Glucose Sensor Based On Co-MOFs-modified Commercial Strip Electrodes
-
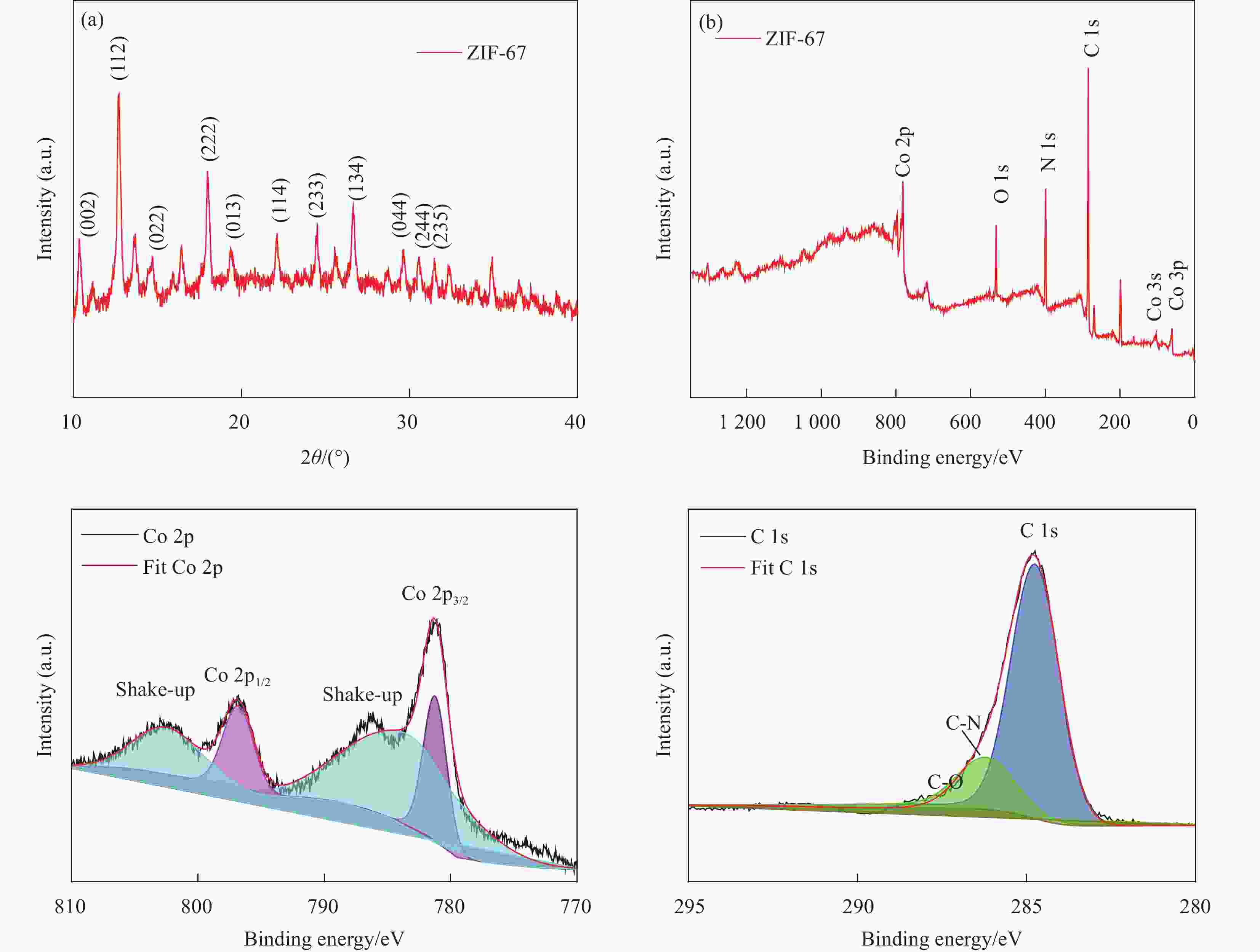
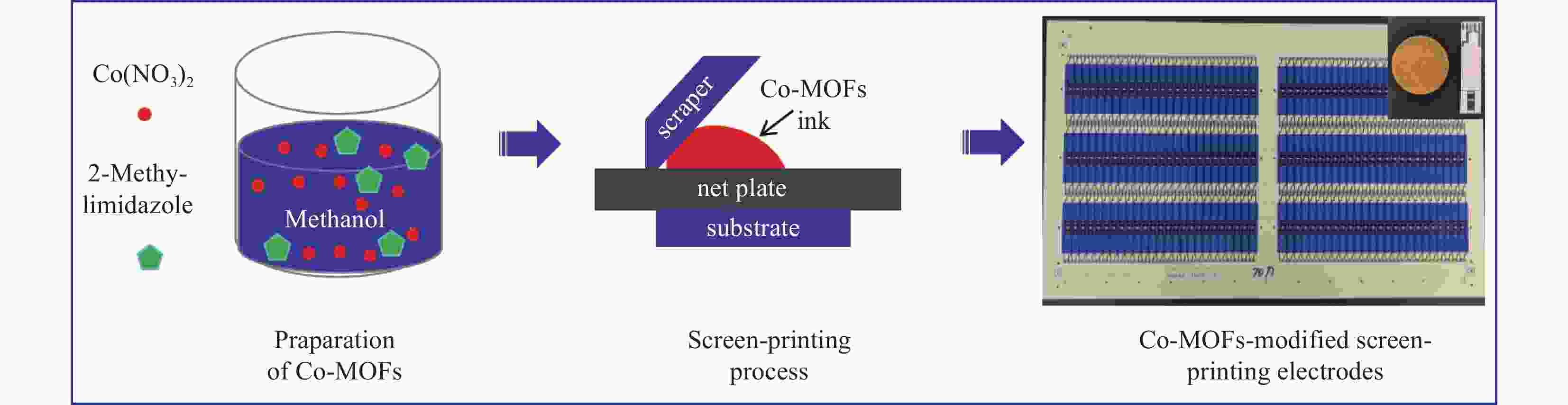
摘要: 第三代葡萄糖传感器中氧化酶存在容易受温度、湿度、酸碱度等影响的缺点。因此,开发低成本、高灵敏度的葡萄糖催化剂具有广阔的应用前景。金属-有机框架(Metal-Origanic Frameworks, MOFs)因具有传质速度快、孔隙率可调和电子转移能力强等优点而受到广泛的关注。丝网印刷技术是一种低成本、批量化制备商业电极工艺。本文采用一种简单、经济的方法室温下成功地合成了Co-MOFs;采用丝网印刷技术将Co-MOFs涂敷在商业化条形银-碳电极上。作为生物传感器的催化剂,Co-MOFs纳米材料对葡萄糖表现出较高的电催化活性。测试结果表明,Co-MOFs基条形电极对葡萄糖检测的灵敏度为1393 nA·L/(mmol·cm2),检测极限为0.58 μmol/L(S/N = 3),线性范围为0.1-0.5 mmol/L。该工作对葡萄糖传感器中批量构筑多功能电极的设计具有一定的指导意义。Abstract: The oxidase catalyst in glucose sensors can be easily affected by temperature, humidity, PH. As a result, developing some catalysts with low cost and high sensitivity has a broad application prospect. Metal-organic Frameworks (MOFs) have attracted much attention because of their high mass transfer rate, adjustable porosity and strong electron transfer ability. The screen-printing technique has been used to fabricate commercial electrodes due to its low cost and batch preparation. Here, Co-MOFs were successfully synthesized by a simple and economical method at room temperature and coated on commercial strip silver-carbon electrode by screen-printing technique. As a biosensor catalyst, Co-MOFs nanomaterials exhibit high electrocatalytic activity for glucose. The results showed that the sensitivity of Co-MOFs-based strip electrode for glucose detection was 1393 nA·L/(mmol·cm2). The detection limit was 0.58 μmol/L (S/N = 3) and the linear range was 0.1-0.5 mmol/L. This work has certain guiding significance for the design of multi-function electrode in glucose sensor.
-
Key words:
- Electrochemical sensor /
- Glucose Detection /
- Co-MOFs /
- Screen printing /
- Metal-organic framework
-
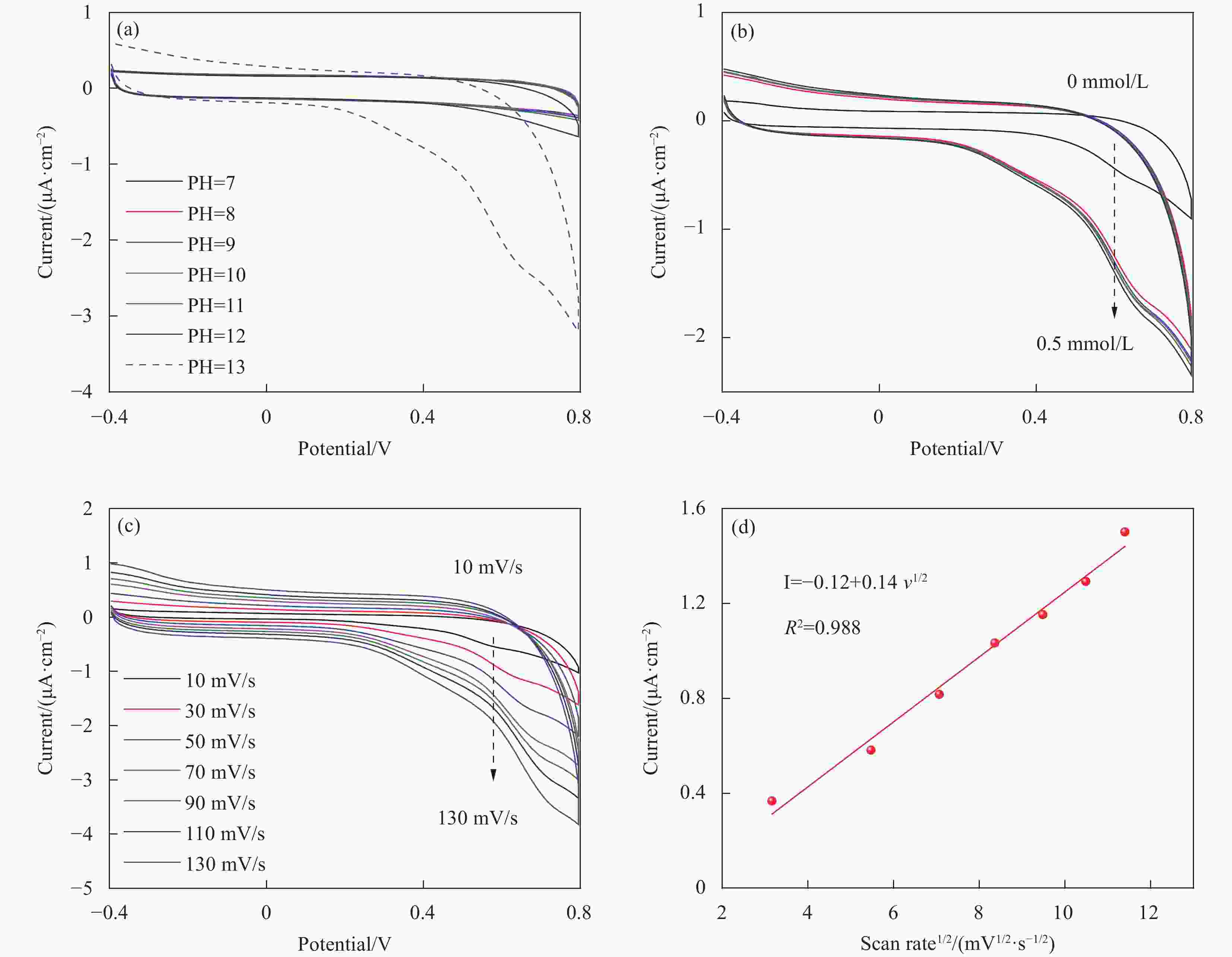
图 4 Co-ZIF-67修饰电极在不同(a)PH值,(b)葡萄糖浓度,(c)扫速对葡萄糖的CV测试曲线;(d)扫速算术平方根与0.5 V电流之间的线性关系。
Figure 4. CV curves of Co-ZIF-67 modified electrodes (a) with different PH and (b) at different glucose concentrations. CV curves (c) at different scan rate and (d) corresponding linear relationship between current and scan rate1/2.
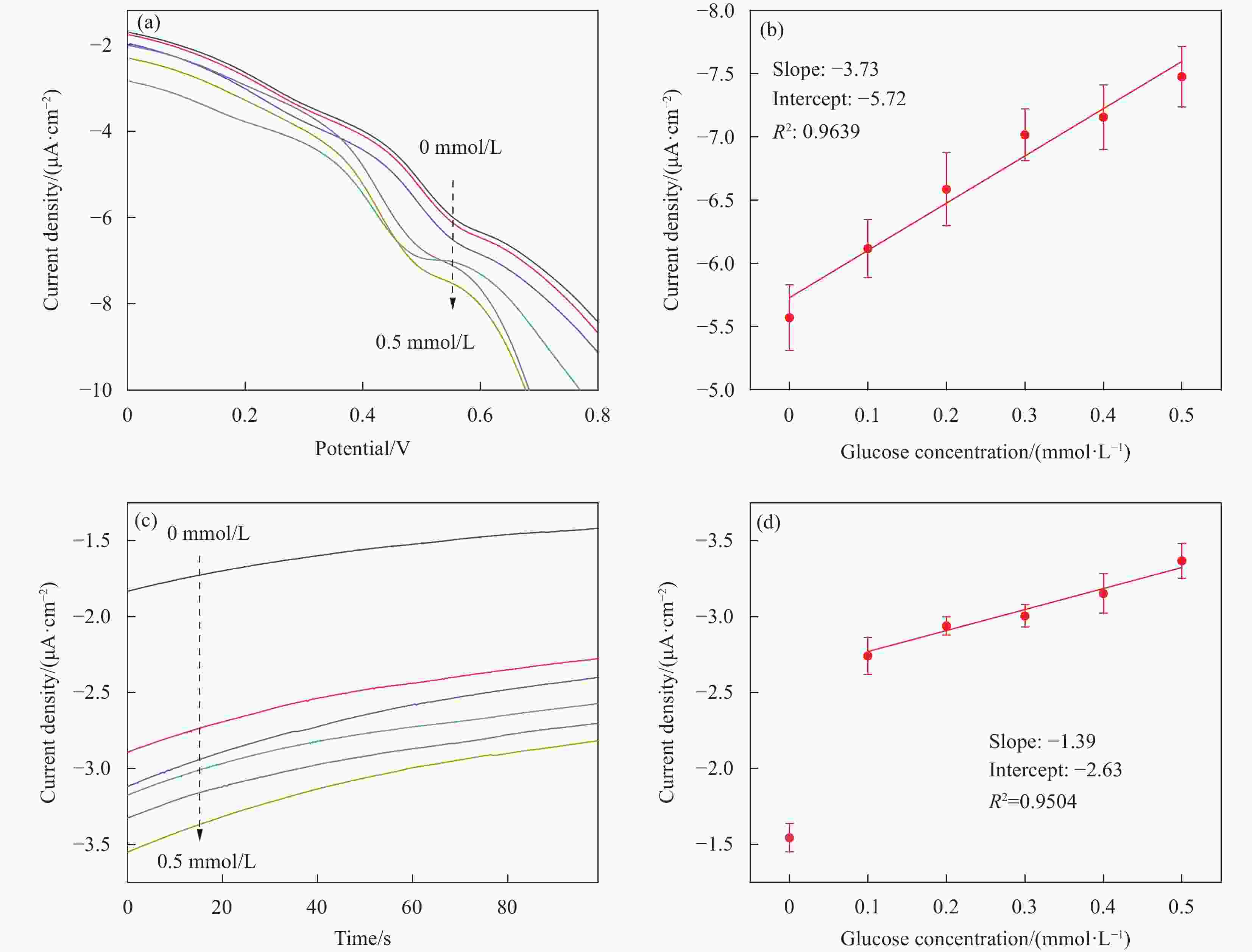
图 5 (a)Co-ZIF-67修饰电极在不同葡萄糖浓度中的DPV测试曲线;(b)每5支Co-ZIF-67修饰电极在0.55 V电位对不同浓度葡萄糖的平均电流响应信号;(c)Co-ZIF-67修饰电极对不同葡萄糖浓度的安培响应。(d)每5支电极对不同葡萄糖浓度的平均响应电流(取第15秒数值)。
Figure 5. (a) DPV curves of Co-ZIF-67 modified electrodes in the presence of glucose; (b) Linear relationship between average DPV current density response and different glucose concentrations of every five electrodes at 0.55 V; (c) Amperometric response of Co-ZIF-67 modified SPEs to different glucose concentration. (d) Corresponding linear curve of average current density of five electrodes in the 15 th second to glucose concentrations.
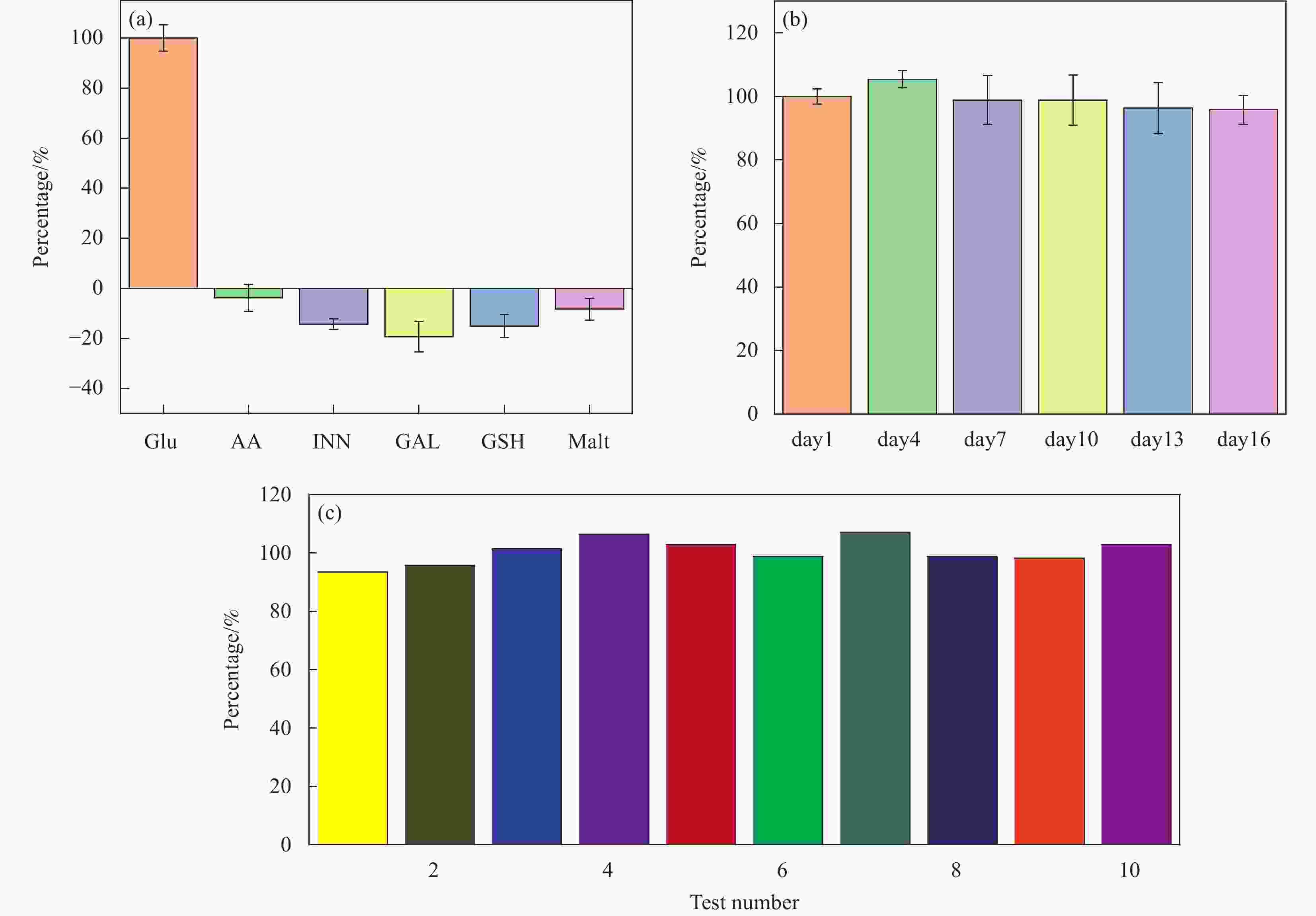
图 6 (a)干扰检查: 五支Co-ZIF-67修饰电极 对0.1 mmol/L 葡萄糖(GLU),0.164 mol/L 艾考糊精(INN),9 mmol/L 半乳糖(GAL),30 mmol/L谷胱甘肽(GSH)和0.584 mol/L 麦芽糖(MAL) 的平均安培响应。(b)稳定性: 每5支Co-ZIF-67修饰电极在第1、4、7、10、13和16天内对0.1 mmol/L 葡萄糖的安培响应信号。(c)重现性: 10支Co-ZIP-67修饰电极对0.1 mmol/L 葡萄糖的响应。
Figure 6. (a) Interference examination: average amperometric responses of five CuO nanomaterials modified SPEs to 0.1 mmol/L Glucose(GLU), 0.164 mol/L Alcodextrin (Inn) , 9 mmol/L Galactose (GAL) , 30 mmol/L Glutathione (GSH) and 0.584 mol/L Maltose (MAL) . (b) Stability of every 5 Co-ZIF-67 modified electrodes to 0.1 mmol/L glucose on the 1 st, 4 th, 7 th, 10 th, 13 th and 16 th days. (c) Reproducibility of Co-ZIF-67 modified electrodes to 0.1 mmol/L glucose.
表 1 Co-ZIF-67修饰电极及其它电极的葡萄糖传感性能
Table 1. Glucose sensing performance of Co-ZIF-67-modified electrodes and other previously reported electrodes
Type of electrode Sensitivity (μA·L/(mmol·cm2) Detection limit (μmol/L) Linear range (mmol/L) Refs AgNPs/MOF-74(Ni) 1290 4.7 0.01-4 [27] NF/NiCo2O4NWs@Co3O4NPs 8163.2 — 0.001-1.7 [29] CuCo-MOF 6861 0.12 — [30] Ni2Co1-BDC/GCE 3925.3 0.29 0.0005-2.8995 [31] Ni/Co(HHTP)MOF/CC 3250 0.1 0.0003-2.312 [32] MIL-88 A@NiFe-PB 1963.2 0.12 0.005-1 [33] Ni3(HHTP)2/CNT 4774 4.1 0.004-3.9 [34] Co-MOFs/SPEs 1.393 0.58 0.1-0.5 This work Abbreviations: BTC: trimesic acid; CC: carbon cloth; BDC: 1,4-benzenedicarboxylic acid; GCE: glassy carbon electrode; HHTP: 2,3,6,7,10,11-hexahydroxytriphenylene; MIL:Materials from Institute Lavoisier; PB:Prussian Blue; CNT: carbon nanotubes; NP: nanoparticle; NF: nickel foam; NWs: nanowires; NPs: nanoparticles; SPEs: screen-printing electrodes 表 2 Co-ZIF-67修饰电极检测血清样品的葡萄糖含量(n = 3)
Table 2. Glucose detection in human serum samples using Co-ZIF-67 modified Ag-C electrodes (n = 3)
Samples Serum glucose (mmol/L) Added glucose (mmol/L) Detected glucose (mmol/L) RSD (%) Recovery rate (%) Human serum 0.12 0.18 0.29 6.20 93.97 0.28 0.39 4.37 101.5 0.36 0.47 3.90 98.97 -
[1] JARNDA K V, WANG D, AIN Q, et al. Recent advances in electrochemical non-enzymatic glucose sensor for the detection of glucose in tears and saliva: a Review[J]. Sensors and Actuators A:Physical, 2023, 363: 114778. doi: 10.1016/j.sna.2023.114778 [2] DO H H, KIM S Y, LE Q V, Development of non-precious metal oxide-based electrodes for enzyme-free glucose detection: a review[J]. Microchemical Journal, 2023, 193: 109202. [3] HE C, ASIF M, LIU Q, et al. Noble metal construction for electrochemical nonenzymatic glucose detection[J]. Advanced Materials Technologies, 2023, 8: 2200272. doi: 10.1002/admt.202200272 [4] TANG X, YUAN X, JIN Yao, et al. A novel hollow CuMn-PBA@NiCo-LDH nanobox for efficient detection of glucose in food[J]. Food Chemistry, 2024, 438: 137969. doi: 10.1016/j.foodchem.2023.137969 [5] DODEVSKA T, LAZAROVA Y, SHTEREV I, Amperometric biosensors for glucose and lactate with applications in food analysis: a brief review[J]. Acta Chimica Slovenica, 2019, 66: 762-776. [6] JIE X, YAN Z, LIU Q, Smartphone-based electrochemical systems for glucose monitoring in biofluids: a review[J]. Sensors, 2022, 22: 5670. [7] OLAYA A, COSTA E, ABEDUL M, 2022. Paper-based enzymatic electrochemical sensors for glucose determination[J]. Sensors, 2022, 22(16): 6232. doi: 10.3390/s22166232 [8] ALI M H, HARATBAR S M, ZARE Y, et al. A review on non-enzymatic electrochemical biosensors of glucose using carbon nanofiber nanocomposites[J]. Biosensors, 2022, 12(11): 1004. doi: 10.3390/bios12111004 [9] PERALES E R, HERNANDEZ P, ROMERO G A, et al. Trends on the development of non-enzymatic electrochemical sensors modified with metal-organic frameworks for the quantification of glucose[J]. Journal of the electrochemical Society, 2023, 170: 087507. doi: 10.1149/1945-7111/aced6f [10] LI P, PENG Y, CAI J, et al. Recent advances in metal-organic frameworks (MOFs) and their composites for non-enzymatic electrochemical glucose sensors[J]. Bioengineering, 2023, 10(6): 733. doi: 10.3390/bioengineering10060733 [11] MUHAMMAD A, KANWAL A, RAHMAN M, et al. Glucose detection devices and methods based on metal-organic frameworks and related materials[J]. Advanced Functional Materials, 2021, 31: 2106023. doi: 10.1002/adfm.202106023 [12] HESSAMADDIN S, MALEKI F, KHAAKI P, et al. Electrochemical-based sensing platforms for detection of glucose and H2O2 by porous metal-organic frameworks: a review of status and prospects[J]. Biosensors, 2023, 13(3): 347. doi: 10.3390/bios13030347 [13] LI Y, XIE M, ZHANG X, et al. Co-MOF nanosheet array: A high-performance electrochemical sensor for non-enzymatic glucose detection[J]. Sensors and Actuators B:Chemical, 2019, 278: 126-132. doi: 10.1016/j.snb.2018.09.076 [14] KHAN I A, BADSHAH A, NADEEM M A, et al. A copper based metal-organic framework as single source for the synthesis of electrode materials for high-performance supercapacitors, glucose sensing applications[J]. International Journal of Hydrogen Energy, 2014, 39: 19609-19620. doi: 10.1016/j.ijhydene.2014.09.106 [15] XIAO X, ZHENG S S, LI X R, et al. Facile synthesis of ultrathin Ni-MOF nanobelts for high-efficiency determination of glucose in human serum[J]. Journal of Materials Chemistry B, 2017, 5: 5234-5239. doi: 10.1039/C7TB00180K [16] ZHA X, YANG W, SHI L, et al. Morphology control strategy of bimetallic MOF nanosheets for upgrading the sensitivity of noninvasive glucose detection[J]. ACS Applied Materials & Interfaces, 2022, 14: 37843-37852. [17] SURESH R R, LAKSHMANAKUMAR M, AROCKIA J, et al. Fabrication of screen-printed electrodes: opportunities and challenges[J]. Journal of Materials Science, 2021, 56: 8951-9006. doi: 10.1007/s10853-020-05499-1 [18] LI X, ZHANG M, Hu Y, et al. Developing a versatile electrochemical platform with optimized electrode configuration through screen-printing technology toward glucose detection[J]. Biomed Microdevices, 2020, 22: 74. doi: 10.1007/s10544-020-00527-y [19] JI D, LIU L, LI S, et al. Smartphone-based cyclic voltammetry system with graphene modified screen printed electrodes for glucose detection[J]. Biosensors and Bioelectronics, 2017, 98: 449-456. doi: 10.1016/j.bios.2017.07.027 [20] FENG X, CARREON M A, Kinetics of transformation on ZIF-67 crystals[J]. Journal of Crystal Growth, 2015, 418: 158-162. [21] GROSS A F, SHERMAN E, VAJO J J, Aqueous room temperature synthesis of cobalt and zinc sodalite zeolitic imidizolate frameworks[J]. Dalton Transactions, 2012, 41: 5458-5460. [22] JAFARINASAB M, AKBARI A, OMIDKHAH M, et al. An efficient Co-based metal-organic framework nanocrystal (Co-ZIF-67) for adsorptive desulfurization of dibenzothiophene: impact of the preparation approach on structure tuning[J]. Energy & Fuels 2020, 34: 12779-12791. [23] TIAN Y, DENG P, WU Y, et al. A Simple and Efficient Molecularly Imprinted Electrochemical Sensor for the Selective Determination of Tryptophan[J]. Biomolecules, 2019, 9(7): 294. doi: 10.3390/biom9070294 [24] WU Y, DENG P, TIAN Y, et al. Rapid recognition and determination of tryptophan by carbon nanotubes and molecularly imprinted polymer-modified glassy carbon electrode[J]. Bioelectrochemistry, 2020, 131: 107393. doi: 10.1016/j.bioelechem.2019.107393 [25] WU Y, LI G, TIAN Y, et al. Electropolymerization of molecularly imprinted polypyrrole film on multiwalled carbon nanotube surface for highly selective and stable determination of carcinogenic amaranth[J]. Journal of Electroanalytical Chemistry, 2021, 895: 115494. doi: 10.1016/j.jelechem.2021.115494 [26] WEI Y, WU Y, FENG J, et al. An ultrasensitive ponceau 4R detection sensor based on molecularly imprinted electrode using pod-like cerium molybdate and multi-walled carbon nanotubes hybrids[J]. Journal of Food Composition and Analysis, 2022, 114: 104849. doi: 10.1016/j.jfca.2022.104849 [27] PENG X, WAN Y, WANG Y, et al. Flower-like Ni(II)-based metal-organic framework-decorated Ag nanoparticles: fabrication, characterization and electrochemical detection of glucose[J]. Electroanalysis, 2019, 31: 2179. doi: 10.1002/elan.201900259 [28] LI S, XIA H, LIU Y, et al. Room-temperature and gram-scale constructed Cu@CuO with promoted kinetics for glucose electrooxidation in the Faraday process[J]. SCIENCE CHINA Materials, 2023, 66(11): 4396-4402. doi: 10.1007/s40843-023-2588-4 [29] LU J, CHENG C, CAO Y, et al. MOFs-derived core-shell structured NiCo2O4NWs@Co3O4NPs for non-enzymatic glucose detection[J]. Ceramics International, 2023, 49: 23958-23966. doi: 10.1016/j.ceramint.2023.04.245 [30] LIU Q, CHEN J, YU F, et al. Multifunctional book-like CuCo-MOF for highly sensitive glucose detection and electrocatalytic oxygen evolution[J]. New Journal of Chemistry, 2021, 45: 16714-16721. doi: 10.1039/D1NJ02931B [31] QI W, JIA Q, HU P, et al. Tunable non-enzymatic glucose electrochemical sensing based on the Ni/Co bimetallic MOFs[J]. Molecules, 2023, 289(15): 5649. [32] XU Z, WANG Q, HUI Z, et al. Carbon cloth-supported nanorod-like conductive Ni/Co bimetal MOF: a stable and high-performance enzyme-free electrochemical sensor for determination of glucose in serum and beverage[J]. Food Chemistry, 2021, 349: 129202. doi: 10.1016/j.foodchem.2021.129202 [33] QIN W, HE L, ZHANG Y, et al. Synchronous wrapping and inward-etching strategy on constructing yolk-shell MIL-88A@NiFe-PB heterostructures for electrochemical non-enzymatic glucose detection[J]. Microchemical Journal, 2024, 196: 109641. doi: 10.1016/j.microc.2023.109641 [34] LUO Y, SHUPLETSOV L, RITA M, et al. Integration of triphenylene-based conductive metal-organic frameworks into carbon nanotube electrodes for boosting nonenzymatic glucose sensing[J]. ACS Applied Materials & Interfaces, 2023, 15(44): 51435-51443. -

 点击查看大图
点击查看大图
计量
- 文章访问数: 56
- HTML全文浏览量: 33
- 被引次数: 0




 下载:
下载: