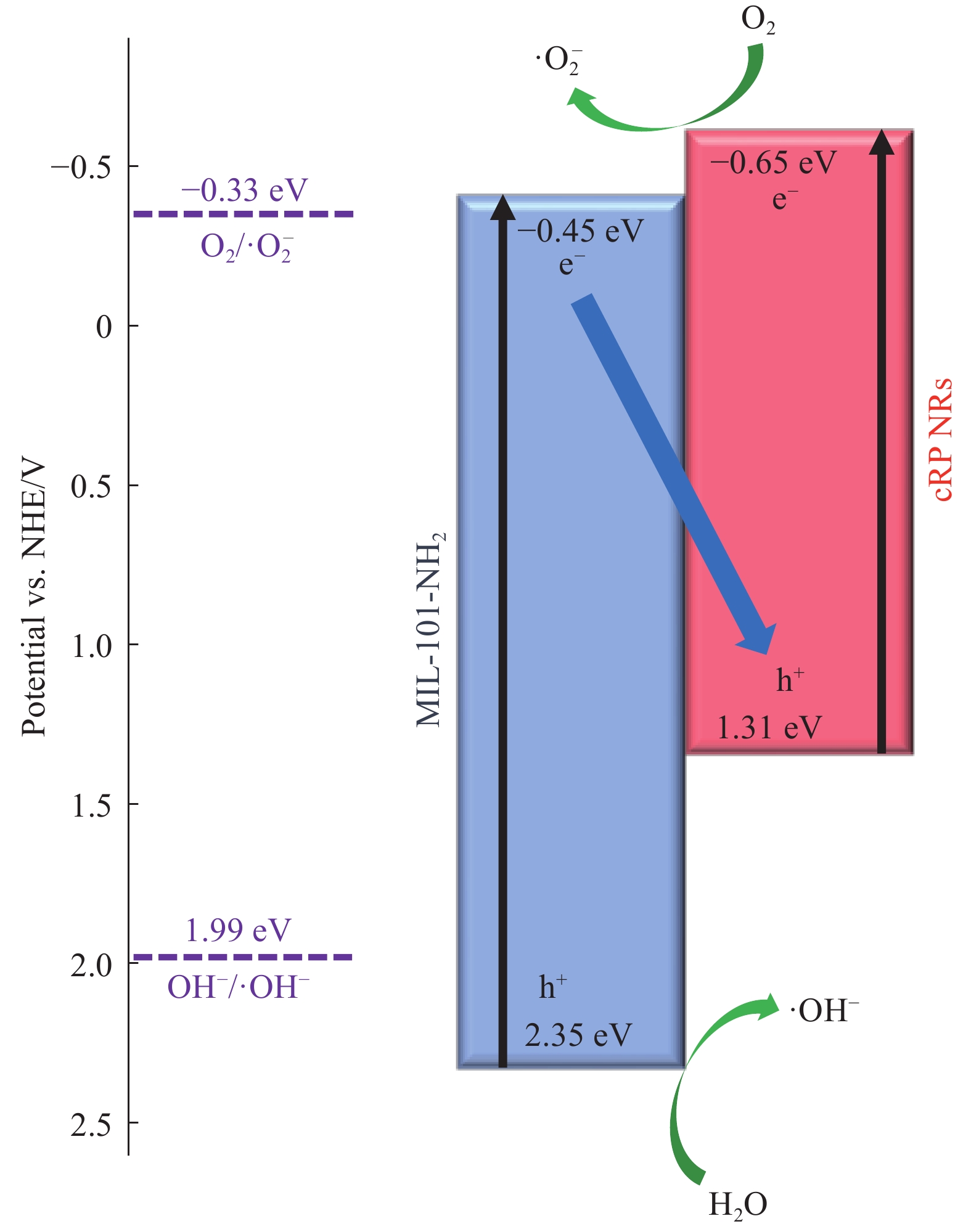
图6(a)中展示了紫外可见漫反射光谱(UV-Vis DRS),可以发现MIL-101-NH2的吸收边最窄,所有材料复合后,都发生了红移,说明了复合材料的光吸收范围扩大。在图6(b)中通过式(1)转换的曲线画出Tauc's 图,通过找到斜率最大处与x轴的交点得到材料的禁带宽度信息。可以得到cRP NRs、MIL-101-NH2和cRP/MIL-101-NH2的带隙宽度分别为1.96 eV、2.80 eV和1.935 eV。通过图6(c)中Mott-Schottky曲线的绘制可以得到几种催化剂的导带情况。可以得到cRP NRs、MIL-101-NH2和cRP/MIL-101-NH2的导带分别为−0.65 eV、−0.45 eV和−0.51 eV,结合前文中的禁带宽度可以计算出这些材料和复合材料的价带。
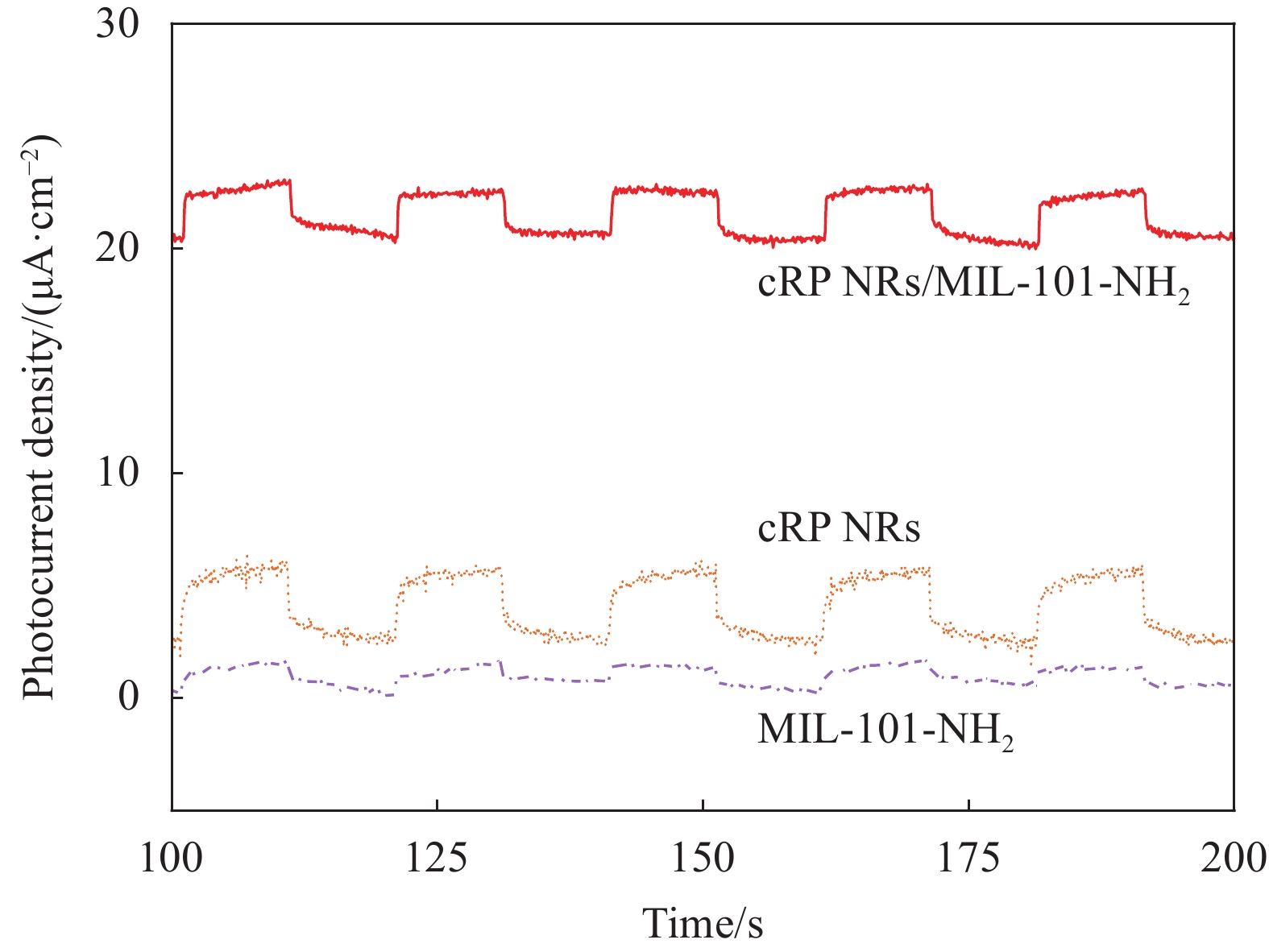
如图7所示,为了分析制备的复合材料的对光的响应情况,对复合材料和原材料一同进行了瞬态光电流响应测试。可以发现, cRP NRs的光响应情况比MIL-101-NH2要高,cRP/MIL-101-NH2复合材料的光响应程度远远高于复合前的原材料。
2.3 样品的光催化性能实验
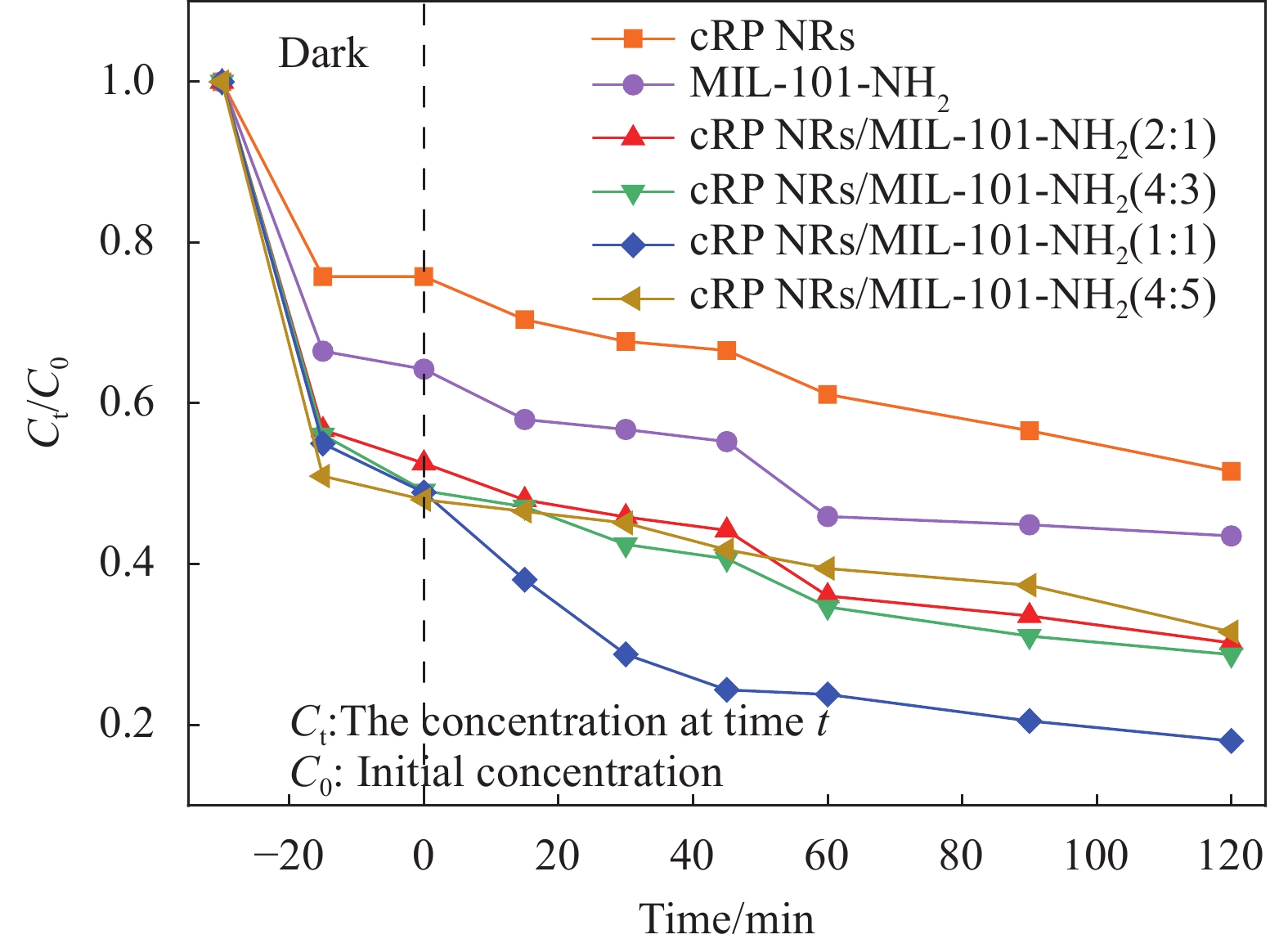
考虑到材料组分比例对光催化降解可能存在影响,并且为了探寻cRP NRs与MIL-101-NH2形成复合材料的最佳比例,设置了原材料质量比分别为2∶1、4∶3、1∶1和4∶5的几种复合材料进行探究。
如图8所示,即为几种不同比例材料和原材料的降解曲线。从图中可以看到复合材料在不同时刻对四环素的降解效果,揭示了复合材料与四环素之间的反应规律。从图中可以看到cRP NRs的吸附效果和光催化效果并不理想,在经过120 min光照后降解效果仅在49%左右。MIL-101-NH2表现出了优于cRP NRs的暗反应吸附性能,同时也展示了其些许的光催化性能。在120 min光照反应后, MIL-101-NH2则在光照60 min后保持平衡,降解了57%左右。经过溶剂热法复合后的材料cRP NRs/MIL-101-NH2表现出了优秀的吸附性能,在暗反应结束时,对TC的去除率最高能够达到52%左右。同时在开始光照后,产生了不错的光催化效果。在结束120 min光照时,几种材料的降解效率排序如下:cRP NRs/MIL-101-NH2 (1∶1)(~82%) > cRP NRs/MIL-101-NH2(4∶3) (~71%) > cRP NRs/MIL-101-NH2 (2∶1)(~70%) > cRP NRs/MIL-101-NH2 (4∶5)(~68%) > MIL-101-NH2 (~57%) > cRP NRs(~49%)。
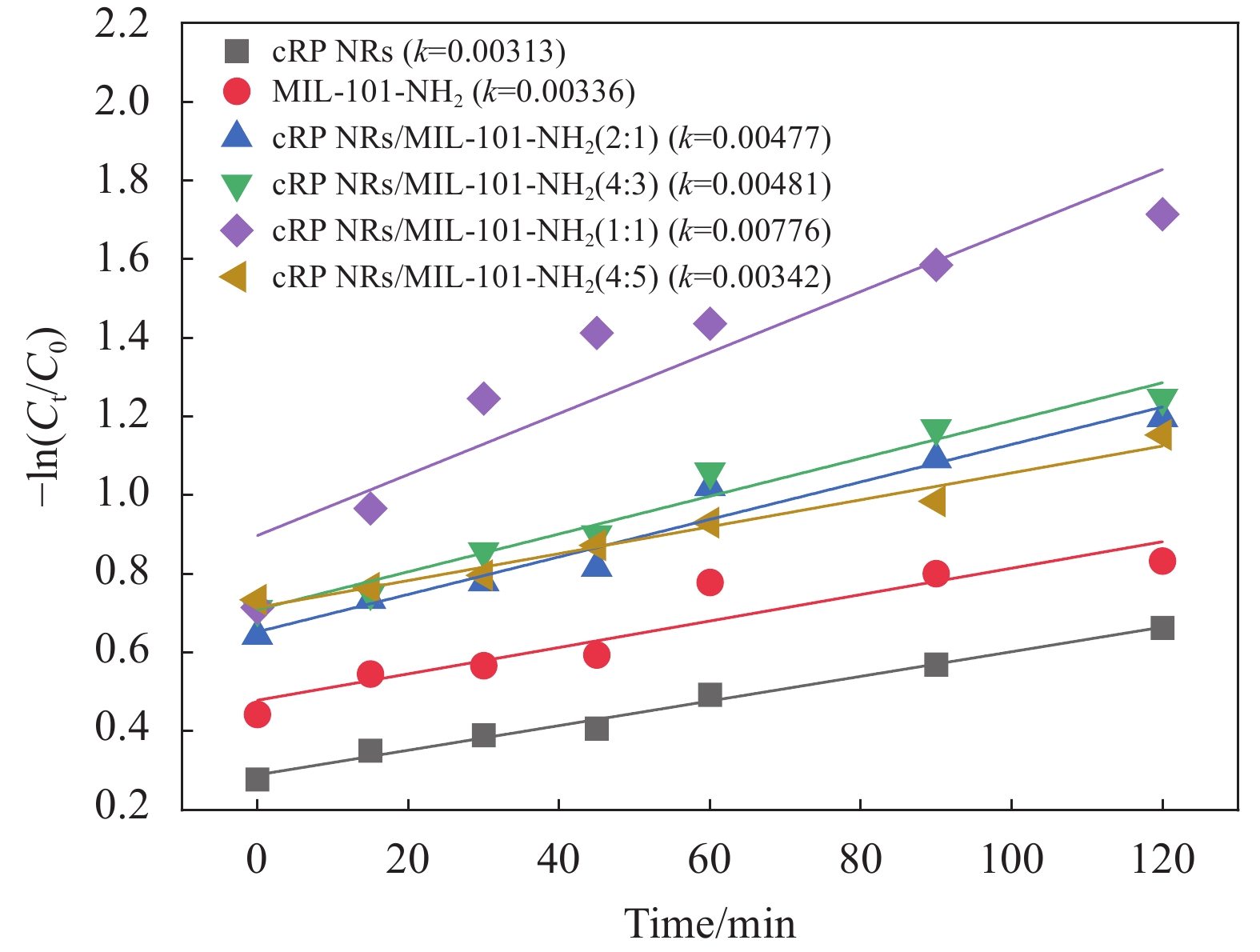
如图9所示,根据方程−ln(Ct/C0)=kt,计算了反应的动力学曲线和相应的反应速率常数k。可以证实,本研究中涉及的所有光催化剂的反应动力学都很好地遵循伪一级反应。其中cRP NRs/MIL-101-NH2 (1∶1)的反应速率常数k值为
0.00776 min−1,高于其余几种比例复合的材料和原材料。毫无疑问,当 cRP NRs和MIL-101-NH2的质量比为1∶1时,复合材料的光催化降解效果最好。因此,在后续实验中继续沿用该比例制备的复合材料,并统一称为cRP NRs/MIL-101-NH2。为了验证复合材料的稳定性,以cRP NRs/MIL-101-NH2作为实验对象,以在热反应下更容易发生形变的MIL-101-NH2作为对比对象来进行循环实验。通过图10可以发现制备出的cRP NRs/MIL-101-NH2催化剂在前3次光催化降解四环素始终保持了优秀的降解效果,在进行第4次降解时虽然效果不如前3次,但是仍在光照120 min后降解效率达到了78%左右。而MIL-101-NH2则在第3次时降解效率就明显下降。经过循环实验的验证,证明了cRP NRs和MIL-101-NH2材料经过复合后,材料的稳定性也得到了明显的提高。
![]() 图 9 cRP NRs、MIL-101-NH2、cRP NRs/MIL-101-NH2 (2∶1)、 cRP NRs/MIL-101-NH2 (4∶3)、cRP NRs/MIL-101-NH2 (1∶1)和cRP NRs/MIL-101-NH2 (4∶5)动力学拟合曲线Figure 9. Kinetic fitting curves of cRP NRs, MIL-101-NH2, cRP NRs/MIL-101-NH2 (2∶1), cRP NRs/MIL-101-NH2 (4∶3), cRP NRs/MIL-101-NH2 (1∶1), and cRP NRs/MIL-101-NH2 (4∶5)k—Reaction rate constant
图 9 cRP NRs、MIL-101-NH2、cRP NRs/MIL-101-NH2 (2∶1)、 cRP NRs/MIL-101-NH2 (4∶3)、cRP NRs/MIL-101-NH2 (1∶1)和cRP NRs/MIL-101-NH2 (4∶5)动力学拟合曲线Figure 9. Kinetic fitting curves of cRP NRs, MIL-101-NH2, cRP NRs/MIL-101-NH2 (2∶1), cRP NRs/MIL-101-NH2 (4∶3), cRP NRs/MIL-101-NH2 (1∶1), and cRP NRs/MIL-101-NH2 (4∶5)k—Reaction rate constant表1为查阅其他文献中的各种材料对四环素的降解情况。通过对比可以发现,制备出的cRP NRs/MIL-101-NH2经过120 min的光照后,其降解效果优于同类催化剂,如CDs/MIL-101(Fe)(81%)、MIL-101(Fe)/MIL-100(Fe)(80%)、TiO2(76.6%)和BiOI/MIL-125(Ti)(70%)。而相对于RP/MIL-101(Fe)、PSCN-50、MIL-101(Fe)/MoS2、RP/HAp以及P-BiOCl这几种材料,cRP NRs/MIL-101-NH2虽然降解效率没有这几种材料高,但是在催化剂浓度的对比上,能够明显发现cRP NRs/MIL-101-NH2的浓度明显低于这些材料。这证明了cRP NRs/MIL-101-NH2具有充分的潜力来进行四环素的光催化降解。
表 1 其他文献材料降解四环素效果对比Table 1. Comparison of tetracycline degradation effects of other materials in literaturesMaterial Catalyst concentration/
(mg·L−1)Dosage/mg TC concentration/
(mg·L−1)Reaction
time/minDegradation
efficiency/%Ref. cRP/MIL-101(Fe)-NH2 0.2 10 50 120 82 This study MIL-101(Fe)/MIL-100(Fe) 0.125 10 50 140 80 [31] TiO2 0.2 20 10 120 76.60 [32] BiOI/MIL-125(Ti) 0.25 25 20 120 70 [33] CDs/MIL-101(Fe) 0.5 50 75 120 81 [34] RP/MIL-101(Fe) 0.5 50 50 80 90.10 [15] PSCN-50 1 100 10 60 85.50 [35] MIL-101(Fe)/MoS2 0.3 30 100 40 85 [36] RP/HAp 1 100 10 30 100 [10] P-BiOCl 0.5 50 20 30 81 [37] Notes: TC is tetracycline; TiO2 is titanium dioxide; BiOI is a compound of bismuth oxide and iodine; CDs are carbon dots; RP is red phosphorus; PSCN is phosphorus-sulfur co-doped g-C3N4; MoS2 is molybdenum disulfide; HAp is a hollow hydroxyapatite; P is phosphorus; BiOCl is a compound of bismuth oxide and chlorine. 2.4 cRP NRs/MIL-101-NH2复合材料的光催化降解机制分析
为了了解在整个光催化进行中因光照可能会产生的自由基对降解的影响,进行了自由基捕获实验。在进行光催化降解实验时加入对苯醌、异丙醇和乙二胺四乙酸二钠,这3种捕获剂分别对超氧自由基(•O−2)、羟基自由基(•OH)和光生空穴(h+)进行捕获。
在图11 cRP NRs/MIL-101-NH2的自由基捕获实验的降解曲线图中可以明显的发现加入的对苯醌、异丙醇和乙二胺四乙酸二钠均对光催化降解有抑制作用。说明在cRP NRs/MIL-101-NH2进行光催化降解四环素的过程中,超氧自由基(•O−2)、羟基自由基(•OH)和光生空穴(h+)都对降解起到了作用。其中对苯醌的加入对四环素降解的抑制效果最为明显,乙二胺四乙酸二钠次之,异丙醇的抑制效果最差但仍起到了部分作用,证明cRP NRs/MIL-101-NH2在光催化降解四环素的过程中超氧自由基(•O−2)对四环素的降解其主要作用,该情况的产生可能是因为MIL-101-NH2和cRP NRs的导带均达到了可产生超氧自由基的电位位置,所以超氧自由基的作用会更将明显。而羟基自由基的产生是由于MIL-101-NH2的价带电位能够达到产生•OH电位(1.99 eV)所造成,因此其在反应中的作用并不显著。
当cRP NRs/MIL-101-NH2受到光照时,cRP NRs和MIL-101-NH2都能产生光激发的e−和h+对。根据典型的II型异质结的假设,cRP NRs/MIL-101-NH2复合材料的氧化还原电位将由MIL-101-NH2的负导带越正和cRP NRs的正价带越负决定[38-39]。虽然已知MIL-101-NH2的导带能量足以将O2分子还原成为•O−2,但cRP NRs的价带 (EVB=1.31 eV)不能从水中产生•OH自由基(E (•OH/OH−)= 1.99 eV)[13]。自由基捕获实验中的测量结果主要证实了反应介质中存在超氧自由基(•O−2)和羟基自由基(•OH)。因此,II型异质结不太可能发生。在图12中,由于MIL-101-NH2的导带位于cRP NRs的导带和价带之间,它很容易使电子(e−)从MIL-101-NH2的导带转移到cRP NRs的价带,并与cRP NRs的价带中的光生空穴(h+)重新结合。当开始光照后,电子向从价带向价带跃迁,同时依据几种材料经过XPS分析中结合能变化,判断出电子是从MIL-101-NH2向cRP NRs进行迁移,因此能够确认cRP NRs/MIL-101-NH2这种复合材料为Z型异质结结构。MIL-101-NH2的高比表面积和其结构特性可以改善cRP NRs团聚现象,降低了cRP NRs自身产生电子空穴对的复合速率,同时增加吸附点和活性位点,活性氧空位和吸附氧为该催化过程提供了更多的活性位点。随着时间的推移,在cRP NRs的导带和MIL-101-NH2的价带上分别积累了大量的e− 和h+。cRP NRs的导带中的e−直接还原溶解的O2分子,生成超氧自由基 (•O−2),光生空穴 (h+)将H2O分子氧化生成羟基自由基 (•OH)。复合材料因其高比表面积和大孔径的特点先将TC吸附到材料表面,而后材料中因光照产生的强氧化物质•O−2 和•OH能够更快地与四环素发生氧化还原反应,在催化剂表面便能生成降解产物,从而提高了反应体系的降解速率。
2.5 cRP NRs/MIL-101-NH2复合材料的光催化降解产物分析
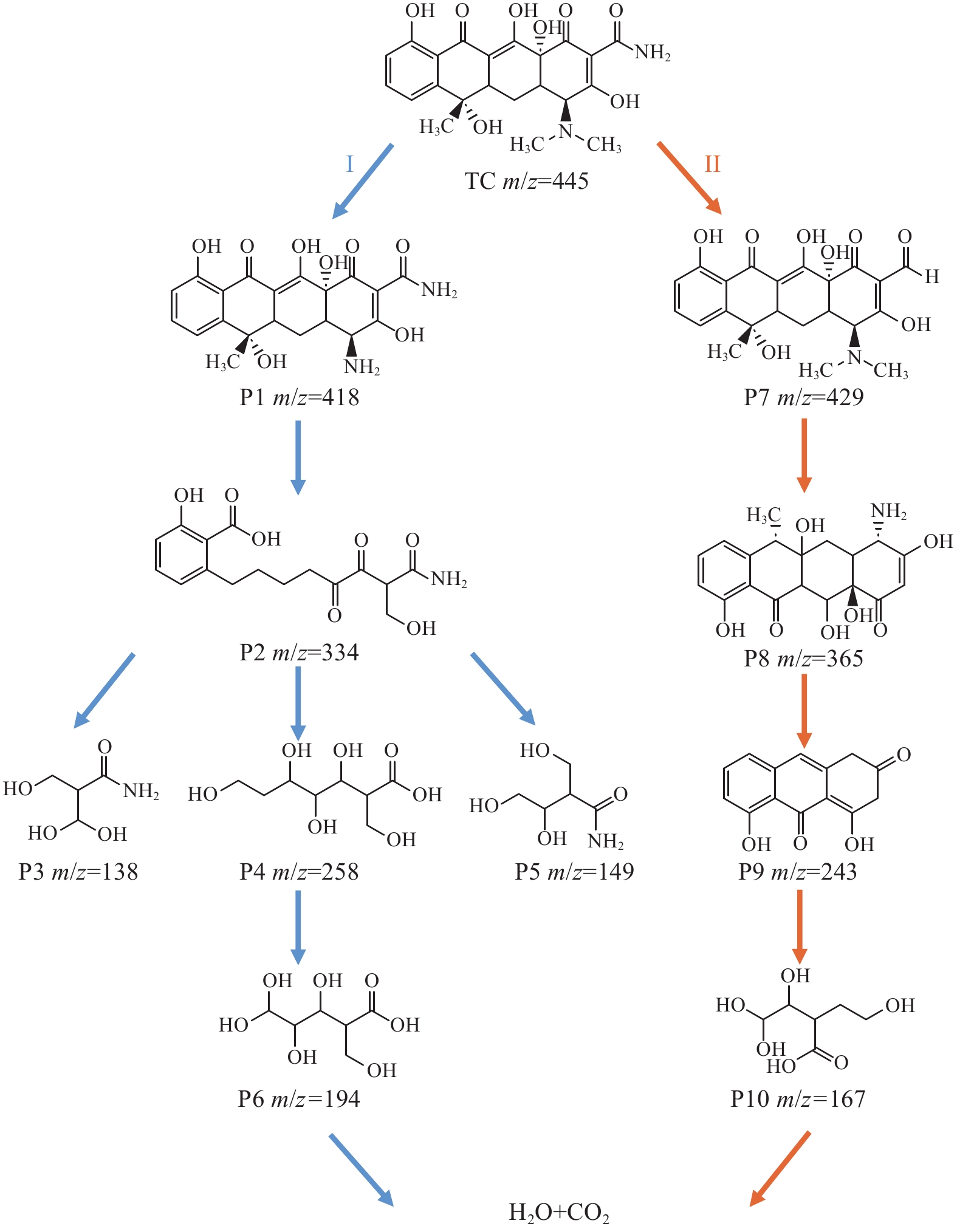
通过高分辨质谱联用技术 (HRMS) 对降解中间产物进行鉴定,可能的反应路径图如图13所示。
在路径I中,开始降解时,TC上的 N—CH3由于N—C的低键能丢失[40]而形成了P1 (m/z=418)。然后在•O−2和 h+的攻击下,进一步•O−2攻击通过羟基和甲基的分离和开环产生 P2 (m/z=334) 的中间体。然后,通过羟基化、脱烷基化、脱水和开环反应进一步降解生成P3 (m/z=138) 、P4 (m/z=258) 、P5 (m/z=149)和P6 (m/z=194)。最终,这些中间产物被矿化CO2和H2O。
在路径II中,—NH2较高的负电荷,使其容易受到h+的攻击[41],导致TC骨架中的氨损失,形成P7 (m/z=429)。随后,由于C的低键能和受到•O−2的攻击等因素,形成了P8 (m/z=365)。接着又由于h+、•OH和•O−2的攻击,通过环的裂解和脱羰反应进一步氧化成较小的化合物P9 (m/z=243) 和P10 (m/z=167)。最终形成无害的CO2和H2O。
3. 总 结
在本研究中,将晶态红磷纳米带(cRP NRs)与MIL-101-NH2通过溶剂热法进行了复合制备。随后,对原材料及其复合材料进行了表征和性能测试。通过对比分析,得到了关于这些材料结构、性能及应用潜力的重要结论,并通过一系列性能测试验证了它们在光催化领域的应用潜力。得到的结论如下:
(1)在SEM和TEM的拍摄下,能够清楚看到多面体的MIL-101-NH2分散包裹在条带状cRP NRs的表面。通过XRD可以直观地看到cRP NRs和MIL-101-NH2的特征峰出现在了复合材料的特征峰上。证明了复合材料cRP NRs/MIL-101-NH2的成功合成。
(2)在相同实验条件下,对原材料和不同比例制备的复合材料开展光催化降解四环素的光催化降解实验。对比实验结果显示,在光照120 min后,cRP NRs降解约49%,MIL-101-NH2 降解约57%,复合材料在质量比1∶1时表现了优于其他比例的光催化降解效果,降解约82%。同时,在cRP NRs/MIL-101-NH2的稳定性实验中,第四次循环中仍能达到78%左右的降解效果。
(3)通过紫外漫反射光谱得到的材料禁带宽度,Mott-Schottky曲线得到的材料导带电位,自由基捕获实验得到的材料表面发生光催化产生的自由基情况和XPS能谱得到材料中可能存在的电子转移和元素价态等实验数据的分析和佐证,最终确定本文所提出的新型复合材料cRP NRs/MIL-101-NH2为Z型异质结结构。
-
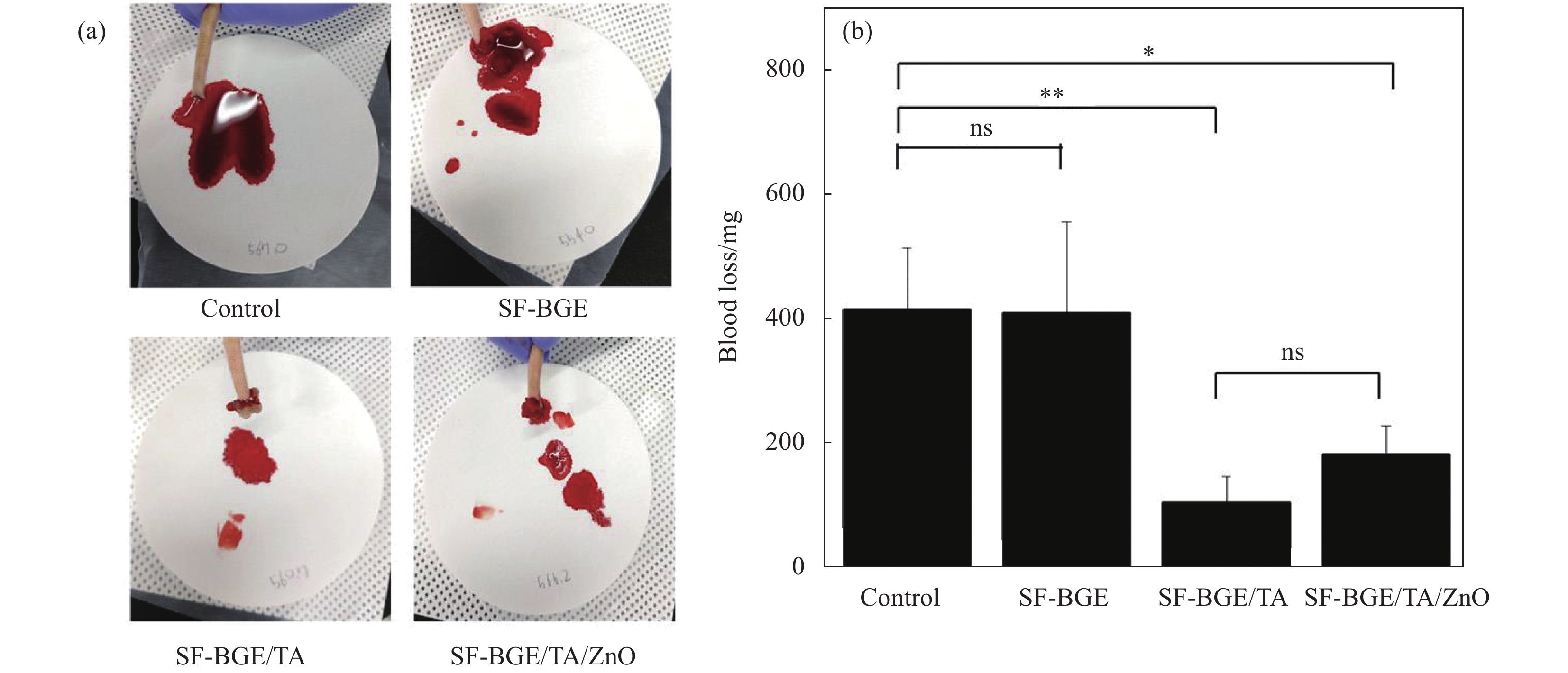
图 2 (a)截尾后给予改性后易溶于水的丝素蛋白(SF-BGE)、加入了单宁酸的易溶于水的丝素蛋白溶液(SFBGE/TA)和SF-BGE/TA/ZnO水凝胶后失血的照片;(b)对照组、SF-BGE、SF-BGE/TA、SF-BGE/TA、SF-BGE/TA/氧化锌水凝胶致伤大鼠的失血量(结果报告为平均值±标准差 (n=3);统计学处理采用单因素方差分析和Tukey's后置检验(*p<0.05,**p<0.01))[48]
Figure 2. (a) Photographs of blood loss after tail amputation and administration of SF-BGE, SFBGE/TA and SF-BGE/TA/ZnO hydrogel; (b) Blood loss from injury rat tail of control, SF-BGE, SF-BGE/TA, and SF-BGE/TA/ZnO hydrogel (The results are reported as the mean±standard deviation (n=3); Statistical significance was analyzed by one-way ANOVA and Tukey's post hoc test (*p<0.05, **p<0.01))[48]
SF—Silk fibroin; BGE—Butyl glycidyl ether; TA—Tannic acid; ns—No significance
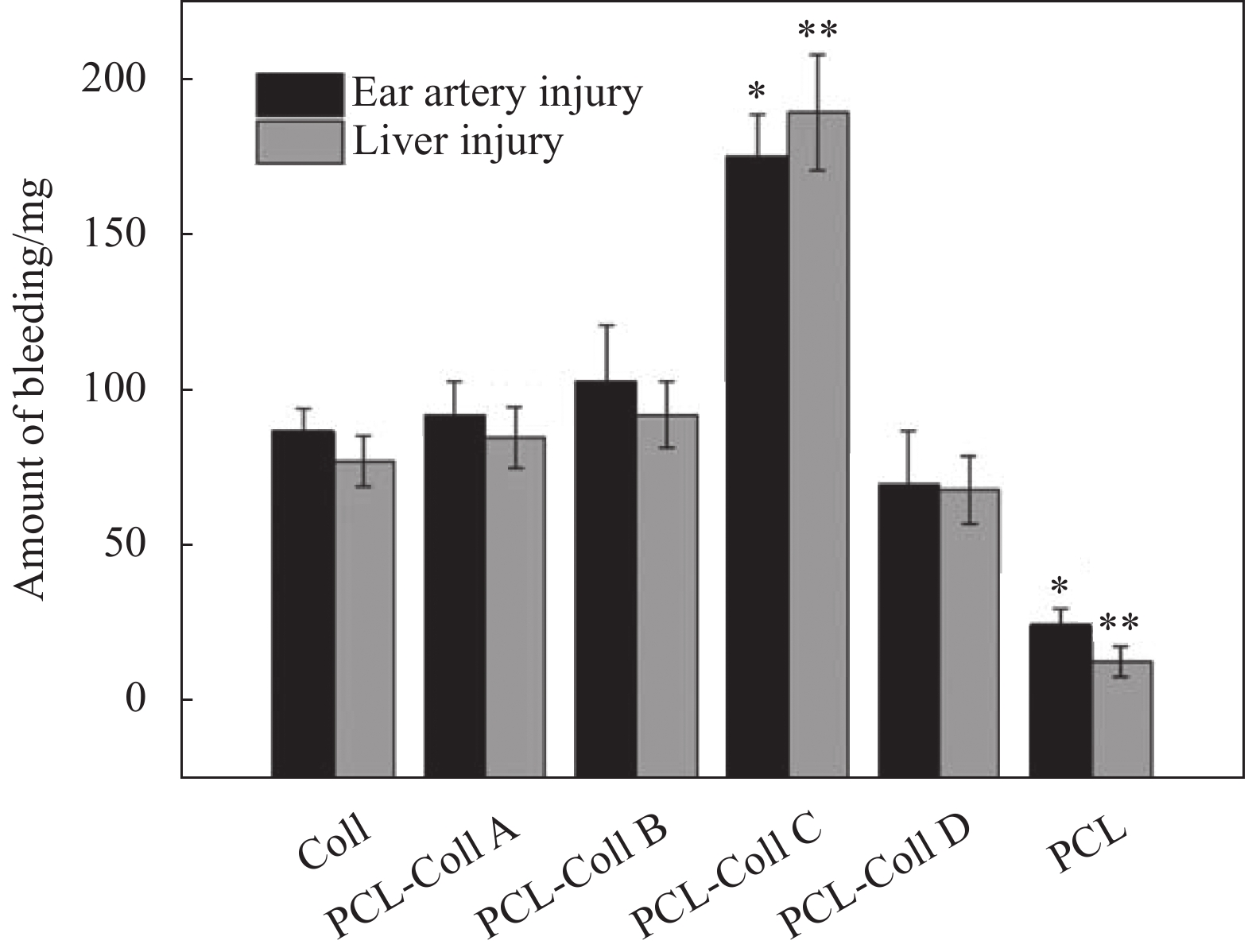
图 3 兔耳动脉和肝脏损伤模型中不同膜片的出血量(PCL-Coll (A~D)组与Coll组之间的统计学意义分别为*p<0.005,**p<0.05)[57]
Figure 3. The amount of bleeding of different films in injury models of rabbit ear artery and liver (Statistical significances between PCL-Coll (A-D) groups and ColI are denoted as *p<0.05, **p<0.005)[57]
PCL—Polycaprolactone
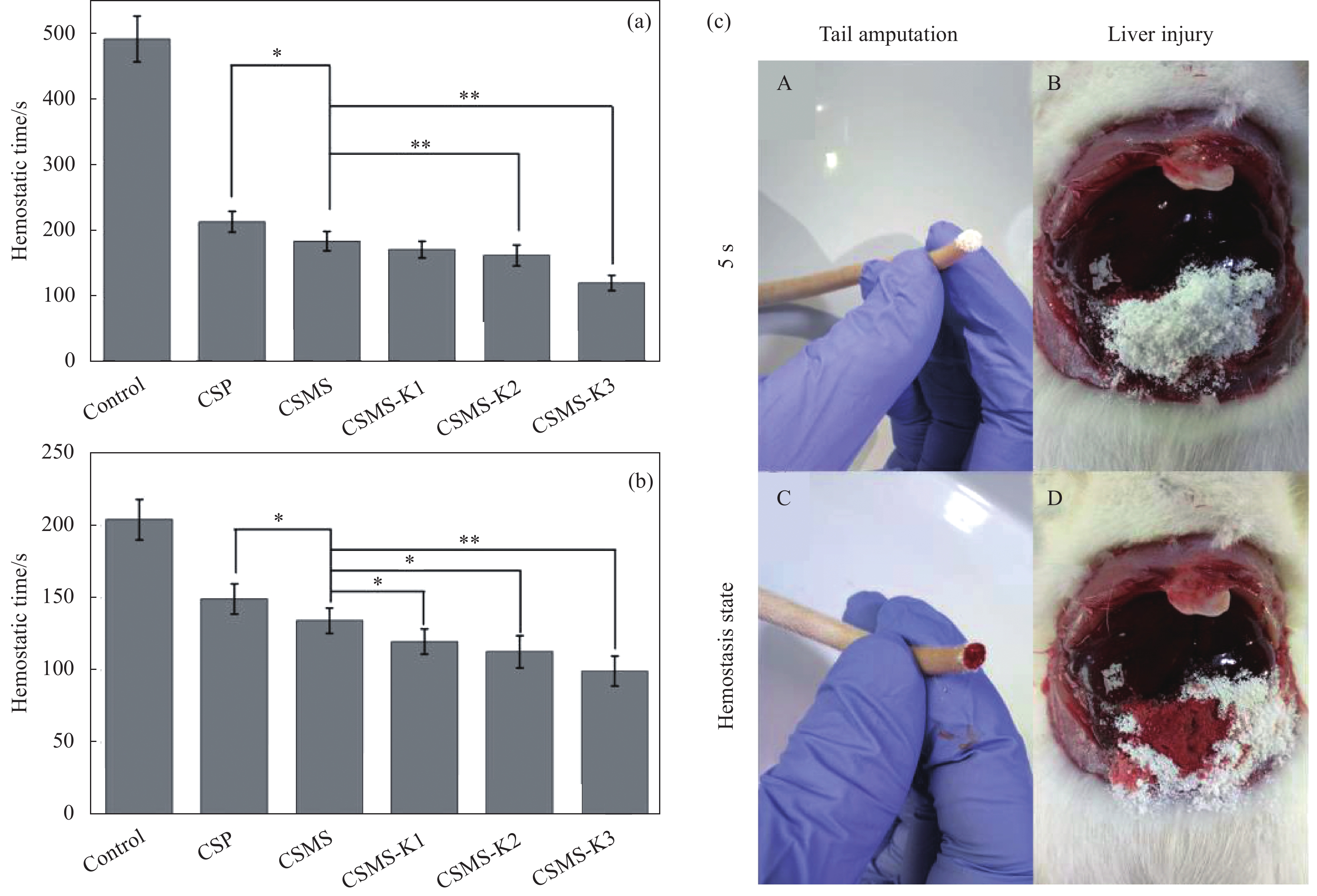
图 4 (a)大鼠断尾模型的止血时间;(b)大鼠肝撕裂伤模型止血时间;(c)大鼠断尾和肝撕裂模型的数字图像(将CSMS-K3涂于A尾部切割处和B肝撕裂伤处;涂抹CSMS-K3 5 min后C大鼠尾部切割处和D肝撕裂伤处;伤处形成暗红色血块,出血停止)[85]
Figure 4. (a) Hemostasis time in rat tail amputation model; (b) Hemostasis time in rat liver laceration model; (c) Digital images of rat tail amputation and liver laceration models (CSMS-K3 was applied onto A the tail cut and B on the liver injury; 5 min after application on the C rat tail cut and D the liver laceration; Dark-red blood clot was formed on the injured sites, bleeding stopped)[85]
CSP—Chitosan particles; CSMS—Chitosan porous microspheres; * indicates difference between the two groups of data and p<0.05; ** indicates significant difference and p<0.01
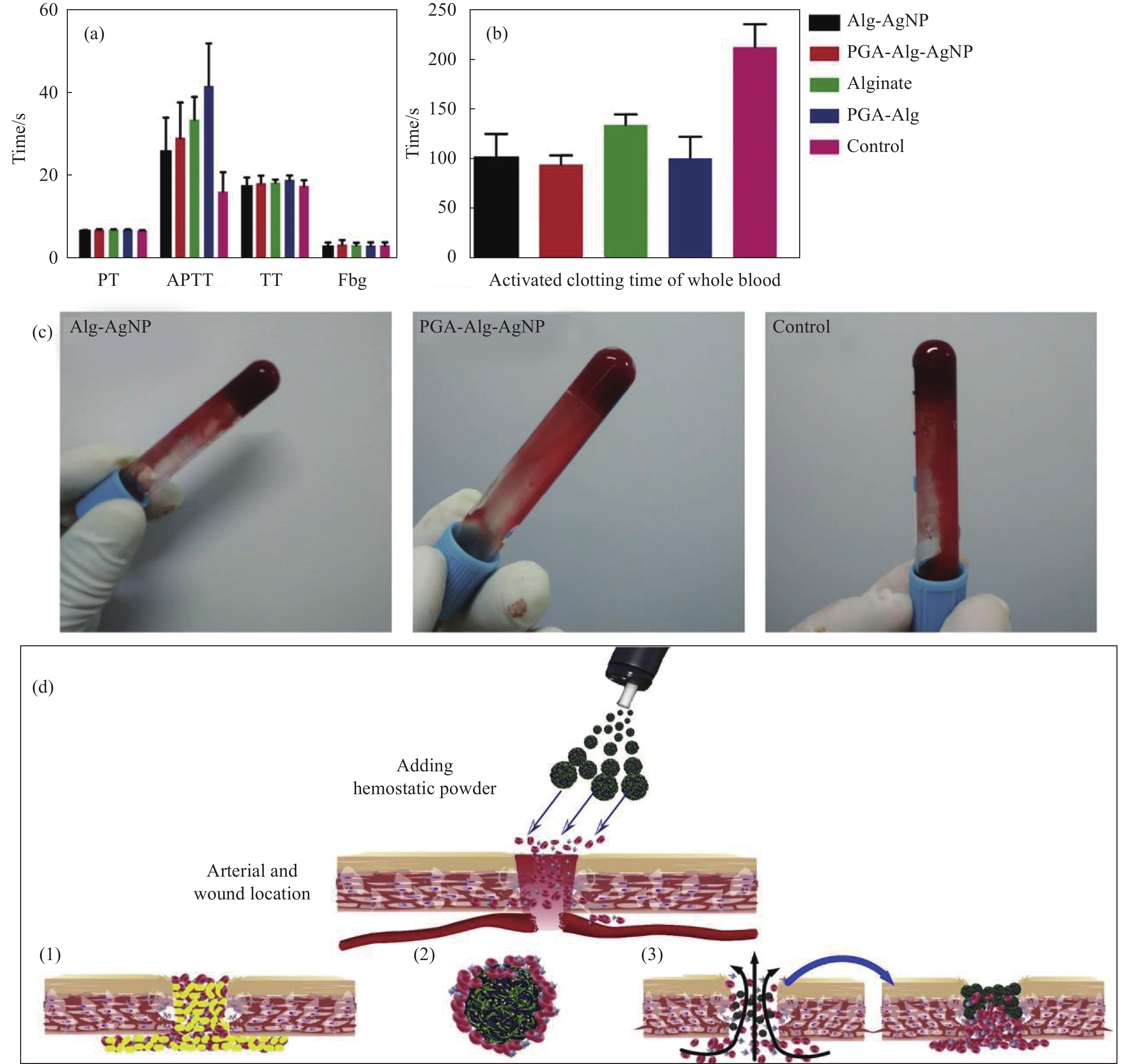
图 5 纯海藻酸微球、海藻酸/银纳米(Alg/AgNPs)、聚(γ-谷氨酸) (PGA)/Alg/AgNPs和PGA/Alg复合微球的止血分析:(a)血浆凝血性能;(b)凝血时间;(c)凝血图像;(d) PGA/Alg/AgNPs微球的止血示意图((1)止血剂提供外源性凝血成分,加速伤口血液凝结;(2)止血剂促进红细胞、凝血因子、血小板聚集,加速凝血;(3)止血剂吸收血液中水分,增加血细胞、血小板、凝血因子浓度,阻断创面,促进聚集止血[93])
Figure 5. Hemostasis assays of pure alginate microspheres, Alg/AgNPs, PGA/Alg/AgNPs, and PGA/Alg composite microspheres: (a) Blood plasma clotting performance; (b) Clotting blood time; (c) Coagulation images; (d) Schematic hemostasis of PGA/Alg/Ag NPs microspheres ((1) The hemostatic agent provides extrinsic coagulation components to accelerate blood clotting on the wound; (2) The hemostatic agent promotes the aggregation of red blood cells, coagulation factors and platelets, and accelerates the coagulation; (3) The hemostatic agent absorbs water in the blood, increases the concentration of blood cells, platelets, and coagulation factors, block the wound area and promotes the aggregation and hemostasis[93])
PT—Prothrombin time; APTT—Activated partial thromboplastin time; TT—Thrombin time; Fbg—Fibrinogen; PGA—Poly(γ-glutamic acid)
表 1 现有商品化生物质止血材料
Table 1 Existing commercial biomass hemostatic materials
Hemostatic material Product name Manufacturer Mechanism of hemostasis Hemostatic effect Limitations or deficiencies Ref. Fibrin sealant Tisseel;
Evicel;
Beriplast PBaxter;
Ethicon;
CSL BehringSimulates the coagulation process in the body and quickly forms blood clots Complete hemostasis was achieved within 30 s (Arterial bypass) to 6 min (Carotid endarterectomy) The price is expensive, and the cost of transportation and storage is high; It is contraindicated in patients with bovine allergies, who have adverse effects including rash, coagulation disorders, anaphylaxis, and death from premade antibodies [16-17] Oxidized cellulose Surgicel;
Cutanplast;
SurgicelJohnson;
B. Braun;
EthiconImmediately after the material encounters blood, the fluid in the blood is extracted and blood proteins, platelets, red blood cells and other active components are captured, resulting in an increased concentration of coagulation factors and an accelerated coagulation process Hemostasis is usually successful within 5 min (Open surgery) Foreign body reactions, minor postoperative complications, as foci of infection or anaphylaxis, manifested mainly by acute dermatitis, eczema, and serous tumors [9, 18] Gelatin sponges Gelfoam;
GelfoamPfizer;
BaxterAttaches to the bleeding site, allowing platelets to stay in uniform pores, activating the coagulation cascade Hemostasis was successfully achieved within 5 min (Reoperative cardiac surgery or emergency resternotomy) Inability to pack bleeding, and the possibility of breaking the clot when the sponge is removed; It may cause thrombosis of small blood vessels or an inflammatory reaction [17, 19] Microporous polysaccharides Arista AH Starch
MedicalIt concentrates blood solids by absorbing water and low-molecular-weight compounds from the blood, thereby providing hemostasis and providing a scaffold for the formation of fibrin clots Haemostasis was completed at 90 s (5 mm pork kidney incision) to 200 s (12 mm pork kidney incision) There will be a slight inflammatory reaction at the beginning of use [20-22] Chitosan sponge HemCon;
ChitoGauze PRO;
CeloxHemCon Medical Technologies;
HemCon Medical Technologies;
Z-MedicaPositively charged chitosan binds to negatively charged red blood cells. As a result, it leads to the formation of sticky clots to promote hemostasis Complete hemostasis usually occurs within 2 min (Tooth extraction surgery) For limb injuries, tourniquets are required [23-24] Notes: CSL—Commonwealth Serum Laboratories; AH—Absorbable hemostasis; PRO—Professional. 表 2 蛋白类止血材料的材料类型、体内模型和止血效果
Table 2 Material types, in vivo models, and hemostatic effects of protein-based hemostatic materials
Hemostatic
materialType of material Model of hemostasis Hemostatic effect Keratin Injectable hydrogel[34] Mouse model of liver injury Occlusion hemostasis was achieved in 90 s Nanoparticles[35] Rat model of liver injury and tail docking The hemostasis time was 60 s and 90 s Silk fibroin Composite sponge[47] Mouse model of liver injury 150 s to completely stop bleeding Nanocomposite hydrogels[48] Rat tail docking model The blood loss was only (183.4±50.1) mg Collagen Porous sponges[56] Rat tail docking model Complete hemostasis in 320 s Nanofiber membranes[57] Rabbit ear artery, liver injury model The hemostasis time was (95.34±10.05) s and
(67.05±7.15) sSelf-healing hydrogel[58] Mouse hemorrhagic liver model The blood loss was only (0.4±0.15) g 表 3 多糖类止血材料的材料类型、体内模型和止血效果
Table 3 Material types, in vivo models, and hemostatic effects of polysaccharide hemostatic materials
Hemostatic material Type of material Model of hemostasis Hemostatic effect Cellulose Composite sponge[67] Rat tail docking and liver injury model The blood loss was 159.46 mg and 80.44 mg Nanocomposite fibers[70] Lemonified human plasma, lemonified bovine whole blood The coagulation time was (143±19) s and (67±5) s Powder[83] Mouse tail docking model The hemostasis time was 158 s, and the blood loss was only 11.0 mg Chitosan Composite microspheres[85] A model of tail docking and liver laceration in rats The hemostasis time is 134 s and 99 s Composite hydrogel[86] Rat model of liver injury and femoral artery injury The hemostasis time was (53±3) s and (189±9) s Alginic acid Composite porous microspheres[91] Rat model of liver laceration and tail breakage The hemostasis time was (73±5) s and (134±5) s Foam[94] Porcine liver injury model The hemostasis time is 5 min Starch Superabsorbent
hydrogel[101]Rat model of femoral artery injury The hemostasis time <10 s Powder[103] Rabbit model of ear vein, dorsal, femoral artery, liver injury The hemostasis time was 52 s, 46 s, 122 s and 102 s Hyaluronic acid Drug-loaded microporous powder[105] Rabbit ear artery, liver injury model The hemostasis time is (108±5) s and (120±6) s Sponge[113] Mouse model of liver injury The hemostasis time was <60 s, and the blood loss was only 23.2 mg Cryogel[114] Rat model of liver injury The hemostasis time is 72 s -
[1] VOS T, LIM S S, ABBAFATI C, et al. Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: A systematic analysis for the Global Burden of Disease Study 2019[J]. The Lancet, 2020, 396(10258): 1204-1222. DOI: 10.1016/S0140-6736(20)30925-9
[2] BAYLIS J R, LEE M M, ST JOHN A E, et al. Topical tranexamic acid inhibits fibrinolysis more effectively when formulated with self-propelling particles[J]. Journal of Thrombosis and Haemostasis, 2019, 17(10): 1645-1654. DOI: 10.1111/jth.14526
[3] LIU C, LIU X, LIU C, et al. A highly efficient, in situ wet-adhesive dextran derivative sponge for rapid hemostasis[J]. Biomaterials, 2019, 205: 23-37. DOI: 10.1016/j.biomaterials.2019.03.016
[4] HICKMAN D A, PAWLOWSKI C L, SEKHON U D S, et al. Biomaterials and advanced technologies for hemostatic management of bleeding[J]. Advanced Materials, 2018, 30(4): 1700859. DOI: 10.1002/adma.201700859
[5] HOLCOMB J B, STANSBURY L G, CHAMPION H R, et al. Understanding combat casualty care statistics[J]. The Journal of Trauma: Injury, Infection, and Critical Care, 2006, 60(2): 397-401. DOI: 10.1097/01.ta.0000203581.75241.f1
[6] ZHONG Y, HU H, MIN N, et al. Application and outlook of topical hemostatic materials: A narrative review[J]. Annals of Translational Medicine, 2021, 9(7): 577. DOI: 10.21037/atm-20-7160
[7] GIDAY S A, KIM Y, KRISHNAMURTY D M, et al. Long-term randomized controlled trial of a novel nanopowder hemostatic agent (TC-325) for control of severe arterial upper gastrointestinal bleeding in a porcine model[J]. Endoscopy, 2011, 43(4): 296-299. DOI: 10.1055/s-0030-1256125
[8] KATSUYAMA S, MIYAZAKI Y, KOBAYASHI S, et al. Novel, infection-free, advanced hemostatic material: Physical properties and preclinical efficacy[J]. Minimally Invasive Therapy & Allied Technologies, 2020, 29(5): 283-292. DOI: 10.1080/13645706.2019.1627373
[9] ZHANG S, LI J, CHEN S, et al. Oxidized cellulose-based hemostatic materials[J]. Carbohydrate Polymers, 2020, 230: 115585. DOI: 10.1016/j.carbpol.2019.115585
[10] MONTAZERIAN H, DAVOODI E, BAIDYA A, et al. Engineered hemostatic biomaterials for sealing wounds[J]. Chemical Reviews, 2022, 122(15): 12864-12903. DOI: 10.1021/acs.chemrev.1c01015
[11] YANG X, LIU W, LI N, et al. Design and development of polysaccharide hemostatic materials and their hemostatic mechanism[J]. Biomaterials Science, 2017, 5(12): 2357-2368. DOI: 10.1039/C7BM00554G
[12] CHEN J, CHENG W, CHEN S, et al. Urushiol-functionalized mesoporous silica nanoparticles and their self-assembly into a Janus membrane as a highly efficient hemostatic material[J]. Nanoscale, 2018, 10(48): 22818-22829. DOI: 10.1039/C8NR05882B
[13] OTROCKA-DOMAGALA I, JASTRZEBSKI P, ADAMIAK Z, et al. Safety of the long-term application of QuikClot Combat Gauze, ChitoGauze PRO and Celox Gauze in a femoral artery injury model in swine—A preliminary study[J]. Polish Journal of Veterinary Sciences, 2016, 19(2): 337-343. DOI: 10.1515/pjvs-2016-0041
[14] SUN X, LI J, SHAO K, et al. A composite sponge based on alkylated chitosan and diatom-biosilica for rapid hemostasis[J]. International Journal of Biological Macromolecules, 2021, 182: 2097-2107. DOI: 10.1016/j.ijbiomac.2021.05.123
[15] YU L, ZHANG H, XIAO L, et al. A bio-inorganic hybrid hemostatic gauze for effective control of fatal emergency hemorrhage in "Platinum Ten Minutes"[J]. ACS Applied Materials & Interfaces, 2022, 14(19): 21814-21821.
[16] ALBALA D M. Fibrin sealants in clinical practice[J]. Cardiovascular Surgery, 2003, 11(Supplement 1): 5-11.
[17] HONG Y M, LOUGHLIN K R. The use of hemostatic agents and sealants in urology[J]. The Journal of Urology, 2006, 176(6): 2367-2374.
[18] AL-ATTAR N, DE JONGE E, KOCHARIAN R, et al. Safety and hemostatic effectiveness of SURGICEL® powder in mild and moderate intraoperative bleeding[J]. Clinical and Applied Thrombosis/Hemostasis, 2023, 29: 10760296231190376.
[19] KTARI O, FRASSANITO P, GESSI M, et al. Gelfoam migration: A potential cause of recurrent hydrocephalus[J]. World Neurosurgery, 2020, 142: 212-217. DOI: 10.1016/j.wneu.2020.06.214
[20] HUMPHREYS M R, LINGEMAN J E, TERRY C, et al. Renal injury and the application of polysaccharide hemospheres: A laparoscopic experimental model[J]. Journal of Endourology, 2008, 22(6): 1375-1381. DOI: 10.1089/end.2008.0008
[21] LYBARGER K S. Review of evidence supporting the Arista™ absorbable powder hemostat[J]. Medical Devices: Evidence and Research, 2024, 17: 173-188.
[22] ZHU J, WU Z, SUN W, et al. Hemostatic efficacy and biocompatibility evaluation of a novel absorbable porous starch hemostat[J]. Surgical Innovation, 2022, 29(3): 367-377. DOI: 10.1177/15533506211046100
[23] AZARGOON H, WILLIAMS B J, SOLOMON E S, et al. Assessment of hemostatic efficacy and osseous wound healing using HemCon dental dressing[J]. Journal of Endodontics, 2011, 37(6): 807-811. DOI: 10.1016/j.joen.2011.02.023
[24] WEDMORE I, MCMANUS J G, PUSATERI A E, et al. A special report on the chitosan-based hemostatic dressing: Experience in current combat operations[J]. Journal of Trauma and Acute Care Surgery, 2006, 60(3): 655-658. DOI: 10.1097/01.ta.0000199392.91772.44
[25] BURNETT L R, RAHMANY M B, RICHTER J R, et al. Hemostatic properties and the role of cell receptor recognition in human hair keratin protein hydrogels[J]. Biomaterials, 2013, 34(11): 2632-2640. DOI: 10.1016/j.biomaterials.2012.12.022
[26] LUSIANA, REICHL S, MULLER-GOYMANN C C. Keratin film made of human hair as a nail plate model for studying drug permeation[J]. European Journal of Pharmaceutics and Biopharmaceutics, 2011, 78(3): 432-440. DOI: 10.1016/j.ejpb.2011.01.022
[27] SHAVANDI A, SILVA T H, BEKHIT A A, et al. Keratin: Dissolution, extraction and biomedical application[J]. Biomaterials Science, 2017, 5(9): 1699-1735. DOI: 10.1039/C7BM00411G
[28] YAN R R, XUE D, SU C, et al. A keratin/chitosan sponge with excellent hemostatic performance for uncontrolled bleeding[J]. Colloids and Surfaces B: Biointerfaces, 2022, 218: 112770. DOI: 10.1016/j.colsurfb.2022.112770
[29] YE W, QIN M, QIU R, et al. Keratin-based wound dressings: From waste to wealth[J]. International Journal of Biological Macromolecules, 2022, 211: 183-197. DOI: 10.1016/j.ijbiomac.2022.04.216
[30] FENG C C, LU W F, LIU Y C, et al. A hemostatic keratin/alginate hydrogel scaffold with methylene blue mediated antimicrobial photodynamic therapy[J]. Journal of Materials Chemistry B, 2022, 10(25): 4878-4888. DOI: 10.1039/D2TB00898J
[31] VERMA V, VERMA P, RAY P, et al. Preparation of scaffolds from human hair proteins for tissue-engineering applications[J]. Biomedical Materials, 2008, 3(2): 025007. DOI: 10.1088/1748-6041/3/2/025007
[32] ABOUSHWAREB T, EBERLI D, WARD C, et al. A keratin biomaterial gel hemostat derived from human hair: Evaluation in a rabbit model of lethal liver injury[J]. Journal of Biomedical Materials Research Part B: Applied Biomaterials, 2009, 90(1): 45-54.
[33] RAHMANY M B, HANTGAN R R, VAN DYKE M. A mechanistic investigation of the effect of keratin-based hemostatic agents on coagulation[J]. Biomaterials, 2013, 34(10): 2492-2500. DOI: 10.1016/j.biomaterials.2012.12.008
[34] TANG A, LI Y, YAO Y, et al. Injectable keratin hydrogels as hemostatic and wound dressing materials[J]. Biomaterials Science, 2021, 9(11): 4169-4177. DOI: 10.1039/D1BM00135C
[35] LUO T, HAO S, CHEN X, et al. Development and assessment of kerateine nanoparticles for use as a hemostatic agent[J]. Materials Science and Engineering: C, 2016, 63: 352-358. DOI: 10.1016/j.msec.2016.03.007
[36] HAGHNIAZ R, GANGRADE A, MONTAZERIAN H, et al. An all-in-one transient theranostic platform for intelligent management of hemorrhage[J]. Advanced Science, 2023, 10(24): e2301406. DOI: 10.1002/advs.202301406
[37] ZHU H, WU B, FENG X, et al. Preparation and characterization of bioactive mesoporous calcium silicate-silk fibroin composite films[J]. Journal of Biomedical Materials Research Part B: Applied Biomaterials, 2011, 98(2): 330-341.
[38] SARAN K, SHI P, RANJAN S, et al. A moldable putty containing silk fibroin yolk shell particles for improved hemostasis and bone repair[J]. Advanced Healthcare Materials, 2015, 4(3): 432-445. DOI: 10.1002/adhm.201400411
[39] WEI W, LIU J, PENG Z, et al. Gellable silk fibroin-polyethylene sponge for hemostasis[J]. Artificial Cells Nanomedicine and Biotechnology, 2020, 48(1): 28-36. DOI: 10.1080/21691401.2019.1699805
[40] GIL E S, PANILAITIS B, BELLAS E, et al. Functionalized silk biomaterials for wound healing[J]. Advanced Healthcare Materials, 2013, 2(1): 206-217. DOI: 10.1002/adhm.201200192
[41] LI X, LI B, MA J, et al. Development of a silk fibroin/HTCC/PVA sponge for chronic wound dressing[J]. Journal of Bioactive and Compatible Polymers, 2014, 29(4): 398-411. DOI: 10.1177/0883911514537731
[42] QIAO Z, LYU X, HE S, et al. A mussel-inspired supramolecular hydrogel with robust tissue anchor for rapid hemostasis of arterial and visceral bleedings[J]. Bioactive Materials, 2021, 6(9): 2829-2840.
[43] SHEFA A A, TAZ M, LEE S Y, et al. Enhancement of hemostatic property of plant derived oxidized nanocellulose-silk fibroin based scaffolds by thrombin loading[J]. Carbohydrate Polymers, 2019, 208: 168-179. DOI: 10.1016/j.carbpol.2018.12.056
[44] LEI C, ZHU H, LI J, et al. Preparation and hemostatic property of low molecular weight silk fibroin[J]. Journal of Biomaterials Science, Polymer Edition, 2016, 27(5): 403-418. DOI: 10.1080/09205063.2015.1136918
[45] BARKUN A N, MOOSAVI S, MARTEL M. Topical hemostatic agents: A systematic review with particular emphasis on endoscopic application in GI bleeding[J]. Gastrointestinal Endoscopy, 2013, 77(5): 692-700. DOI: 10.1016/j.gie.2013.01.020
[46] HUANG X, FU Q, DENG Y, et al. Surface roughness of silk fibroin/alginate microspheres for rapid hemostasis in vitro and in vivo[J]. Carbohydrate Polymers, 2021, 253: 117256. DOI: 10.1016/j.carbpol.2020.117256
[47] LEE J, CHOI H N, CHA H J, et al. Microporous hemostatic sponge based on silk fibroin and starch with increased structural retentivity for contact activation of the coagulation cascade[J]. Biomacromolecules, 2023, 24(4): 1763-1773. DOI: 10.1021/acs.biomac.2c01512
[48] YANG C M, LEE J, LEE S Y, et al. Silk fibroin/tannin/ZnO nanocomposite hydrogel with hemostatic activities[J]. Gels, 2022, 8(10): 650. DOI: 10.3390/gels8100650
[49] SUN L, LI B, SONG W, et al. Comprehensive assessment of Nile tilapia skin collagen sponges as hemostatic dressings[J]. Materials Science and Engineering: C, 2020, 109: 110532. DOI: 10.1016/j.msec.2019.110532
[50] LIU X, ZHENG M, WANG X, et al. Biofabrication and characterization of collagens with different hierarchical architectures[J]. ACS Biomaterials Science & Engineering, 2019, 6(1): 739-748.
[51] JIANG X, WANG Y, FAN D, et al. A novel human-like collagen hemostatic sponge with uniform morphology, good biodegradability and biocompatibility[J]. Journal of Biomaterials Applications, 2017, 31(8): 1099-1107. DOI: 10.1177/0885328216687663
[52] WANG Q, CHEN J, WANG D, et al. Rapid hemostatic biomaterial from a natural bath sponge skeleton[J]. Marine Drugs, 2021, 19(4): 220. DOI: 10.3390/md19040220
[53] BROEKEMA F I, VAN OEVEREN W, BOERENDONK A, et al. Hemostatic action of polyurethane foam with 55% polyethylene glycol compared to collagen and gelatin[J]. Bio-Medical Materials and Engineering, 2016, 27(2-3): 149-159. DOI: 10.3233/BME-161578
[54] LUO J, MENG Y, ZHENG L, et al. Fabrication and characterization of Chinese giant salamander skin composite collagen sponge as a high-strength rapid hemostatic material[J]. Journal of Biomaterials Science, Polymer Edition, 2018, 30(4): 247-262.
[55] SHI X, FANG Q, DING M, et al. Microspheres of carboxymethyl chitosan, sodium alginate and collagen for a novel hemostatic in vitro study[J]. Journal of Biomaterials Applications, 2015, 30(7): 1092-1102.
[56] DAO M, CHENG X, SHAO Z, et al. Isolation, characterization and evaluation of collagen from jellyfish Rhopilema esculentum Kishinouye for use in hemostatic applications[J]. PLOS One, 2017, 12(1): e0169731. DOI: 10.1371/journal.pone.0169731
[57] CHENG W, ZHANG Z, XU R, et al. Incorporation of bacteriophages in polycaprolactone/collagen fibers for antibacterial hemostatic dual-function[J]. Journal of Biomedical Materials Research Part B: Applied Biomaterials, 2018, 106(7): 2588-2595. DOI: 10.1002/jbm.b.34075
[58] DING C, TIAN M, FENG R, et al. Novel self-healing hydrogel with injectable, pH-responsive, strain-sensitive, promoting wound-healing, and hemostatic properties based on collagen and chitosan[J]. ACS Biomaterials Science & Engineering, 2020, 6(7): 3855-3867.
[59] CIDREIRA A C M, DE CASTRO K C, HATAMI T, et al. Cellulose nanocrystals-based materials as hemostatic agents for wound dressings: A review[J]. Biomedical Microdevices, 2021, 23(4): 43. DOI: 10.1007/s10544-021-00581-0
[60] FAN X, LI M, YANG Q, et al. Morphology-controllable cellulose/chitosan sponge for deep wound hemostasis with surfactant and pore-foaming agent[J]. Materials Science and Engineering: C, 2021, 118: 111408. DOI: 10.1016/j.msec.2020.111408
[61] FAN X, LI Y, LI N, et al. Rapid hemostatic chitosan/cellulose composite sponge by alkali/urea method for massive haemorrhage[J]. International Journal of Biological Macromolecules, 2020, 164: 2769-2778. DOI: 10.1016/j.ijbiomac.2020.07.312
[62] LAURENCE S, BAREILLE R, BAQUEY C, et al. Development of a resorbable macroporous cellulosic material used as hemostatic in an osseous environment[J]. Journal of Biomedical Materials Research Part A, 2005, 73A(4): 422-429. DOI: 10.1002/jbm.a.30280
[63] LI B, PAN W, SUN X, et al. Hemostatic effect and safety evaluation of oxidized regenerated cellulose in total knee arthroplasty—A randomized controlledtrial[J]. BMC Musculoskeletal Disorders, 2023, 24(1): 797. DOI: 10.1186/s12891-023-06932-7
[64] AYDEMIR SEZER U, SAHIN İ, ARU B, et al. Cytotoxicity, bactericidal and hemostatic evaluation of oxidized cellulose microparticles: Structure and oxidation degree approach[J]. Carbohydrate Polymers, 2019, 219: 87-94. DOI: 10.1016/j.carbpol.2019.05.005
[65] HERNÁNDEZ-BONILLA S, RODRÍGUEZ-GARCÍA A M, JIMÉNEZ-HEFFERNAN J A, et al. FNA cytology of postoperative pseudotumoral lesions induced by oxidized cellulose hemostatic agents[J]. Cancer Cytopathology, 2019, 127(12): 765-770. DOI: 10.1002/cncy.22194
[66] VELÁZQUEZ-AVIÑA J, MÖNKEMÜLLER K, SAKAI P, et al. Hemostatic effect of oxidized regenerated cellulose in an experimental gastric mucosal resection model[J]. Endoscopy, 2014, 46(10): 878-882. DOI: 10.1055/s-0034-1365494
[67] BIAN J, BAO L, GAO X, et al. Bacteria-engineered porous sponge for hemostasis and vascularization[J]. Journal of Nanobiotechnology, 2022, 20(1): 47. DOI: 10.1186/s12951-022-01254-7
[68] OHTA S, NISHIYAMA T, SAKODA M, et al. Development of carboxymethyl cellulose nonwoven sheet as a novel hemostatic agent[J]. Journal of Bioscience and Bioengineering, 2015, 119(6): 718-723. DOI: 10.1016/j.jbiosc.2014.10.026
[69] LIU R, DAI L, SI C, et al. Antibacterial and hemostatic hydrogel via nanocomposite from cellulose nanofibers[J]. Carbohydrate Polymers, 2018, 195: 63-70. DOI: 10.1016/j.carbpol.2018.04.085
[70] UDANGAWA R N, MIKAEL P E, MANCINELLI C, et al. Novel cellulose—Halloysite hemostatic nanocomposite fibers with a dramatic reduction in human plasma coagulation time[J]. ACS Applied Materials & Interfaces, 2019, 11(17): 15447-15456.
[71] WU Z, ZHOU W, DENG W, et al. Antibacterial and hemostatic thiol-modified chitosan-immobilized AgNPs composite sponges[J]. ACS Applied Materials & Interfaces, 2020, 12(18): 20307-20320.
[72] RADWAN-PRAGŁOWSKA J, PIĄTKOWSKI M, DEINEKA V, et al. Chitosan-based bioactive hemostatic agents with antibacterial properties—Synthesis and characterization[J]. Molecules, 2019, 24(14): 2629.
[73] LAN G, LU B, WANG T, et al. Chitosan/gelatin composite sponge is an absorbable surgical hemostatic agent[J]. Colloids and Surfaces B: Biointerfaces, 2015, 136: 1026-1034. DOI: 10.1016/j.colsurfb.2015.10.039
[74] GHEORGHIȚĂ D, MOLDOVAN H, ROBU A, et al. Chitosan-based biomaterials for hemostatic applications: A review of recent advances[J]. International Journal of Molecular Sciences, 2023, 24(13): 10540. DOI: 10.3390/ijms241310540
[75] HU Z, ZHANG D Y, LU S T, et al. Chitosan-based composite materials for prospective hemostatic applications[J]. Marine Drugs, 2018, 16(8): 273. DOI: 10.3390/md16080273
[76] YAN T, CHENG F, WEI X, et al. Biodegradable collagen sponge reinforced with chitosan/calcium pyrophosphate nanoflowers for rapid hemostasis[J]. Carbohydrate Polymers, 2017, 170: 271-280. DOI: 10.1016/j.carbpol.2017.04.080
[77] DOWLING M B, KUMAR R, KEIBLER M A, et al. A self-assembling hydrophobically modified chitosan capable of reversible hemostatic action[J]. Biomaterials, 2011, 32(13): 3351-3357. DOI: 10.1016/j.biomaterials.2010.12.033
[78] KUMAR A, VIMAL A, KUMAR A. Why chitosan? From properties to perspective of mucosal drug delivery[J]. International Journal of Biological Macromolecules, 2016, 91: 615-622. DOI: 10.1016/j.ijbiomac.2016.05.054
[79] BELLICH B, D'AGOSTINO I, SEMERARO S, et al. "The Good, the Bad and the Ugly" of chitosans[J]. Marine Drugs, 2016, 14(5): 99. DOI: 10.3390/md14050099
[80] KHAN M A, MUJAHID M. A review on recent advances in chitosan based composite for hemostatic dressings[J]. International Journal of Biological Macromolecules, 2019, 124: 138-147. DOI: 10.1016/j.ijbiomac.2018.11.045
[81] LORD M S, CHENG B, MCCARTHY S J, et al. The modulation of platelet adhesion and activation by chitosan through plasma and extracellular matrix proteins[J]. Biomaterials, 2011, 32(28): 6655-6662. DOI: 10.1016/j.biomaterials.2011.05.062
[82] FISCHER T, THATTE H, NICHOLS T, et al. Synergistic platelet integrin signaling and factor XII activation in polyacetyl glucosamine fiber-mediated hemostasis[J]. Biomaterials, 2005, 26(27): 5433-5443.
[83] WU S, HUANG Z, YUE J, et al. The efficient hemostatic effect of Antarctic krill chitosan is related to its hydration property[J]. Carbohydrate Polymers, 2015, 132: 295-303. DOI: 10.1016/j.carbpol.2015.06.030
[84] LI B, WANG J, GUI Q, et al. Continuous production of uniform chitosan beads as hemostatic dressings by a facile flow injection method[J]. Journal of Materials Chemistry B, 2020, 8(35): 7941-7946. DOI: 10.1039/D0TB01462A
[85] SUN X, TANG Z, PAN M, et al. Chitosan/kaolin composite porous microspheres with high hemostatic efficacy[J]. Carbohydrate Polymers, 2017, 177: 135-143. DOI: 10.1016/j.carbpol.2017.08.131
[86] SUNDARAM M N, AMIRTHALINGAM S, MONY U, et al. Injectable chitosan-nano bioglass composite hemostatic hydrogel for effective bleeding control[J]. International Journal of Biological Macromolecules, 2019, 129: 936-943. DOI: 10.1016/j.ijbiomac.2019.01.220
[87] ZHENG Y, PAN N, LIU Y, et al. Novel porous chitosan/N-halamine structure with efficient antibacterial and hemostatic properties[J]. Carbohydrate Polymers, 2021, 253: 117205. DOI: 10.1016/j.carbpol.2020.117205
[88] LEE K Y, MOONEY D J. Alginate: Properties and biomedical applications[J]. Progress in Polymer Science, 2012, 37(1): 106-126. DOI: 10.1016/j.progpolymsci.2011.06.003
[89] CHEN Y, WU L, LI P, et al. Polysaccharide based hemostatic strategy for ultrarapid hemostasis[J]. Macromolecular Bioscience, 2020, 20(4): e1900370. DOI: 10.1002/mabi.201900370
[90] ALAVI M, RAI M. Recent progress in nanoformulations of silver nanoparticles with cellulose, chitosan, and alginic acid biopolymers for antibacterial applications[J]. Applied Microbiology and Biotechnology, 2019, 103(21-22): 8669-8676. DOI: 10.1007/s00253-019-10126-4
[91] PAN M, TANG Z, TU J, et al. Porous chitosan microspheres containing zinc ion for enhanced thrombosis and hemostasis[J]. Materials Science and Engineering: C, 2018, 85: 27-36. DOI: 10.1016/j.msec.2017.12.015
[92] ZHONG W. Efficacy and toxicity of antibacterial agents used in wound dressings[J]. Cutaneous and Ocular Toxicology, 2015, 34(1): 61-67. DOI: 10.3109/15569527.2014.890939
[93] TONG Z, YANG J, LIN L, et al. In situ synthesis of poly(γ- glutamic acid)/alginate/AgNP composite microspheres with antibacterial and hemostatic properties[J]. Carbohydrate Polymers, 2019, 221: 21-28. DOI: 10.1016/j.carbpol.2019.05.035
[94] CHOUDHARY H, RUDY M B, DOWLING M B, et al. Foams with enhanced rheology for stopping bleeding[J]. ACS Applied Materials & Interfaces, 2021, 13(12): 13958-13967.
[95] ZHENG Y, SHARIATI K, GHOVVATI M, et al. Hemostatic patch with ultra-strengthened mechanical properties for efficient adhesion to wet surfaces[J]. Biomaterials, 2023, 301: 122240. DOI: 10.1016/j.biomaterials.2023.122240
[96] YANG X, LIU W, XI G, et al. Fabricating antimicrobial peptide-immobilized starch sponges for hemorrhage control and antibacterial treatment[J]. Carbohydrate Polymers, 2019, 222: 115012.
[97] CHEN J, CHEN S, CHENG W, et al. Fabrication of porous starch microspheres by electrostatic spray and supercritical CO2 and its hemostatic performance[J]. International Journal of Biological Macromolecules, 2019, 123: 1-9. DOI: 10.1016/j.ijbiomac.2018.10.219
[98] PAVLOVIC S, BRANDAO P R G. Adsorption of starch, amylose, amylopectin and glucose monomer and their effect on the flotation of hematite and quartz[J]. Minerals Engineering, 2003, 16(11): 1117-1122. DOI: 10.1016/j.mineng.2003.06.011
[99] LIU G, GU Z, HONG Y, et al. Electrospun starch nanofibers: Recent advances, challenges, and strategies for potential pharmaceutical applications[J]. Journal of Controlled Release, 2017, 252: 95-107. DOI: 10.1016/j.jconrel.2017.03.016
[100] CHEN F, CAO X, YU J, et al. Quaternary ammonium groups modified starch microspheres for instant hemorrhage control[J]. Colloids and Surfaces B: Biointerfaces, 2017, 159: 937-944. DOI: 10.1016/j.colsurfb.2017.08.024
[101] MIRZAKHANIAN Z, FAGHIHI K, BARATI A, et al. Synthesis and characterization of fast-swelling porous superabsorbent hydrogel based on starch as a hemostatic agent[J]. Journal of Biomaterials Science, Polymer Edition, 2015, 26(18): 1439-1451. DOI: 10.1080/09205063.2015.1100496
[102] AWASTHI G P, ADHIKARI S P, KO S, et al. Facile synthesis of ZnO flowers modified graphene like MoS2 sheets for enhanced visible-light-driven photocatalytic activity and antibacterial properties[J]. Journal of Alloys and Compounds, 2016, 682: 208-215. DOI: 10.1016/j.jallcom.2016.04.267
[103] HOU Y, XIA Y, PAN Y, et al. Influences of mesoporous zinc-calcium silicate on water absorption, degradability, antibacterial efficacy, hemostatic performances and cell viability to microporous starch based hemostat[J]. Materials Science and Engineering: C, 2017, 76: 340-349. DOI: 10.1016/j.msec.2017.03.094
[104] QIAN J, CHEN Y, YANG H, et al. Preparation and characterization of crosslinked porous starch hemostatic[J]. International Journal of Biological Macromolecules, 2020, 160: 429-436. DOI: 10.1016/j.ijbiomac.2020.05.189
[105] SU H, WEI S, CHEN F, et al. Tranexamic acid-loaded starch hemostatic microspheres[J]. RSC Advances, 2019, 9(11): 6245-6253. DOI: 10.1039/C8RA06662K
[106] CUI R, CHEN F, ZHAO Y, et al. A novel injectable starch-based tissue adhesive for hemostasis[J]. Journal of Materials Chemistry B, 2020, 8(36): 8282-8293. DOI: 10.1039/D0TB01562H
[107] SU Y, CHEN H, LIU Q, et al. Thermoresponsive gels with embedded starch microspheres for optimized antibacterial and hemostatic properties[J]. ACS Applied Materials & Interfaces, 2024, 16(10): 12321-12331.
[108] MASTERS K S, SHAH D N, LEINWAND L A, et al. Crosslinked hyaluronan scaffolds as a biologically active carrier for valvular interstitial cells[J]. Biomaterials, 2005, 26(15): 2517-2525. DOI: 10.1016/j.biomaterials.2004.07.018
[109] YAMANLAR S, SANT S, BOUDOU T, et al. Surface functionalization of hyaluronic acid hydrogels by polyelectrolyte multilayer films[J]. Biomaterials, 2011, 32(24): 5590-5599. DOI: 10.1016/j.biomaterials.2011.04.030
[110] WANG Y, LIU G, WU L, et al. Rational design of porous starch/hyaluronic acid composites for hemostasis[J]. International Journal of Biological Macromolecules, 2020, 158: 1319-1329. DOI: 10.1016/j.ijbiomac.2020.05.018
[111] ABALLAY A, HERMANS M H E. Neodermis formation in full thickness wounds using an esterified hyaluronic acid matrix[J]. Journal of Burn Care & Research, 2019, 40(5): 585-589. DOI: 10.1093/jbcr/irz057
[112] ROEHRS H, STOCCO J G, POTT F, et al. Dressings and topical agents containing hyaluronic acid for chronic wound healing[J]. The Cochrane Database of Systematic Reviews, 2023, 7(7): Cd012215.
[113] LIU J Y, LI Y, HU Y, et al. Hemostatic porous sponges of cross-linked hyaluronic acid/cationized dextran by one self-foaming process[J]. Materials Science and Engineering: C, 2018, 83: 160-168. DOI: 10.1016/j.msec.2017.10.007
[114] WANG M, HU J, OU Y, et al. Shape-recoverable hyaluronic acid-waterborne polyurethane hybrid cryogel accelerates hemostasis and wound healing[J]. ACS Applied Materials & Interfaces, 2022, 14(15): 17093-17108.
[115] CHEN Z, YE S Y, YANG Y, et al. A review on charred traditional Chinese herbs: Carbonization to yield a haemostatic effect[J]. Pharmaceutical Biology, 2019, 57(1): 498-506. DOI: 10.1080/13880209.2019.1645700
[116] DONG X, LIANG W, MEZIANI M J, et al. Carbon dots as potent antimicrobial agents[J]. Theranostics, 2020, 10(2): 671-686. DOI: 10.7150/thno.39863
[117] LI D, XU K Y, ZHAO W P, et al. Chinese medicinal herb-derived carbon dots for common diseases: Efficacies and potential mechanisms[J]. Frontiers in Pharmacology, 2022, 13: 815479.
[118] SZCZEPANKOWSKA J, KHACHATRYAN G, KHACHATRYAN K, et al. Carbon dots—Types, obtaining and application in biotechnology and food technology[J]. International Journal of Molecular Sciences, 2023, 24(19): 14984.
[119] VILLALBA-RODRÍGUEZ A M, GONZÁLEZ-GONZÁLEZ R B, MARTÍNEZ-RUIZ M, et al. Chitosan-based carbon dots with applied aspects: New frontiers of international interest in a material of marine origin[J]. Marine Drugs, 2022, 20(12): 782. DOI: 10.3390/md20120782
[120] KRYSTYJAN M, KHACHATRYAN G, KHACHATRYAN K, et al. Polysaccharides composite materials as carbon nanoparticles carrier[J]. Polymers, 2022, 14(5): 948. DOI: 10.3390/polym14050948
[121] YAN X, ZHAO Y, LUO J, et al. Hemostatic bioactivity of novel pollen typhae carbonisata-derived carbon quantum dots[J]. Journal of Nanobiotechnology, 2017, 15(1): 60. DOI: 10.1186/s12951-017-0296-z
[122] LI S, GUO Z, ZHANG Y, et al. Blood compatibility evaluations of fluorescent carbon dots[J]. ACS Applied Materials & Interfaces, 2015, 7(34): 19153-19162.
[123] HAN B, SHEN L, XIE H, et al. Synthesis of carbon dots with hemostatic effects using traditional Chinese medicine as a biomass carbon source[J]. ACS Omega, 2023, 8(3): 3176-3183. DOI: 10.1021/acsomega.2c06600
[124] WU T, LI M, LI T, et al. Natural biomass-derived carbon dots as a potent solubilizer with high biocompatibility and enhanced antioxidant activity[J]. Frontiers in Molecular Biosciences, 2023, 10: 1284599. DOI: 10.3389/fmolb.2023.1284599
[125] ZHANG J, ZOU L, LI Q, et al. Carbon dots derived from traditional Chinese medicines with bioactivities: A rising star in clinical treatment[J]. ACS Applied Bio Materials, 2023, 6(10): 3984-4001. DOI: 10.1021/acsabm.3c00462
[126] SUN Z, LU F, CHENG J, et al. Haemostatic bioactivity of novel Schizonepetae Spica Carbonisata-derived carbon dots via platelet counts elevation[J]. Artificial Cells, Nanomedicine, and Biotechnology, 2018, 46(sup3): 308-317. DOI: 10.1080/21691401.2018.1492419
[127] LIU X, WANG Y, YAN X, et al. Novel Phellodendri Cortex (Huang Bo)-derived carbon dots and their hemostatic effect[J]. Nanomedicine: Nanotechnology, Biology, and Medicine, 2018, 13(4): 391-405.
[128] ZHANG M, CHENG J, LUO J, et al. Protective effects of Scutellariae Radix Carbonisata-derived carbon dots on blood-heat and hemorrhage rats[J]. Frontiers in Pharmacology, 2023, 14: 1118550. DOI: 10.3389/fphar.2023.1118550
[129] ALTINTIG E, SARICI B, KARATAŞ S. Prepared activated carbon from hazelnut shell where coated nanocomposite with Ag+ used for antibacterial and adsorption properties[J]. Environmental Science and Pollution Research, 2022, 30(5): 13671-13687. DOI: 10.1007/s11356-022-23004-w
[130] CHENG P, XUE X, SU J, et al. 1H NMR-based metabonomic revealed protective effect of Moutan Cortex charcoal on blood-heat and hemorrhage rats[J]. Journal of Pharmaceutical and Biomedical Analysis, 2019, 169: 151-158. DOI: 10.1016/j.jpba.2019.02.044
[131] ZHANG Y, GE X L, ZHANG J, et al. Effect of the oxygenic groups on activated carbon on its hemocompatibility[J]. Colloids and Surfaces B: Biointerfaces, 2024, 233: 113655. DOI: 10.1016/j.colsurfb.2023.113655
[132] WANG S, ZHANG Y, SHI Y, et al. Rhubarb charcoal-crosslinked chitosan/silk fibroin sponge scaffold with efficient hemostasis, inflammation, and angiogenesis for promoting diabetic wound healing[J]. International Journal of Biological Macromolecules, 2023, 253(Pt 2): 126796.
-
目的
全世界每年约有6000万人遭受严重创伤,其中约200万人因为受创后无法控制的严重出血而死亡,意外创伤造成的严重出血的黄金急救时间只有几分钟。因此,伤口的快速止血和愈合对于解决意外事故造成的出血具有重要意义。本文以止血材料的生物质来源为依托,探索生物质止血材料的种类、制备方法与止血性能之间的相关性,同时关注其在具体应用中的止血效果。
方法利用生物质材料在临床止血领域的发展现状为依托,采用分类法来探究不同生物质原材料及制备方法对止血材料的止血性能和生物相容性的影响,并分析其相应的止血应用效果,即从止血时间和失血量来分析其止血效果。
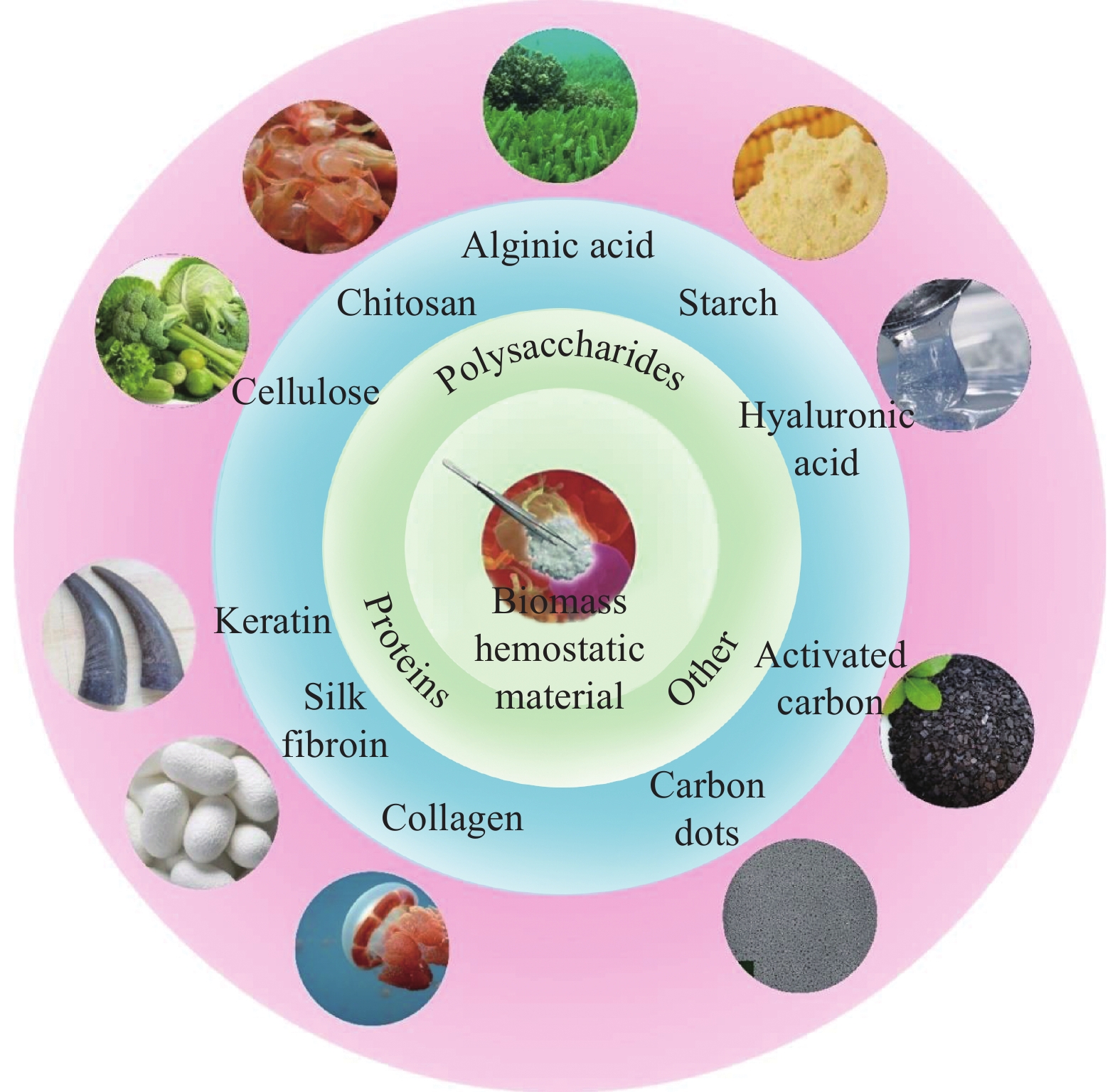
结果依据原材料来源的不同,生物质止血材料主要可分为以下几种:(1)蛋白类止血材料,包括角蛋白类、丝素蛋白类和胶原蛋白类;(2)多糖类止血材料,包括纤维素类、壳聚糖类、海藻酸类、淀粉类和透明质酸类;(3)其他生物质止血材料,包括中药类碳点和中药类活性炭。以角蛋白、丝素蛋白、胶原蛋白为代表的蛋白类和以纤维素、壳聚糖、海藻酸为代表的多糖类等生物质材料,因其无毒性、低抗原性、良好的生物相容性、生物可降解性等优点在止血领域展现了前所未有的应用价值。同时,复合生物质止血材料能够综合各种生物质材料的优点,具备更好的止血效果和生物相容性。
结论生物质止血材料相比其他止血材料具有独特的天然优势,如角蛋白类止血材料能够促使纤维蛋白原聚合成纤维蛋白,加速凝血;丝素蛋白类止血材料能够促进成纤维细胞增殖、再上皮化和胶原合成,以此促进伤口的止血和愈合;胶原蛋白类止血材料的抗原性低;纤维素类止血材料具备优异的机械性能和可再生性;壳聚糖类止血材料表面的阳离子簇可以与红细胞上的阴离子相互作用,诱导血小板聚集,实现止血;海藻酸类止血材料具有优异的吸水性和生物降解性;淀粉类止血材料和透明质酸类止血材料具有优异的生物相容性和可降解性。但无论哪一类生物质止血材料都很难实现完美止血性、高抗菌性及低毒和低抗原性的并存,复合生物质止血材料必然是未来的发展趋势。此外,新型生物质止血材料的开发必需综合考虑各种生物质材料的物理性质、化学性质和生理学性质,以赋予其优异的止血性、低毒性、低抗原性、抗菌性和生物相容性等综合性能。





 下载:
下载: