Preparation and corrosion resistance of SiO2 or TiO2 nano particles/fluorinated polyacrylate polymer composite coatings
-
摘要: 通过在含氟聚丙烯酸酯(PFHI)溶液中添加固体纳米粒子,经涂覆热固化后得到了厚度约为1 μm的SiO2或TiO2纳米粒子/PFHI复合涂层,考察了SiO2或TiO2两种纳米粒子质量分数对复合涂层表面性质和防腐蚀性能的影响。利用Tafel极化曲线和电化学交流阻抗(EIS)测试研究了复合涂层在3.5wt% NaCl溶液中的电化学防腐蚀性能,并运用XPS、衰减全反射傅里叶变换红外光谱(ATR-FTIR)、TG-DTA、SEM、光学接触角(OCA)手段对复合涂层进行表征。结果表明,添加SiO2或TiO2纳米粒子均可大幅提高PFHI涂层的电化学防腐蚀性能,SiO2与PFHI质量比为0.3的SiO2/PFHI复合涂层电荷转移阻抗值Rct与PFHI涂层相比上升了2个数量级。SiO2或TiO2纳米粒子增大了涂层表面粗糙度,与PFHI紧密结合形成致密的复合涂层,提高了涂层的疏水性和致密性,从而改善了涂层的抗腐蚀性能。Abstract: SiO2 or TiO2 nanoparticles/fluorinated polyacrylate polymer (PFHI) composite coatings with thickness of 1 μm were prepared by incorporating nanoparticles into PFHI solution followed by dip-coating and thermal curing. The effects of the amount of SiO2 or TiO2 nanoparticles on the surface properties and corrosion resistance of the composite coatings were investigated. The corrosion electrochemical property of the SiO2 or TiO2/PFHI composite coatings in 3.5wt% NaCl solution was investigated by Tafel curves and electrochemical impedance spectroscopy (EIS). XPS, attenuated total reflection Fourier transform infrared (ATR-FTIR), TG-DTA, SEM, optical contact angle (OCA) were used to characterize the composite coatings. The results show that the addition of SiO2 or TiO2 nanoparticles could greatly improve the corrosion resistance of the PFHI coatings. The SiO2/PFHI composite coating with SiO2 to PFHI of 0.3 in mass ratio exhibits the best electrochemical corrosion resistance performance and the penetration resistance Rct is increased by two order of magnitude compared with the neat PFHI coating. The SiO2 or TiO2 nanoparticles combine with the PFHI tightly and increase the coating surface roughness to form the compact composite coatings, improving the hydrophobicity and compactness and the corrosion resistance of SiO2 or TiO2/PFHI composite coatings.
-
表 1 SiO2或TiO2/含氟聚丙烯酸酯(PFHI)涂层配方
Table 1. Recipe of SiO2 or TiO2/fluorinated polyacrylate polymer (PFHI) coatings
Sample 5wt% PFHI
solution/gNano particle/g Nano particle:
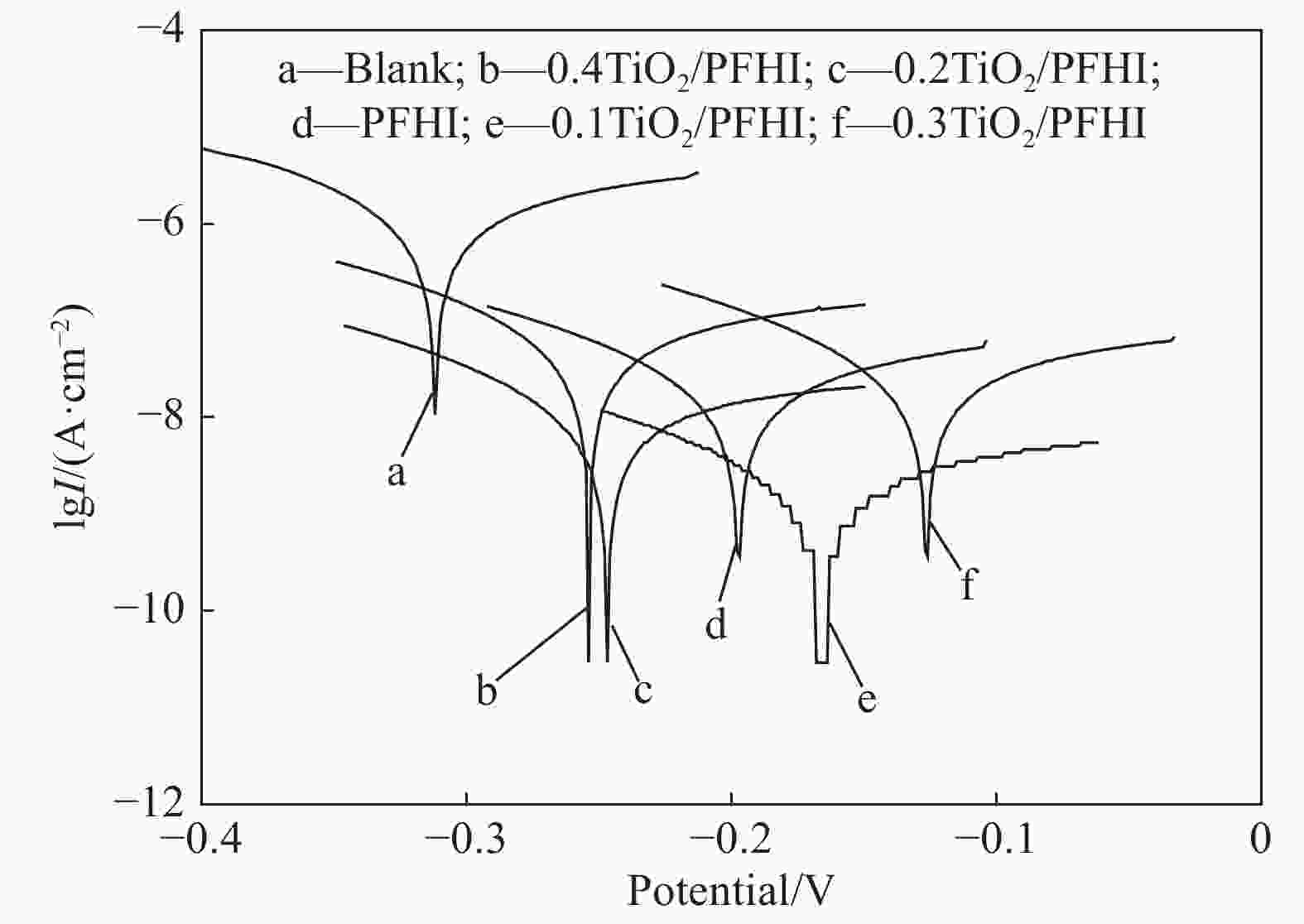
PFHI (Mass ratio)0.1SiO2/PFHI 10 0.05 0.1 0.2SiO2/PFHI 0.10 0.2 0.3SiO2/PFHI 0.15 0.3 0.4SiO2/PFHI 0.20 0.4 0.1TiO2/PFHI 10 0.05 0.1 0.2TiO2/PFHI 0.10 0.2 0.3TiO2/PFHI 0.15 0.3 0.4TiO2/PFHI 0.20 0.4 表 2 TiO2/PFHI复合材料涂层的Tafel极化曲线拟合数据
Table 2. Fitting data of Tafel polarization curves for TiO2/PFHI composite coatings
Sample Icorr/10−9 (A·cm−2) Ecorr/
mVCorrosion rate/10−5
(mm·a−1)βc βa Pe/
%Blank 1 501 −312 1 741 7.35 6.620 — PFHI 7.570 −197 8.780 8.12 37.93 99.5 0.1TiO2/PFHI 3.110 −168 3.610 8.03 3.620 99.8 0.2TiO2/PFHI 16.08 −247 18.66 8.20 2.910 99.0 0.3TiO2/PFHI 12.59 −126 14.61 8.78 28.21 99.2 0.4TiO2/PFHI 91.10 −254 105.7 7.76 3.200 94.0 Notes: Icorr—Corrosion current density; Ecorr—Corrosion electric potential; βc—Cathode Tafel slope; βa—Anode Tafel slope; Pe—Protection efficiency in the electrochemical corrosion experiments. 表 3 SiO2/PFHI和TiO2/PFHI复合涂层的EIS曲线拟合数据
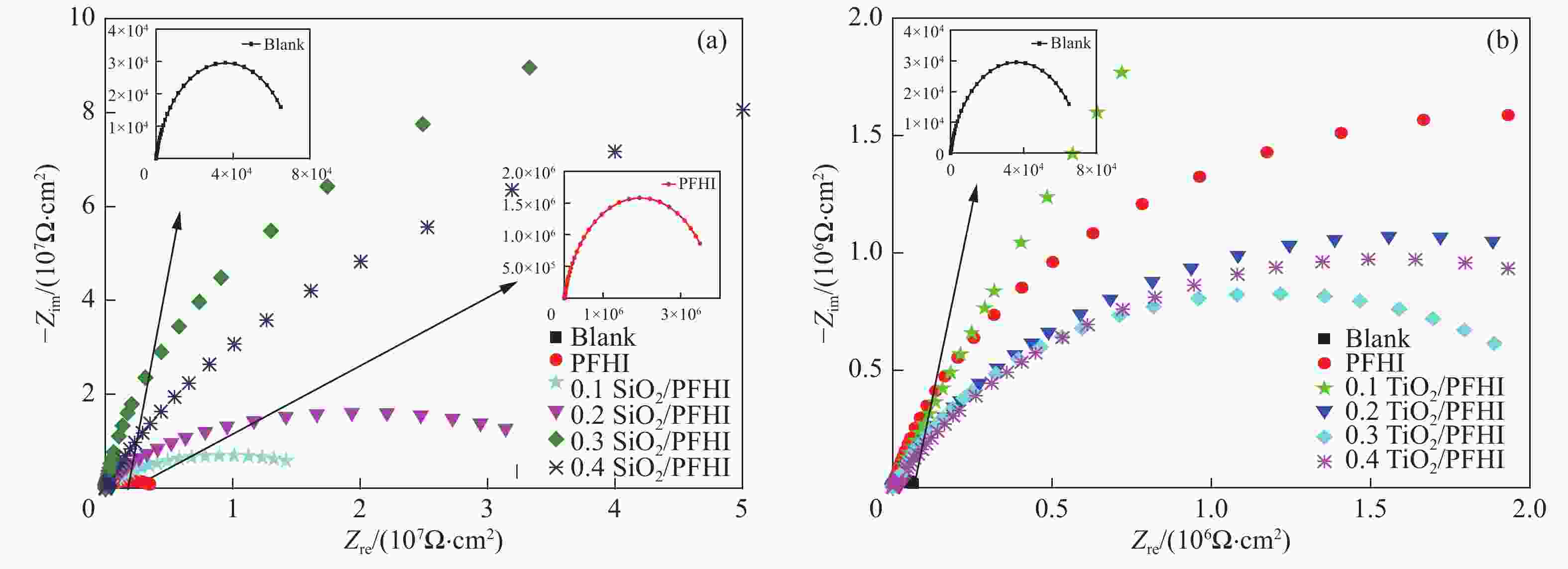
Table 3. Fitting data of EIS curves of SiO2/PFHI and TiO2/PFHI composite coatings
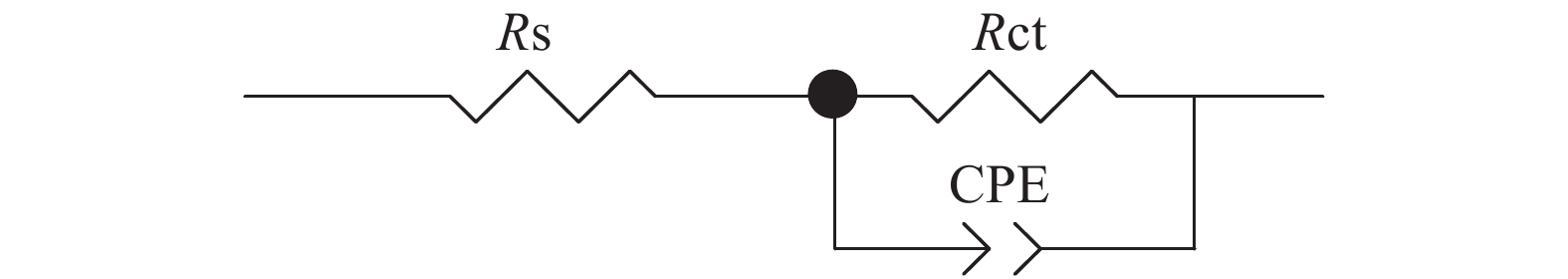
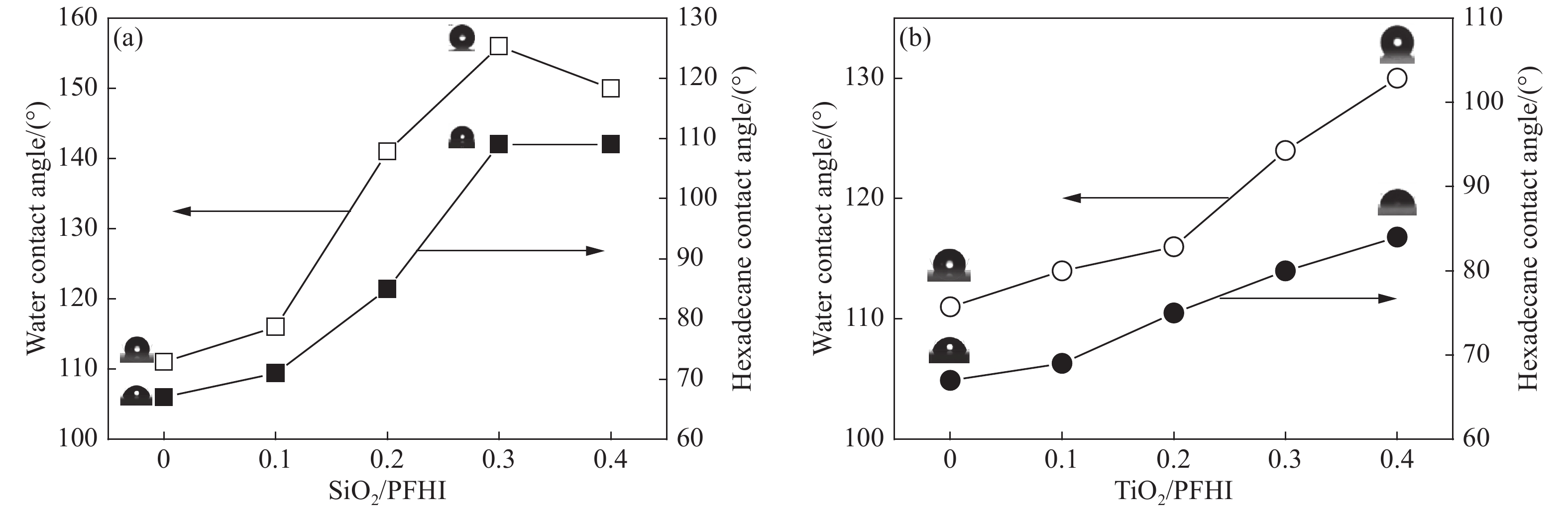
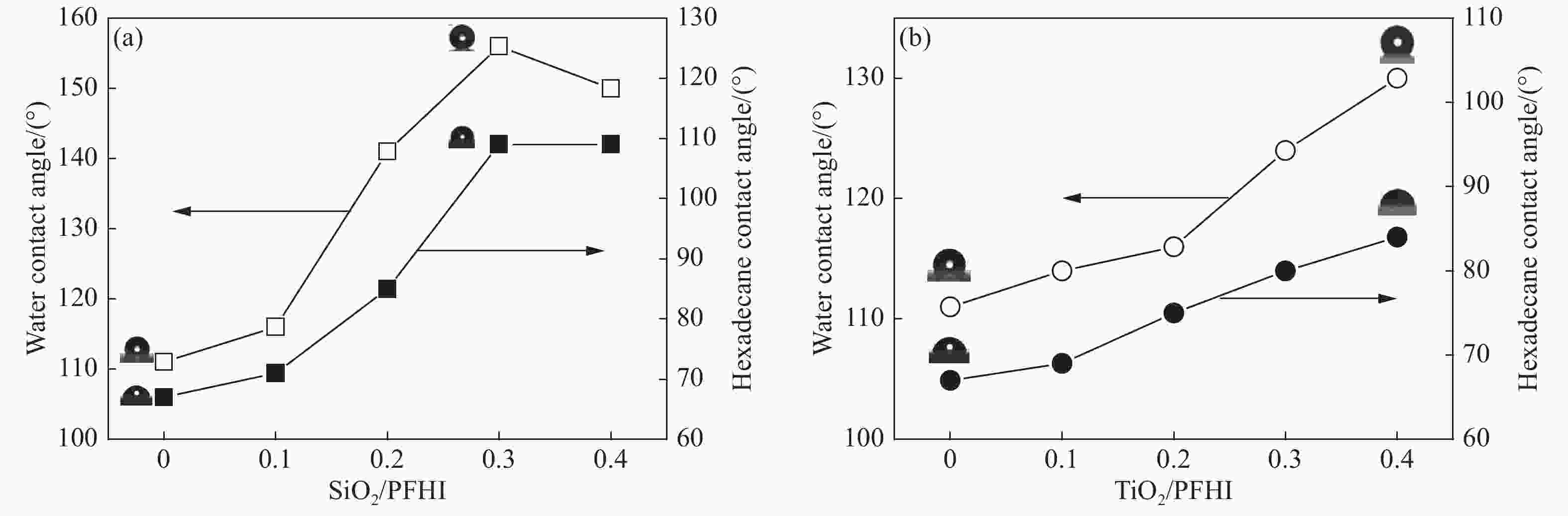
Sample Rs/(Ω·cm2) Rct/106 (Ω·cm2) Y0/10−7 n Blank 6.36 0.07 321 0.874 PFHI 74.68 3.91 5.93 0.869 0.1SiO2/PFHI 125.4 25.2 1.06 0.844 0.2SiO2/PFHI 146.3 56.2 0.262 0.856 0.3SiO2/PFHI 126.2 328 0.016 0.962 0.4SiO2/PFHI 135.8 263 12.8 0.875 0.1TiO2/PFHI 159.2 42.5 59.0 0.781 0.2TiO2/PFHI 154.0 3.23 1.32 0.747 0.3TiO2/PFHI 147.7 3.53 1.72 0.706 0.4TiO2/PFHI 118.1 2.37 1.04 0.774 Notes: Rs—Solution impedance; Rct—Charge transfer resistance; Y0—Constant of constant phase element(CPE); n—Surface roughness of the surface of phase shifting working electrode. 表 4 涂覆涂层碳钢在6wt% FeCl3溶液中的质量损失
Table 4. Mass loss of coated carbon steel in 6wt% FeCl3 solution
Sample mloss/mg RM /% Pc/% Blank 149.9 1.75 0 PFHI 126.0 1.47 15.9 0.1SiO2/PFHI 109.6 1.28 26.9 0.2SiO2/PFHI 106.6 1.25 28.9 0.3SiO2/PFHI 77.3 0.90 48.4 0.4SiO2/PFHI 108.7 1.28 27.5 0.1TiO2/PFHI 94.8 1.10 36.8 0.2TiO2/PFHI 127.0 1.47 15.3 0.3TiO2/PFHI 125.8 1.47 16.1 0.4TiO2/PFHI 137.6 1.60 8.2 Notes: mloss—Mass loss of the sample after the chemical corrosion experiments; RM—Percentage of mass loss; Pc—Protection efficiency in the chemical corrosion experiments. 表 5 PFHI、0.1TiO2/PFHI和0.3SiO2/PFHI涂层表面元素含量
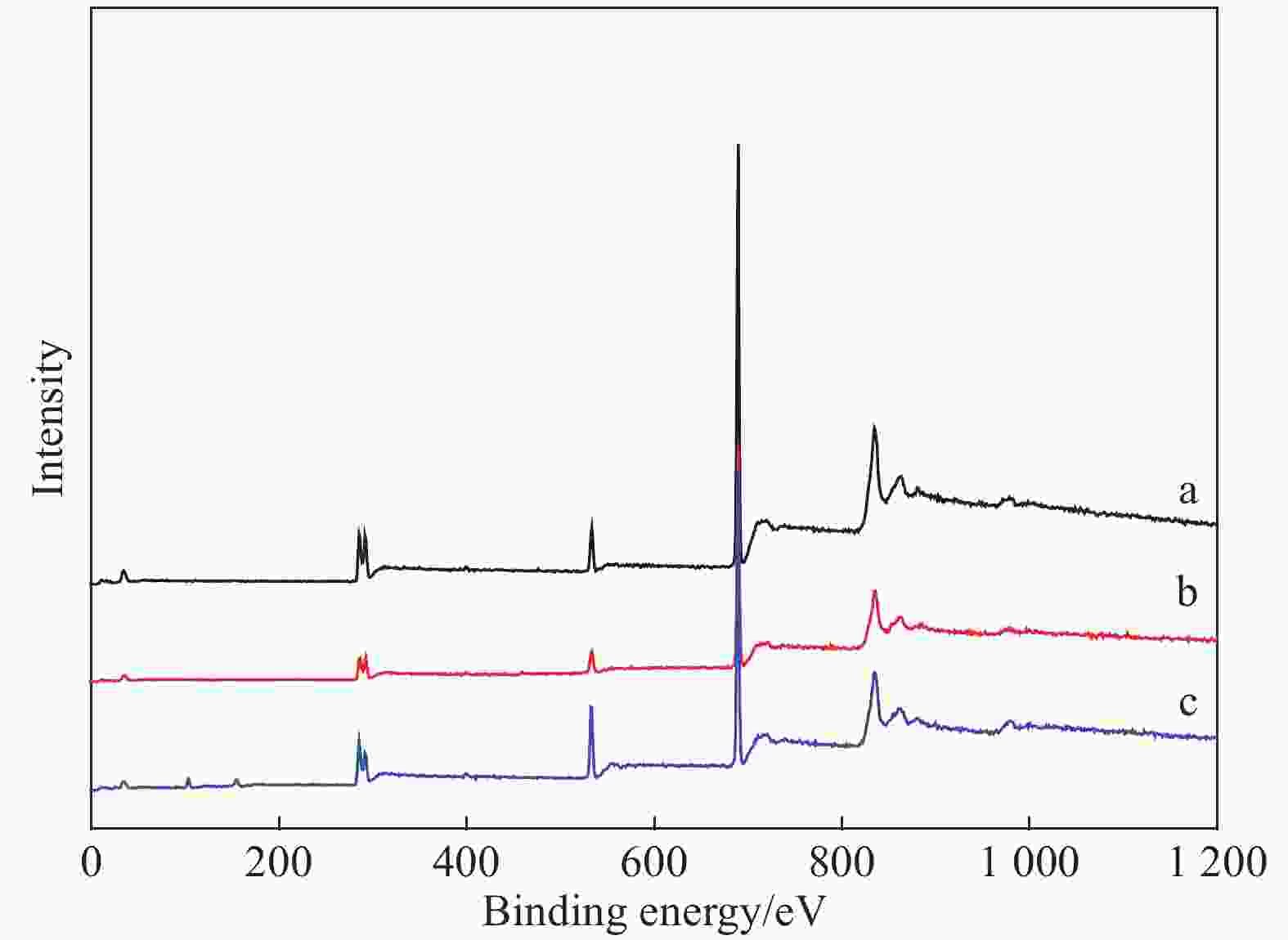
Table 5. Surface element contents of PFHI, 0.1TiO2/PFHI and 0.3SiO2/PFHI coatings
Coating F/at% C/at% O/at% F/C Ti or Si/at% PFHI 47.37 43.42 7.96 1.09 — 0.1TiO2/PFHI 46.64 42.90 8.64 1.09 0.76 0.3SiO2/PFHI 38.66 39.86 15.24 0.97 4.66 -
[1] 刘登良. 涂料工艺[M]. 北京: 化学工业出版社, 2010.LIU Dengliang. Coating process[M]. Beijing: Chemical Industry Press, 2010(in Chinese). [2] 李俊, 陈庆民. 含氟丙烯酸酯聚合物研究进展[J]. 高分子材料科学与工程, 2005(5):14-18. doi: 10.3321/j.issn:1000-7555.2005.05.004LI Jun, CHEN Qingmin. Research progress in fluorine-containing acrylate polymer[J]. Polymer Materials Science & Engineering,2005(5):14-18(in Chinese). doi: 10.3321/j.issn:1000-7555.2005.05.004 [3] 李玉峰, 祝晶晶, 高晓辉. 水性聚苯胺/叔氟丙烯酸酯复合防腐涂层的制备及性能[J]. 复合材料学报, 2016, 33(9):1859-1867.LI Yufeng, ZHU Jingjing, GAO Xiaohui. Preparation and properties of waterborne polyaniline/versatate-fluoro-acrylate composite anticorrosion coatings[J]. Acta Materiae Compositae Sinica,2016,33(9):1859-1867(in Chinese). [4] 冷长松, 邓瑾妮, 殷绿, 等. 基于自分层效应可高温固化的含氟丙烯酸酯低表面能涂料的制备与性能[J]. 高分子材料科学与工程, 2015, 31(11):151-155.LENG Changsong, DENG Jinni, YIN Lü, et al. Synthesis and characterization of high-temperature curable fluorinated polyacrylate/polyacrylate low-surface energy coating based on self-stratification[J]. Polymer Materials Science & Engineering,2015,31(11):151-155(in Chinese). [5] TUNNEY M M, GORMAN S P, PATRICK S. Infection associated with medical devices[J]. Reviews in Medical Microbiology,1996,7(4):195-206. doi: 10.1097/00013542-199610000-00002 [6] TSIBOUKLIS J, STONE M, THORPE A A, et al. Preventing bacterial adhesion onto surfaces: The low-surface-energy approach[J]. Biomaterials,1999,20(13):1229-1235. doi: 10.1016/S0142-9612(99)00023-X [7] 久保田浩治, 椛泽诚, 森田正道, 等. 耐久型防水-防湿性涂敷组合物: 中国, 104220541B[P]. 2017-09-26.HIROYUKI K, JAN T, MASAHIRO M, et al. Durable waterproof-moisture resistant coating composition: China, 104220541B[P]. 2017-09-26(in Chinese). [8] 姜兆辉, 李志迎, 王婧, 等. 纳米无机粒子/聚合物共混体系分散机理研究进展[J]. 化工新型材料, 2015, 43(2):27-29,170.JIANG Zhaohui, LI Zhiying, WANG Qian, et al. Research progress on dispersion mechanism of nanoparticles/polymer system[J]. New Chemical Materials,2015,43(2):27-29,170(in Chinese). [9] KANGO S, KALIA S, CELLI A, et al. Surface modification of inorganic nanoparticles for development of organic–inorganic nanocomposites: A review[J]. Progress in Polymer Science,2013,38(8):1232-1261. doi: 10.1016/j.progpolymsci.2013.02.003 [10] 赵金榜. 有机-无机复合体系的制备及其在涂料领域中的应用[J]. 现代涂料与涂装, 2017, 20(9):33-36,56. doi: 10.3969/j.issn.1007-9548.2017.09.009ZHAO Jinbang. Preparation of organic-inorganic compo-site system and its application in coatings[J]. Modern Paint Finishing,2017,20(9):33-36,56(in Chinese). doi: 10.3969/j.issn.1007-9548.2017.09.009 [11] 马正峰, 姬忠莹, 王晓龙. 水性UV聚氨酯丙烯酸酯/二氧化硅复合材料的制备及涂膜性能[J]. 表面技术, 2018, 47(10):283-288.MA Zhengfeng, JI Zhongying, WANG Xiaolong. Preparation and film properties of UV-curable waterborne polyurethane modified acrylate/silica composites[J]. Surface Technology,2018,47(10):283-288(in Chinese). [12] 邱守季, 杨磊, 张娅, 等. 微滴乳液聚合制备纳米SiO<sub>2</sub>/聚丙烯酸酯复合材料[J]. 复合材料学报, 2013, 30(5):29-33. doi: 10.3969/j.issn.1000-3851.2013.05.005QIU Shouji, YANG Lei, ZHANG Ya, et al. Preparation of nano SiO<sub>2</sub>/polyacrylates composites via miniemulsion polymerization[J]. Acta Materiae Compositae Sinica,2013,30(5):29-33(in Chinese). doi: 10.3969/j.issn.1000-3851.2013.05.005 [13] BOINOVICH L B, GNEDENKOV S V, ALPYSBAEVA D A, et al. Corrosion resistance of composite coatings on low-carbon steel containing hydrophobic and superhydrophobic layers in combination with oxide sublayers[J]. Corrosion Science,2012,55:238-245. doi: 10.1016/j.corsci.2011.10.023 [14] HSIEH C T, CHEN J M, KUO R R, et al. Influence of surface roughness on water- and oil-repellent surfaces coated with nanoparticles[J]. Applied Surface Science,2005,240(1):318-326. [15] ZHOU CL, LU X, XIN Z, et al. Corrosion resistance of novel silanefunctional polybenzoxazine coating on steel[J]. Corrosion Science,2013,70:145-151. doi: 10.1016/j.corsci.2013.01.023 [16] ZHOU S, WU L, SUN J, et al. The change of the properties of acrylic-based polyurethane via addition of nano-silica[J]. Progress in Organic Coatings,2002,45(1):33-42. doi: 10.1016/S0300-9440(02)00085-1 [17] 随林林, 刘芳, 陈晓蕊, 等. 纳米SiO<sub>2</sub>-氧化石墨烯/环氧涂层的制备及其防腐蚀性能[J]. 复合材料学报, 2018, 35(7):1716-1724.SUI Linlin, LIU Fang, CHEN Xiaorui, et al. Preparation and corrosion resistance of nano SiO<sub>2</sub>-graphene oxide/epoxy composite coating[J]. Acta Materiae Compositae Sinica,2018,35(7):1716-1724(in Chinese). [18] 贾涉, 姚正军, 张莎莎, 等. 硅烷改性纳米TiO<sub>2</sub>-Zn-Al/水性环氧涂层的防腐性能[J]. 复合材料学报, 2018, 35(9):2405-2413.JIA She, YAO Zhengjun, ZHANG Shasha, et al. Anti-corrosion performance of silane modified nano TiO<sub>2</sub>-Zn-Al/waterborne epoxy coatings[J]. Acta Materiae Compositae Sinica,2018,35(9):2405-2413(in Chinese). [19] 邢翠娟, 于良民, 张志明. 超疏水性聚苯胺微/纳米结构的合成及防腐蚀性能[J]. 高等学校化学学报, 2013, 34(8):1999-2004.XING Cuijuan, YU Liangmin, ZHANG Zhiming. Superhydrophobic polyaniline micro/nano structures as anticorrosion coating[J]. Chemical Journal of Chinese Universities,2013,34(8):1999-2004(in Chinese). [20] 曹楚南, 张鉴清. 电化学阻抗谱导论[M]. 北京: 科学出版社, 2002.CAO Chunan, ZHANG Jianqing. An introduction to electrochemical impedance spectroscopy[M]. Beijing: Science Press, 2002(in Chinese). [21] 何毅, 陈春林, 罗智, 等. TiO<sub>2</sub>-MWCNTs的制备及TiO<sub>2</sub>-MWCNTs/epoxy复合涂层性能[J]. 复合材料学报, 2014, 31(2):429-435.HE Yi, CHEN Chunlin, LUO Zhi, et al. Preparation of TiO<sub>2</sub>-MWCNTs and properties of TiO<sub>2</sub>-MWCNTs/epoxy compo-site epoxy coatings[J]. Acta Materiae Compositae Sinica,2014,31(2):429-435(in Chinese). [22] BERNETT M K, ZISMAN W A. Wetting properties of tetrafluoroethylene and hexafluoropropylene copolymers[J]. The Journal of Physical Chemistry,1960,64(9):1292-1294. doi: 10.1021/j100838a040 [23] NISHINO T, MEGURO M, NAKAMAE K, et al. The lowest surface free energy based on-CF<sub>3</sub> alignment[J]. Langmuir,1999,15(13):4321-4323. doi: 10.1021/la981727s [24] HONDA K, MORITA M, OTSUKA H, et al. Molecular aggregation structure and surface properties of poly(fluoroalkyl acrylate) thin films[J]. Macromolecules,2005,38(13):5699-5705. doi: 10.1021/ma050394k -





 下载:
下载: