Advanced Ti3C2@ε-MnO2 cathode as rechargeable aqueous zinc-ion batteries
-
摘要: 可充电水性锌-氧化锰(Zn-MnOx)电池具有成本低、安全性高、易于安装等特点,成为太阳能及风能储能装置的最佳选择。由于MnOx导电性欠佳,导致电池循环性能较差,为解决此问题,本文采用导电性优异、具有丰富化学终端(Tx,如=O、—F、—OH)的二维层状过渡金属碳化物(MXene) Ti3C2Tx材料作为MnOx颗粒的良好载体。基于化学终端的电负性,Mn2+能够与其产生强静电吸引,从而嵌入Ti3C2Tx MXene材料层间并吸附在其表面,使生成的Mn3O4颗粒牢牢地锚定在Ti3C2Tx MXene上,形成了Ti3C2@Mn3O4复合材料。当作为水性锌离子电池的正极材料时,Ti3C2@Mn3O4在第1次充电过程中,完全转化为Ti3C2@ε-MnO2。由于Ti3C2Tx MXene材料优异的导电性及层状结构,使Ti3C2@ε-MnO2电极展现出了优异的动力学和电化学性能,在0.2 C (1 C=308 mA·h·g−1)倍率下放电时,比容量高达440 mA·h·g−1,能量密度为607 W·h·kg−1,在1 C倍率下循环150次后,容量从270 mA·h·g−1增长至480 mA·h·g−1。优异的电池性能,简单的材料制备方法再加上低成本、高安全性及易于组装的特性,使可充电水性Zn-MnOx电池在大规模储能装置上的应用成为可能。
-
关键词:
- 可充电水性锌离子电池 /
- Mn3O4 /
- Ti3C2Tx MXene /
- Ti3C2@ε-MnO2电极 /
- 可充电电池
Abstract: Due to the low cost, high safety and easy assembly, rechargeable aqueous zinc-manganese oxide (Zn-MnOx) batteries are the best devices for energy storage. However, poor conductivity of MnOx results in the bad cycle performance. Herein, highly conductive and layered Ti3C2Tx MXene with rich terminations (Tx, for example, =O, —F, —OH) were used as carriers for MnOx particles. Due to the electronegativity of the terminations, Mn2+ was intercalated into the layers and adsorbed on the surface of Ti3C2Tx MXene, making the generated Mn3O4 particles can firmly anchored, forming the Ti3C2@Mn3O4 composites. As for the cathode of zinc-ion batteries, Ti3C2@Mn3O4 was fully converted to Ti3C2@ε-MnO2 during the 1st charge process. Thanks to the excellent conductivity and layered structure of Ti3C2Tx MXene, Ti3C2@ε-MnO2 cathode presents excellent kinetic properties and electrochemical performance with a high specific capacity of 440 mA·h·g−1 and high energy density (607 W·h·kg−1) at 0.2 C (1 C=308 mA·h·g−1), and the capacities increase from 270 mA·h·g−1 to 480 mA·h·g−1 after 150 cycles at 1 C. Excellent electrochemical performance, simple material preparation methods, combined with the low cost, high safety and easy assembly characteristics, enable the possible application of rechargeable aqueous Zn-MnOx batteries in large-scale energy storage. -
-
图 1 Ti3AlC2、Ti3C2Tx MXene、Mn3O4、Ti3C2@Mn3O4的XRD图谱 ((a), (b));Ti3C2@Mn3O4 (c)和Mn3O4 (d) 的Brunner-Emmet-Teller曲线;Ti3C2@Mn3O4中Ti2p (e)、C1s (f)、Mn3s (g)、O1s (h) 的XPS图谱
Figure 1. XRD patterns of Ti3AlC2, Ti3C2Tx MXene, Mn3O4, Ti3C2@Mn3O4 ((a), (b)); Brunner-Emmet-Teller curves of Ti3C2@Mn3O4 (c) and Mn3O4 (d); XPS patterns of Ti2p (e), C1s (f), Mn3s (g) and O1s (h) in Ti3C2@Mn3O4
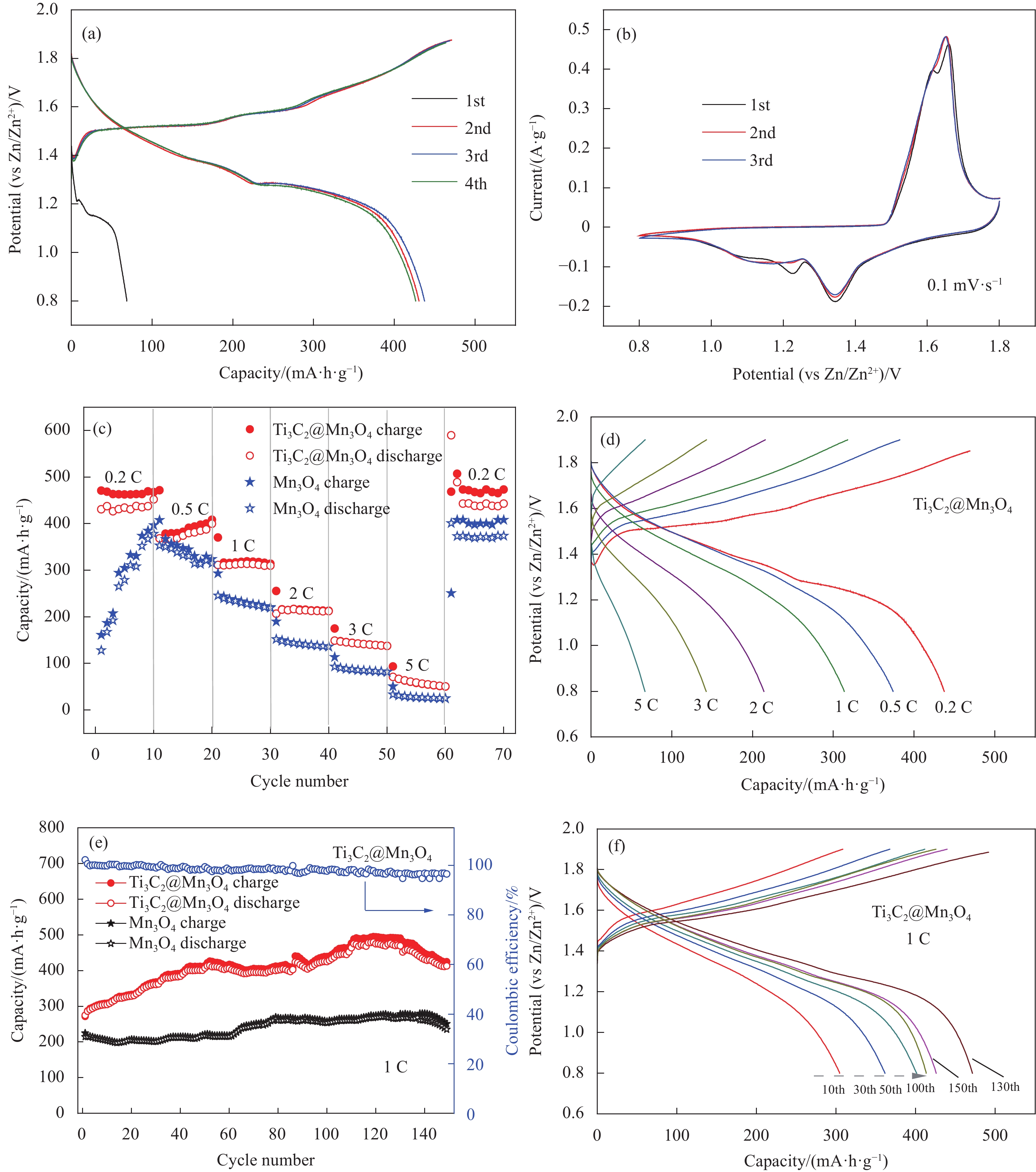
图 3 Ti3C2@Mn3O4的电化学性能:(a) 0.2 C (1 C=308 mA·h·g−1)倍率下的充放电曲线;(b) 0.1 mV·s−1扫速下的循环伏安(CV)曲线;((c), (d)) 倍率性能及不同倍率下的充放电曲线;((e), (f)) 1 C倍率下的循环性能及不同循环次数的充放电曲线
Figure 3. Electrochemical performance of Ti3C2@Mn3O4: (a) Galvanostatic discharge/charge curves at 0.2 C; (b) Cyclic voltammetry curves at 0.1 mV·s−1; ((c), (d)) Rate performance and corresponding charge/discharge curves at different ratios; ((e), (f)) Cycling performance at 1 C and corresponding charge/discharge curves at different cycles
图 4 Ti3C2@Mn3O4电极经过不同循环次数后的XRD图谱 (a);Ti3C2@Mn3O4电极充电至1.9 V的SEM微观形貌图:(b) 在0.2 C倍率下经过第1次充电后;(c) 在0.2 C倍率下循环20次后再在1 C倍率下循环3次;(d) 继续在1 C倍率下循环150次
Figure 4. XRD patterns of Ti3C2@Mn3O4 cathode after different cycles (a) ; SEM images of Ti3C2@Mn3O4 cathode recharged to 1.9 V: (b) After 1st charge at 0.2 C; (c) After 20 cycles at 0.2 C and 3 cycles at 1 C; (d) 150 cycles at 1 C
SUS—Steel use stainless
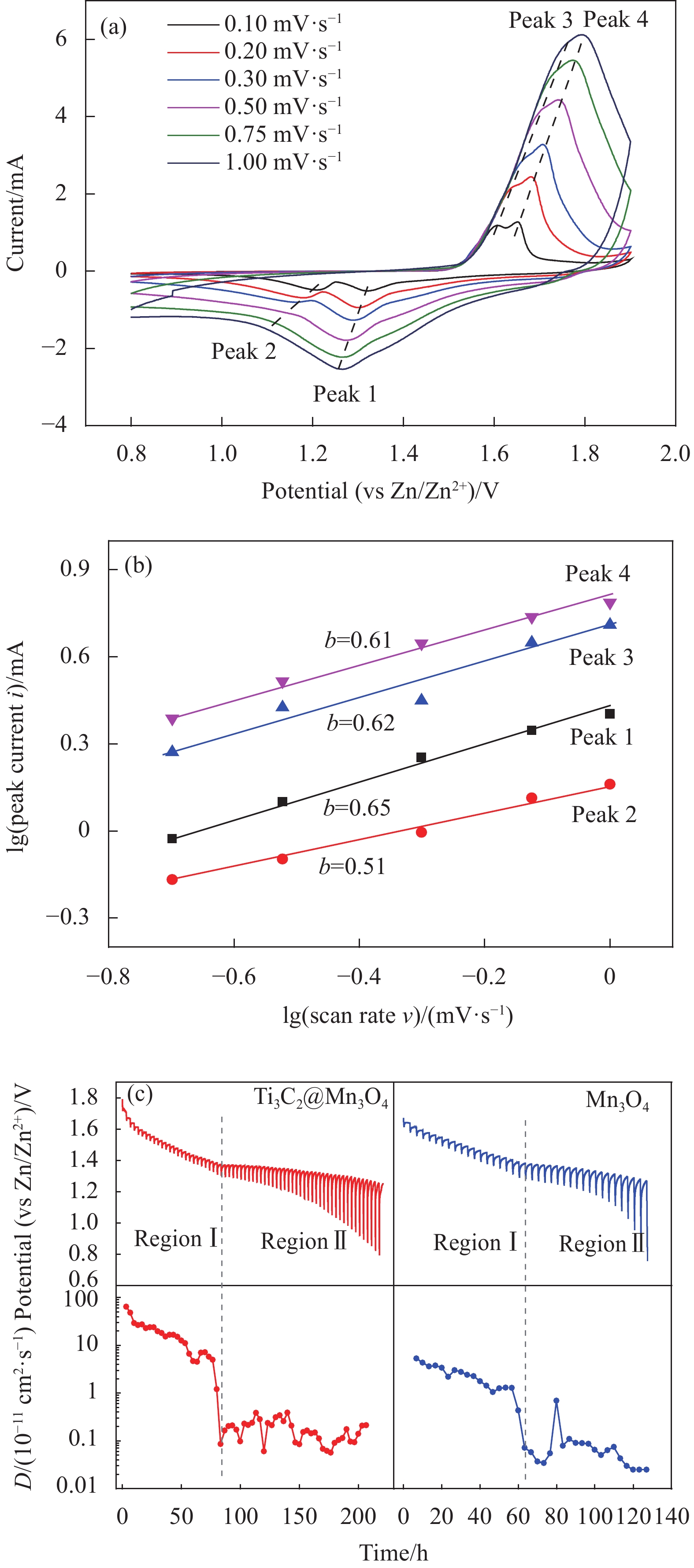
图 5 (a) Ti3C2@Mn3O4电极在不同扫速下的CV曲线;(b)不同峰位lgi和lgυ的拟合曲线;(c) Ti3C2@Mn3O4和Mn3O4电极的恒流间歇滴定(GITT)曲线及相应的离子扩散系数D
Figure 5. (a) CV curves of Ti3C2@Mn3O4 cathode at different scan rates; (b) lgi and lgυ plots at specific peak currents; (c) Galvanostatic intermittent titration technique curves and the corresponding ion diffusion coefficients D of Ti3C2@Mn3O4 and Mn3O4 cathode
b—Slope of the fitted curve
图 6 (a) Ti3C2@Mn3O4电极在0.2 C倍率下循环20次后放电至0.8 V的XRD图谱;(b) Ti3C2@Mn3O4电极在0.2 C倍率下循环20次后完全放电至0.8 V和充电至1.9 V的Mn2p XPS图谱
Figure 6. (a) XRD pattern of Ti3C2@Mn3O4 cathode after fully discharged to 0.8 V at 0.2 C after 20 cycles; (b) XPS spectrums of Mn2p for Ti3C2@Mn3O4 cathode fully discharged to 0.8 V and recharged to 1.9 V after 20 cycles at 0.2 C
-
[1] GOODENOUGH J B, KIM Y. Challenges for rechargeable Li batteries[J]. Chemistry of Materials,2010,22(3):587-603. DOI: 10.1021/cm901452z
[2] POSADA J O G, RENNIE A J R, VILLAR S P, et al. Aqueous batteries as grid scale energy storage solutions[J]. Renewable & Sustainable Energy Reviews,2017,68(2):1174-1182. DOI: 10.1016/j.rser.2016.02.024
[3] PARKER J F, CHERVIN C N, PALA I R, et al. Rechargeable nickel-3D zinc batteries: An energy-dense, safer alternative to lithium-ion[J]. Science,2017,356(6336):415-418. DOI: 10.1126/science.aak9991
[4] ALFARUQI M H, MATHEW V, SONG J, et al. Electrochemical zinc intercalation in lithium vanadium oxide: A high-capacity zinc-ion battery cathode[J]. Chemistry of Materials,2017,29(4):1684-1694. DOI: 10.1021/acs.chemmater.6b05092
[5] PAN H L, SHAO Y Y, YAN P F, et al. Reversible aqueous zinc/manganese oxide energy storage from conversion reactions[J]. Nature Energy,2016,1:16039. DOI: 10.1038/nenergy.2016.39
[6] ZHANG N, CHENG F, LIU J, et al. Rechargeable aqueous zinc-manganese dioxide batteries with high energy and power densities[J]. Nature Communications,2017,8(1):405-414. DOI: 10.1038/s41467-017-00467-x
[7] HUANG J, WANG Z, HOU M, et al. Polyaniline-intercalated manganese dioxide nanolayers as a high-perfor-mance cathode material for an aqueous zinc-ion battery[J]. Nature Communications,2018,9(1):2906-2914. DOI: 10.1038/s41467-018-04949-4
[8] GAO X, WU H, LI W, et al. H+-insertion boosted α-MnO2 for an aqueous Zn-ion battery[J]. Small,2020,16(5):1905842. DOI: 10.1002/smll.201905842
[9] CHEN C, SHI M, ZHAO Y, et al. Al-intercalated MnO2 cathode with reversible phase transition for aqueous Zn-ion batteries[J]. Chemical Engineering Journal,2021,422:130375. DOI: 10.1016/j.cej.2021.130375
[10] LIU W, ZHANG X, HUANG Y, et al. β-MnO2 with proton conversion mechanism in rechargeable zinc ion battery[J]. Journal of Energy Chemistry,2021,56(1):365-373. DOI: 10.1016/j.jechem.2020.07.027
[11] LI L, HOANG T K A, ZHI J, et al. Functioning mechanism of the secondary aqueous Zn-β-MnO2 battery[J]. ACS Applied Materials & Interfaces,2020,12(11):12834-12846. DOI: 10.1021/acsami.9b22758
[12] ALFARUQI M H, MATHEW V, GIM J, et al. Electrochemically induced structural transformation in a γ-MnO2 cathode of a high capacity zinc-ion battery system[J]. Chemistry of Materials,2015,27(10):3609-3620. DOI: 10.1021/cm504717p
[13] ALFARUQI M H, GIM J, KIM S, et al. A layered δ-MnO2 nanoflake cathode with high zinc-storage capacities for eco-friendly battery applications[J]. Electrochemistry Communications,2015,60(1):121-125. DOI: 10.1016/j.elecom.2015.08.019
[14] YUAN C, ZANG Y, PAN Y, et al. Investigation of the intercalation of polyvalent cations (Mg2+, Zn2+) into λ-MnO2 for rechargeable aqueous battery[J]. Electrochimica Acta,2014,116(2):404-412. DOI: 10.1016/j.electacta.2013.11.090
[15] ZHANG M, WU W, LUO J, et al. A high-energy-density aqueous zinc-manganese battery with a La–Ca co-doped ε-MnO2 cathode[J]. Journal of Materials Chemistry A,2020,8(23):11642-11648. DOI: 10.1039/D0TA03706K
[16] HUANG L, LUO X, CHEN C, et al. A high specifc capacity aqueous zinc-manganese battery with a ε-MnO2 cathode[J]. Ionics,2021,27(9):3933-3941. DOI: 10.1007/s11581-021-04160-4
[17] ZHOU Z, WANG L, LIANG J, et al. Two-dimensional hierarchical Mn2O3@graphene as high rate and ultrastable cathode for aqueous zinc-ion batteries[J]. Journal of Materials Chemistry C,2021,9(1):1326-1332. DOI: 10.1039/D0TC04984K
[18] MAO M, WU X, HU Y, et al. Charge storage mechanism of MOF-derived Mn2O3 as high performance cathode of aqueous zinc-ion batteries[J]. Journal of Energy Che-mistry,2021,52(1):277-283. DOI: 10.1016/j.jechem.2020.04.061
[19] LIU N, WU X, YIN Y, et al. Constructing the efficient ion diffusion pathway by introducing oxygen defects in Mn2O3 for high-performance aqueous zinc-ion batteries[J]. ACS Applied Materials & Interfaces,2020,12(25):28199-28205. DOI: 10.1021/acsami.0c05968
[20] DHIMAN A, IVEY D G. Electrodeposited manganese oxide on carbon paper for zinc-ion battery cathodes[J]. Batteries & Supercaps,2020,3(3):293-305. DOI: 10.1002/batt.201900150
[21] TAN Q, LI X, ZHANG B, et al. Valence engineering via in situ carbon reduction on octahedron sites Mn3O4 for ultra-long cycle life aqueous Zn-ion battery[J]. Advanced Energy Materials,2020,10(38):2001050. DOI: 10.1002/aenm.202001050
[22] CHEN H, ZHOU W, ZHU D, et al. Porous cube-like Mn3O4@C as an advanced cathode for low-cost neutral zinc-ion battery[J]. Journal of Alloys and Compounds,2020,813:151812. DOI: 10.1016/j.jallcom.2019.151812
[23] LONG J, YANG Z, YANG F, et al. Electrospun core-shell Mn3O4/carbon fibers as high-performance cathode materials for aqueous zinc-ion batteries[J]. Electrochimica Acta,2020,344(1):136155. DOI: 10.1016/j.electacta.2020.136155
[24] ANASORI B, LUKATSKAYA M R, GOROTSI Y. 2D metal carbides and nitrides (MXenes) for energy storage[J]. Nature Review Materials,2017,2(2):16098. DOI: 10.1038/natrevmats.2016.98
[25] KAMYSBAYEV V, FILATOV A S, HU H, et al. Covalent surface modifications and superconductivity of two-dimensional metal carbide MXenes[J]. Science,2020,369(6506):979-983. DOI: 10.1126/science.aba8311
[26] LI M, LU J, LUO K, et al. Element replacement approach by reaction with Lewis acidic molten salts to synthesize nanolaminated MAX phases and MXenes[J]. Journal of Ameri-can Chemical Society,2019,141(11):4730-4737. DOI: 10.1021/jacs.9b00574
[27] BI S, WU Y, CAO A, et al. Free-standing three-dimensional carbon nanotubes/amorphous MnO2 cathodes for aqueous zinc-ion batteries with superior rate performance[J]. Materials Today Energy,2020,18(1):100548. DOI: 10.1016/j.mtener.2020.100548
[28] TONG H, LI T, LIU J, et al. Fabrication of the oxygen vacancy amorphous MnO2/carbon nanotube as cathode for advanced aqueous zinc-ion batteries[J]. Energy Technology,2021,9(2):2000769. DOI: 10.1002/ente.202000769
[29] CHEN X, LIA W, ZENG Z, et al. Engineering stable Zn-MnO2 batteries by synergistic stabilization between the carbon nanofiber core and birnessite-MnO2 nanosheets shell[J]. Chemical Engineering Journal,2021,405(1):126969. DOI: 10.1016/j.cej.2020.126969
[30] WEI C, TAO Y, AN Y, et al. Recent advances of emerging 2D MXene for stable and sendrite-free metal anodes[J]. Advanced Functional Materials,2020,30(45):2004613. DOI: 10.1002/adfm.202004613
[31] ZHU X, CAO Z, WANG W, et al. Superior-performance aqueous zinc-ion batteries based on the in situ growth of MnO2 nanosheets on V2CTx MXene[J]. ACS Nano,2021,15(2):2971-2983. DOI: 10.1021/acsnano.0c09205
[32] WANG L, CAO X, XU L, et al. Transformed akhtenskite MnO2 from Mn3O4 as cathode for a rechargeable aqueous zinc ion battery[J]. ACS Sustainable Chemistry & Engineering,2018,6(12):16055-16063. DOI: 10.1021/acssuschemeng.8b02502
[33] SUN W, WANG F, HOU S, et al. Zn/MnO2 battery chemistry with H+ and Zn2+ coinsertion[J]. Journal of the American Chemical Society,2017,139(29):9775-9778. DOI: 10.1021/jacs.7b04471
[34] ZHAO Q, CHEN X, WANG Z, et al. Unravelling H+/Zn2+ synergistic intercalation in a novel phase of manganese oxide for high-performance aqueous rechargeable battery[J]. Small,2019,15(47):1904545. DOI: 10.1002/smll.201904545
[35] CHEN W, LI G, PEI A , et al. A manganese-hydrogen battery with potential for grid-scale energy storage[J]. Nature Energy,2018,3(5):428-435. DOI: 10.1038/s41560-018-0147-7
[36] CHAO D, ZHOU W, YE C, et al. An electrolytic Zn-MnO2 battery for high-voltage and scalable energy storage[J]. Angewandte Chemie International Edition,2019,58(23):7823-7828. DOI: 10.1002/anie.201904174
[37] LIU M, ZHAO Q, LIU H, et al. Tuning phase evolution of β-MnO2 during microwave hydrothermal synthesis for high-performance aqueous Zn ion battery[J]. Nano Energy,2019,64(1):103942. DOI: 10.1016/j.nanoen.2019.103942
-
期刊类型引用(8)
1. 徐康康,程蹈,刘点,陈玉霞,涂道伍,郭勇,吴自成. 无损检测技术在纤维增强聚合物复合材料机械损伤监测中的应用进展. 塑料工业. 2024(02): 8-15 .  百度学术
百度学术
2. 孟凌霄,石文泽,卢超,黄良,凌建. 碳纤维增强树脂基复合材料气瓶电磁超声在线监测方法及失效机制. 复合材料学报. 2024(04): 1820-1829 .  本站查看
本站查看
3. 熊政辉,陈俊超,肖树坤,荆砚,何喜,陈尧. 应用于斜楔块的子孔径虚拟源全聚焦成像. 应用声学. 2024(03): 625-634 .  百度学术
百度学术
4. 曹欢庆,朱启民,赵培含,何梓科,郭师峰. 复杂型面结构超声成像检测研究进展. 仪器仪表学报. 2024(06): 42-53 .  百度学术
百度学术
5. 李荣光,朱甜甜,孙伶,陈斯迅,周文彬,周正干. 管道碳纤维复合材料修复层阵列超声检测方法提高检测精度. 石油钻采工艺. 2024(06): 728-742 .  百度学术
百度学术
6. 夏玉秀,张义凤,薛峰. 相控阵超声检测技术在航空领域应用研究进展. 无损探伤. 2023(04): 6-10+38 .  百度学术
百度学术
7. 杨红娟,杨正岩,杨雷,单一男,林奎旭,武湛君. 碳纤维复合材料损伤的超声检测与成像方法研究进展. 复合材料学报. 2023(08): 4295-4317 .  本站查看
本站查看
8. 陈学宽,龙盛蓉,宋奕霖,邹越豪,李志农. 基于二维等效声速的频域全聚焦超声成像研究. 仪器仪表学报. 2023(08): 130-140 .  百度学术
百度学术
其他类型引用(5)
-





 下载:
下载: