Basic scientific problems of nickel-rich cathode for lithium-ion battery: Regulation and formation mechanism of radially oriented parties
-
摘要: 一次晶粒径向有序组装的颗粒结构能够较好抑制微裂纹的产生并提供良好的Li+扩散路径,是高端多晶富镍锂电正极材料理想的形态。近年来,研究人员通过对前驱体沉淀结晶和正极高温锂化结晶调控,获得具有较大长宽比晶粒有序组装的富镍正极材料。然而,关于富镍正极径向有序结构调控方法及形成机制的论述,特别是关于径向有序氢氧化物前驱体的调控方法、关键参数对有序结构的影响等并无详细的阐述。本文首先介绍了多晶富镍正极径向有序结构调控的必要性及其电化学性能提升的作用机制;其次,介绍了径向有序多晶富镍正极调控方法及形成机制,包括沉淀结晶过程关键参数(pH、氨浓度及固含量)对径向有序前驱体的影响、高温锂化过程温度及掺杂元素对正极继承前驱体径向有序性的影响;最后,对径向有序富镍正极调控面临的问题进行探讨,以期为相关专业人员开发富镍正极提供参考。Abstract: Secondary particle assembed with radial oriented primary grains can inhibit the formation of microcracks and provide a good Li+ diffusion path, and it is an ideal morphology for high-end polycrystalline Ni-rich cathode materials. In recent years, some researchers have obtained nickel-rich cathode materials assembed with grains with large length-width ratio by regulating precursor precipitation crystallization and high temperature lithium crystallization. However, the regulation method and formation mechanism of the radially oriented structure of Ni-rich cathode, especially the regulation method of the radially oriented hydroxide precursor and the influence of the key parameters on the radially oriented structure, have not been elaborated. In this paper, the necessity of regulating the radially oriented structure of polycrystalline Ni-rich cathode and the mechanism on enhancing electrochemical performance are introduced. Secondly, the regulation method and formation mechanism of the radially oriented polycrystalline Ni-rich cathode are introduced, including the influence of the key parameters of precipitation crystallization process (pH, ammonia concentration and solid content) on the radially oriented precursor, and the influence of temperature and doping elements induced in calcination process on the maintenance of the oriented structure of precursor. Finally, the challenges facing for the regulation of radially oriented Ni-rich cathode are discussed.
-
图 1 (a) 3种不同镍含量的富镍正极材料晶胞参数a、c及晶胞体积V随充电电压的变化情况[8];(b) 无序多晶及径向有序多晶充放电循环过程稳定性示意图[14];(c) 径向有序多晶明场STEM图像[14];(d) 径向有序多晶晶粒放大图、选取电子衍射及晶体结构示意图[14]
Figure 1. (a) Variations of a and c lattice parameters and normalized unit cell volume V as a function of the cell voltage[8]; (b) Schematic illustration of mechanical stability of the cathodes with randomly oriented particles and radially oriented particles during charge and discharge cycling[14]; (c) Bright-field STEM images of radially oriented particles[14]; (d) Magnified TEM image of textured primary grains, electron diffraction pattern from region marked by circle, and schematic drawing of crystal structure of oriented primary grains[14]
NCA—Li[Ni1−x−y-CoxAly]O2 (1−x−y=0.8, 0.88 and 0.95); TM—Transition metal
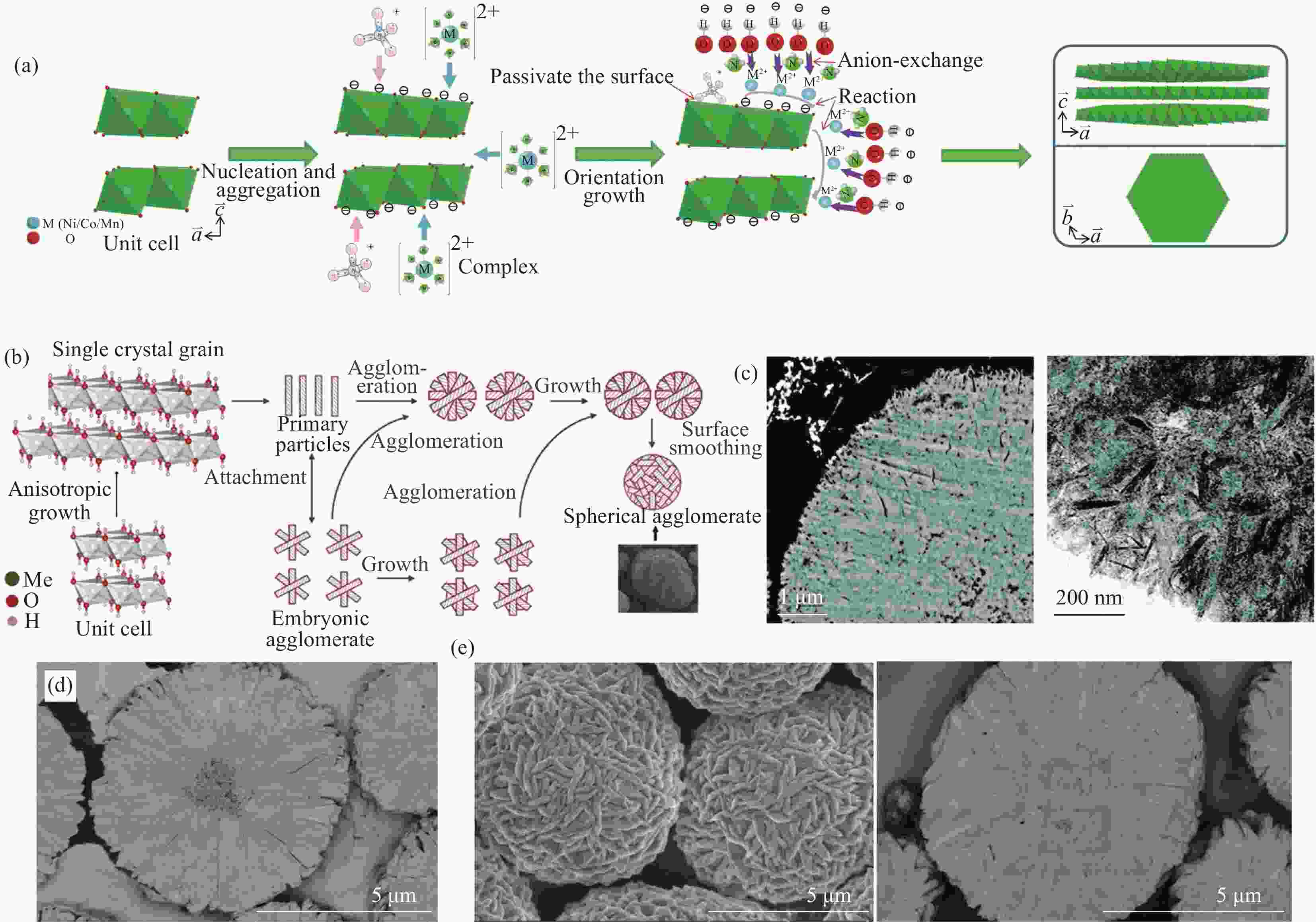
图 2 (a) 氨存在下氢氧化物前驱体一次晶粒(001)晶面择优生长机制[18];(b) 球形二次颗粒团聚体的形成示意图[20];(c) 无序多晶横截面SEM图像及TEM图像[2];(d) 具有无序堆积内核的二次颗粒的截面SEM图像[25];(e) 晶粒密实生长的颗粒表面及截面SEM图像[25]
Figure 2. (a) Preferential growth mechanism of (001) crystal plane for primary grain in the presence of ammonia[18]; (b) Formation mechanism of spherical secondary particle agglomerates[20]; (c) Cross-sectional SEM and TEM images of randomly oriented precursors[2]; (d) SEM image of the particle with randomly oriented core[25]; (e) SEM images of the particle with compact assembled grains[25]
图 4 ((a), (b)) 具有不同内核大小的径向有序放射状内核及树突状外部区域的前驱体[26];((c), (d)) 具有不同孔隙率区域的径向有序前驱体及其示意图[24]
Figure 4. ((a), (b)) Precursors with a radially oriented radial core and a dendritic outer region[26]; ((c), (d)) Radial oriented precursor with different porosity regions and its corresponding schematic diagram[24]
图 5 ((a), (b)) 具有纳米晶粒的核壳前驱体的SEM及TEM图像[27];((c), (d))具有径向有序纳米棒晶粒的核壳前驱体的SEM及TEM图像[27];((e), (f)) 径向有序浓度梯度前驱体的截面SEM及TEM图像[2];((g), (h)) 间断进料制备的无序浓度梯度前驱体的截面SEM及TEM图像[2]
Figure 5. ((a), (b)) SEM and TEM images of the core-shell precursor with a nanoparticle shell[27]; ((c), (d)) SEM and TEM images of the core-shell precursor with a nanorod shell[27]; ((e), (f)) Cross-sectional SEM and TEM images of radially oriented precursors with concentration gradient[2]; ((g), (h)) Cross-sectional SEM and TEM images of randomly oriented precursors with concentration gradient[2]
图 6 ((a)~(c)) 径向有序前驱体及其掺杂2mol%Al和4mol%Al的正极截面SEM图像[2];((d), (e)) 径向有序前驱体Ni0.9Co0.1(OH)2未掺杂及掺杂Ta在不同温度下煅烧的LiNi0.9Co0.1O2 截面SEM图像[29]
Figure 6. ((a)-(c)) Cross-sectional SEM images of radially oriented precursor and corresponding cathodes with 2mol%Al and 4%molAl[2]; ((d), (e)) Cross-sectional SEM images of pristine and Ta doped LiNi0.9Co0.1O2 cathodes, lithiated at different temperatures[29]
-
[1] PARK G T, RYU H H, NOH T C, et al. Microstructure-optimized concentration-gradient NCM cathode for long-life Li-ion batteries[J]. Materials Today,2022,52:9-18. doi: 10.1016/j.mattod.2021.11.018 [2] PARK G T, PARK N Y, NOH T C, et al. High-performance Ni-rich LiNi0.9−xCo0.1AlxO2 cathodes via multi-stage microstructural tailoring from hydroxide precursor to the lithiated oxide[J]. Energy & Environmental Science,2021,14(9):5084-5095. [3] NGUYEN T T, KIM U H, YOON C S, et al. Enhanced cycling stability of Sn-doped LiNi0.9Co0.05Mn0.05O2 via optimization of particle shape and orientation[J]. Chemical Engineering Journal, 2021, 405: 126887. [4] 曹梦圆, 王明星, 邓中莉, 等. 锂离子电池富镍正极基础科学问题: 晶粒形态及组装方式调控[J]. 复合材料学报, 2023, 40(5):2525-2535.CAO Mengyuan, WANG Mingxing, DENG Zhongli, et al. Basic scientific problems of nickel-rich cathode for lithium-ion battery: Morphology and accumulation regulation of primary particles[J]. Acta Materiae Compositae Sinica,2023,40(5):2525-2535(in Chinese). [5] XIAO P H, SHI T, HUANG W X, et al. Understanding surface densified phases in Ni-rich layered compounds[J]. ACS Energy Letters,2019,4(4):811-818. doi: 10.1021/acsenergylett.9b00122 [6] LI J Y, MANTHIRAM A. A comprehensive analysis of the interphasial and structural evolution over long-term cycling of ultrahigh-nickel cathodes in lithium-ion batteries[J]. Advanced Energy Materials,2019,9(45):1902731. doi: 10.1002/aenm.201902731 [7] PARK K J, HWANG J Y, RYU H H, et al. Degradation mechanism of Ni-enriched NCA cathode for lithium batteries: Are microcracks really critical?[J]. ACS Energy Letters,2019,4(6):1394-1400. doi: 10.1021/acsenergylett.9b00733 [8] NAM G W, PARK N Y, PARK K J, et al. Capacity fading of Ni-rich NCA cathodes: Effect of microcracking extent[J]. ACS Energy Letters, 2019, 4: 2995-3001. [9] RYU H H, PARK N Y, SEO J H, et al. A highly stabilized Ni-rich NCA cathode for high-energy lithium-ion batteries[J]. Materials Today, 2020, 36: 73-82. [10] JUNG S H, KIM U H, KIM J H, et al. Ni-rich layered cathode materials with electrochemo-mechanically compliant microstructures for all-solid-state Li batteries[J]. Advanced Energy Materials,2020,10(6):1903360. doi: 10.1002/aenm.201903360 [11] KIM U H, RYU H H, KIM J H, et al. Microstructure-controlled Ni-rich cathode material by microscale compositional partition for next-generation electric vehicles[J]. Advanced Energy Materials,2019,9(15):1803902. doi: 10.1002/aenm.201803902 [12] NOH H J, CHEN Z H, YOON C S, et al. Cathode material with nanorod structure—An application for advanced high-energy and safe lithium batteries[J]. Chemistry of Materials,2013,25(10):2109-2115. doi: 10.1021/cm4006772 [13] YOON C S, KIM S J, KIM U H, et al. Microstructure evolution of concentration gradient LiNi0.75Co0.1Mn0.15O2 cathode for lithium-ion batteries[J]. Advanced Functional Materials,2018,28(28):1802090. doi: 10.1002/adfm.201802090 [14] PARK K J, JUNG H G, KUO L Y, et al. Improved cycling stability of LiNi0.9Co0.05Mn0.05O2 through microstructure modification by boron doping for Li-ion batteries[J]. Advanced Energy Materials,2018,8(25):1801202. doi: 10.1002/aenm.201801202 [15] XU X, HUO H, JIAN J Y, et al. Radially oriented single-crystal primary nanosheets enable ultrahigh rate and cycling properties of LiNi0.8Co0.1Mn0.1O2 cathode material for lithium-ion batteries[J]. Advanced Energy Materials,2019,9(15):1803963. doi: 10.1002/aenm.201803963 [16] PARK G T, YOON D R, KIM U H, et al. Ultrafine-grained Ni-rich layered cathode for advanced Li-ion batteries[J]. Energy & Environmental Science, 2021, 14: 6616-6626. [17] KIM U H, PARK J H, AISHOVA A, et al. Microstructure engineered Ni-rich layered cathode for electric vehicle batteries[J]. Advanced Energy Materials,2021,11(25):2100884. doi: 10.1002/aenm.202100884 [18] HUA W B, LIU W Y, CHEN M Z, et al. Unravelling the growth mechanism of hierarchically structured Ni1/3Co1/3Mn1/3(OH)2 and their application as precursors for high-power cathode materials[J]. Electrochimica Acta, 2017, 232: 123-131. [19] 王鑫, 任莉, 王硕, 等. 锂离子电池富镍正极基础科学问题: 氢氧化物前驱体结晶调控及机制[J]. 复合材料学报, 2022, 39(5):1995-2013.WANG Xin, REN Li, WANG Shuo, et al. Basic scientific problems of nickel rich cathode materials for Li-ion battery: Regulation and mechanism for crystallization of hydroxide precursor[J]. Acta Materiae Compositae Sinica,2022,39(5):1995-2013(in Chinese). [20] YANG Y, XU S M, XIE M, et al. Growth mechanisms for spherical mixed hydroxide agglomerates prepared by co-precipitation method: A case of Ni1/3Co1/3Mn1/3(OH)2[J]. Journal of Alloys and Compounds, 2015, 619: 846-853. [21] CHEN X L, LI D, MO Y, et al. Cathode materials with cross-stack structures for suppressing intergranular cracking and high-performance lithium-ion batteries[J]. Electrochimica Acta, 2018, 261: 513-520. [22] 王碧武, 黄晓笑, 于建, 等. 一种高镍前驱体材料、其制备方法及应用: 中国, 202110565272.6[P]. 2022-11-15.WANG Biwu, HUANG Xiaoxiao, YU Jian, et al. A precursor material for high nickel cathode and its preparation method and application: CN, 202110565272.6[P]. 2022-11-15(in Chinese). [23] 熊俊, 卞益鹏, 高文政, 等. 一种三元前驱体晶须形貌的控制方法: 中国, 202210726493.1[P]. 2022-09-20.XIONG Jun, BIAN Yipeng, GAO Wenzheng, et al. A method for controlling the whisker morphology of precursor material for ternary cathode: CN, 202210726493.1[P]. 2022-09-20(in Chinese). [24] 訚硕, 王乐, 张雨英, 等. 三元正极材料前驱体及制备方法、正极材料、正极浆料、锂离子电池及正极和涉电设备: 中国, 202111623510.0[P]. 2023-01-24.YAN Shuo, WANG Le, ZHANG Yuying, et al. A precursor material for ternary cathode and its preparation method, cathode, cathode paste, lithium ion battery and electric-related equipment: CN, 202111623510.0[P]. 2023-01-24(in Chinese). [25] 余春林, 陈旭东. 球形高镍三元前驱体材料、其制备方法与高镍三元正极材料: 中国, 202110948253.1[P]. 2021-11-16.YU Chunlin, CHEN Xudong. A spherical precursor material for high nickel ternary cathode and its preparation method and corresponding high nickel ternary cathode: CN, 202110948253.1[P]. 2021-11-16(in Chinese). [26] 余春林, 陈旭东. 高镍三元前驱体材料、其制备方法与高镍三元正极材料: 中国, 202110949247.8[P]. 2021-11-16.YU Chunlin, CHEN Xudong. A high-nickel ternary precursor material, its preparation method and high-nickel ternary positive electrode material: CN, 202110949247.8[P]. 2021-11-16(in Chinese). [27] YOON S J, MYUNG S T, NOH H J, et al. Nanorod and nanoparticle shells in concentration gradient core-shell lithium oxides for rechargeable lithium batteries[J]. ChemSusChem,2014,7(12):3295-3303. doi: 10.1002/cssc.201402389 [28] KIM U H, KIM J H, HWANG J Y, et al. Compositionally and structurally redesigned high-energy Ni-rich layered cathode for next-generation lithium batteries[J]. Materials Today, 2018, 23: 26-36. [29] KIM U H, PARK G T, SON B K, et al. Heuristic solution for achieving long-term cycle stability for Ni-rich layered cathodes at full depth of discharge[J]. Nature Energy, 2021, 5: 860-869. [30] YANG W, LI H D, WANG D, et al. Ta induced fine tuning of microstructure and interface enabling Ni-rich cathode with unexpected cyclability in pouch-type full cell[J]. Nano Energy,2022,104:107880. doi: 10.1016/j.nanoen.2022.107880 [31] SUN H H, KIM U H, PARK J H, et al. Transition metal-doped Ni-rich layered cathode materials for durable Li-ion batteries[J]. Nature Communications,2021,12(1):6552. doi: 10.1038/s41467-021-26815-6 [32] KIM U H, PARK N Y, PARK G T, et al. High-energy W-doped LiNi0.95Co0.04Al0.01O2 cathodes for next-generation electric vehicles[J]. Energy Storage Materials, 2020, 33: 399-407. [33] PARK N Y, RYU H H, KUO L Y, et al. High-energy cathodes via precision microstructure tailoring for next-generation electric vehicles[J]. ACS Energy Letters,2021,6(12):4195-4202. doi: 10.1021/acsenergylett.1c02281 [34] ZHU C Q, CAO M Y, ZHANG H Y, et al. Synergistic effect of microstructure engineering and local crystal structure tuning to improve the cycling stability of Ni-rich cathodes[J]. ACS Applied Materials & Interfaces,2021,13(41):48720-48729. -






 下载:
下载: