Flame retardant effect of phosphotungstic acid intercalated ZnAl layered double hydroxides and intumescent flame retardant on epoxy-polyamide resin
-
摘要: 采用[PW12O40]3−离子柱撑插层共沉淀法合成的ZnAl硝酸根(NO3-ZnAl)层状双金属氢氧化物(LDHs),制备了PW12O40-ZnAl LDHs,并利用XRD、FTIR、电感耦合等离子体(ICP)、SEM等进行组成和结构的表征。将NO3-ZnAl LDHs和PW12O40-ZnAl LDHs分别与含聚磷酸铵、三聚氰胺、季戊四醇的膨胀阻燃剂(IFRs)复合阻燃环氧-聚酰胺树脂(EP-PA),采用TGA、背温实验和锥形量热实验评价不同ZnAl LDHs与IFRs复合阻燃EP-PA的热及烟气的释放规律。TGA结果表明,PW12O40-ZnAl-IFRs/(EP-PA)复合材料的最大降解速率最小,残炭率最高,说明PW12O40-ZnAl LDHs提高了IFRs/(EP-PA)复合材料高温下的抗氧化能力。背温实验表明,相同热辐射强度下,PW12O40-ZnAl-IFRs/(EP-PA)复合材料的背温达到200℃和300℃用时最长,具有最低的背温升温速率,说明PW12O40-ZnAl LDHs使IFRs/(EP-PA)复合材料耐火能力明显增强。从锥形量热实验数据可知,PW12O40-ZnAl-IFRs使PW12O40-ZnAl-IFRs/(EP-PA)复合材料具有最低的热释放速率峰值(PHRR)、平均热释放速率(MHRR)、平均有效燃烧热(MEHC)和总热释放量(THR),其火势增长指数(FGI)仅为IFRs/(EP-PA)复合材料的14.5%,烟释放总量(TSP)比NO3-ZnAl-IFRs/(EP-PA)复合材料减少了27.6%,比IFRs/(EP-PA)复合材料减少了55.3%。说明PW12O40-ZnAl-IFRs比NO3-ZnAl-IFRs更能有效地减少EP-PA的热量释放,抑制烟气生成。
-
关键词:
- ZnAl层状双金属氢氧化物 /
- 插层改性 /
- 环氧-聚酰胺树脂 /
- 阻燃 /
- 背温速率
Abstract: The PW12O40-ZnAl layered double hydroxides(LDHs) was prepared by using [PW12O40]3− ion pillared intercalation NO3-ZnAl LDHs. The composition and structure were analyzed by XRD, FTIR, inductively coupled plasma(ICP) and SEM. The flame retardant epoxy-polyamide resin(EP-PA) were prepared by NO3-ZnAl LDHs or PW12O40-ZnAl LDHs compound with intumescent flame retardants(IFRs) containing ammonium polyphosphate, melamine, pentaerythritol. The heat and smoke release rules of different ZnAl LDHs-IFRs flame retardant EP-PA were evaluated by back temperature experiment and cone calorimetry experiment. TGA result shows that the maximum degradation rate of PW12O40-ZnAl-IFRs/(EP-PA) composite is the lowest, and the carbon residue rate is the highest, which indicate that PW12O40-ZnAl LDHs improve the oxidation resistance of PW12O40-ZnAl-IFRs/(EP-PA) composite at high temperature. The back temperature experiment results show that under the same heat radiation intensity, the back temperature of PW12O40-ZnAl-IFRs/(EP-PA) composite reaches to 200℃ and 300℃ with the longest time and the lowest rate of back temperature rise. The results show that PW12O40-ZnAl LDHs can obviously enhance the fire resistance of EP-PA. From cone calorimetry experimental data, it can be seen that PW12O40-ZnAl-IFRs makes PW12O40-ZnAl-IFRs/(EP-PA) composite have the lowest peak of heat release rate(PHRR), mean heat release rate(MHRR), mean effective heat of combustion(MEHC) and total heat release(THR). Its fire growth index (FGI) is only 14.5% of IFRs/(EP-PA) composite, and the total smoke production (TSP) is 27.6% lower than NO3-ZnAl-IFRs/(EP-PA) composite and 55.3% lower than IFRs/(EP-PA) composite. The results suggest that PW12O40-ZnAl-IFRs is more effective than NO3-ZnAl-IFRs in reducing the heat release and inhibiting the generation of flue gas. -
表 1 层状双金属氢氧化物-膨胀阻燃剂/(环氧-聚酰胺树脂)(LDHs-IFRs/(EP-PA))复合材料配比
Table 1. Proportion of layered double hydroxides- intumescent flame retardants/(epoxy-polyamide resin) (LDHs-IFRs/(EP-PA)) composites
Sample EP/g PA/g IFRs/g LDHs/g NO3-ZnAl PW12O40-ZnAl IFRs/(EP-PA) 10 10 5.00 0 0 NO3-ZnAl-IFRs/(EP-PA) 10 10 3.75 1.25 0 PW12O40-ZnAl-IFRs/(EP-PA) 10 10 3.75 0 1.25 表 2 NO3-ZnAl LDHs 和 PW12O40-ZnAl LDHs的XRD衍射参数
Table 2. XRD parameters of NO3-ZnAl LDHs and PW12O40-ZnAl LDHs
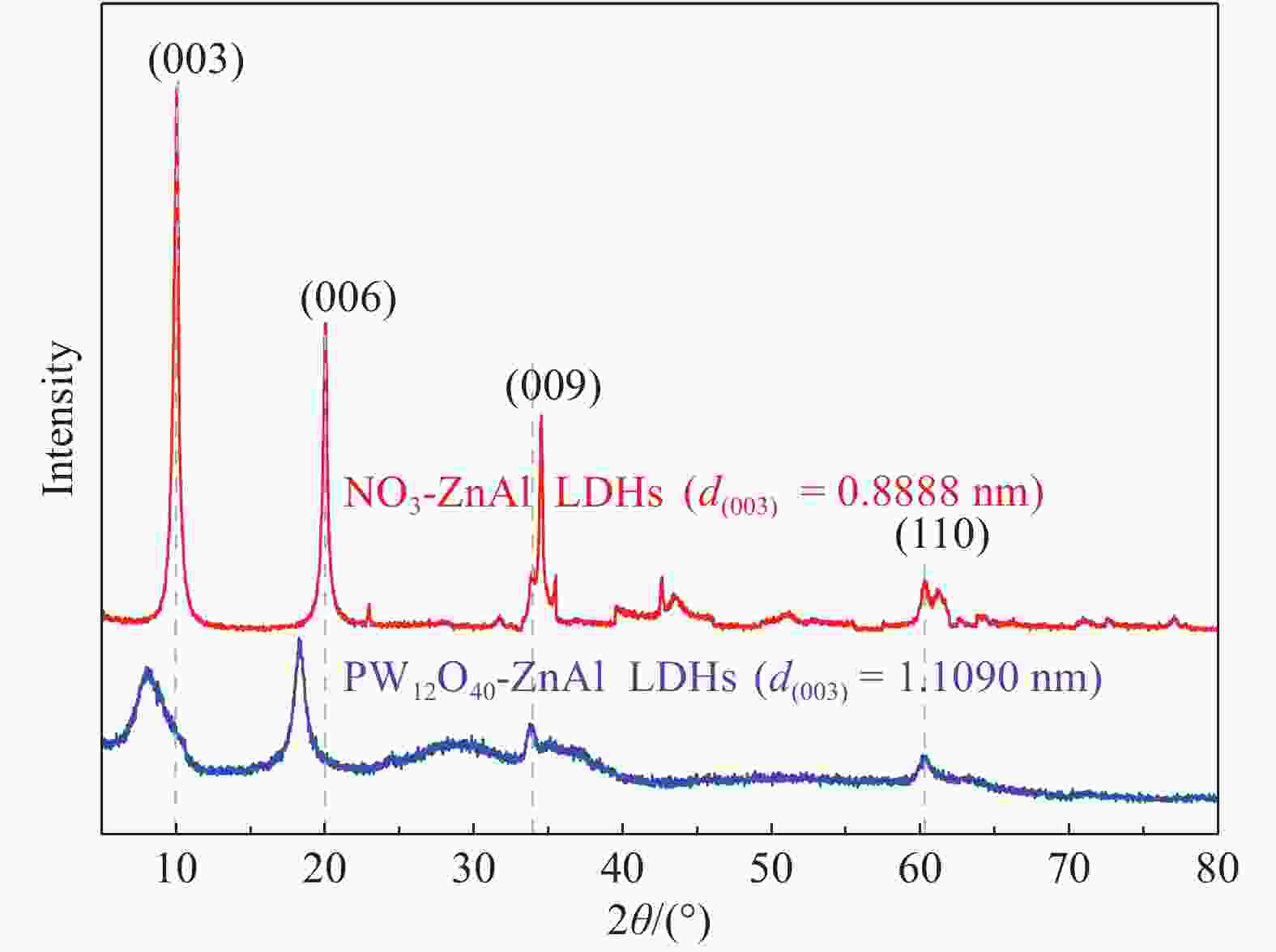
Crystal face NO3-ZnAl LDHs PW12O40-ZnAl LDHs 2θ/(°) d/nm 2θ/(°) d/nm (003) 9.94 0.8888 7.96 1.1090 (006) 19.92 0.4452 18.24 0.4858 (009) 33.76 0.2651 33.36 0.2682 (110) 60.30 0.1533 60.40 0.1531 a/nm 0.3066 0.3061 c/nm 2.6660 3.3270 Notes:d—Interplanar distance; θ—Angle of deviation; a=2d(110); c=3d( 003). 表 3 NO3-ZnAl LDHs和PW12O40-ZnAl LDHs的元素分析结果
Table 3. Elemental analysis results of NO3-ZnAl LDHs and PW12O40-ZnAl LDHs
Sample Zn/wt% Al/wt% W/wt% Chemical formula NO3-ZnAl LDHs 5.57 42.80 – ${\rm{Z}}{{\rm{n}}_{{\rm{0}}{\rm{.76}}}}{\rm{A}}{{\rm{l}}_{{\rm{0}}{\rm{.24}}}}{{\rm{(OH)}}_{\rm{2}}}{{\rm{(N}}{{\rm{O}}_{\rm{3}}}{\rm{)}}_{{\rm{0}}{\rm{.24}}}} \cdot {\rm{0}}{\rm{.59}}{{\rm{H}}_{\rm{2}}}{\rm{O}}$ PW12O40-ZnAl LDHs 3.47 22.50 37.13 ${\rm{Z}}{{\rm{n}}_{{\rm{0}}{\rm{.73}}}}{\rm{A}}{{\rm{l}}_{{\rm{0}}{\rm{.27}}}}{{\rm{(OH)}}_{\rm{2}}}{{\rm{(P}}{{\rm{W}}_{{\rm{12}}}}{{\rm{O}}_{{\rm{40}}}}{\rm{)}}_{{\rm{0}}{\rm{.037}}}}{{\rm{(N}}{{\rm{O}}_{\rm{3}}}{\rm{)}}_{{\rm{0}}{\rm{.16}}}}$ $ \cdot {\rm{0}}{\rm{.58}}{{\rm{H}}_{\rm{2}}}{\rm{O}}$ 表 4 IFRs/(EP-PA)、NO3-ZnAl-IFRs/(EP-PA)和PW12O40-ZnAl-IFRs/(EP-PA)复合材料的TG数据
Table 4. TG data of IFRs/(EP-PA), NO3-ZnAl-IFRs/(EP-PA) and PW12O40-ZnAl-IFRs/(EP-PA) composites
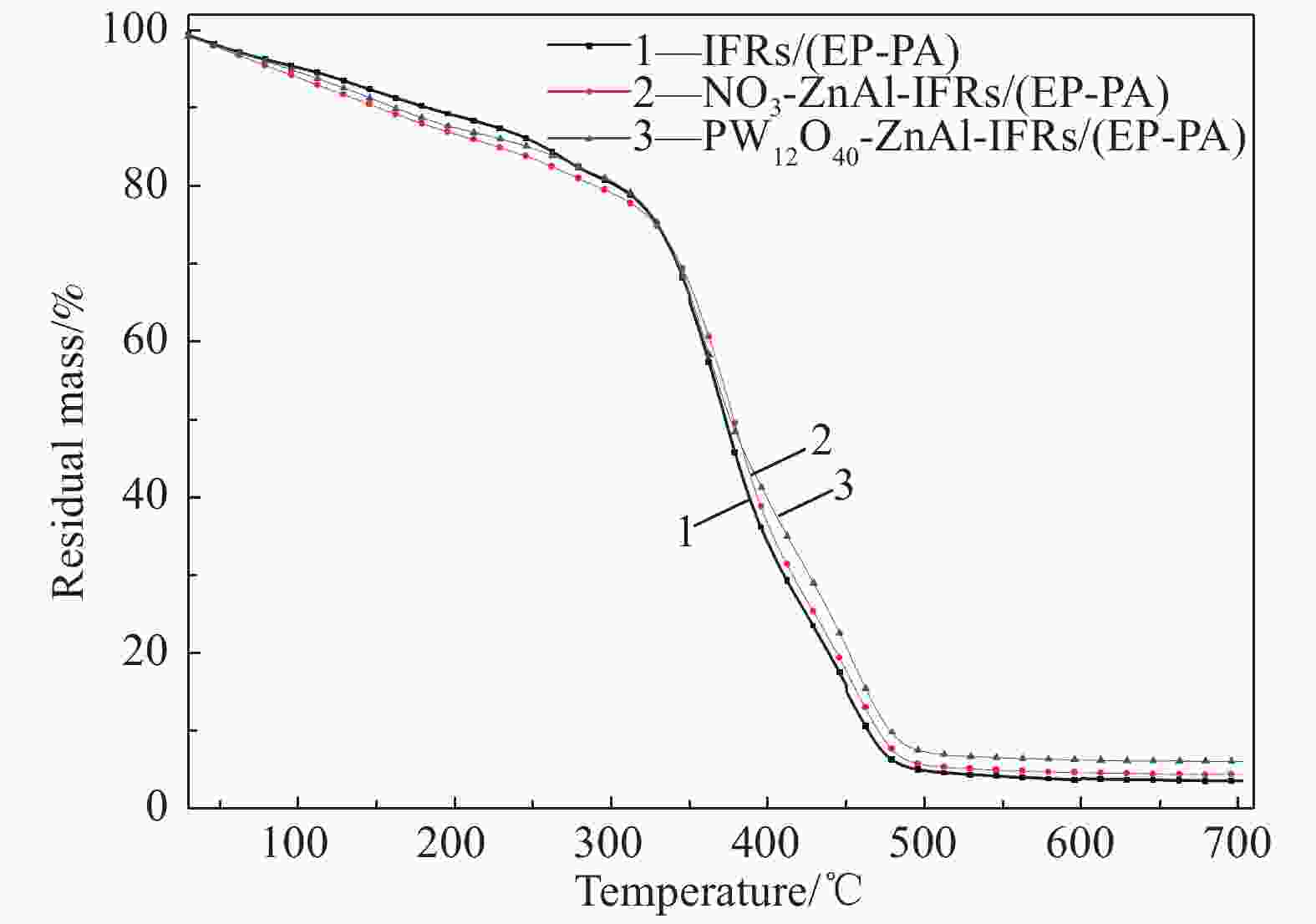
Sample T5%/℃ T50%/℃ T70%/℃ Rmax1/(%·℃−1) Residue at 700℃/% IFRs/(EP-PA) 104.9 329.1 410.2 0.71 3.47 NO3-ZnAl-IFRs/(EP-PA) 86.1 328.3 415.7 0.69 4.42 PW12O40-ZnAl-IFRs/(EP-PA) 95.1 330.1 426.2 0.66 6.05 Notes: T5%, T50%, T70%—Temperature corresponding to 5%, 50% and 70% of degradation mass, respectively; Rmax1—Maximum degradation rate. 表 5 IFRs/(EP-PA)、NO3-ZnAl-IFRs/(EP-PA)和PW12O40-ZnAl-IFRs/(EP-PA)复合材料的背温实验结果
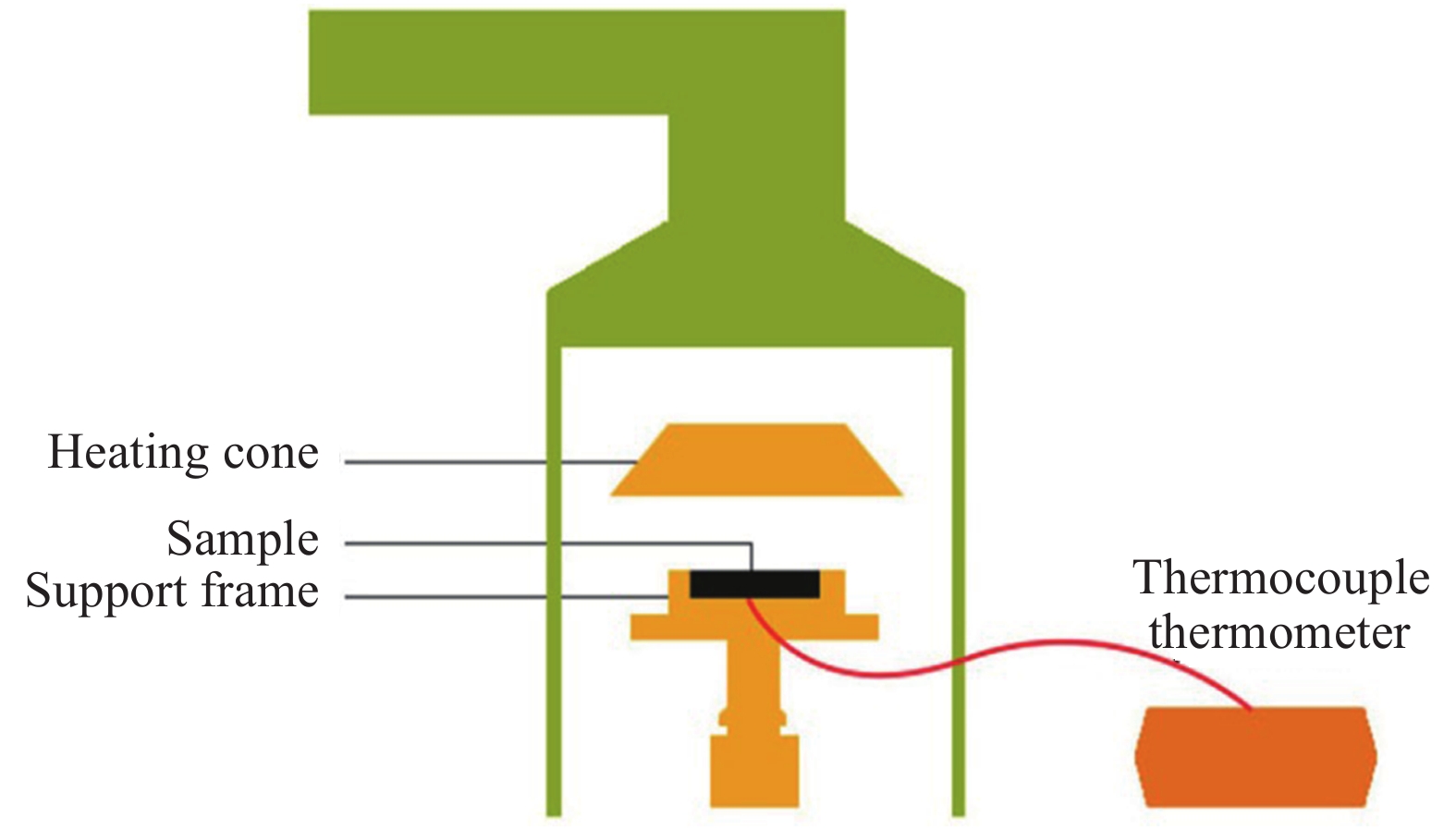
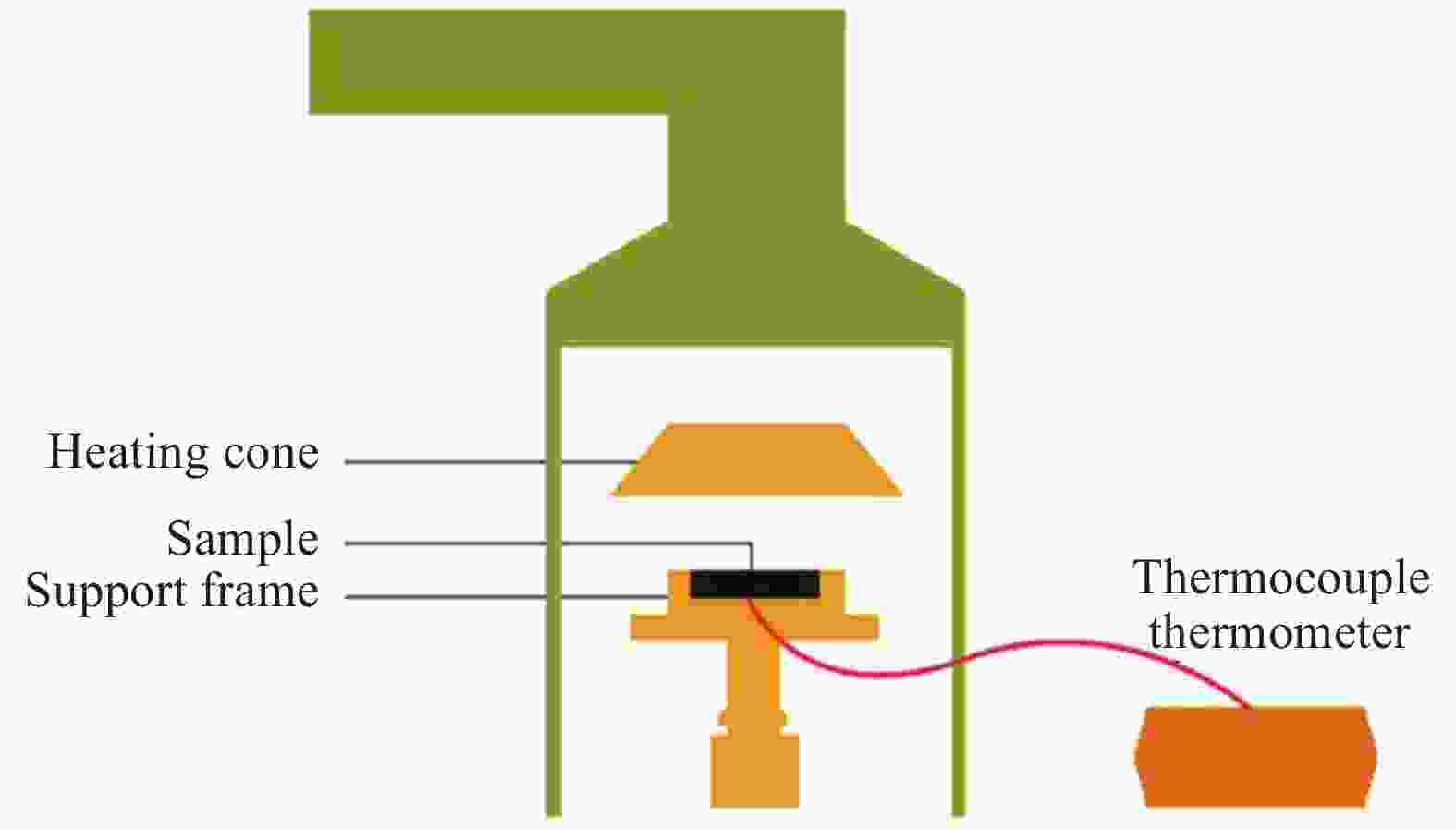
Table 5. Back temperature test results of IFRs/(EP-PA), NO3-ZnAl-IFRs/(EP-PA) and PW12O40-ZnAl-IFRs/(EP-PA) composites
Sample Fire resistance time/s V200℃/(℃·s−1) V300℃/(℃·s−1) t200℃ t300℃ IFRs/(EP-PA) 459.5 942.5 0.44 0.33 NO3-ZnAl-IFRs/(EP-PA) 618.0 916.0 0.32 0.32 PW12O40-ZnAl-IFRs/(EP-PA) 764.0 1 526.0 0.26 0.20 Notes: V200℃—Back temperature rising rate at 200℃;V300℃—Back temperature rising rate at 300℃. 表 6 IFRs/(EP-PA)、NO3-ZnAl-IFRs/(EP-PA)和PW12O40-ZnAl-IFRs/(EP-PA)复合材料的锥形量热实验数据
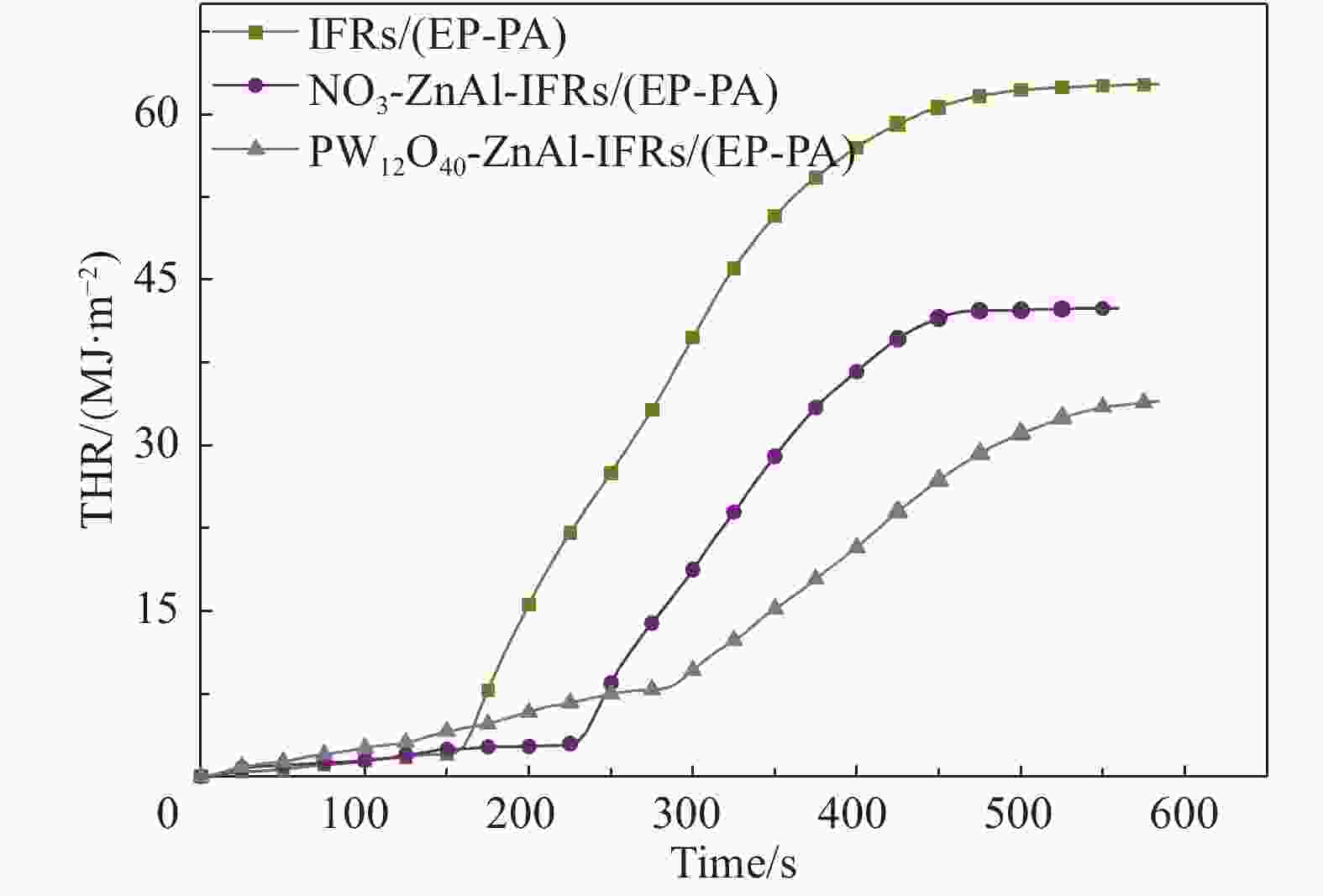
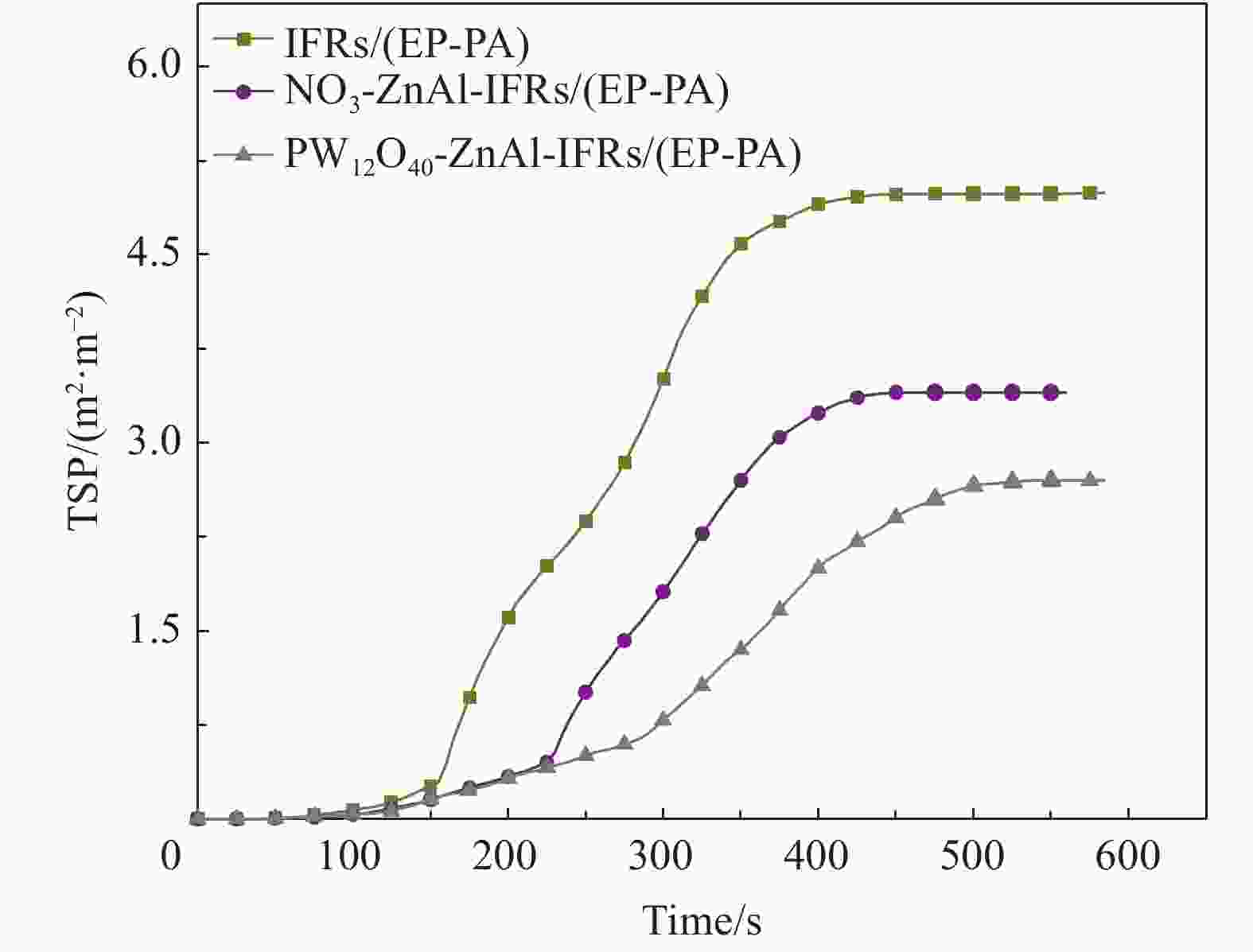
Table 6. Cone calorimetry test data of IFRs/(EP-PA), NO3-ZnAl-IFRs/(EP-PA) and PW12O40-ZnAl-IFRs/(EP-PA) composites
Sample TTI/$ \mathrm{s} $ PHRR/
(kW·m−2)MHRR/
(kW·m−2)THR/
(MJ·m−2)MEHC/
(MJ·kg−1)TSP/
(m2·m−2)FGI/
(kW·(s·m2)−1)IFRs/(EP-PA) 154.0 388.3 140.8 60.6 20.3 4.7 2.35 NO3-ZnAl-IFRs/(EP-PA) 227.0 317.5 118.5 39.4 20.2 2.9 1.48 PW12O40-ZnAl-
IFRs/(EP-PA)281.0 136.3 62.3 28.2 14.5 2.1 0.34 Notes: TTI—Ignition time; PHRR—Peak of heat release rate; MHRR—Mean heat release rate; THR—Total heat release; MEHC—Mean effective heat of combustion; TSP—Total smoke produce; FGI—Fire growth index. 表 7 EP-PA、IFRs/(EP-PA)、NO3-ZnAl-IFRs/(EP-PA)和PW12O40-ZnAl-IFRs/(EP-PA)复合材料的力学性能
Table 7. Mechanical properties of EP-PA, IFRs/(EP-PA), NO3-ZnAl-IFRs/(EP-PA) and PW12O40-ZnAl-IFRs/(EP-PA)composites
Sample Tensile strength/MPa Tear strength/(N·mm−1) Elongation at break/% EP-PA 11.47 42.83 10.87 IFRs/(EP-PA) 8.12 31.40 16.93 NO3-ZnAl-IFRs/(EP-PA) 7.86 30.23 19.60 PW12O40-ZnAl-IFRs/(EP-PA) 8.03 30.62 19.39 -
[1] WILLIAMS G R, O'HARE D. Towards understanding, control and application of layered double hydroxide chemistry[J]. Journal of Materials Chemistry,2006,16(30):3065-3074. doi: 10.1039/b604895a [2] 段雪, 陆军. 二维纳米复合氢氧化物结构、组装与功能[M]. 北京: 科学出版社, 2013.DUAN Xue, LU Jun. Structure, assembly and function of two-dimensional nano composite hydroxide[M]. Beijing: Science Press, 2013(in Chinese). [3] 张新可. 改性锌铝水滑石的制备及其协同膨胀阻燃聚烯烃的研究[D]. 哈尔滨: 哈尔滨理工大学, 2015.ZHANG Xinke. Preparation of modified ZnAl hydrotalcite and study on its synergistic expansion and flame retardant polyolefin[D]. Harbin: Harbin University of Science and Technology, 2015(in Chinese). [4] GUO D X, SONG X M, TAN L C, et al. A facile dissolved and reassembled strategy towards sandwich-like rGO@NiCoAl-LDHs with excellent supercapacitor performance[J]. Chemical Engineering Journal,2019,356:955-963. doi: 10.1016/j.cej.2018.09.101 [5] SHEN Y L, YIN K J, AN C H, et al. Design of a difunctional Zn-Ti LDHs supported PdAu catalyst for selective hydrogenation of phenylacetylene[J]. Applied Surface Science,2018,456:1-6. doi: 10.1016/j.apsusc.2018.06.091 [6] 丁娴, 殷凡文, 彭成栋, 等. Mg-Ni-Al-EDTA柱撑LDHs层状材料的水热合成、结构及性能[J]. 无机化学学报, 2012, 28(4):763-772.DING Xian, YIN Fanwen, PENG Chengdong, et al. Hydrothermal synthesis, structural analysis and performance of EDTA pillared Mg-Ni-Al layered double hydroxides (LDHs)[J]. Chinese Journal of Inorganic Chemistry,2012,28(4):763-772(in Chinese). [7] 刘跃军, 高鑫, 刘亦武, 等. 多元LDHs/EVA纳米复合材料的制备及性能研究[J]. 功能材料, 2012, 43(15):2009-2013. doi: 10.3969/j.issn.1001-9731.2012.15.010LIU Yuejun, GAO Xin, LIU Yiwu, et al. Preparation of plurality LDHs/EVA nanocomposites and its properties[J]. Journal of Functional Materials,2012,43(15):2009-2013(in Chinese). doi: 10.3969/j.issn.1001-9731.2012.15.010 [8] 尚松川, 杨保俊, 张睿辰, 等. Sb<sub>2</sub>O<sub>3</sub>-ZnMgAl类水滑石的制备及其在软聚氯乙烯阻燃中的应用[J]. 复合材料学报, 2017, 34(8):1667-1673.SHANG Songchuan, YANG Baojun, ZHANG Ruichen, et al. Preparation of Sb<sub>2</sub>O<sub>3</sub>-LDHs and its assisting flame retardant effects on soft polyvinyl chloride[J]. Acta Materiae Compositae Sinica,2017,34(8):1667-1673(in Chinese). [9] 杨杰, 谢光银. 聚合物-层状双氢氧化物纳米复合材料的研究进展[J]. 合成纤维, 2014, 43(6):41-47.YANG Jie, XIE Guangyin. Progress in polymer-layered double hydroxides nano composites[J]. Synthetic Fiber in China,2014,43(6):41-47(in Chinese). [10] 郑怡磊, 陈晓, 陈英红, 等. 固相剪切碾磨制备聚丙烯/层状双金属氢氧化物纳米复合材料的力学性能及热稳定性[J]. 高分子材料科学与工程, 2011, 27(5):47-50.ZHENG Yilei, CHEN Xiao, CHEN Yinghong, et al. Mechanical properties and thermal stability of polypropylene/layered double hydroxides nanocomposites prepared by solid state shear milling[J]. Polymer Materials Science <italic>&</italic> Engineering,2011,27(5):47-50(in Chinese). [11] 徐文总, 李冲冲, 汪贵松, 等. 不同阴离子插层的ZnMgAl层状双氢氧化物对聚氨酯弹性体阻燃抑烟性能的影响[J]. 复合材料学报, 2017, 34(8):1683-1692.XU Wenzong, LI Chongchong, WANG Guisong, et al. Effect of ternary layered double hydroxide with different interlayer anions as flame retardant and smoke suppressant on polyurethane elastomer[J]. Acta Materiae Compositae Sinica,2017,34(8):1683-1692(in Chinese). [12] 李茜, 杨保俊, 王百年, 等. ZnMgAl-LDHs阻燃剂的改性及其在聚丙烯中的应用[J]. 合肥工业大学学报(自然科学版), 2014, 37(11):1294-1299, 1340.LI Xi, YANG Baojun, WANG Bainian, et al. Surface modification of flame retardant ZnMgAl-LDHs and its application to polypropylene[J]. Journal of Hefei University of Technology (Natural Science),2014,37(11):1294-1299, 1340(in Chinese). [13] 杨保俊, 薛中华, 王百年,等. 类水滑石的制备与改性及其在聚丙烯阻燃中的应用[J]. 复合材料学报, 2014, 31(2):353-361.YANG Baojun, XUE Zhonghua, WANG Bainian, et al. Preparation and modification of layered double hydroxides and application in polypropylene as flame retardant[J]. Acta Materiae Compositae Sinica,2014,31(2):353-361(in Chinese). [14] DING P, KANG B, ZHANG J, et al. Phosphorus-containing flame retardant modified layered double hydroxides and their applications on polylactide film with good transparency[J]. Journal of Colloid And Interface Science,2015,440:46-52. doi: 10.1016/j.jcis.2014.10.048 [15] HUANG G B, FEI Z D, CHEN X Y, et al. Functionalization of layered double hydroxides by intumescent flame retardant: Preparation, characterization, and application in ethylene vinyl acetate copolymer[J]. Applied Surface Science,2012,258(24):10115-10122. doi: 10.1016/j.apsusc.2012.06.088 [16] WANG P J, HU X P, LIAO D J, et al. Dual fire retardant action: The combined gas and condensed phase effects of Azo-modified NiZnAl layered double hydroxide on intumescent polypropylene[J]. Industrial <italic>&</italic> Engineering Chemistry Research,2017,56(4):920-932. [17] 周友, 刘秀, 王芳, 等. 金属氧化物对膨胀阻燃涂层耐火及成炭性能的影响[J]. 无机材料学报, 2014, 29(9):972-978. doi: 10.15541/jim20130686ZHOU You, LIU Xiu, WANG Fang, et al. Effect of metal oxides on fire resistance and char formation of intumescent flame retardant coating[J]. Journal of Inorganic Materials,2014,29(9):972-978(in Chinese). doi: 10.15541/jim20130686 [18] International Organization for Standardization. Reaction-to-fire tests: Heat release, smoke production and mass loss rate Part 1: Heat release rate (cone calorimeter method): ISO 5660—1—2002[S]. Geneva: International Organization for Standardization, 2002. [19] 中国国家标准化管理委员会. 硫化橡胶或热塑性橡胶 拉伸应力应变性能的测定: GB/T 528—2009[S]. 北京: 中国标准出版社, 2009.Standardization Administration of the People’s Republic of China. Rubber, vulcanized or thermoplastic: Determination of tensile stress-strain properties: GB/T 528—2009[S]. Beijing: China Standards Press, 2009(in Chinese). [20] 中国国家标准化管理委员会. 硫化橡胶或热塑性橡胶撕裂强度的测定(裤形、直角形和新月形试样): GB/T 529—2008[S]. 北京: 中国标准出版社, 2008.Standardization Administration of the People’s Republic of China. Rubber vulcanized or thermoplastic: Determination of tear strength (Trouser, angle and crescent test pieces): GB/T 529—2008[S]. Beijing: China Standards Press, 2008(in Chinese). [21] MARIAPPAN T. Recent developments of intumescent fire protection coatings for structural steel: A review[J]. Journal of Fire Sciences,2016,34(2):120-163. doi: 10.1177/0734904115626720 [22] BOURIGOT S, LE-BRAS M, DELOBEL R, et al. Synergistic effect of zeolite in an intumescence process. Study of the interactions between the polymer and the additives[J]. Journal of the Chemical Society Faraday Transactions,1996,92(18):3435-3444. doi: 10.1039/FT9969203435 [23] XUE Y D, ZHANG S P, YANG W L. Influence of expanded vermiculite on fire protection of intumescent fireproof coatings for steel structures[J]. Journal of Coatings Technology and Research,2015,12(2):357-364. doi: 10.1007/s11998-014-9626-3 [24] COSTANTINO U, GALLIPOLIA A, NOCCHETTIA M, et al. New nanocomposites constituted of polyethylene and organically modified ZnAl-hydrotalcites[J]. Polymer Degradation and Stability,2005,90(3):586-590. doi: 10.1016/j.polymdegradstab.2005.05.019 [25] LIN Y J, WANG J R, G. EVANS D, et al. Layered and intercalated hydrotalcite-like materials as thermal stabilizers in PVC resin[J]. Journal of Physics and Chemistry of Solids,2006,67(5):998-1001. -





 下载:
下载: