Classification of conductive composite hydrogels and their application in flexible wearable devices
-
摘要: 近年来,基于水凝胶的导电材料及其作为柔性可穿戴设备的应用引起了人们的广泛关注。柔性可穿戴设备不仅可以采集人体生理信号用于远程健康监测,还在人机界面、软机器人等方面展示出巨大的应用潜力。导电水凝胶所具有的良好导电性、高延伸性、可调柔韧性、生物兼容性和多重刺激响应性等优点使其成为制备柔性可穿戴设备的理想材料。到目前为止,各种导电材料被广泛用于制作导电复合水凝胶。本文根据导电材料对导电复合水凝胶进行分类,包括离子导电水凝胶(基于盐离子、离子液体、聚电解质等导电物质)、电子导电水凝胶(基于导电聚合物基、碳材料、MXene和金属等导电物质)两大类,并介绍了导电水凝胶在人体运动监测、健康监测、人机界面等柔性可穿戴设备中的应用进展。Abstract: In recent years, hydrogel-based conductive materials as flexible wearable devices have attracted wide attention. Flexible wearable devices can not only be used to collect physiological signals of human body for remote health monitoring, but also show great application potential in man-machine interface, soft robot and other aspects. Conductive hydrogels with good electrical conductivity, high extensibility, adjustable flexibility, biocompatibility and multiple stimulus response become an ideal material for flexible wearable devices. Up to now, various conductive materials have been widely used to prepare conductive hydrogels. In this paper, conductive composite hydrogels are classified according to conductive materials, including ionic conductive hydrogels (based on salt ions, ionic liquids, polyelectrolytes) and electronic conductive hydrogels (based on conductive polymers, carbon materials, MXene and metal), and then introduce the application progress of conductive hydrogels in flexible wearable devices such as human movement monitoring, health monitoring and human-machine interface.
-
Key words:
- hydrogel /
- conductivity /
- wearable devices /
- intelligent sensor /
- human motion monitoring
-
图 1 相变可调凝胶 (PTTGC凝胶)的设计、制备和性能:(a) PTTGC凝胶的合成与构成;(b) PTTGC凝胶内部成分之间的强氢键和静电相互作用[35]
Figure 1. Design, preparation, and properties of the phase-transition-tunable gel (PTTGC gel): (a) Synthesis and formation of a PTTGC gel; (b) Strong hydrogen bonds and electrostatic interactions between internal components of PTTGC gel[35]
PAAm—Polyacrylamide; GC—Clay composite
图 2 (a) 通过溶胶-凝胶法从乙烯基三乙氧基硅烷(VTES)合成乙烯基官能化二氧化硅纳米粒子(VSNPs);(b) 甲基丙烯酸磺基甜菜碱 (SBMA)两性离子单体的合成;(c) P(AM-SBMA-AMPS)-SiO2水凝胶的合成示意图[38]
Figure 2. (a) Synthesis of vinyl-functionalized silica nanoparticles (VSNPs) from vinyltriethoxysilane (VTES) by a sol-gel process; (b) Synthesis of sulfobetaine methacrylate (SBMA) zwitterionic monomer; (c) Schematic fabrication of P(AM-SBMA-AMPS)-SiO2 hydrogels[38]
AM—Acrylamide; AMPS—2-acrylamido-2-methyl-1-propanesulfonic acid
图 4 制备导电还原氧化石墨烯/甲基丙烯酰化明胶(r(GO/GelMA))水凝胶管的示意图,该管道由GO/GelMA复合水凝胶聚合并随后温和化学还原成r(GO/GelMA)[49]
Figure 4. Schematic outline of the production of a conductive reduced GO/GelMA (r(GO/GelMA)) hydrogel conduit consisting of polymerization of GO/GelMA compositehydrogel and its subsequent mild chemical reduction to r(GO/GelMA)[49]
图 5 碳纳米管/疏水缔合聚丙烯酰胺(CNTs/HAPAAm) 水凝胶的制备过程示意图[51]
Figure 5. Schematic illustration of the preparation process for the CNTs/hydrophobically associated polyacrylamide (CNTs/HAPAAm) hydrogel[51]
APS—Ammonium persulfate; TEMED—N,N,N',N'-tetramethylethylenediamine; SDS—Sodium dodecyl sulfate; LMA—Lauryl methacrylate; AAm—Acrylamide
图 9 (a) 大面积水凝胶薄膜的照片、SEM图像及肘部运动过程中的电流-时间曲线[64];(b) 具有坚固界面的结构凝胶复合材料(SGC)的制造及水下实时脉冲检测器[66]
Figure 9. (a) Photograph and SEM image of large-area hydrogel film, and current-time curve during elbow motion[64]; (b) Fabrication of structural gel composite (SGC) with robust interface and an underwater real-time pulse detector[66]
ΔI—Current variation under certain pressure; I0—Current without pressure; ΔR—Relative resistance change; R0—Resistance without applied strain
图 10 (a) 两性离子导电水凝胶薄膜的制备;(b) 表皮触控面板的结构和操作。(表皮集成触控面板示意图。通过表皮触摸面板玩愤怒的小鸟的操作照片:当手指接触面板时,弹弓被弹弓捕获;当手指离开面板时,弹弓被释放,小鸟起飞;目标被小鸟打断。使用面板下棋的照片记录:手指点击触摸面板上的棋子位置时选择了一个棋子;棋子选择了棋盘的另一个位置,棋子移动;棋子移动到了目标位置)[69]
Figure 10. (a) Preparation of zwitterionic conducting hydrogel films; (b) Structure and operation of an epidermal touch panel. (Schematic illustration of an epidermal integrated touch panel. Operational photographs of playing Angry Birds by an epidermal touch panel. An Angry Bird was captured by the slingshot when a finger touched the panel. The slingshot was released when the finger left the panel, and the bird took off, the target was broken by the bird. Photographs record of playing Chess using the panel. A chess piece was selected when the finger clicked the piece position on the touch panel. Another position of the chessboard was chosen for the chess piece, and the chess piece shifted. The chess piece was shift to the target position)[69]
PDMS—Poly(dimethylsiloxane); VHB—Dielectric elastomer very high bond
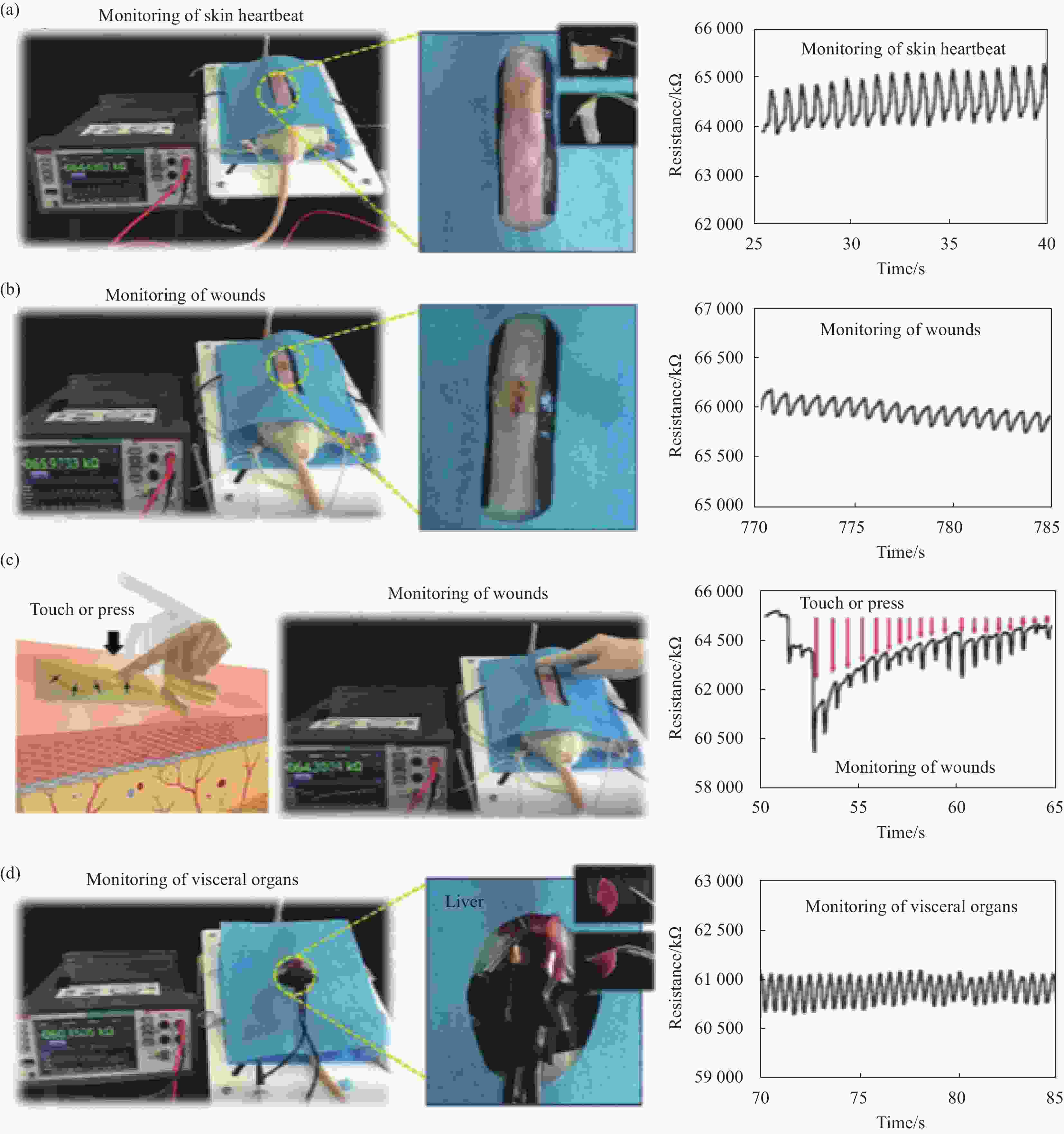
图 11 实时人体健康监测:(a) 监测麻醉大鼠心脏的照片并记录阻力随时间变化以监测心跳的变化;(b) 监测麻醉大鼠腹部创伤的照片并记录阻力随时间的变化以监测腹部伤口的变化;(c) 监测腹部伤口触摸或按压的照片并记录的阻力随时间变化,用于监测腹部伤口触摸或按压的变化(箭头表示当时短暂按压水凝胶);(d) 监测内脏器官(肝脏)的照片并记录阻力随时间变化以监测肝脏的变化[72]
Figure 11. Real-time human health monitoring: (a) Photographs of monitoring the hearts of anesthetized rats and recorded resistance with time to monitor changes in heartbeats; (b) Photographs of monitoring the abdominal wounds in anesthetized rats and recorded resistance with time to monitor changes in abdominal wounds; (c) Photographs of monitoring the touching or pressing of abdominal wounds and recorded resistance with time for monitoring changes in touching or pressing on abdominal wounds (Arrow indicates a brief press on the hydrogel at that time point); (d) Photographs of monitoring a visceral organ (liver) and recorded resistance with time for monitoring changes in the liver[72]
表 1 基于各种导电材料的导电水凝胶的综合性能比较
Table 1. Comprehensive performance comparison of conductive hydrogels based on various conductive materials
Conductive
hydrogelConductive
materialComponents of
polymer networksConductivity/
(S·m−1)Tensile
strain/%Temperature
rangeRef. Saline solution AlCl3 CMCP(AA-AM) 2 963 — [34] ZnCl2 Gelatin/PAAm-GC 1.68 — −60-60℃ [35] CaCl2 PAA/ISP 1.86 890 −20℃-RT [36] IL [BMIM]Cl P(AM-HEA)/SSS 8.4 1310 −25-75℃ [37] Polyelectrolyte SBMA P(AM-AMPS)-SiO2 3.4 1000 −20-100℃ [38] Conductive Polymer PANI PNIPAm/AMPS ~0.027 — — [43] PPy GelMA-PAM ~0.071 — — [44] PANI PVA 20.5 100 — [45] PANI ANF-PVA 2.02 140 — [46] Carbon-based materials GO GelMA ~0.87 — — [49] CNT PS/TC — 83.2 — [50] CNT HAPAAm ~1.7 3000 — [51] MXene MXene PAA-ACC — 450 — [58] MXene Chitosan 0.04 1900 — [57] MXene PAA/PAM/TA — 560 — [59] Metal/LMs Ag/TA@CNCs PVA 4.61 4000 — [60] — Au/SiO2 GelMA — ~70 — [61] — AgNW PNIPAM ~0.025 300 — [62] — LMPs PVA 0.375 — — [63] Notes: IL—Ionic liquid; LMs—Liquid metal; [BMIM]Cl—1-butyl-3-methylimidazolium chloride; PPy—Polypyrrole; TA—Tannic acid; AA—Acrylic acid; CMC—Carboxymethyl cellulose; ISP—Ice structuring proteins; HEA—2-hydroxyethyl acrylate; SSS—Sodium p-styrene sulfonate; ANF—Aramid nanofiber; PS—Pigskin; TC—Oxidized-nanocellulose; RT—Room temperature. -
[1] CHENG Y M, WANG K, XU H, et al. Recent developments in sensors for wearable device applications[J]. Analytical and Bioanalytical Chemistry,2021,413(24):6037-6057. doi: 10.1007/s00216-021-03602-2 [2] 鲁燕燕, 谢红珍. 可穿戴设备在医疗领域的应用[J]. 中国医疗器械杂志, 2017, 41(3):213-215. doi: 10.3969/j.issn.1671-7104.2017.03.015LU Yanyan, XIE Hongzhen. Application of wearable devices in medical field[J]. Chinese Journal of Medical Instrumentation,2017,41(3):213-215(in Chinese). doi: 10.3969/j.issn.1671-7104.2017.03.015 [3] WANG L L, LOU Z, JIANG K, et al. Bio-multifunctional smart wearable sensors for medical devices[J]. Advanced Intelligent Systems, 2019, 1(5): 1900040. [4] GAO W, OTA H, KIRIYA D, et al. Flexible electronics toward wearable sensing[J]. Accounts of Chemical Research,2019,52(3):523-533. doi: 10.1021/acs.accounts.8b00500 [5] KENRY, YEO J C, LIM C T. Emerging flexible and wearable physical sensing platforms for healthcare and biomedical applications[J]. Microsystems & Nanoengineering,2016,2:16043. doi: 10.1038/micronano.2016.43 [6] GUESS M, ZAVANELLI N, YEO W H. Recent advances in materials and flexible sensors for arrhythmia detection[J]. Materials (Basel), 2022, 15(3): 724. [7] LA T G, LE L H. Flexible and wearable ultrasound device for medical applications: A review on materials, structural designs, and current challenges[J]. Advanced Materials Technologies, 2022, 7(3): 2100798. [8] SERVATI A, ZOU L, WANG Z J, et al. Novel flexible wearable sensor materials and signal processing for vital sign and human activity monitoring [J]. Sensors (Basel), 2017, 17(7): 1622. [9] ANSAR R, SAQIB S, MUKHTAR A, et al. Challenges and recent trends with the development of hydrogel fiber for biomedical applications[J]. Chemosphere,2022,287:131956. doi: 10.1016/j.chemosphere.2021.131956 [10] XU C, XU Y, YANG M, et al. Black-phosphorus-incorporated hydrogel as a conductive and biodegradable platform for enhancement of the neural differentiation of mesenchymal stem cells[J]. Advanced Functional Materials, 2020, 30(39): 2000177. [11] XU Y, CUI M Y, PATSIS P A, et al. Reversibly assembled electroconductive hydrogel via a host-guest interaction for 3D cell culture[J]. ACS Applied Materials & Interfaces,2019,11(8):7715-7724. [12] HASANI-SADRABADI M M, SARRION P, POURAGHAEI S, et al. An engineered cell-laden adhesive hydrogel promotes craniofacial bone tissue regeneration in rats[J]. Science Translational Medicine, 2020, 12(534): eaay6853. [13] FAN L, HE Z J, PENG X L, et al. Injectable, intrinsically antibacterial conductive hydrogels with self-healing and pH stimulus responsiveness for epidermal sensors and wound healing[J]. ACS Applied Materials & Interfaces,2021,13(45):53541-53552. [14] ZHU Y T, ZENG Q, ZHANG Q, et al. Temperature/near-infrared light-responsive conductive hydrogels for controlled drug release and real-time monitoring[J]. Nanoscale,2020,12(16):8679-8686. doi: 10.1039/D0NR01736A [15] DHIBAR S, DEY A, GHOSH D, et al. Mechanically tuned molybdenum dichalcogenides (MoS2 and MoSe2) dispersed supramolecular hydrogel scaffolds[J]. Journal of Molecular Liquids,2019,276:184-193. doi: 10.1016/j.molliq.2018.11.159 [16] ZHOU Y, WAN C J, YANG Y S, et al. Highly stretchable, elastic, and ionic conductive hydrogel for artificial soft electronics[J]. Advanced Functional Materials, 2019, 29(1): 1806220. [17] WANG Y F, TEBYETEKERWA M, LIU Y, et al. Extremely stretchable and healable ionic conductive hydrogels fabricated by surface competitive coordination for human-motion detection[J]. Chemical Engineering Journal,2021,420:127637. doi: 10.1016/j.cej.2020.127637 [18] SHI Y, PAN L J, LIU B R, et al. Nanostructured conductive polypyrrole hydrogels as high-performance, flexible supercapacitor electrodes[J]. Journal of Materials Chemistry A,2014,2(17):6086-6091. doi: 10.1039/C4TA00484A [19] SU G H, YIN S Y, GUO Y H, et al. Balancing the mechanical, electronic, and self-healing properties in conductive self-healing hydrogel for wearable sensor applications[J]. Materials Horizons,2021,8(6):1795-1804. doi: 10.1039/D1MH00085C [20] YAN T, WANG Z, PAN Z J. Flexible strain sensors fabricated using carbon-based nanomaterials: A review[J]. Current Opinion in Solid State & Materials Science,2018,22(6):213-228. [21] ZHOU C, ZHAO X H, XIONG Y S, et al. A review of etching methods of mxene and applications of mxene conductive hydrogels[J]. European Polymer Journal,2022,167:111063. doi: 10.1016/j.eurpolymj.2022.111063 [22] MIN J H, PATEL M, KOH W G. Incorporation of conductive materials into hydrogels for tissue engineering applications[J]. Polymers, 2018, 10(10): 1078. [23] XIA S, SONG S X, JIA F, et al. A flexible, adhesive and self-healable hydrogel-based wearable strain sensor for human motion and physiological signal monitoring[J]. Journal of Materials Chemistry B,2019,7(30):4638-4648. doi: 10.1039/C9TB01039D [24] PAN M F, WU M, SHUI T, et al. Highly stretchable, elastic, antimicrobial conductive hydrogels with environment-adaptive adhesive property for health monitoring[J]. Journal of Colloid and Interface Science,2022,622:612-624. doi: 10.1016/j.jcis.2022.04.119 [25] HUANG H L, HAN L, FU X B, et al. Multiple stimuli responsive and identifiable zwitterionic ionic conductive hydrogel for bionic electronic skin[J]. Advanced Electronic Materials, 2020, 6(7): 2000239. [26] HSIAO L Y, JING L, LI K R, et al. Carbon nanotube-integrated conductive hydrogels as multifunctional robotic skin[J]. Carbon,2020,161:784-793. doi: 10.1016/j.carbon.2020.01.109 [27] DENG Z X, YU R, GUO B L. Stimuli-responsive conductive hydrogels: Design, properties, and applications[J]. Materials Chemistry Frontiers,2021,5(5):2092-2123. doi: 10.1039/D0QM00868K [28] ZHANG W R, JIANG H Y, CHANG Z G, et al. Recent achievements in self-healing materials based on ionic liquids: A review[J]. Journal of Materials Science,2020,55(28):13543-13558. doi: 10.1007/s10853-020-04981-0 [29] WANASINGHA N, DORISHETTY P, DUTTA N K, et al. Polyelectrolyte gels: Fundamentals, fabrication and applications[J]. Gels, 2021, 7(3): 148. [30] HUANG H L, HAN L, LI J F, et al. Super-stretchable, elastic and recoverable ionic conductive hydrogel for wireless wearable, stretchable sensor[J]. Journal of Materials Chemistry A,2020,8(20):10291-10300. doi: 10.1039/D0TA02902E [31] ZHANG W, WU B H, SUN S T, et al. Skin-like mechanoresponsive self-healing ionic elastomer from supramolecular zwitterionic network[J]. Nature Communications, 2021, 12(1): 4082. [32] GAN D L, HAN L, WANG M H, et al. Conductive and tough hydrogels based on biopolymer molecular templates for controlling in situ formation of polypyrrole nanorods[J]. ACS Applied Materials & Interfaces,2018,10(42):36218-36228. [33] HAN L, LIU K Z, WANG M H, et al. Mussel-inspired adhesive and conductive hydrogel with long-lasting moisture and extreme temperature tolerance[J]. Advanced Functional Materials, 2018, 28(3): 1704195. [34] CHEN Z, LIU J, CHEN Y J, et al. Multiple-stimuli-responsive and cellulose conductive ionic hydrogel for smart wearable devices and thermal actuators[J]. ACS Applied Materials & Interfaces,2021,13(1):1353-1366. doi: 10.1021/acsami.0c16719 [35] ZHU Y, LIN L, CHEN Y, et al. Extreme temperature-tolerant conductive gel with antibacterial activity for flexible dual-response sensors[J]. ACS Applied Materials & Interfaces,2020,12(50):56470-56479. doi: 10.1021/acsami.0c17242 [36] LU C Y, QIU J H, ZHAO W, et al. Low-temperature adaptive conductive hydrogel based on ice structuring proteins/CaCl2 anti-freeze system as wearable strain and temperature sensor[J]. International Journal of Biological Macromolecules,2021,188:534-541. doi: 10.1016/j.ijbiomac.2021.08.060 [37] ZHANG X Y, ZHANG G, HUANG X H, et al. Antifreezing and nondrying sensors of ionic hydrogels with a double-layer structure for highly sensitive motion monitoring[J]. ACS Applied Materials & Interfaces,2022,14(26):30256-30267. doi: 10.1021/acsami.2c08589 [38] DIAO W J, WU L L, MA X F, et al. Reversibly highly stretchable and self-healable zwitterion-containing polyelectrolyte hydrogel with high ionic conductivity for high-performance flexible and cold-resistant supercapacitor[J]. Journal of Applied Polymer Science, 2020, 137(34): 48995. [39] GAO Z J, LI Y F, SHANG X L, et al. Bio-inspired adhesive and self-healing hydrogels as flexible strain sensors for monitoring human activities[J]. Materials Science and Engineering: C,2020,106:110168. doi: 10.1016/j.msec.2019.110168 [40] FANG Y L, XU J H, GAO F, et al. Self-healable and recyclable polyurethane-polyaniline hydrogel toward flexible strain sensor[J]. Composites Part B: Engineering,2021,219:108965. doi: 10.1016/j.compositesb.2021.108965 [41] FU F F, WANG J L, YU J. Interpenetrating PAA-PEDOT conductive hydrogels for flexible skin sensors[J]. Journal of Materials Chemistry C,2021,9(35):11794-11800. doi: 10.1039/D1TC01578H [42] WANG Z W, CONG Y, FU J. Stretchable and tough conductive hydrogels for flexible pressure and strain sensors[J]. Journal of Materials Chemistry B,2020,8(16):3437-3459. doi: 10.1039/C9TB02570G [43] RIVERO R E, MOLINA M A, RIVAROLA C R, et al. Pressure and microwave sensors/actuators based on smart hydrogel/conductive polymer nanocomposite[J]. Sensors and Actuators B: Chemical,2014,190:270-278. doi: 10.1016/j.snb.2013.08.054 [44] HU S Q, ZHOU L, TU L J, et al. Elastomeric conductive hybrid hydrogels with continuous conductive networks[J]. Journal of Materials Chemistry B,2019,7(15):2389-2397. doi: 10.1039/C9TB00173E [45] LI L, ZHANG Y, LU H Y, et al. Cryopolymerization enables anisotropic polyaniline hybrid hydrogels with superelasticity and highly deformation-tolerant electrochemical energy storage[J]. Nature Communications,2020,11(1):62. doi: 10.1038/s41467-019-13959-9 [46] WANG J, LIN Y K, MOHAMED A, et al. High strength and flexible aramid nanofiber conductive hydrogels for wearable strain sensors[J]. Journal of Materials Chemistry C,2021,9(2):575-583. doi: 10.1039/D0TC02983A [47] ARNDT C, HAUCK M, WACKER I, et al. Microengineered hollow graphene tube systems generate conductive hydrogels with extremely low filler concentration[J]. Nano Letters,2021,21(8):3690-3697. doi: 10.1021/acs.nanolett.0c04375 [48] LEE I J, KIM K E, MA H S, et al. Preparation of carbon nanotube fiber/conductive materials composites and their electrical properties[J]. Textile Science and Engineering,2019,56(1):35-40. [49] PARK J, JEON J, KIM B, et al. Electrically conductive hydrogel nerve guidance conduits for peripheral nerve regeneration[J]. Advanced Functional Materials, 2020, 30(39): 2003759. [50] WU Y, SUN S M, GENG A B, et al. Using TEMPO-oxidized-nanocellulose stabilized carbon nanotubes to make pigskin hydrogel conductive as flexible sensor and supercapacitor electrode: Inspired from a Chinese cuisine[J]. Composites Science and Technology,2020,196:108226. doi: 10.1016/j.compscitech.2020.108226 [51] QIN Z H, SUN X, YU Q Y, et al. Carbon nanotubes/hydrophobically associated hydrogels as ultrastretchable, highly sensitive, stable strain, and pressure sensors[J]. ACS Applied Materials & Interfaces,2020,12(4):4944-4953. doi: 10.1021/acsami.9b21659 [52] ZHOU H, WANG F Q, WANG Y W, et al. Study on contact angles and surface energy of MXene films[J]. RSC Advances,2021,11(10):5512-5520. doi: 10.1039/D0RA09125A [53] WANG Q H, PAN X F, WANG X P, et al. Fabrication strategies and application fields of novel 2D Ti3C2Tx (MXene) composite hydrogels: A mini-review [J]. Ceramics International, 2021, 47(4): 4398-4403. [54] CHANG T H, ZHANG T R, YANG H T, et al. Controlled crumpling of two-dimensional titanium carbide (MXene) for highly stretchable, bendable, efficient supercapacitors[J]. ACS Nano,2018,12(8):8048-8059. doi: 10.1021/acsnano.8b02908 [55] LIN X P, LI Z J, QIU J M, et al. Fascinating MXene nanomaterials: Emerging opportunities in the biomedical field[J]. Biomaterials Science,2021,9(16):5397-5431. doi: 10.1039/D1BM00430A [56] LUAN H X, ZHANG D Z, XU Z Y, et al. Mxene-based composite double-network multifunctional hydrogels as highly sensitive strain sensors[J]. Journal of Materials Chemistry C, 2022,10(19): 7604-7613. [57] LIU Y Q, XU D R, DING Y, et al. A conductive polyacrylamide hydrogel enabled by dispersion-enhanced MXene@chitosan assembly for highly stretchable and sensitive wearable skin[J]. Journal of Materials Chemistry B,2021,9(42):8862-8870. doi: 10.1039/D1TB01798E [58] LI X B, HE L Z, LI Y F, et al. Healable, degradable, and conductive MXene nanocomposite hydrogel for multifunctional epidermal sensors[J]. ACS Nano,2021,15(4):7765-7773. doi: 10.1021/acsnano.1c01751 [59] QIN M, YUAN W F, ZHANG X M, et al. Preparation of PAA/PAM/MXene/TA hydrogel with antioxidant, healable ability as strain sensor[J]. Colloids and Surfaces B: Biointerfaces,2022,214:112482. doi: 10.1016/j.colsurfb.2022.112482 [60] LIN F C, WANG Z, SHEN Y P, et al. Natural skin-inspired versatile cellulose biomimetic hydrogels[J]. Journal of Materials Chemistry A,2019,7(46):26442-26455. doi: 10.1039/C9TA10502F [61] MAHARJAN B, KUMAR D, AWASTHI G P, et al. Synthesis and characterization of gold/silica hybrid nanoparticles incorporated gelatin methacrylate conductive hydrogels for H9C2 cardiac cell compatibility study[J]. Composites Part B: Engineering,2019,177:107415. doi: 10.1016/j.compositesb.2019.107415 [62] WANG T J, SONG J, LIU R J, et al. Motion detecting, temperature alarming, and wireless wearable bioelectronics based on intrinsically antibacterial conductive hydrogels[J]. ACS Applied Materials & Interfaces,2022,14(12):14596-14606. doi: 10.1021/acsami.2c00713 [63] LIAO M H, LIAO H, YE J J, et al. Polyvinyl alcohol-stabilized liquid metal hydrogel for wearable transient epidermal sensors[J]. ACS Applied Materials & Interfaces,2019,11(50):47358-47364. doi: 10.1021/acsami.9b16675 [64] GE G, ZHANG Y Z, SHAO J J, et al. Stretchable, transparent, and self-patterned hydrogel-based pressure sensor for human motions detection[J]. Advanced Functional Materials, 2018, 28(32): 1802576. [65] HE X L, DONG J, ZHANG X N, et al. Self-healing, anti-fatigue, antimicrobial ionic conductive hydrogels based on choline-amino acid polyionic liquids for multi-functional sensors[J]. Chemical Engineering Journal,2022,435:135168. [66] WANG Z B, ZHOU H H, LIU D, et al. A structural gel composite enabled robust underwater mechanosensing strategy with high sensitivity[J]. Advanced Functional Materials, 2022, 32(25): 2201396. [67] HANG C Z, ZHAO X F, XI S Y, et al. Highly stretchable and self-healing strain sensors for motion detection in wireless human-machine interface[J]. Nano Energy,2020,76:105064. doi: 10.1016/j.nanoen.2020.105064 [68] GAO Y, WANG Y R, XIA S, et al. An environment-stable hydrogel with skin-matchable performance for human-machine interface[J]. Science China Materials,2021,64(9):2313-2324. doi: 10.1007/s40843-020-1624-y [69] GUO X K, YANG F, SUN X L, et al. Anti-freezing self-adhesive self-healing degradable touch panel with ultra-stretchable performance based on transparent triboelectric nanogenerators[J]. Advanced Functional Materials, 2022, 32(31): 2201230. [70] ZHANG Q, LIU X, DUAN L J, et al. Nucleotide-driven skin-attachable hydrogels toward visual human-machine interfaces[J]. Journal of Materials Chemistry A,2020,8(8):4515-4523. doi: 10.1039/C9TA14224J [71] LIU X, CHEN X F, CHI X Y, et al. Biomimetic integration of tough polymer elastomer with conductive hydrogel for highly stretchable, flexible electronic[J]. Nano Energy,2022,92:106735. doi: 10.1016/j.nanoen.2021.106735 [72] LEI H, ZHAO J, MA X X, et al. Antibacterial dual network hydrogels for sensing and human health monitoring[J]. Advanced Healthcare Materials,2021,10(21):2101089. doi: 10.1002/adhm.202101089 -






 下载:
下载: