Designed growth of hollow WO3/PEDOT bilayer hybrid nanosphere arrays film with superior electrochromic and capacitive performance
-
摘要: 本文采用模板辅助磁控溅射和电化学沉积技术,构建了三氧化钨/聚噻吩双层复合空心纳米球阵列。独特的空心纳米阵列结构可以提供与电解液较大的接触面积,有利于电极与电解液之间的离子传输,所制备的三氧化钨层属于对小离子具有大容量的非晶相,提供了大量的离子结合位点,聚噻吩层则形成了一个独特的导电网络,可以有效地促进电子传递并连接原本分离的色心。所制备的空心纳米球复合薄膜在可见光波段具有较高光学调制能力(633 nm 处77.4%),较快的响应速度(着色3.2 s,褪色4.2 s),良好的循环稳定性(2000次循环后失去14.5%的光调制幅度),在低电位下−1.0/1.0 V下具有良好的着色效率(116.2 cm2·C−1),所获得的有机无机复合材料还具有较高的面电容(54.6 mF/cm2)、出色的倍率性能和循环稳定性(经历2000次循环后的面积电容仍保持为原来的79.6%)。Abstract: In this work, hollow tungsten trioxide (WO3)/poly (3,4-ethylenedioxythiophene)(PEDOT)bilayer hybrid nanosphere arrays film is constructed by template-assisted magnetron sputtering combined with electrochemical deposition. The hollow nanosphere arrays can provide large contact area with electrolyte to benefit ion exchange. The prepared WO3 layer is responsible for offering a lot of ions binding sites due to the large capacity of amorphous WO3 for small ions, and the PEDOT layer is prepared to construct a unique conductive network which can effectively facilitate electron transportation and connect separated color centers. The obtained hollow hybrid nanosphere film exhibits outstanding electrochromic performance with high contrast (77.4% at 633 nm), fast response speed (3.2 s for coloring and 4.2 s for bleaching), good cycling stability (lose 14.5% optical modulation after 2000 cycles) and decent coloring efficiency (116.2 cm2·C−1) at low colored/bleached potentials (−1.0/1.0 V). The hollow hybrid nanospheres also display high areal capacitance (54.6 mF/cm2), superior rate capability and cyclic stability (areal capacitance remains 79.6% after 2000 cycles).
-
Key words:
- WO3 /
- PEDOT /
- nanostructure /
- magnetron sputtering /
- electrodeposition /
- electrochromic /
- energy storage
-
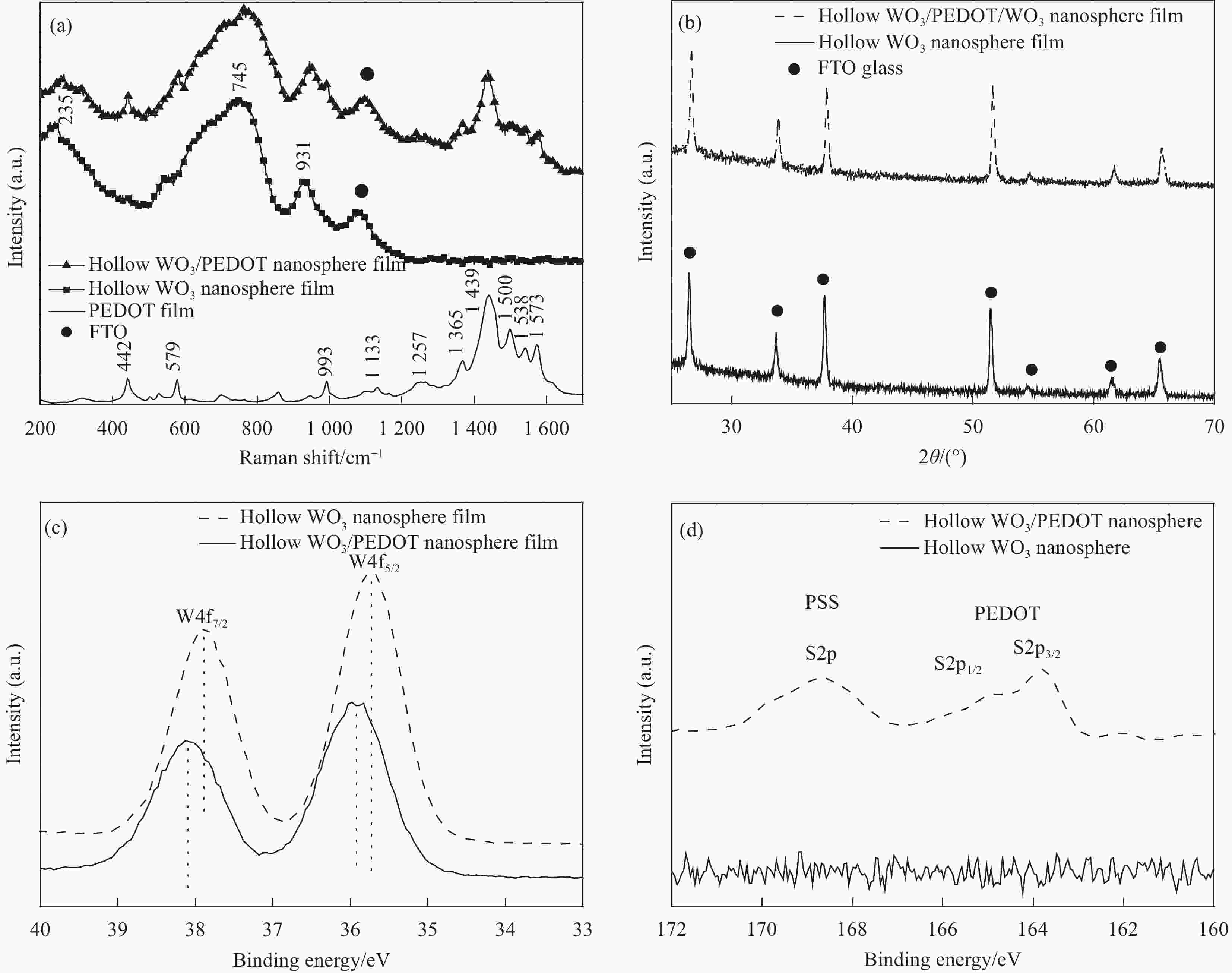
图 3 (a)单纯PEDOT薄膜、WO3空心纳米球阵列以及WO3/PEDOT双层复合空心纳米球阵列拉曼光谱;(b)WO3空心纳米球阵列以及WO3/PEDOT双层复合空心纳米球阵列XRD图谱;(c)WO3空心纳米球阵列以及WO3/PEDOT双层复合空心纳米球阵列 XPS的W元素核心光谱和(d)S元素核心光谱
Figure 3. The Raman spectra(a)of pristine PEDOT film, hollow WO3 nanosphere film and hollow WO3/PEDOT nanosphere film. (b)XRD patterns of hollow WO3 nanosphere film and hollow WO3/PEDOT nanosphere film. XPS spectra in(c)W 4 f region and(d)S 2 p region for hollow WO3 nanoarrays and hollow WO3/PEDOT nanoarrays
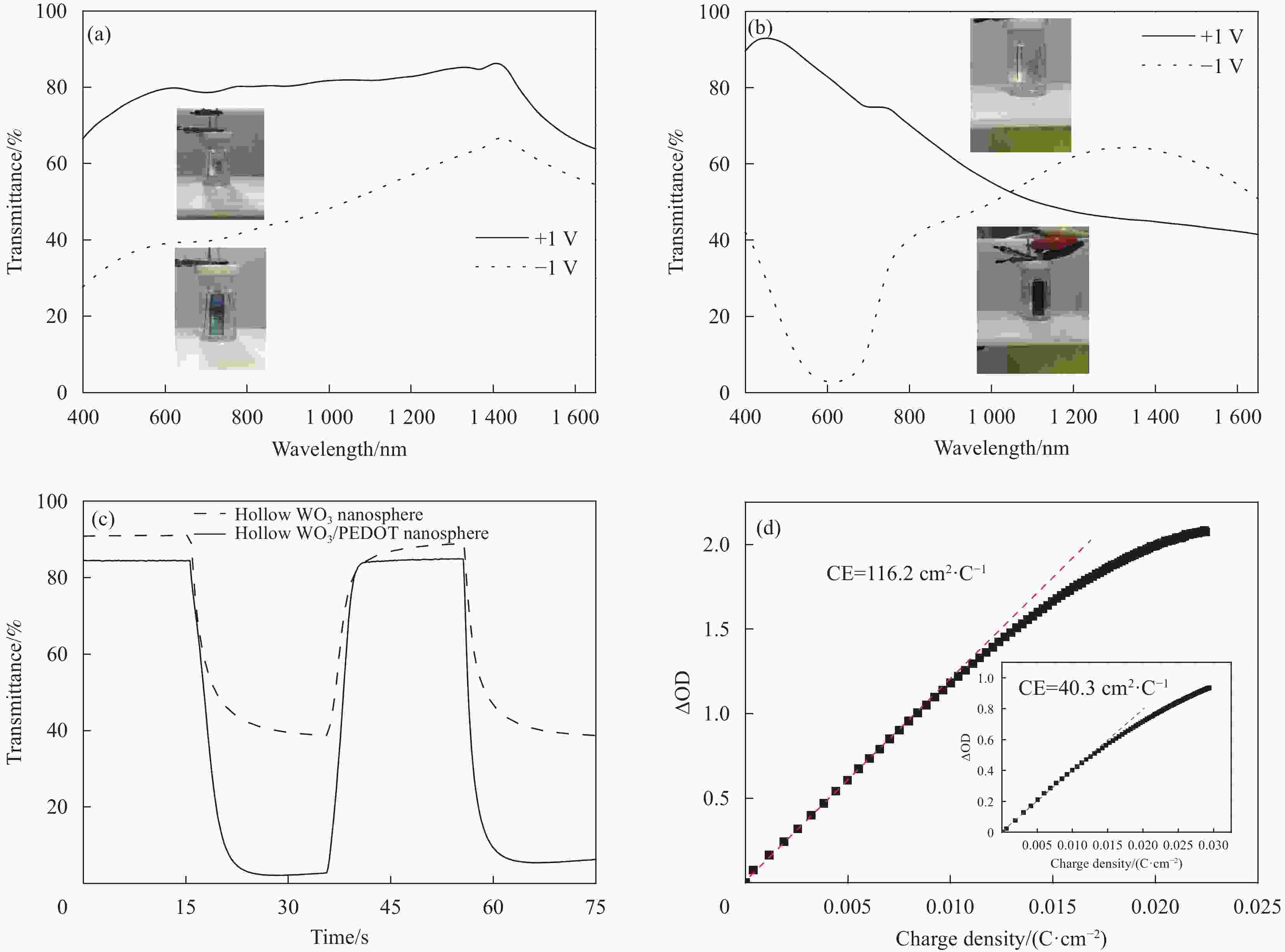
图 4 (a) WO3空心纳米球阵列、(b) WO3/PEDOT双层复合空心纳米球阵列在不同电位下的透射光谱以及相应的实物照片;(c) WO3空心纳米球阵列(虚线)和WO3/PEDOT双层复合空心纳米球阵列(实线)在+1.0到−1.0 V时间间隔为20 s的方波电位作用下的633 nm波长处动力学光谱;(d) WO3空心纳米球阵列(插图)以及WO3/PEDOT双层复合空心纳米球阵列原位光学密度变化相对于电荷密度变化曲线
Figure 4. Transmittance spectra under different applied potentials of (a) hollow WO3 nanoarrays and (b) hollow WO3/PEDOT nanoarrays with the inset showing the corresponding digital photographs; (c) dynamic optical transmittance spectra at 633 nm of the (Ⅰ) hollow WO3 nanoarrays (dashed line) and (Ⅱ) hollow WO3/PEDOT nanoarrays (solid line) under a square-wave potential oscillating between +1.0 and −1.0 V and a time step of 20 s; (d) variation of the in situ optical density (OD) vs. charge density for hollow WO3 nanospheres (in the insert) and hollow WO3/PEDOT nanosphere
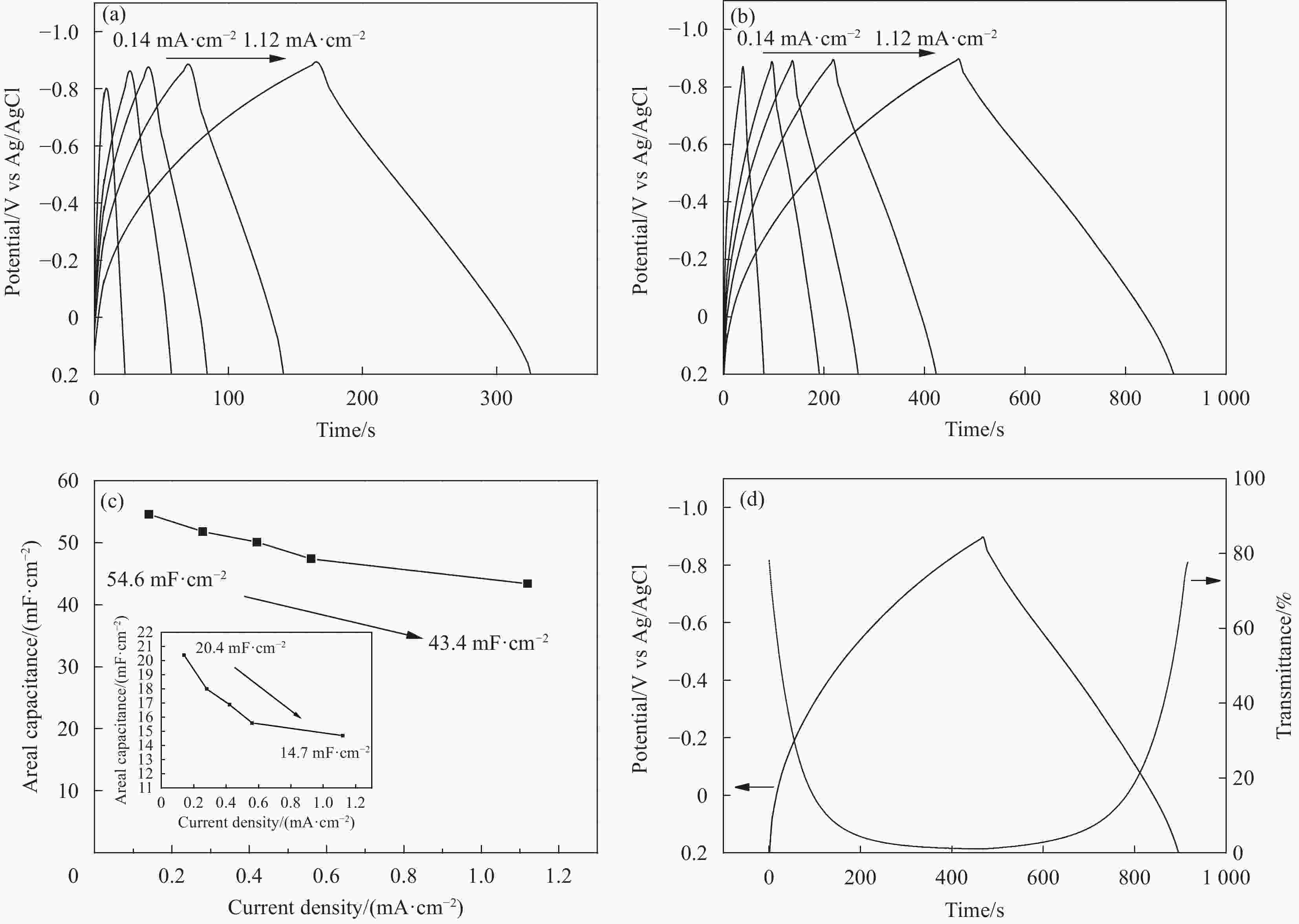
图 5 (a) WO3空心纳米球阵列和(b) WO3/PEDOT双层复合空心纳米球阵列在不同电流密度下的恒电流充放电曲线;(c) WO3空心纳米球阵列(插图)和WO3/PEDOT双层复合空心纳米球阵列面电容对应电流密度点线图; (d) WO3/PEDOT双层复合空心纳米球阵列在电压范围0.2到−0.9 V电流密度0.14 mA cm−2条件下恒流充放电曲线及相对应的633 nm波长下原位透过率曲线
Figure 5. Galvanostatic charge/discharge curves at different current densities of (a) hollow WO3 nanoarrays and (b) hollow WO3/PEDOT nanoarrays; (c) Areal capacitance as a function of the current density of hollow WO3/PEDOT nanoarrays; (d) Galvanostatic charge/discharge curves at 0.14 mA cm−2 in the voltage range of 0.2 to −0.9 V and the corresponding in situ optical response at 633 nm
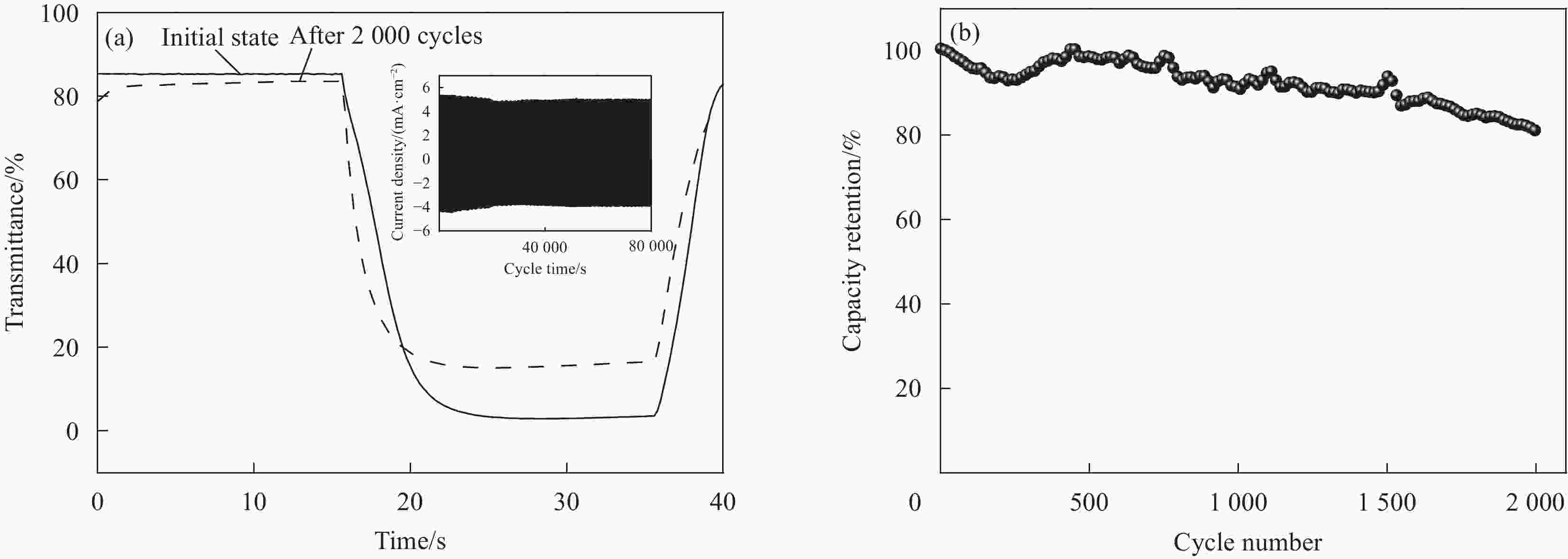
图 6 (a) WO3/PEDOT双层复合空心纳米球阵列的动力学透过率测试图谱(方波电位+1.0 和 −1.0 V之间转换,单步时间为20 s,固定波长为633 nm,电解液为1 M 高氯酸锂/碳酸丙烯酯,分别为初始态和2000圈循环后),插图为动力学相对应的电流数据;(b)在0.42 mA·cm−2电流密度测试条件下的WO3/PEDOT双层复合空心纳米球阵列电容循环性能
Figure 6. Dynamic transmittance data of (a) hollow WO3/PEDOT nanoarrays under a square-wave potential oscillating between +1.0 and −1.0 V at a time interval of 20 s at 633 nm tests in 1 M LiClO4/PC solution, before and after 2000 cycles. Inset: corre sponding current data of hollow WO3/PEDOT nanoarrays; (b) capacitive cycle performance of hollow WO3/PEDOT nanoarrays measured under a current density of 0.42 mA·cm−2
表 1 WO3空心纳米球阵列和WO3/PEDOT双层复合空心纳米球阵列电致变色电容性能汇总
Table 1. Electrochromic and capacitive performance data for hollow WO3 nanoarrays and hollow WO3/PEDOT nanoarrays
Optical modulation range/%633 nm Coloring time/s Bleaching time/s Coloration efficiency/(cm2·C−1) Areal capacitance/
(mF· cm−2)Rate capability/% Hollow WO3 nanoarrays 40.2 8.0 7.1 40.3 20.4 79.5 Hollow WO3/PEDOT nanoarrays 77.4 3.2 4.2 116.2 54.6 72.4 表 2 本工作中所制备的WO3/PEDOT双层复合空心纳米球阵列电致变色电容性能与其它报道过的工作对比表
Table 2. The summarized comparison of electrochromic and capacitive properties of our hollow WO3/PEDOT nanoarrays film with the previously reported works.
Film material Optical modulation
range/%Coloring time/s /
Bleaching time/sColoration
efficiency/(cm2·C−1)Areal capacitance/
( mF·cm−2)Ref. m-WO3-x nanowires 93.2 16/13 121 — [27] hexagonal/amorphousWO3
nanorod arrays67.7 15/21 101 108 [28] MoO3-W0.71Mo0.29O3 50 >3 20.8 — [29] Polyaniline (PANI)//WO3 ~75 9.9/13.6 98.4 25 [30] WO3/ZnO nanocomposites 68.2 6.2/2.8 80.6 15.24 [31] Mo-doped WO3 nanowire ~56 3.2/2.6 123.5 ~66.5 [32] PEDOT–Au@WO3 ~71 ~7.4/1.7 ~707 42 [33] hollow WO3/PEDOT nanoarrays 77.4 3.2/4.2 116.2 54.6 This work -
[1] 贾岩, 刘东青, 程海峰, 等. 基于金属氧化物纳米晶的电致变色材料研究进展[J]. 复合材料学报, 2023, 40(9): 4863-4879.JIA Yan, LIU Dongqing, CHENG Haifeng, et al. A review of electrochromic materials based on metal oxide nanocrystals[J]. Acta Materiae Compositae Sinica, 2023, 40(9): 4863-4879(in Chinese). [2] CAI G F, DARMAWAN P, CUI M Q, Et al. Inkjet-printed all solid-state electrochromic devices based on NiO/WO3 nanoparticle complementary electrodes[J]. Nanoscale, 2016, 8(1): 348-357. doi: 10.1039/C5NR06995E [3] 汤凯, 管康威, 刘淑婧, 等. Ti掺杂核壳结构晶态@非晶WO3纳米线复合薄膜的制备与电致变色性能[J]. 复合材料学报, 2023, 40(6): 3539-3552.TANG Kai, GUAN Kangwei, LIU Shujing, et al. Synthesis and electrochromic properties of Ti-doped core-shell crystalline@amorphous WO3 nanowire composite films[J]. Acta Materiae Compositae Sinica, 2023, 40(6): 3539-3552(in Chinese). [4] 成明, 杨继凯, 杨馥瑜, 等. WO3/TiO2复合薄膜的制备及其电致变色性能[J]. 复合材料学报, 2019, 36(4): 914-920.CHEN Ming, YANG Jikai, YANG Fuyu, et al. Preparation and electrochromic properties of WO3/TiO2 composite films[J]. Acta Materiae Compositae Sinica, 2019, 36(4): 914-920 (in Chinese). [5] KUMAR K S, CHOUDHARY N, JUNG Y, et al. Recent advances in two-dimensional nanomaterials for supercapacitor electrode applications[J]. ACS Energy Letters, 2018, 3(2): 482-495. doi: 10.1021/acsenergylett.7b01169 [6] SANKARAN B, REYNOLDS J R. High-Contrast Electrochromic Polymers from Alkyl-Derivatized Poly(3, 4-ethylenedioxythiophenes)[J]. Macromolecules, 1997, 30(9): 2582-2588. doi: 10.1021/ma961607w [7] WANG G P, ZHANG L, ZHANG J J. A review of electrode materials for electrochemical supercapacitors[J]. Chemical Society Reviews, 2012, 41(2): 797-828. doi: 10.1039/C1CS15060J [8] TANG K, ZHANG Y, SHI Y D, et al. Preparation of V2O5 dot-decorated WO3 nanorod arrays for high performance multi-color electrochromic devices[J]. Journal of Materials Chemistry C, 2018, 6(45): 12206-12216. doi: 10.1039/C8TC04247K [9] MA D Y, SHI G Y, WANG H Z, et al. Morphology-tailored synthesis of vertically aligned 1D WO3 nano-structure films for highly enhanced electrochromic performance[J]. Journal of Materials Chemistry A, 2013, 1(3): 684-691. doi: 10.1039/C2TA00090C [10] LEE S H, DESHPANDE R, PARILLA P A, et al. Crystalline WO3 nanoparticles for highly improved electrochromic applications[J]. Advanced Materials, 2006, 18(6): 763-766. doi: 10.1002/adma.200501953 [11] SONG Y Y, GAO Z D, WANG J H, et al. Multistage coloring electrochromic device based on TiO2 nanotube arrays modified with WO3 nanoparticles[J]. Advanced Functional Materials, 2011, 21(10): 1941-1946. doi: 10.1002/adfm.201002258 [12] GUNBAS G, TOPPARE L. Electrochromic conjugated polyheterocycles and derivatives--highlights from the last decade towards realization of long lived aspirations[J]. Chemical Communication, 2012, 48(8): 1083-1101. [13] LIU S P, XU L, LI F Y, et al. Enhanced electrochromic performance of composite films by combination of polyoxometalate with poly(3, 4-ethylenedioxythiophene)[J]. Journal of Materials Chemistry, 2011, 21(6): 1946-1952. doi: 10.1039/C0JM02412K [14] TAO Y J, ZHANG K, ZHANG Z Y, et al. Synthesis, characterizations and electrochromic properties of polymers based on functionalized anthracene[J]. Chemical Engineering Journal, 2016, 293(1): 34-43. [15] LING H, LU J L, PHUA S, et al. One-pot sequential electrochemical deposition of multilayer poly(3, 4-ethylenedioxythiophene): poly(4-styrenesulfonic acid)/tungsten trioxide hybrid films and their enhanced electrochromic properties[J]. Journal of Materials Chemistry A, 2014, 2(8): 2708-2717. doi: 10.1039/C3TA14781A [16] SHI Y D, ZHANG Y, TANG K, et al. Designed growth of WO3 /PEDOT core/shell hybrid nanorod arrays with modulated electrochromic properties[J]. Chemical Engineering Journal, 2019, 355: 942-951. doi: 10.1016/j.cej.2018.08.163 [17] TAMBURRI E, ORLANDUCCI S, TOSCHI F, et al. Growth mechanisms, morphology, and electroactivity of PEDOT layers produced by electrochemical routes in aqueous medium[J]. Synthetic Metals, 2009, 159(5): 406-414. [18] ROUGIER A, PORTEMER F, QUÉDÉ A, et al. Characterization of pulsed laser deposited WO3 thin films for electrochromic devices[J]. Applied Surface Science, 1999, 153(1): 1-9. doi: 10.1016/S0169-4332(99)00335-9 [19] DANIEL M F, DESBAT B, LASSEGUES J C, et al. Infrared and Raman study of WO3 tungsten trioxides and WO3, xH2O tungsten trioxide tydrates[J]. Journal of solid state chemistry, 1987, 67(2): 235-247. doi: 10.1016/0022-4596(87)90359-8 [20] SHPAK A P, KORDUBAN A M, MEDVEDSKIJ M M, et al. XPS studies of active elements surface of gas sensors based on WO3−x nanoparticles[J]. Journal of Electron Spectroscopy and Related Phenomena, 2007, 156-158: 172-175. doi: 10.1016/j.elspec.2006.12.059 [21] PHURUANGRAT A, HAM D J, HONG S J, et al. Synthesis of hexagonal WO3 nanowires by microwave-assisted hydrothermal method and their electrocatalytic activities for hydrogen evolution reaction[J]. Journal of Materials Chemistry, 2010, 20(9): 1683-1690. doi: 10.1039/B918783A [22] DALAVI D S, DEVAN R S, PATIL R A, et al. Efficient electrochromic performance of nanoparticulate WO3 thin films[J]. Journal of Materials Chemistry C, 2013, 1(23): 3722-3728. doi: 10.1039/c3tc30378k [23] WANG G M, LING Y C, WANG H Y, et al. Hydrogen-treated WO3 nanoflakes show enhanced photostability[J]. Energy & Environmental ence, 2012, 5(3): 6180-6187. [24] SPANNINGA S A, MARTIN D C, CHEN Z, X-ray photoelectron spectroscopy study of counterion incorporation in poly(3, 4-ethylenedioxythiophene)[J]. J. Phys. Chem. C, 2009, 113(14): 5585–5592. [25] SHI Y D, ZHANG Y, TANG K, et al. In situ growth of PEDOT/graphene oxide nanostructures with enhanced electrochromic performance[J]. RSC Advances, 2018, 8(25): 13679-13685. doi: 10.1039/C8RA01153B [26] SHI Y D, SUN M J, ZHANG Y, et al. Structure modulated amorphous/crystalline WO3 nanoporous arrays with superior electrochromic energy storage performance, Solar Energy Materials and Solar Cells, 212(2020): 110579-110588. [27] ZHANG S L, CAO S, ZHANG T R, et al. Al3+ intercalation/de-intercalation-enabled dual-band electrochromic smart windows with a high optical modulation, quick response and long cycle life[J]. Energy & Environmental Science, 2018, 11(10): 2884-2892. [28] HUO X T, ZHANG H Y, SHEN W G, et al. Bifunctional aligned hexagonal/amorphous tungsten oxide core/shell nanorod arrays with enhanced electrochromic and pseudocapacitive performance[J]. Journal of Materials Chemistry A, 2019, 7(28): 16867-16875. doi: 10.1039/C9TA03725J [29] LI H Z, MCRAE L, FIRBY C J, et al. Nanohybridization of molybdenum oxide with tungsten molybdenum oxide nanowires for solution-processed fully reversible switching of energy storing smart windows[J]. Nano Energy, 2018, 47: 130-139. doi: 10.1016/j.nanoen.2018.02.043 [30] WEI H G, Yan X R, Wu S J, LUO Z P, et al. Electropolymerized polyaniline stabilized tungsten oxide nanocomposite films: electrochromic behavior and electrochemical energy storage[J]. The Journal of Physical Chemistry C, 2012, 116(47): 25052-25064. doi: 10.1021/jp3090777 [31] BI Z J, LI X M, CHEN Y B, Et al. Bi-functional flexible electrodes based on tungsten trioxide/zinc oxide nanocomposites for electrochromic and energy storage applications[J]. Electrochimica Acta, 2017, 227: 61-68. doi: 10.1016/j.electacta.2017.01.003 [32] ZHOU D, SHI F, XIE D, et al. Bi-functional Mo-doped WO3 nanowire array electrochromism-plus electrochemical energy storage[J]. Journal of colloid and interface science, 2016, 465: 112-120. doi: 10.1016/j.jcis.2015.11.068 [33] REDDY B N, KUMAR P N, DEEPA M. A Poly (3, 4-ethylenedioxypyrrole)-Au@ WO3-Based Electrochromic Pseudocapacitor[J]. ChemPhysChem, 2015, 16(2): 377-389. doi: 10.1002/cphc.201402625 -





 下载:
下载: