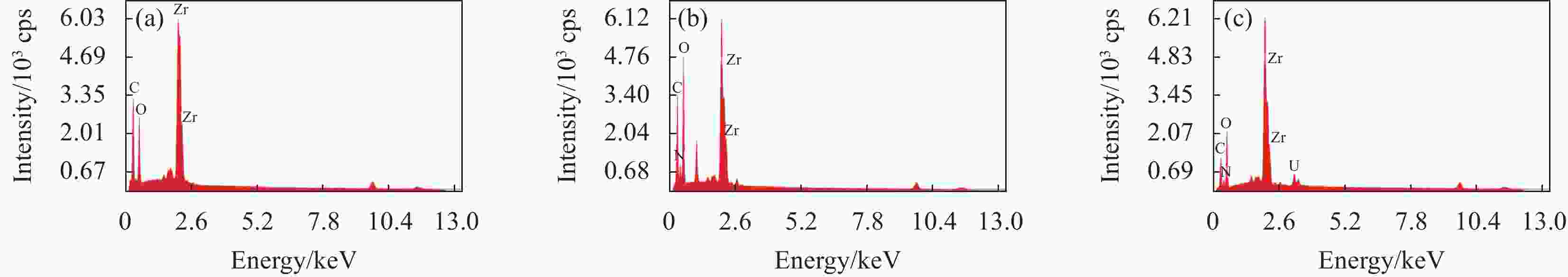
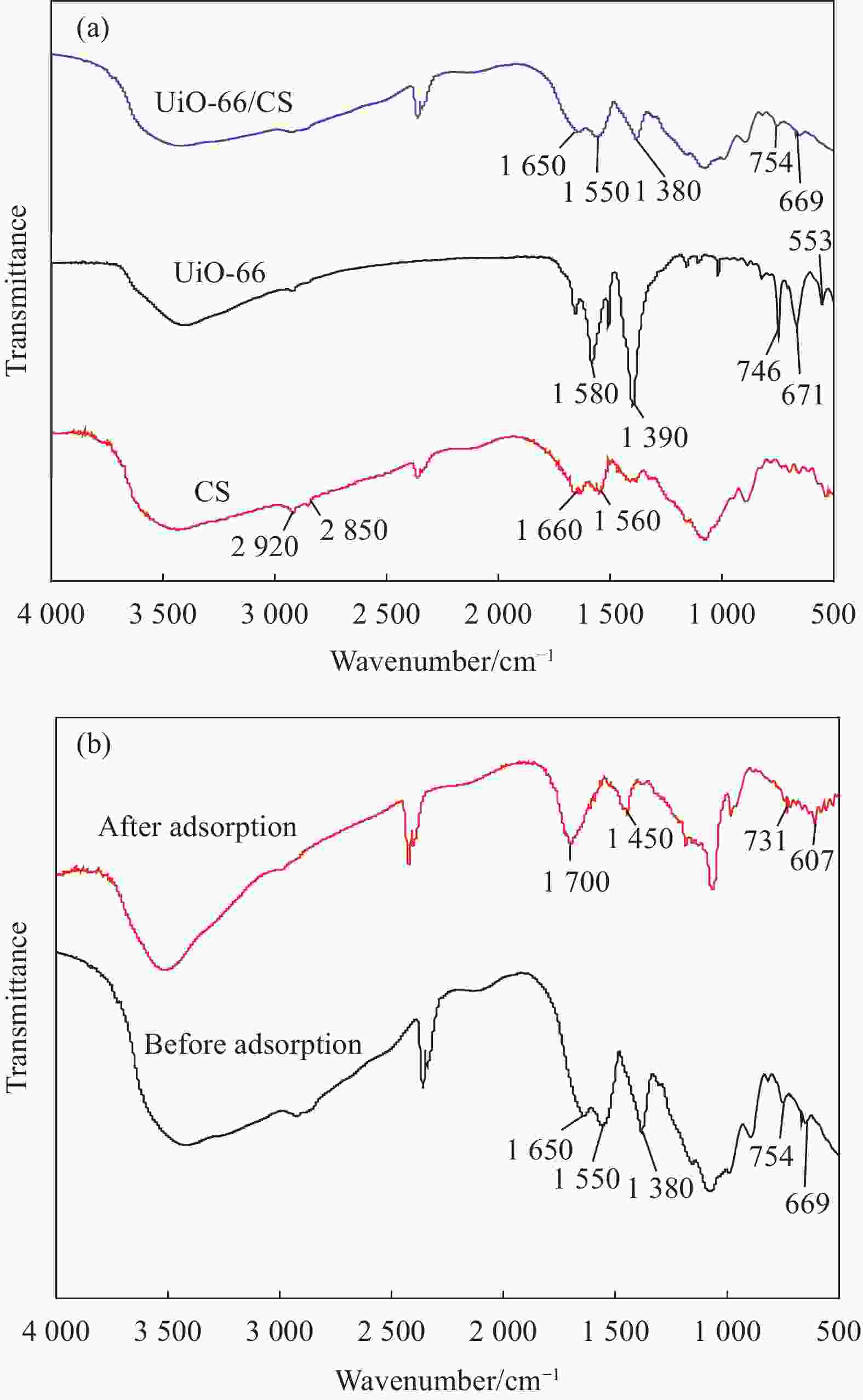
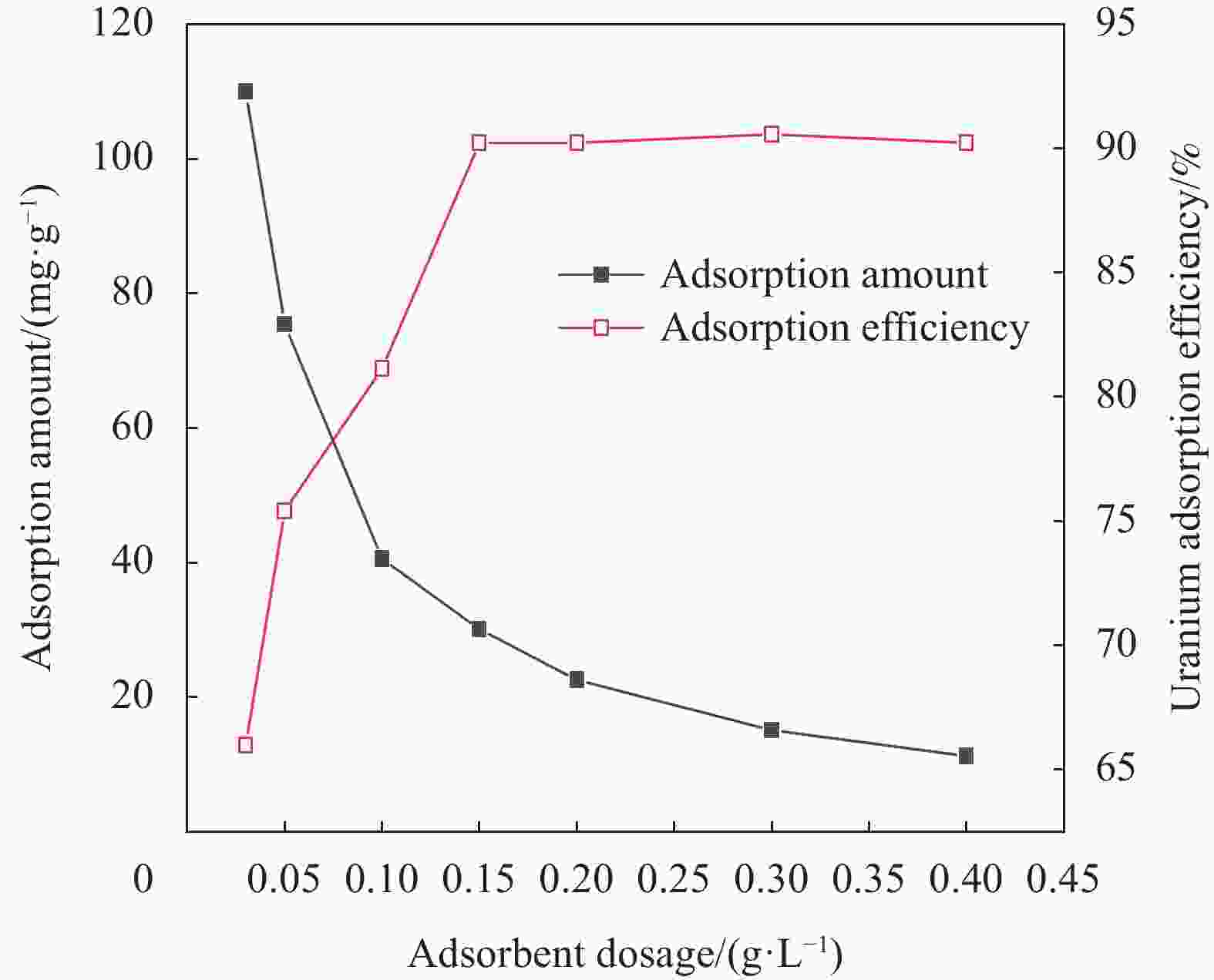
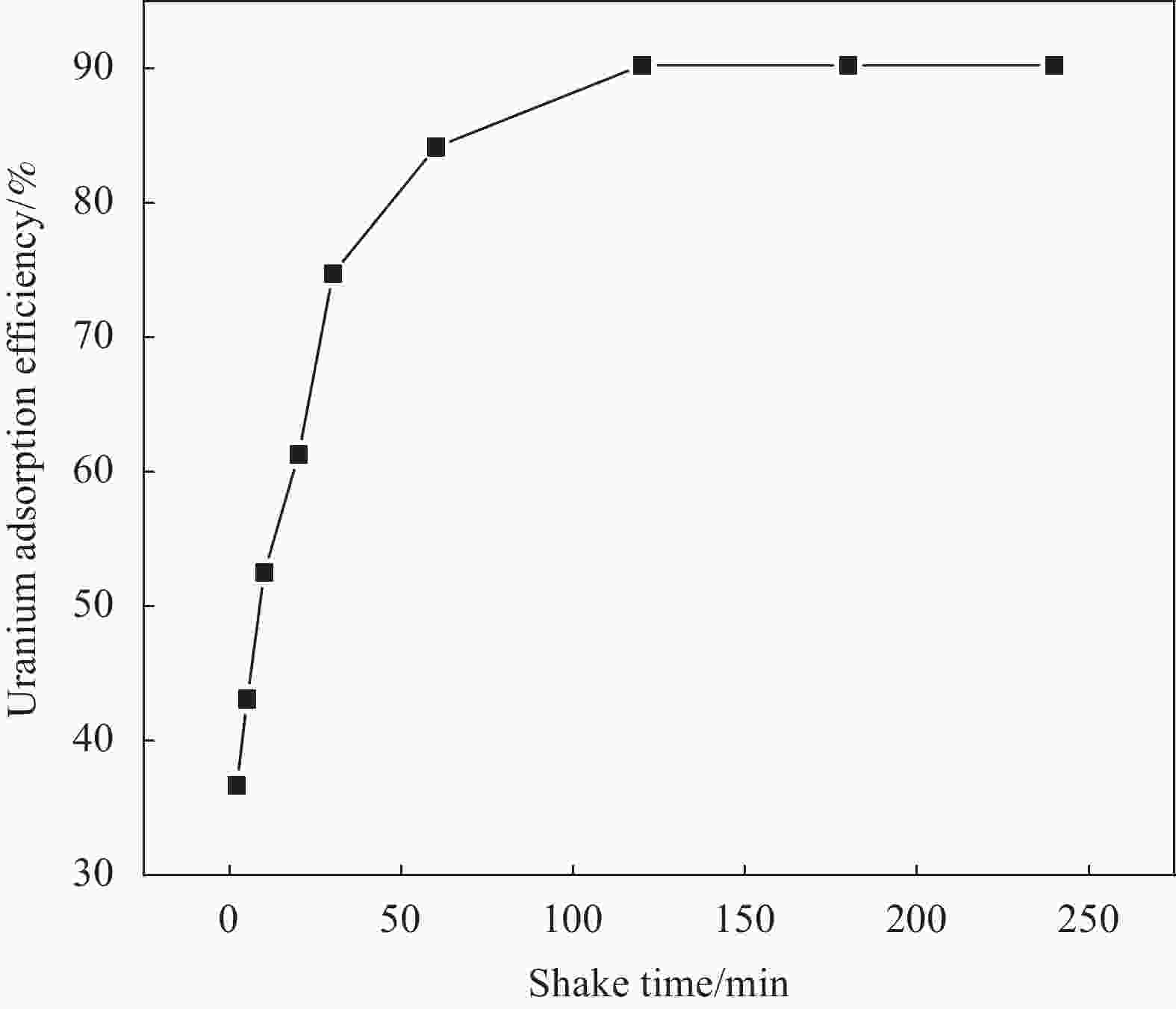
Preparation of UiO-66/chitosan and its adsorption mechanism of U(VI)
-
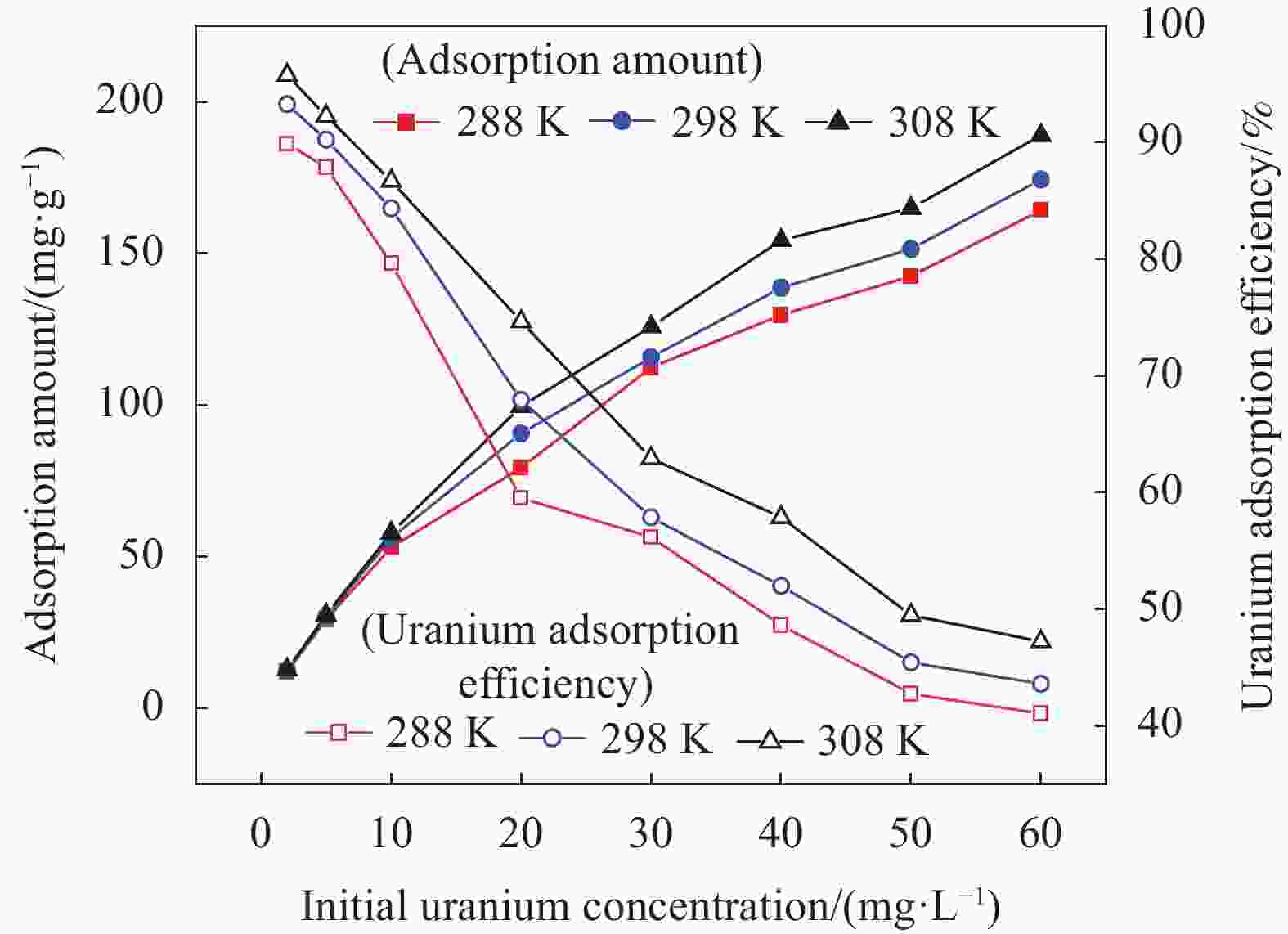
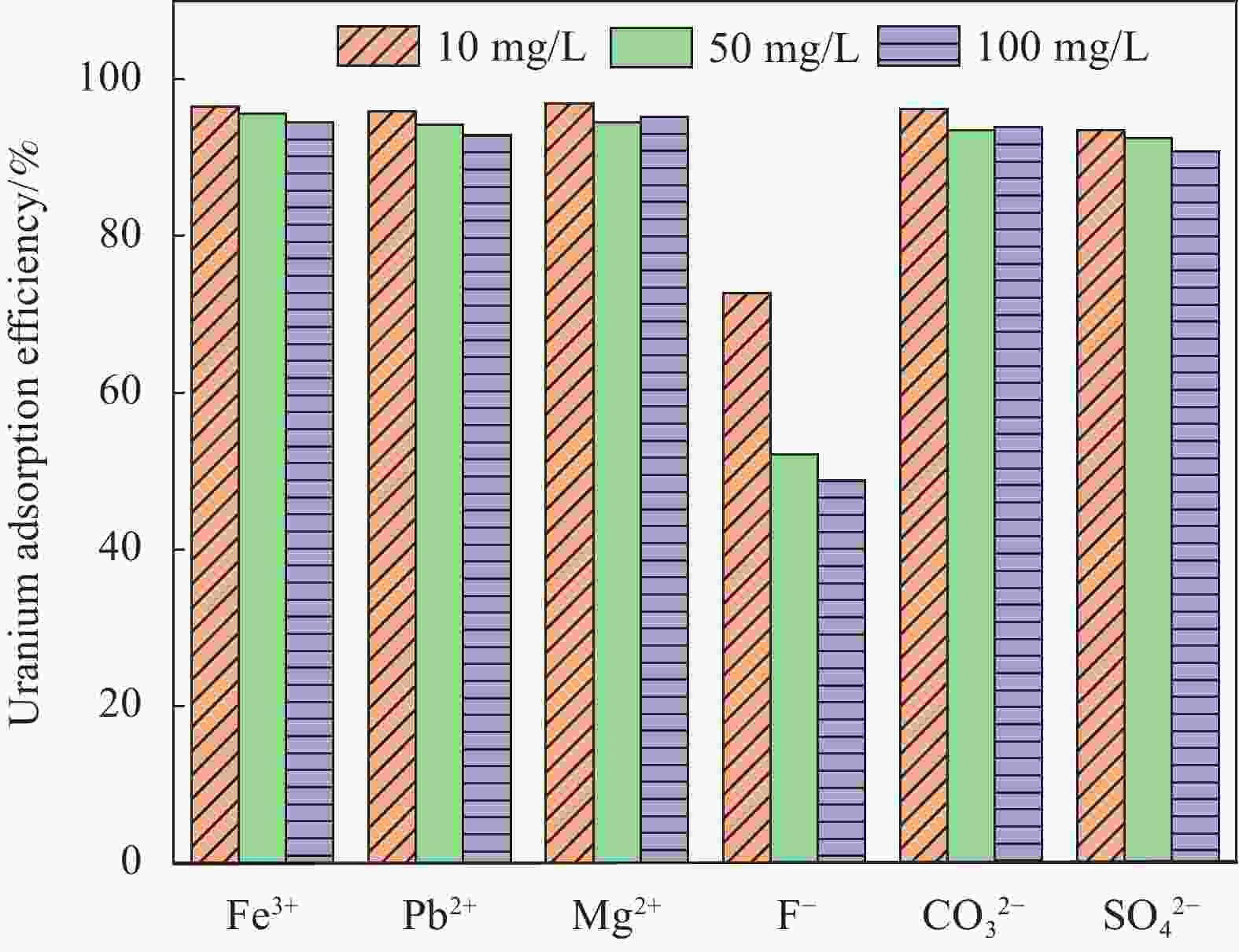
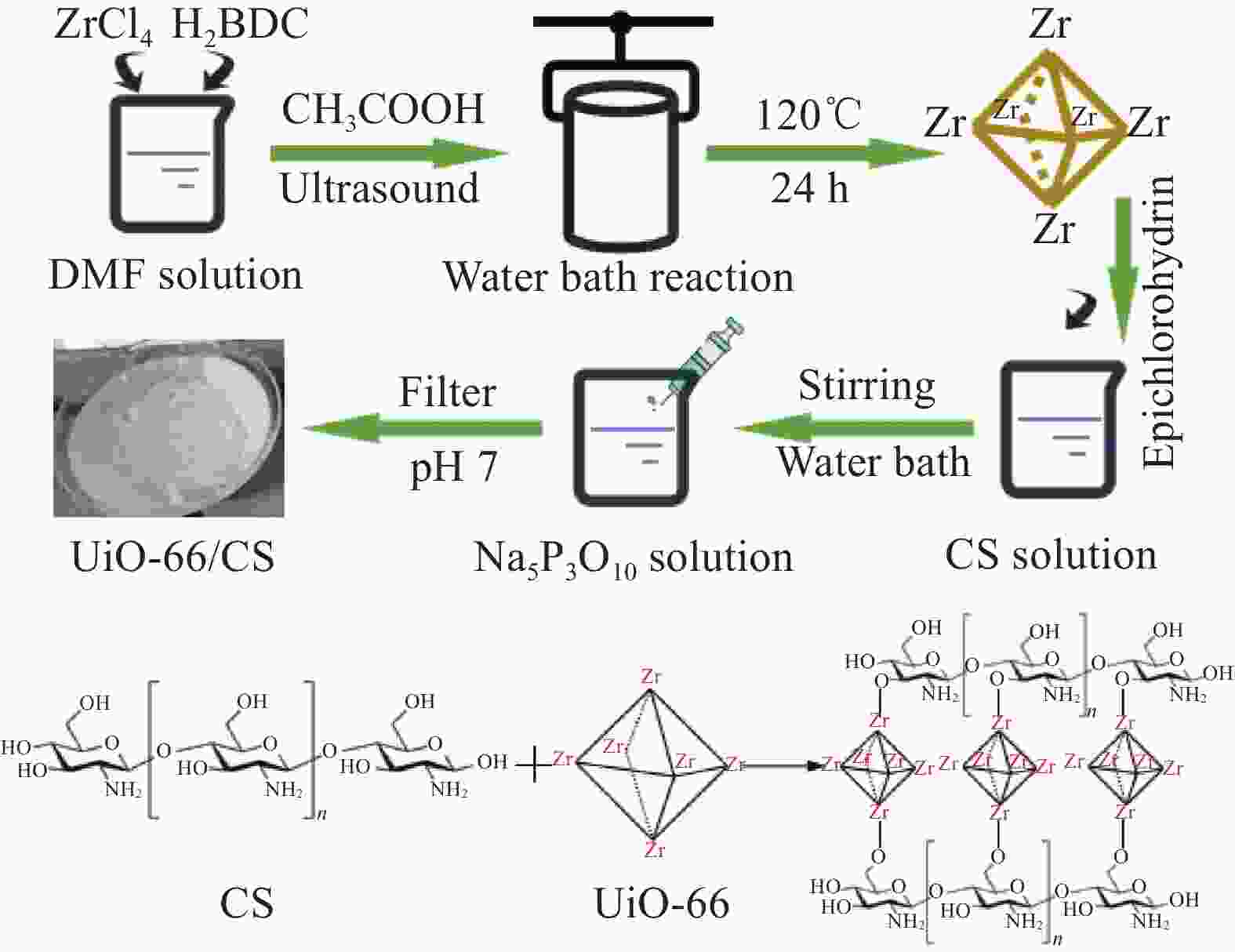
摘要: 铀矿开采和冶炼等工艺产生大量低浓度铀废水,危害着生态环境和人类健康,从含铀废水中去除铀(VI)迫在眉睫。本文以UiO-66、壳聚糖(CS)为原料,采用交联法制备UiO-66/CS新型复合材料,通过静态吸附实验,考察不同pH值、吸附剂投加量、吸附时间及铀初始浓度等外部因素对U(VI)去除率的影响。通过SEM、FTIR、XPS等对UiO-66/CS材料进行表征分析,揭示吸附剂去除U(VI)的机制。结果表明:在铀初始浓度为5 mg/L,温度为298 K,pH为5,投加量为0.15 g/L,吸附时间120 min条件下,UiO-66/CS对U(VI)的去除率可达90.24%。吸附过程符合准二级动力学模型和Freundlich等温吸附模型。U(VI)吸附去除机制主要是—NH、—COOH、Zr—O、—OH等官能团与U(VI)发生络合作用。Abstract: Uranium mining and smelting processes produce a large amount of low-concentration uranium wastewater, which endangers the ecological environment and human health. It is urgent to remove uranium(VI) from uranium-containing wastewater. In this work, UiO-66 and chitosan (CS) were used as raw materials to prepare UiO-66/CS new composite materials by cross-linking method. Through static adsorption experiments, different pH values, adsorbent dosage, adsorption time and initial uranium concentration were investigated of the influence of external factors on U(VI) removal rate. The UiO-66/CS material was characterized and analyzed by SEM, FTIR, XPS, etc., revealing the mechanism of adsorbent removal of U(VI). The results show that when the initial uranium concentration is 5 mg/L, the temperature is 298 K, the pH is 5, the dosage is 0.15 g/L, and the adsorption time is 120 min, the removal rate of U(VI) by UiO-66/CS can reach 90.24%. The adsorption process conforms to the quasi-second-order kinetic model and Freundlich isotherm adsorption model. The adsorption and removal mechanism of U(VI) is mainly the complexation of —NH, —COOH, Zr—O,—OH and other functional groups with U(VI).
-
Key words:
- UiO-66 /
- chitosan /
- uranium /
- adsorption /
- mechanism /
- quasi-second-order kinetic
-
表 1 UiO-66/CS吸附U(VI)的动力学方程拟合参数
Table 1. Fitting parameters of the kinetic equation of UiO-66/CS adsorption of U(VI)
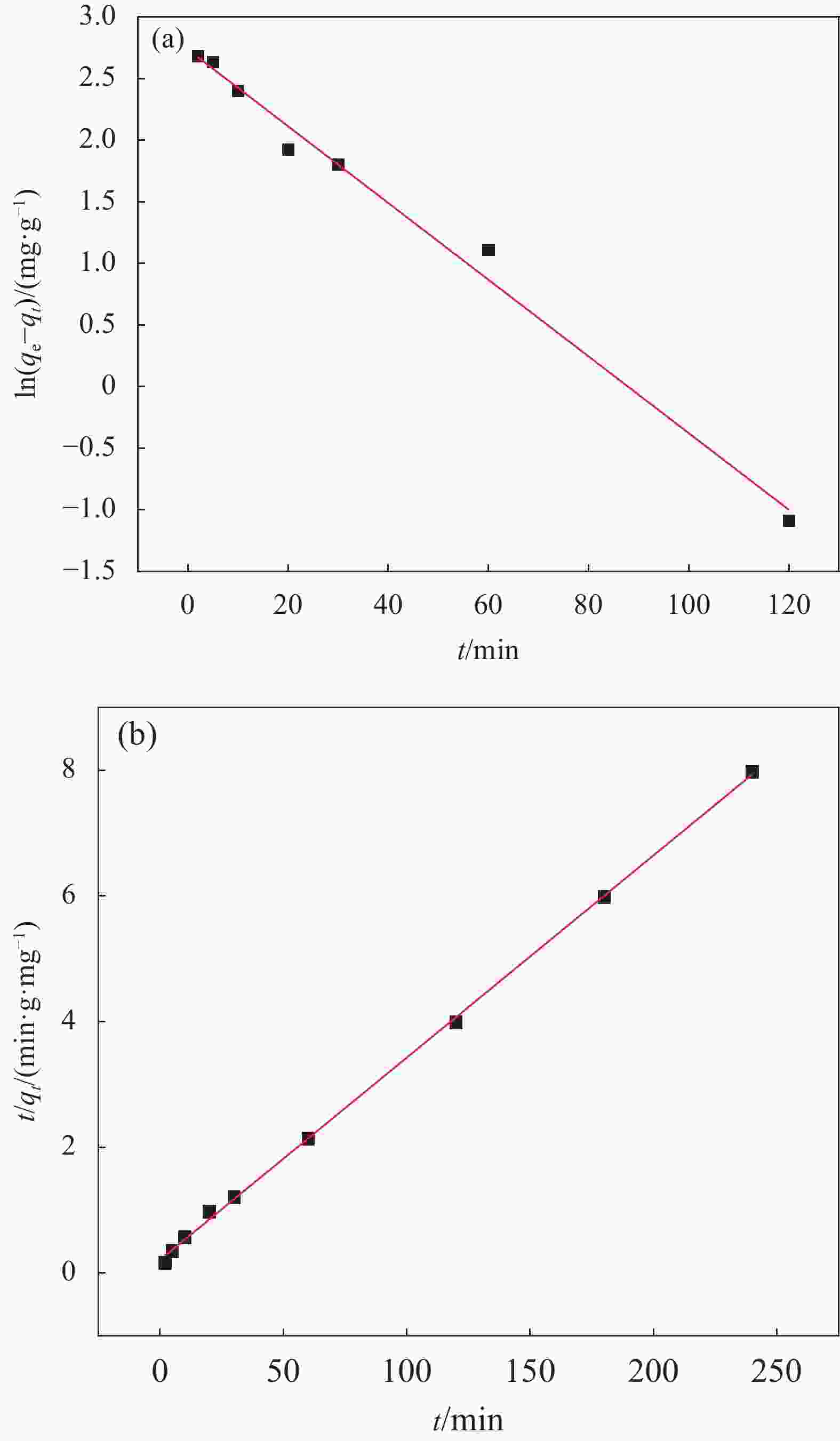
Adsorbent qe,exp/(mg·g−1) Quasi-first-order dynamics model Quasi-second-order dynamics model k1/min−1 qe/(mg·g−1) R2 k2/(g·(mg·min)−1) qe/(mg·g−1) R2 UiO-66/CS 30.078 0.380 18.793 0.986 0.005 31.056 0.999 Notes: qe,exp—Experimental adsorption capacity; qe—Equilibrium adsorption capacity; k1—Quasi-first-order adsorption rate constant; k2—Quasi-second-order adsorption rate constant; R2—Linear correlation coefficient. 表 2 UiO-66/CS吸附U(VI)的动力学方程拟合参数
Table 2. Fitting parameters of the kinetic equation of UiO-66/CS adsorption of U(VI)
Temperature/K Langmuir Freundlich qmax/(mg·g−1) b R2 KF n R2 288 173.310 0.190 0.956 31.592 2.122 0.976 298 179.211 0.246 0.967 37.424 2.228 0.979 308 193.798 0.299 0.963 44.022 2.275 0.987 Notes: qmax—Maximum adsorption capacity; b—Adsorption equilibrium constant of the Langmuir model; KF—Adsorption equilibrium constant of Freundlich model; 1/n—Empirical parameter related to the adsorption strength. -
[1] NING S Y, WANG X P, LIU R Q, et al. Evaluation of Me2-CA-BTP/SiO2-P adsorbent for the separation of minor actinides from simulated HLLW[J]. Journal of Radioanalytical and Nuclear Chemistry,2015,303(3):2011-2017. doi: 10.1007/s10967-014-3651-7 [2] LU W, TANG S Y, LI L, et al. Adsorption and recovery of amidoxime modified nano-Fe3O4-aspergillus niger for U(VI) from low concentration uranium solution[J]. Nanoscience and Nanotechnology Letters,2019,11(3):337-345. doi: 10.1166/nnl.2019.2896 [3] PANAGIOTOU N, LISATAOU I, POURNARA A, et al. Water-stable 2D Zr MOFs with exceptional UO22+ sorption capability[J]. Journal of Materials Chemistry A,2020,8(4):1849-1857. doi: 10.1039/C9TA10701K [4] MA F Q, DONG B R, GUI Y Y, et al. Adsorption of low-concentration uranyl ion by amidoxime polyacrylonitrile fibers[J]. Industrial & Engineering Chemistry Research,2018,57(51):17384-17393. doi: 10.1021/acs.iecr.8b03509 [5] World Health Organization. Guidelines for drinking-water quality[M]. Geneva: World Health Organization, 2004. [6] 王建龙, 刘海洋. 放射性废水的膜处理技术研究进展[J]. 环境科学学报, 2013, 33(10):2639-2656. doi: 10.13671/j.hjkxxb.2013.10.010WANG Jianlong, LIU Haiyang. Research progress on membrane treatment technology of radioactive waste water[J]. Journal of Environmental Science,2013,33(10):2639-2656(in Chinese). doi: 10.13671/j.hjkxxb.2013.10.010 [7] NORO S I, KITAGAWA S, AKUTAGAWA T, et al. Coordination polymers constructed from transition metal ions and organic N-containing heterocyclic ligands: Crystal structures and microporous properties[J]. Progress in Polymer Science,2009,34(3):240-279. doi: 10.1016/j.progpolymsci.2008.09.002 [8] LIU J M, YIN X H, LIU T. Amidoxime-functionalized metal-organic frameworks UiO-66 for U(VI) adsorption from aqueous solution[J]. Journal of the Taiwan Institute of Chemical Engineers,2019,95:416-423. doi: 10.1016/j.jtice.2018.08.012 [9] WU Z L, WANG C H, ZHAO B, et al. A semi-conductive copper-organic framework with two types of photocatalytic activity[J]. Angewandte Chemie International Edition,2016,55(16):4938-4942. doi: 10.1002/anie.201508325 [10] HU Z C, DEIBERT B J, LI J. Luminescent metal-organic frameworks for chemical sensing and explosive detection[J]. Chemical Society Reviews,2014,43(16):5815-5840. doi: 10.1039/C4CS00010B [11] LAZARO I A, WELLS C J R, FORGAN R S. Multivariate modulation of the Zr MOF UiO-66 for defect-controlled combination anticancer drug delivery[J]. Angewandte Chemie International Edition,2020,59(13):5211-5217. doi: 10.1002/anie.201915848 [12] ZHAO B, YUAN L Y, WANG Y, et al. Carboxylated UiO-66 tailored for U(VI) and Eu(III) trapping: From batch adsorption to dynamic column separation[J]. ACS Applied Materials & Interfaces,2021,13(14):16300-16308. doi: 10.1021/acsami.1c00364 [13] QIU J H, FENG Y, ZHANG X F, et al. Acid-promoted synthesis of UiO-66 for highly selective adsorption of anionic dyes: Adsorption performance and mechanisms[J]. Jour-nal of Colloid and Interface Science,2017,499:151-158. doi: 10.1016/j.jcis.2017.03.101 [14] KANDIAH M, NILSEN M H, USSEGLIO S, et al. Synthesis and stability of tagged UiO-66 Zr-MOFs[J]. Chemistry of Materials,2010,22(24):6632-6640. doi: 10.1021/cm102601v [15] NIE J, YI S Z. Neutralization of acid wastewater and magnesium hydroxide slurry from seawater electrolytic pretreatment[J]. Advanced Materials Research,2015,1073:949-954. doi: 10.4028/www.scientific.net/AMR.1073-1076.949 [16] DANDIL S, SAHBAZ D A, ACIKGOZ C. Adsorption of Cu(II) ions onto crosslinked chitosan/waste active sludge char (WASC) beads: Kinetic, equilibrium, and thermodynamic study[J]. International Journal of Biological Macromolecules,2019,136:668-675. doi: 10.1016/j.ijbiomac.2019.06.063 [17] 杨爱丽. 氧化石墨烯-壳聚糖复合吸附剂的制备及其吸附性能[J]. 稀有金属材料与工程, 2018, 47(5):1583-1588.YANG Aili. Preparation and adsorption performance of graphene oxide-chitosan composite adsorbent[J]. Rare Metal Materials and Engineering,2018,47(5):1583-1588(in Chinese). [18] YANG W X, CHENG M J, HAN Y, et al. Heavy metal ions' poisoning behavior-inspired etched UiO-66/CTS aerogel for Pb(II) and Cd(II) removal from aqueous and apple juice[J]. Journal of Hazardous Materials,2021,401:123318. doi: 10.1016/j.jhazmat.2020.123318 [19] JAMSHIDIFARD S, KOUSHKBAGHI S, HOSSEINI S, et al. Incorporation of UiO-66-NH2 MOF into the PAN/chitosan nanofibers for adsorption and membrane filtration of Pb(II), Cd(II) and Cr(VI) ions from aqueous solutions[J]. Journal of Hazardous Materials,2019,368:10-20. doi: 10.1016/j.jhazmat.2019.01.024 [20] ZHU X Y, GU J L, WANG Y, et al. Inherent anchorages in UiO-66 nanoparticles for efficient capture of alendronate and its mediated release[J]. Chemical Communications,2014,50(63):8779-8782. doi: 10.1039/C4CC02570A [21] AKYUZ L, KAYA M, KOC B, et al. Diatomite as a novel composite ingredient for chitosan film with enhanced physicochemical properties[J]. International Journal of Biological Macromolecules,2017,105(Pt 2):1401-1411. doi: 10.1016/j.ijbiomac.2017.08.161 [22] 高翔. 生物炭-壳聚糖复合物的制备及其对低浓度含铀废水的吸附试验研究[D]. 衡阳: 南华大学, 2019.GAO Xiang. Preparation of biochar-chitosan complex and its adsorption experiment on low-concentration uranium-containing wastewater[D]. Hengyang: University of South China, 2019(in Chinese). [23] GUO D X, SONG X M, ZHANG L L, et al. Recovery of uranium(VI) from aqueous solutions by the polyethyleneimine-functionalized reduced graphene oxide/molybdenum disulfide composition aerogels[J]. Journal of the Taiwan Institute of Chemical Engineers,2020,106:198-205. doi: 10.1016/j.jtice.2019.09.029 [24] DUAN S X, XU X T, LIU X, et al. Highly enhanced adsorption performance of U(VI) by non-thermal plasma modified magnetic Fe3O4 nanoparticles[J]. Journal of Colloid and Interface Science,2018,513:92-103. doi: 10.1016/j.jcis.2017.11.008 [25] LI X, LIU Y, ZHANG C L, et al. Porous Fe2O3 microcubes derived from metal organic frameworks for efficient elimination of organic pollutants and heavy metal ions[J]. Chemical Engineering Journal,2018,336:241-252. doi: 10.1016/j.cej.2017.11.188 [26] HAN M N, KONG L J, HU X L, et al. Phase migration and transformation of uranium in mineralized immobilization by wasted bio-hydroxyapatite[J]. Journal of Cleaner Production,2018,197:886-894. doi: 10.1016/j.jclepro.2018.06.253 [27] 俞坤, 刘金香, 谢水波, 等. 聚吡咯/石墨相氮化碳复合材料吸附铀(VI)的性能与机制[J]. 材料导报, 2020, 34(23):23020-23026. doi: 10.11896/cldb.19110056YU Kun, LIU Jinxiang, XIE Shuibo, et al. Adsorption performance and mechanism of polypyrrole/graphite carbon nitride composite material for uranium(VI)[J]. Materials Review,2020,34(23):23020-23026(in Chinese). doi: 10.11896/cldb.19110056 [28] YANG P P, LIU Q, LIU J Y, et al. Interfacial growth of a metal-organic framework (UiO-66) on functionalized graphene oxide (GO) as a suitable seawater adsorbent for extraction of uranium(VI)[J]. Journal of Materials Che-mistry A,2017,5(34):17933-17942. doi: 10.1039/C6TA10022H [29] PLAZINSKI W, RUDZINSKI W, PLAZINSKA A. Theoretical models of sorption kinetics including a surface reaction mechanism: A review[J]. Advances in Colloid and Interface Science,2009,152(1-2):2-13. doi: 10.1016/j.cis.2009.07.009 [30] 杨金辉, 胡世琴, 杨斌, 等. 氨化烟末生物碳吸附剂的制备及对Cr(VI)的吸附行为[J]. 复合材料学报, 2022, 39(1):222-231. doi: 10.13801/j.cnki.fhclxb.20210320.001YANG Jinhui, HU Shiqin, YANG Bin, et al. Preparation of bio-carbon adsorbent for ammoniated tobacco dust and its adsorption behavior for Cr(VI)[J]. Acta Materiae Compositae Sinica,2022,39(1):222-231(in Chinese). doi: 10.13801/j.cnki.fhclxb.20210320.001 [31] IRVING L. The adsorption of gases on plane surfaces of glass, mica and platinum[J]. Journal of the American Chemical Society,1918,40(9):1361-1403. doi: 10.10.1021/ja02242a004 [32] HERBERT F. Uber die adsorption in lösungen[J]. Zeitschrift für Physikalische Chemie,1907,57U(1):385-470. doi: 10.1515/zpch-1907-5723 [33] LIU W J, ZHANG L Y, CHEN F M, et al. Efficiency and mechanism of adsorption of low-concentration uranium from water by a new chitosan/aluminum sludge compo-site aerogel[J]. Dalton Transactions,2020,49(10):3209-3221. doi: 10.1039/C9DT04670D -





 下载:
下载: