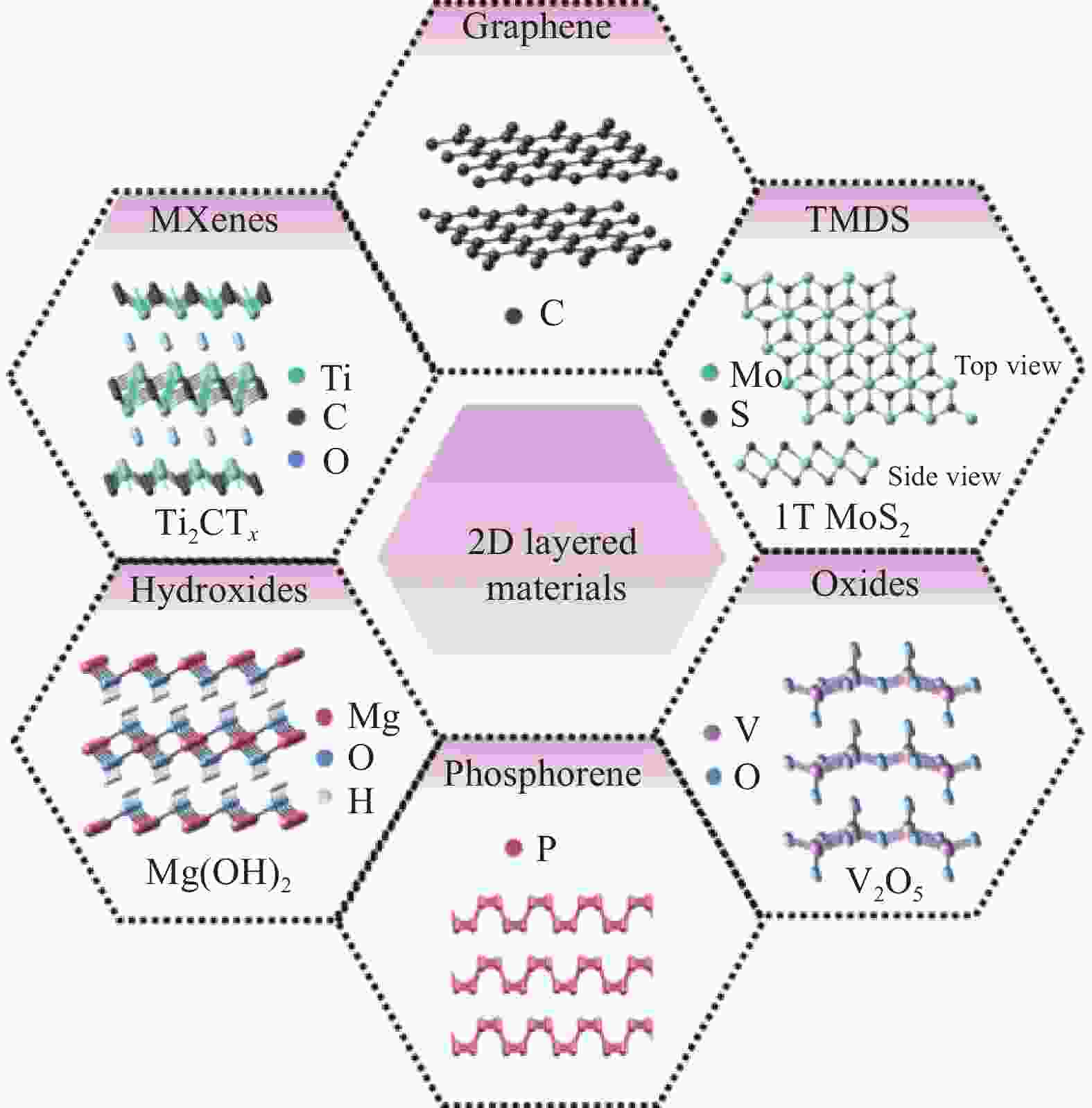
Research progress of two-dimensional materials in the field of lithium extraction from salt lake
-
摘要: 近年来,随着新能源行业的大力发展,锂资源的供需矛盾加剧,合理开发利用锂资源正成为我国新能源发展的关键因素。盐湖卤水因其具备成本低、储量多的优势逐渐成为我国提锂行业的发展方向。二维材料是指电子仅可在两个维度的非纳米尺度(1~100 nm)上平面运动的材料。由于二维纳米材料具备原子级尺度的特殊片层结构、高比表面积、优异的机械强度、丰富的活性位点及良好的可修饰性等特点,得到了科学界的广泛关注。本文从不同二维材料的特点出发,归纳了二维材料在吸附法提锂和膜分离法提锂两个领域的应用,总结了两种方法的特点,并指出其目前面临的问题及今后的发展趋势,为二维材料在盐湖提锂领域的研究与应用提供参考。Abstract: In recent years, with the vigorous development of new energy industry, the contradiction between the supply and demand of lithium materials has intensified, and the rational development and utilization of lithium resources are gradually becoming key factors in the development of new energy in China. Salt lake brine is gradually becoming the development direction of China's lithium extraction industry due to its low cost and abundant reserves. Two-dimensional (2D) materials refer to materials in which electrons can only move in two dimensions on a non-nanometer scale (1-100 nm). 2D nanomaterials have received extensive attention in the scientific community due to their special lamellar structure at the atomic scale, high specific surface area, good mechanical strength, rich active sites and good modifiability. This article summarizes the applications of 2D materials as adsorbents and membrane separation materials for lithium extraction in terms of their characteristics, and also points out the current problems and future development trends. This review provides references for the research and application of 2D materials in the field of lithium extraction from salt lake.
-
图 2 (a) 交联聚(N-异丙基丙烯酰胺-共-丙烯酰胺苯并-18-冠醚-6)功能化MOF-808(pNCE/MOF-808)吸附剂对单价金属离子的选择性吸附;(b) 适宜温度下的解吸示意图[44]
Figure 2. (a) Selective adsorption of monovalent metal ions by A crosslinked poly (N-isopropylacrylamide-co-acryloylamidobenzo-18-crown-6) functionalized MOF-808 (pNCE/MOF-808) adsorbent; (b) Schematic diagram of desorption at suitable temperature[44]
AmB18C6—acryloylamidobenzo-18-crown-6; pNIPAM—Poly N-isopropylacrylamide
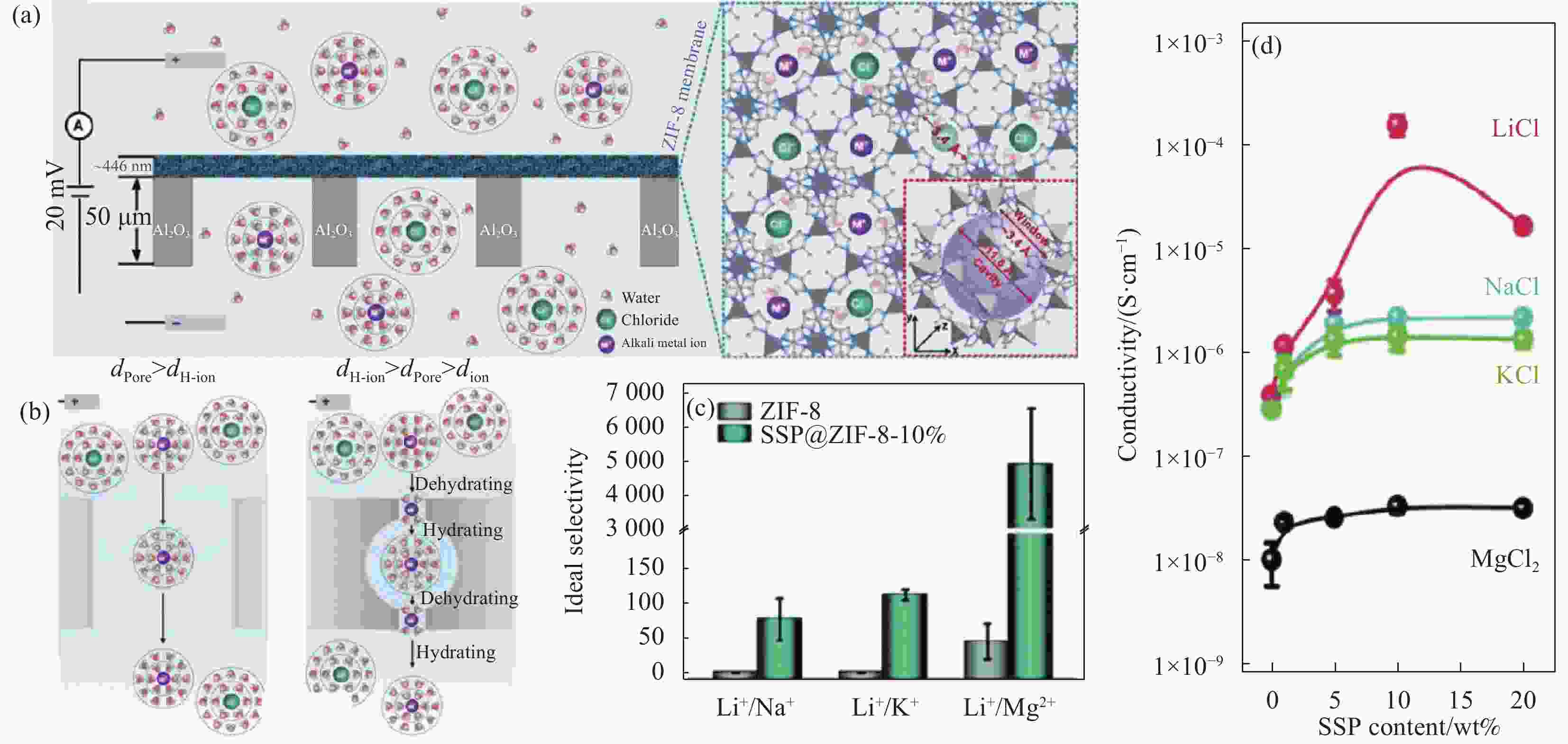
图 5 (a) ZIF-8的晶体结构示意图;(b) ZIF-8离子传输及脱水-水合机制示意图;(c) 原始ZIF-8膜的离子选择性和磺化螺吡喃(SSP)@ZIF-8膜选择性比较;(d) 不同SSP含量的SSP@ZIF-8膜的不同离子电导率[58-59]
Figure 5. (a) Schematic diagram of the crystal structure of ZIF-8; (b) Schematic diagram of the ion transport and dehydration-hydration mechanism of ZIF-8; (c) Comparison of ion selectivity of pristine ZIF-8 membrane and sulfonated spiropyran (SSP)@ZIF-8 membrane selectivity; (d) Different ionic conductivities of SSP@ZIF-8 membrane with different SSP contents[58-59]
dion—Dehydrated ionic diameters; dPore—Diameter of the ion transport pore; dH-ion—Hydrated ionic diameter; SSP@ZIF-8-10%—SSP@ZIF-8 with 10wt%SSP content
图 6 (a) MXene/聚4-苯乙烯磺酸钠(PSS)复合膜中Li+的快速传输通道示意图;(b) PSS含量对复合膜分离性能的影响;(c) MXene/PSS复合膜的长期稳定性[63]
Figure 6. (a) Schematic diagram of the fast transport channel of Li+ in the MXene/polysodium 4-styrene sulfonate (PSS) composite membrane; (b) Effect of PSS content on the separation performance of the composite membrane; (c) Long-term stability of MXene/PSS composite membrane[63]
-
[1] 周久龙, 树银雪. 我国盐湖卤水提锂产业化现状及发展建议[J]. 化工矿物与加工, 2022, 52(1):57-62.ZHOU Jiulong, SHU Yinxue. Present situation and development suggestions of lithium extraction from salt lake brine in China[J]. Iindustrial Minerals& Processing,2022,52(1):57-62(in Chinese). [2] AMBROSE H, KENDALL A. Understanding the future of lithium: Part 1, resource model[J]. Journal of Industrial Ecology,2020,24(1):80-89. doi: 10.1111/jiec.12949 [3] KESLER S E, GRUBER P W, MEDINA P A, et al. Global lithium resources: Relative importance of pegmatite, brine and other deposits[J]. Ore Geology Reviews,2012,48:55-69. doi: 10.1016/j.oregeorev.2012.05.006 [4] 赵旭, 张琦, 武海虹, 等. 盐湖卤水提锂[J]. 化学进展, 2017, 29(7):796-808.ZHAO Xu, ZHANG Qi, WU Haihong, et al. Extraction of lithium from salt lake brine[J]. Progress in Chemistry,2017,29(7):796-808(in Chinese). [5] 钟辉, 殷辉安. 偏钛酸型锂离子交换剂表面性质与选择吸附性研究[J]. 离子交换与吸附, 2003(1):55-60. doi: 10.3321/j.issn:1001-5493.2003.01.009ZHONG Hui, YIN Huian. Study on the properties of the surface and absorpt of Li+ ion- exchanger of H2TiO3 type[J]. Ion Exchange and Adsorption,2003(1):55-60(in Chinese). doi: 10.3321/j.issn:1001-5493.2003.01.009 [6] 韩继龙, 曾祥杰, 王奎虎, 等. 复合膜材料在盐湖提锂中的研究进展和展望[J]. 复合材料学报, 2022, 39(5):2106-2120.HAN Jilong, ZENG Xiangjie, WANG Kuihu, et al. Research progress and prospect of membrane method in seawater/brine extraction of lithium[J]. Acta Materiae Compositae Sinica,2022,39(5):2106-2120(in Chinese). [7] 葛涛, 徐亮, 孟金伟, 等. 盐湖卤水提锂工艺技术研究进展[J]. 有色金属工程, 2021, 11(2):55-62.GE Tao, XU Liang, MENG Jinwei, et al. Research progress of lithium extraction technology from salt lake brine[J]. Nonferrous Metals Engineering,2021,11(2):55-62(in Chinese). [8] 李志录, 王敏, 赵有璟, 等. 膜特征对锂资源提取过程的影响[J]. 化工进展, 2021, 40(9):5061-5072.LI Zhilu, WANG Min, ZHAO Youjing, et al. Effects of membrane characteristics for lithium extraction[J]. Chemical Industry and Engineering Progress,2021,40(9):5061-5072(in Chinese). [9] MANNIX A J, KIRALY B, HERSAM M C, et al. Synthesis and chemistry of elemental 2D materials[J]. Nature Reviews Chemistry,2017,1(2):1-14. doi: 10.1038/s41570-016-0014 [10] MOMENI K, JI Y, WANG Y, et al. Multiscale computational understanding and growth of 2D materials: A review[J]. npj Computational Materials,2020,6(1):1484-1501. doi: 10.1038/s41524-020-0280-2 [11] 曾广勇, 王彬, 张俊, 等. 二维MXene膜的构筑及在水处理应用中的研究进展[J]. 复合材料学报, 2021, 38(7):2078-2091.ZENG Guangyong, WANG Bin, ZHANG Jun, et al. Construction of two-dimensional MXene membrane and its research progress of application in water treatment[J]. Acta Materiae Compositae Sinica,2021,38(7):2078-2091(in Chinese). [12] 赵而法, 华旭江, 亓东美, 等. 二维插层功能材料的制备及应用研究进展[J]. 化工新型材料, 2022, 50(5):32-37.ZHAO Erfa, HUA Xujiang, QI Dongmei, et al. Advance in the preparation and application of 2D intercalated functional material[J]. New Chemical Materials,2022,50(5):32-37(in Chinese). [13] QUE H, JIANG H, WANG X, et al. Utilization of the van der waals gap of 2D materials[J]. Acta Physico-Chimica Sinica, 2021, 37(11): 2010051. [14] WANG J, ZHANG L, ZHANG T, et al. Selective removal of heavy metal ions in aqueous solutions by sulfide-selector intercalated layered double hydroxide adsorbent[J]. Jour-nal of Materials Science & Technology, 2019, 35(9): 1809-1816. [15] LI X, WANG J. One-dimensional and two-dimensional synergized nanostructures for high-performing energy storage and conversion[J]. InfoMat, 2020, 2(1): 3-32. [16] ZHANG P, MA L, FAN F, et al. Fracture toughness of graphene[J]. Nature Communications,2014,5(1):3782. doi: 10.1038/ncomms4782 [17] SU Y, KRAVETS V G, WONG S L, et al. Impermeable barrier films and protective coatings based on reduced graphene oxide[J]. Nature Communications, 2014, 5(1): 4843. [18] 胡晓兰, 周川, 代少伟, 等. 氧化石墨烯改性不同表面性质的碳纤维/环氧树脂复合材料的微观形貌与动态热力学性能[J]. 复合材料学报, 2020, 37(5):1070-1080.HU Xiaolan, ZHOU Chuan, DAI Shaowei, et al. Micro-structures and dynamic thermal mechanical properties of graphene oxide modified carbon fiber/epoxy composites with different fiber surface properties[J]. Acta Materiae Compositae Sinica,2020,37(5):1070-1080(in Chinese). [19] ZHANG Q, HOU Q, HUANG G, et al. Removal of heavy metals in aquatic environment by graphene oxide compo-sites: A review[J]. Environmental Science and Pollution Research,2020,27(1):190-209. doi: 10.1007/s11356-019-06683-w [20] YUAN S, HUANG L, HUANG Z, et al. Continuous variation of lattice dimensions and pore sizes in metal-organic frameworks[J]. Journal of the American Chemical Society,2020,142(10):4732-4738. doi: 10.1021/jacs.9b13072 [21] PENG Y, ZHAO M, CHEN B, et al. Hybridization of MOFs and COFs: A new strategy for construction of MOF@COF core-shell hybrid materials[J]. Advanced Materials,2018,30(3):1705454. doi: 10.1002/adma.201705454 [22] LU Y Q, LIU W, LIU J T, et al. A review on 2D porous organic polymers for membrane-based separations: Processing and engineering of transport channels[J]. Advanced Membranes,2021,1:100014. [23] 刘超, 李茜, 郝丽芬, 等. MXene的功能化改性及其应用研究进展[J]. 复合材料学报, 2021, 38(4):1020-1028.LIU Chao, LI Xi, HAO Lifen, et al. Research progress of functional modification of MXene and its applications[J]. Acta Materiae Compositae Sinica,2021,38(4):1020-1028(in Chinese). [24] ZOU H, HE B, KUANG P, et al. Metal-organic framework-derived nickel-cobalt sulfide on ultrathin Mxene nanosheets for electrocatalytic oxygen evolution[J]. ACS Applied Materials & Interfaces,2018,10(26):22311-22319. [25] LIN Q, ZENG G, YAN G, et al. Self-cleaning photocatalytic MXene composite membrane for synergistically enhanced water treatment: Oil/water separation and dyes removal[J]. Chemical Engineering Journal,2022,427:131668. doi: 10.1016/j.cej.2021.131668 [26] JOHN Z C, SERGIO P, NIALL M, et al. Oxidation stability of colloidal two-dimensional titanium carbides (MXenes)[J]. Chemistry of Materials, 2017, 29(11): 4848-4856. [27] ZHANG X, YUAN X, JIANG L, et al. Powerful combination of 2D g-C3N4 and 2D nanomaterials for photocatalysis: Recent advances[J]. Chemical Engineering Journal,2020,390:124475. doi: 10.1016/j.cej.2020.124475 [28] ZENG G, HE Z, WAN T, et al. A self-cleaning photocatalytic composite membrane based on g-C3N4@MXene nanosheets for the removal of dyes and antibiotics from wastewater[J]. Separation and Purification Technology,2022,292:121037. doi: 10.1016/j.seppur.2022.121037 [29] GUSAIN R, KUMAR N, FOSSO-KANKEU E, et al. Efficient removal of Pb(II) and Cd(II) from industrial mine water by a hierarchical MoS2/SH-MWCNT nanocomposite[J]. ACS Omega,2019,4(9):13922-13935. doi: 10.1021/acsomega.9b01603 [30] 王艳芝, 张旺玺, 孙长红, 等. 氮化硼系列材料的合成制备及应用研究进展[J]. 陶瓷学报, 2018, 39(6): 661-671.WANG Yanzhi, ZHANG Wangxi, SUN Changhong, et al. The development of the applications and synthesis of boron nitride materials[J]. Journal of Ceramics, 2018, 39(6): 661-671(in Chinese). [31] VIKRANT Y, VAIBHAV K. Boron nitride: A promising material for proton exchange membranes for energy applications[J]. Nanoscale, 2019, 11(27): 12755-12773. [32] 崔世强, 阚洪敏, 张宁, 等. 六方氮化硼的制备应用及研究进展[J]. 功能材料, 2020, 51(8):8072-8077. doi: 10.3969/j.issn.1001-9731.2020.08.011CUI Shiqiang, KAN Hongmin, ZHANG Ning, et al. Preparation, application and research progress of hexagonal boron nitride[J]. Jorunal of Functional Materials,2020,51(8):8072-8077(in Chinese). doi: 10.3969/j.issn.1001-9731.2020.08.011 [33] 杨正芳, 张悦, 蔡金霄, 等. 层状双金属氢氧化物及其复合材料的制备与应用研究新进展[J]. 材料导报, 2021, 35(19):19062-19069. doi: 10.11896/cldb.19120181YANG Zhengfang, ZHANG Yue, CAI Jinxiao, et al. Recent advances in the preparation and application of layered double hydroxides and their composites[J]. Materials Reports,2021,35(19):19062-19069(in Chinese). doi: 10.11896/cldb.19120181 [34] LAIPAN M, YU J, ZHU R, et al. Functionalized layered double hydroxides for innovative applications[J]. Materials Horizons,2020,7(3):715-745. doi: 10.1039/C9MH01494B [35] MAO N, ZHOU C H, TONG D S, et al. Exfoliation of layered double hydroxide solids into functional nanosheets[J]. Applied Clay Science,2017,144:60-78. [36] 吕维扬, 孙继安, 姚玉元, 等. 层状双金属氢氧化物的控制合成及其在水处理中的应用[J]. 化学进展, 2020, 32(12):2049-2063. doi: 10.7536/PC200404LV Weiyang, SUN Ji'an, YAO Yuyuan, et al. Morphology control of layered double hydroxide and its application in water remediation[J]. Progress in Chemistry,2020,32(12):2049-2063(in Chinese). doi: 10.7536/PC200404 [37] SUN Y, WANG Q, WANG Y, et al. Recent advances in magnesium/lithium separation and lithium extraction technologies from salt lake brine[J]. Separation and Purification Technology,2021,256:117807. doi: 10.1016/j.seppur.2020.117807 [38] KONG Q, PREIS S, LI L, et al. Relations between metal ion characteristics and adsorption performance of graphene oxide: A comprehensive experimental and theoretical study[J]. Separation and Purification Technology, 2020, 232: 115956. [39] PENG W, LI H, LIU Y, et al. A review on heavy metal ions adsorption from water by graphene oxide and its compo-sites[J]. Journal of Molecular Liquids,2017,230:496-504. [40] CHENG M M, YAO C X, SU Y, et al. Synthesis of membrane-type graphene oxide immobilized manganese dioxide adsorbent and its adsorption behavior for lithium ion[J]. Chemosphere,2021,279:130487. [41] YANG X, WAN Y, ZHENG Y, et al. Surface functional groups of carbon-based adsorbents and their roles in the removal of heavy metals from aqueous solutions: A critical review[J]. Chemical Engineering Journal,2019,366:608-621. [42] LI X W, CHEN L, CHAO Y H, et al. Highly selective separation of lithium with hierarchical porous lithium-ion sieve microsphere derived from MXene[J]. Desalination,2022,537:115847. [43] PARK S H, KIM K, LIM J H, et al. Selective lithium and magnesium adsorption by phosphonate metal-organic framework-incorporated alginate hydrogel inspired from lithium adsorption characteristics of brown algae[J]. Separation and Purification Technology,2019,212:611-618. [44] ZHANG S, OU R, MA H, et al. Thermally regenerable metal-organic framework with high monovalent metal ion selectivity[J]. Chemical Engineering Journal,2021,405:127037. doi: 10.1016/j.cej.2020.127037 [45] ZHANG Y J, WANG L, ZHANG N N, et al. Adsorptive environmental applications of MXene nanomaterials: A review[J]. RSC Advances, 2018, 8(36): 2046-2069. [46] MA C, DU Y, DU B, et al. Investigation of an eco-friendly aerogel as a substrate for the immobilization of MoS2 nanoflowers for removal of mercury species from aqueous solutions[J]. Journal of Colloid and Interface Science,2018,525:251-259. doi: 10.1016/j.jcis.2018.04.079 [47] NUNES S P, CULFAZ-EMECEN P Z, RAMON G Z, et al. Thinking the future of membranes: Perspectives for advanced and new membrane materials and manufacturing processes[J]. Journal of Membrane Science,2020,598:117761. doi: 10.1016/j.memsci.2019.117761 [48] ZENG G, WEI K, ZHANG H, et al. Ultra-high oil-water separation membrane based on two-dimensional MXene (Ti3C2Tx) by co-incorporation of halloysite nanotubes and polydopamine[J]. Applied Clay Science,2021,211:106177. doi: 10.1016/j.clay.2021.106177 [49] TANSEL B. Significance of thermodynamic and physical characteristics on permeation of ions during membrane separation: Hydrated radius, hydration free energy and viscous effects[J]. Separation and Purification Technology,2012,86:119-126. [50] CHENG L, LIU G, ZHAO J, et al. Two-dimensional-material membranes: Manipulating the transport pathway for molecular separation[J]. Accounts of Materials Research,2021,2(2):114-128. doi: 10.1021/accountsmr.0c00092 [51] GUO Y, YING Y L, MAO Y Y, et al. polystyrene sulfonate threaded through a metal-organic framework membrane for fast and selective lithium-ion separation[J]. Angewandte Chemie, 2016, 128(48): 15120-15124. [52] XIE L, TANG J, QIN R, et al. Surface charge modification on 2D nanofluidic membrane for regulating ion transport[J]. Advanced Functional Materials,2022,33(4):2208959. [53] AN D, YANG L, WANG T, et al. Separation performance of graphene oxide membrane in aqueous solution[J]. Industrial & Engineering Chemistry Research,2016,55(17):4803-4810. [54] HADI A, MOHAMMAD Z, EHSAN H, et al. Heterogeneous asymmetric passable cavities within graphene oxide nanochannels for highly efficient lithium sieving[J]. Desalination,2022,538:115888. [55] WEI Z, QINGBO H, SONG L, et al. Graphene oxide membrane regulated by surface charges and interlayer channels for selective transport of monovalent ions over divalent ions[J]. Separation and Purification Technology,2022,291:120938. [56] HADI A, EHSAN H, WITHITA C, et al. Incorporation of natural lithium-ion trappers into graphene oxide nanosheets[J]. Advanced Materials Technologies, 2020, 6(10): 2000665. [57] XU T T, SHEHZAD M A, YU D B, et al. Highly cation permselective metal-organic framework membranes with leaf-like morphology[J]. ChemSusChem, 2019, 12(12): 2593-2597. [58] ZHANG H, HOU J, HU Y, et al. Ultrafast selective transport of alkali metal ions in metal organic frameworks with subnanometer pores[J]. Science Advances, 2018, 4(2): eaaq0066. [59] LIANG H, GUO Y, PENG X, et al. Light-gated cation-selective transport in metal-organic framework membranes[J]. Journal of Materials Chemistry A,2020,8(22):11399-11405. doi: 10.1039/D0TA02895A [60] SHENG F M, WU B, LI X Y, et al. Efficient ion sieving in covalent organic framework membranes with sub-2-nanometer channels[J]. Advanced Materials (Deerfield Beach, Fla. ), 2021, 33(44): e2104404. [61] BING S S, XIAN W P, CHEN S F, et al. Bio-inspired construction of ion conductive pathway in covalent organic framework membranes for efficient lithium extraction[J]. Matter, 2021, 4(6): 2027-2038. [62] ZENG G, LIU Y, LIN Q, et al. Constructing composite membranes from functionalized metal organic frameworks integrated MXene intended for ultrafast oil/water emulsion separation[J]. Separation and Purification Technology,2022,293:121052. doi: 10.1016/j.seppur.2022.121052 [63] WANG H, LU Z, WU Y, et al. A lamellar MXene (Ti3C2Tx)/PSS composite membrane for fast and selective lithium-ion separation[J]. Angewandte Chemie, 2021, 60(41): 22265-22269. [64] ZHANG C, MU Y, ZHANG W, et al. PVC-based hybrid membranes containing metal-organic frameworks for Li+/Mg2+ separation[J]. Journal of Membrane Science,2020,596:117724. doi: 10.1016/j.memsci.2019.117724 [65] BI Q Y, ZHANG C, LIU J D, et al. Positively charged zwitterion-carbon nitride functionalized nanofiltration membranes with excellent separation performance of Mg2+/Li+ and good antifouling properties[J]. Separation and Purification Technology, 2020, 257: 117959. -





 下载:
下载: