H2 production performance of photocatalyst and mechanism of WS2/g-C3N4 heterojunction
-
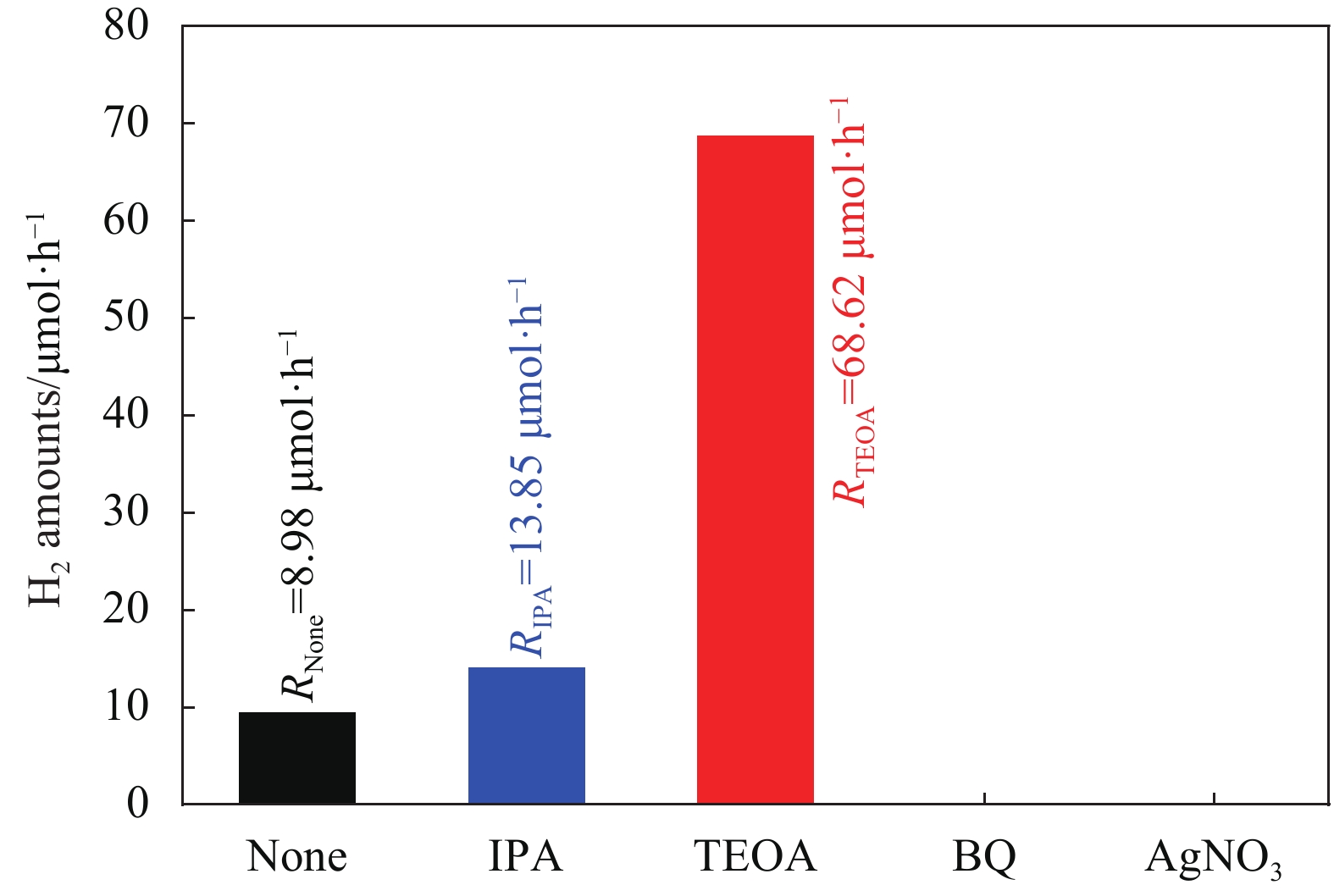
摘要: 通过溶剂蒸发和二次高温煅烧石墨相碳化氮(g-C3N4)纳米片和WS2纳米片混合物构建WS2/g-C3N4异质结,该异质结保留g-C3N4和WS2主体结构的同时,在界面处形成化学键,确保该异质结的化学稳定性和热稳定性。光催化分解水制氢实验表明,WS2纳米片含量为3wt%时光催化制氢速率高达68.62 μmol/h,分别是g-C3N4纳米片和WS2纳米片的2.53倍和15.29倍,表明异质结的构建可大幅提升g-C3N4的光催化性能,循环实验表明该异质结在5次循环实验后光催化性能没有明显下降,表明该异质结的稳定性较好。光电性能测试表明异质结的构建不仅提高激发电子的转移效率,同时抑制激发电子空穴的复合率,大幅提升激发电子的利用效率,致使光催化分解水制氢速率较g-C3N4纳米片和WS2纳米片大幅提升。Abstract: The WS2/graphite phase nitrogen carbide(g-C3N4) heterojunction was established through the solvent evaporation and second calcinations the mixture of g-C3N4 nanosheets and WS2 nanosheets. The main structure of g-C3N4 and WS2 in the heterojunction is not destroyed in the calcinations process and the interface is connected by chemical bond, which enhances the stability of heterojunction. The photocatalysis results indicate that the H2 production rate reaches to 68.62 μmol/h while the content of WS2 is 3wt%, which are 2.53 times and 15.29 times as that of g-C3N4 nanosheets and WS2 nanosheets, respectively. Besides, the H2 production rate is not decreased distinctly after 5 times circulation experiments, which reveals that the WS2/g-C3N4 heterojunction has a good chemical stability. Photoelectric property indicates that the establish of heterojunction structure can not only enhance the transport rate of excited electrons, but also suppress the recombination rate of charge carriers. Thus, the H2 production rate is enhanced distinctly compared with that of pure g-C3N4 nanosheets and WS2 nanosheets.
-
Keywords:
- g-C3N4 /
- WS2 /
- heterojunction /
- photocatalysis /
- hydrogen
-
随着飞机材料的更迭,复合材料现已成为民用飞机主要结构的常规材料[1]。玻璃纤维/环氧树脂蜂窝夹层复合材料由于其重量轻、较高的比强度/比刚度、优异的力学性能/阻燃性能和良好的隔热性能,被广泛应用于航空航天和交通运输等领域[2-4]。在航空航天领域,例如机身、舱门、机翼、尾翼、地板、整流罩和雷达罩等区域都采用蜂窝夹层结构复合材料[5-7]。在高温下,材料达到一定温度会发生热解,严重影响材料的力学性能,增大了火灾发生的可能性,因此蜂窝夹层结构复合材料的导热性研究至关重要。
国内外学者对蜂窝结构的导热性进行了相关研究,目前主要的研究方法有半经验公式理论预算、有限元模拟计算和试验测定3种[8]。Swann等[9]在忽略上下面板对蜂窝传热贡献的前提下,通过建立一维有限差分传热模型,推导测定蜂窝结构有效导热系数的Swan-Pittman半经验公式。Daryabeigi[10]采用有限差分法模拟了辐射耦合作用下蜂窝结构的导热系数。解维华等[11]将蜂窝结构内部热传导简化为一维稳态传热问题,建立铝蜂窝板结构有效热导率的数值计算模型,并将模型测试值与用Fourier定律计算得到的蜂窝板有效导热系数进行比较,证明了一维稳态传热适用于蜂窝结构。吴大方等[12]对合金蜂窝板结构在800℃非线性加热环境下进行隔热性能研究,研究了加热板温度升降速率与材料隔热性能之间的关系。李玮等[13]采用有限元分析了低温下铝合金蜂窝芯体的导热性能变化。王丹玥等[14]采用有限元软件分析了碳纤维/环氧树脂层压板厚度变化对材料导热性的影响。潘园艺等[15]探究了单向碳纤维/环氧树脂预浸料在低于230℃的高温环境下导热系数的变化趋势。Yuan等[16]采用稳态法研究金属蜂窝夹层结构在100~400℃不同温度下导热性的变化趋势。
由此可见,国内外学者对于蜂窝夹层结构导热性能的研究主要集中在金属蜂窝夹层结构上。对于纤维增强树脂基复合材料的研究主要针对其力学性能上,导热性能的研究相对较少,且主要针对层压板结构,关于树脂基复合材料蜂窝夹层结构板导热性能的研究相对匮乏。美国《联邦航空条例》(Federal Aviation Regulations,FAR)明确表示目前飞机所有内饰面板均由树脂体系和纤维增强材料组成[17]。目前绝大多数飞机内饰壁板为夹层结构,整体式层压板结构则使用较少。尽管玻璃纤维/环氧树脂蜂窝夹层复合材料具有优异的阻燃性能,但在高温下仍然具有一定的火灾危险性。为了避免火灾造成人员伤亡和财产损失,对其导热性能的研究就显得非常重要。
在计算蜂窝夹层结构导热系数时,Swan-Pittman半经验公式仅考虑蜂窝芯子的导热系数,忽略了夹层结构中上下面板对其导热性的影响。目前,关于面板对蜂窝夹层结构导热性影响的研究较少。本文通过结合Fourier定律与Swan-Pittman半经验公式,考虑上下面板对蜂窝芯导热系数的影响,建立适用于树脂基复合材料蜂窝夹层结构板热传导的理论模型。以玻璃纤维/环氧树脂预浸料和芳纶纸蜂窝芯为原料制备蜂窝夹层复合材料,探究面板和蜂窝芯对蜂窝夹层复合材料导热系数的影响规律,以期为飞机防火救援提供参考依据,为开发新型轻质蜂窝夹层壁板材料提供数据支撑。
1. 蜂窝夹层板导热性的理论计算
蜂窝夹层结构内部的传热较为复杂,如图1所示。蜂窝夹层材料主要由上下面板和中间的蜂窝芯构成,其传热过程主要包括蜂窝芯体材料的热传导、蜂窝芯体胞壁与蜂窝面板之间的热辐射、蜂窝腔体内的空气导热及自然对流换热[18]。
1.1 蜂窝芯子导热系数计算
Swann-Pittman经验方程作为蜂窝夹层材料传热特性的标准模型[19],可得蜂窝芯的等效热导率ke为
ke=kfΔAA+kg(1−ΔAA)+kr (1) 其中:kf是蜂窝芯体材料的导热系数(W/(m⋅K));kg是蜂窝腔体内气体的导热系数(W/(m⋅K));kr是蜂窝芯体辐射换热等效导热系数(W/(m⋅K));ΔA/A是蜂窝芯体胞壁截面积与整个截面积之比。对于胞壁厚度相同的正六边形蜂窝:
ΔAA=2√3Δ3l (2) 其中:Δ是蜂窝胞壁的厚度(mm);l是蜂格边长(mm)。
采用Swann-Pittman给出的等效热导率公式计算辐射导热系数[20]:
kr=4ξσT3avghc (3) 其中:Tavg是整个蜂窝夹芯板温度的平均值(K);σ是斯特潘-玻尔兹曼常数(W⋅m−2⋅K−4);hc是蜂窝芯子的高度(mm)。式中:
ξ=0.664(η+0.3)(−0.69)ε1.63(η+1)(−0.89) (4) 其中:ε是蜂窝结构内部统一的辐射率;η=hc/d是蜂窝芯子高度与蜂窝内切圆直径之比。
气体导热率可由下式得到[19]:
kg=k∗g1+2(2−αα)(2γγ+2)(1Pr)(λLc) (5) \其中: {k}_{{\mathrm{g}}} 是气体导热系数( \mathrm{W}/\left(\mathrm{m}\cdot \mathrm{K}\right) ); k_{\mathrm{g}}^{\ast} 是随温度$变化的空气气体热导率 \left(\mathrm{W}/\left(\mathrm{m}\cdot\mathrm{K}\right)\right) ;$ \alpha 是适应系数,可在0 1之间取值; \gamma 是比热率; {P}_{{\mathrm{r}}} 是普朗特数; {L}_{{\mathrm{c}}} 是蜂窝高度(mm); \lambda $是平均分子自由程。
λ=KBT√2πd2gP (6) 其中:KB是波尔兹曼常数(J⋅K−1);dg是气体的分子平均碰撞直径(m);T是热力学温度(K);P是压力(Pa)。
1.2 蜂窝夹层材料导热系数计算
以下部分概述了蜂窝夹层材料导热性能的估计方法。分析中使用的参数如图2所示。在传热过程中,蜂窝夹层材料上端面受热使其温度高于下端面产生热量的传递。对于该蜂窝夹层结构复合材料而言,格拉晓夫数Gr分别为6.05×104、2.8×105、2.2×106,均小于109,因此该蜂窝芯体空气的流动状态为层流,这种状态下气体的对流换热系数较低,在传热过程中对流的影响几乎不存在,因此可忽略自然对流换热对蜂窝芯导热系数的影响[21]。此外,蜂窝芯体内空气的热传导系数约为
0.0269 W/(m⋅K),相较于蜂窝芯体等效导热系数较小[22],且蜂窝腔内空气体积有限,因此忽略空气的热传导对蜂窝芯体导热系数的影响。因此,在计算过程中考虑上下面板与蜂窝芯体材料的热传导,蜂窝芯体胞壁与蜂窝面板之间的热辐射。根据导热系数测试仪的原理,在实验过程中,蜂窝夹层材料上下表面温度为定值且存在温度差,热量由高温向低温方向传递[23-24]。可以将蜂窝夹层结构看作三层平壁的普通夹层板,如图3所示,将蜂窝夹层材料的传热过程分解为两部分:(1)将蜂窝芯子看作均质材料,将蜂窝夹层材料的导热看作三层平壁的稳态导热问题[25];(2)将蜂窝芯子的传热问题分解为蜂窝芯体材料的热传导和蜂窝芯体胞壁之间的热辐射。
上下面板和蜂窝芯子的导热系数分别为k1、k3、ke,可以通过导热系数测试仪进行测定;厚度分别为h1、h3、hc;各层之间的接触紧密,因此相互接触的表面具有相同的温度,分别为T2、T3;平壁两侧外表面分别保持恒定的温度,分别为T1、T4;显然通过三层平壁的热流量相等,由Fourier定律[26]可得:
ϕ=qA=T1−T4h1/Ak1+hc/Ake+h3/Ak3=T1−T4R1+R2+R3 (7) 其中:q是平壁内任意位置的热流密度(W/(s⋅m2));ϕ是通过整个平壁的热量(W);A是平壁的表面积(m2)。
由此可见,三层平壁稳态导热的总导热热阻R为各层导热热阻之和,见下式:
h1+hc+h3Ak=h1Ak1+hcAke+h3Ak3 (8) 将公式(8)化简可得蜂窝夹层材料的导热系数k的计算公式:
k=k1kek3hh1kek3+hck1k3+h3k1ke (9) 其中:h=h1+hc+h3,为整个蜂窝夹层材料的总厚度(mm);k为蜂窝夹层材料的等效热导率(W/(m⋅K))。
ke=kfΔAA+kr=kfΔAA+4(0.664(η+0.3)(−0.69)ε1.63(η+1)(−0.89))σT3avg hc (10) 2. 实验部分
2.1 主要原料
玻璃纤维/环氧树脂预浸料和芳纶纸蜂窝来自复材易购(北京)科技有限公司。其中玻璃纤维/环氧树脂预浸料规格为XPREG@GXC120 SW210 B1高强玻璃纤维预浸料,环氧树脂含量在38%左右;高度为3 mm、5 mm、10 mm的航空级Nomex纸蜂窝的规格分别为孔径4 mm,边长2.29 mm;孔径4.8 mm,边长2.75 mm;孔径8 mm,边长4.5 mm,其耐火性满足联邦航空条例FAR.25.583[17]。
2.2 实验方法
(1)材料的制备与合成
采用凝胶时间测试仪测定预浸料的凝胶时间和凝胶温度,根据此特性确定固化压力、固化温度及固化时间3个关键参数,制定合适的热压程序。其具体的热压程序为仪器设定压力为5 MPa,温度为80℃的条件下保持30 min,仪器设定压力10 MPa,温度为125℃的条件下保持90 min,撤去仪器施加的压力和温度,待树脂基复合材料面板冷却到室温后取出;在施加压力为0.6 MPa,温度为150℃的条件下进行树脂基复合材料上下面板与蜂窝芯的粘结,从而制备出不同厚度蜂窝夹层材料,使用的仪器为东菀迅拓自动化科技有限公司生产的STA气液增压缸,其制备过程见图4。
通过专用模具控制预浸料叠加层数从而控制树脂基复合材料面板的厚度,将其与不同高度纸蜂窝芯进行合成。其中,下面板厚度均为0.3 mm,上面板厚度为0.7、0.9、1.1 mm,蜂窝芯高度分别为3、5、10 mm,共制备出9种不同厚度的蜂窝夹层材料作为实验样品,如表1所示。
表 1 实验样品参数Table 1. Experimental sample parametersSample number h1/mm hc/mm h3/mm h/mm l/mm 1-1 0.70 3.00 0.30 4.00 2.29 1-2 0.90 3.00 0.30 4.20 2.29 1-3 1.10 3.00 0.30 4.40 2.29 2-1 0.70 5.00 0.30 6.00 2.75 2-2 0.90 5.00 0.30 6.20 2.75 2-3 1.10 5.00 0.30 6.40 2.75 3-1 0.70 10.00 0.30 11.00 4.50 3-2 0.90 10.00 0.30 11.20 4.50 3-3 1.10 10.00 0.30 11.40 4.50 Note: h—thickness of the sandwich structure. (2)导热性能测试
将不同厚度的蜂窝夹层材料和树脂基复合材料面板制备为20 mm×20 mm的方型试样。采用上热下冷的工况条件,热板和冷板的温度分别设置为80℃和20℃,根据预先设定的载荷值,将试样紧紧的夹紧在冷板和热板之间,系统自动记录试件厚度。为减小测量时导热数值的不确定度,每次测试前采用标准板进行系数标定,且每种材料在相同环境下测试3次,实验结果取平均值。为减小环境温湿度对测试结果的影响,将样品提前放置于100℃和相对湿度为45%~55%的DHG-9140A干燥箱(上海飞越实验仪器有限公司)内处理4 h。采用湘潭湘仪仪器有限公司生产的DRL系列导热系数测试仪分别测试9组蜂窝夹层材料和4组树脂基复合材料面板的导热系数,其面板的导热系数见表2。所制备面板厚度上下误差不超过0.01 mm。图5为不同面板导热系数的误差范围。
表 2 不同面板导热系数测定值Table 2. Measurement values of thermal conductivity of different panelsPanel thickness/mm 0.3 0.7 0.9 1.1 Thermal conductivity/
(W·m−1·K−1)0.589 0.460 0.364 0.338 (3)隔热性能测试
选取上面板为0.9 mm的3种蜂窝夹层材料,制备尺寸为25 mm×25 mm样品,将上海向潜电气有限公司生产的ES-34燃烧器固定在距离蜂窝夹层材料500 mm的位置,同时使用德国InfraTec GmbH公司生产的VarioCAM-900型红外热成像仪记录蜂窝夹层材料背面的温度变化。为了确保燃烧器获得稳定的燃烧状况,打开丙烷气体,调整气体阀门压力范围在 2.8~3.2 kPa,此时试样距离燃烧器圆形桶出火口(500±3) mm,调整空气流量并打开风机 35 s,点火将燃烧器预热 2~3 min,待稳定后开始测试。
2.3 模型构建
从结构上看,蜂窝夹层结构复合材料是由一个个蜂窝单元排列组成,为简化模型,从整个蜂窝夹层结构的中部切取一个蜂窝单元来进行模型的构建,所选蜂窝单元进行周期性复制后必须能重现原来的蜂窝芯体[27]。由于蜂窝夹层复合材料相邻的蜂窝腔被蜂窝芯壁相隔,在切取单元实体模型时,每个蜂窝单元模型的壁厚应为试验材料蜂窝芯结构壁厚的一半,即为0.189 mm。
基于以上假设,定义材料属性,其设定如表3所示。
表 3 试样属性参数Table 3. Sample attribute parametersName of material Density/
(kg·m−3)Thermal conductivity/
(W·m−1·K−1)0.3 mm panel 2.58×103 0.589 0.7 mm panel 2.4×103 0.460 0.9 mm panel 2.04×103 0.364 1.1 mm panel 1.87×103 0.338 3 mm honeycomb core 40 0.993 5 mm honeycomb core 45 1.162 10 mm honeycomb core 80 1.289 设定第一类边界条件:
(1)上面板温度为80℃,下面板温度为20℃;
(2)分析步长300 s,样品周边为绝热边界条件。
在对蜂窝单元模型进行网格划分时,网格类型DC3D8为八节点线性传热的六面体单元,网格划分如图6所示。
3. 结果与讨论
3.1 蜂窝夹层结构理论模型分析
利用ABAQUS有限元分析软件模拟了该蜂窝夹层结构材料的热量传递过程,得到了热量传递过程中蜂窝芯体纵向的温度场分布图,如图7所示。
通过Origin软件对数据进行积分处理,得到各蜂窝夹层结构材料蜂窝芯体的平均温度,见表4。
表 4 各厚度蜂窝芯体平均温度Table 4. Average temperature of honeycomb cores with different thicknessesSample number hc/mm h/mm Temperature/℃ 1-1 3.00 4.00 49.5 1-2 3.00 4.20 48.6 1-3 3.00 4.40 48.3 2-1 5.00 6.00 46.8 2-1 5.00 6.20 50.8 2-3 5.00 6.40 48.8 3-1 10.00 11.00 46.8 3-2 10.00 11.20 45.1 3-3 10.00 11.40 42.5 将蜂窝芯体的平均温度代入公式(9)中,计算出各厚度蜂窝夹层结构材料导热系数的理论值,并与实验值进行比较,如表5所示。该蜂窝夹层结构材料导热系数的误差是由空气的热传导、对流换热以及复杂的真实环境所带来的,其中最大误差不超过26%,由于材料本身的导热系数较小,因此误差的波动范围不大,说明该玻璃纤维/环氧树脂的蜂窝夹层材料的导热系数可以通过公式(9)进行预测。
隔热性能作为材料发生火灾后抵挡热量传递的能力,与材料的导热性能相关,是评价材料阻燃性能的一个重要指标。分析加热状态下不同厚度蜂窝夹层材料的背面温度,可探究材料的隔热性能。
根据Fourier定律可知:
dQdt=kSdThdl (11) 在稳定导热情况下,即热传导速率在空间中任意一点均相等,那么对于介质中任意两点,可以将式(11)简化为
k=QlS(T′1−T′2) (12) 其中:Q是稳态时通过试样有效传热面积的热量(W);t是时间(s);k是导热系数(W/(m⋅K));S是截面面积(m2);Th是测点纵剖面的温度(K);l是试样厚度(m);T′1是表面温度(K);T′2是背面温度(K)。
当材料加热时间相同,夹层材料背面温度越低,材料的垂直温度差越大。由式(12)可知,同一材料上下表面的温度差与该材料的导热系数呈负相关[28],因此背面温度越低,导热系数越小,材料的隔热性能越强。
为最大程度保证结果的准确性,选取夹层板中间区域的温度,分别截取初始时刻、60 s、120 s、180 s时的温度图像,如图8所示。
表 5 蜂窝夹层材料的导热系数Table 5. Thermal conductivity of honeycomb sandwich materialsSample number A1-1 A1-2 A1-3 A2-1 A2-2 A2-3 A3-1 A3-2 A3-3 Geometric dimensions/mm Bee grid edge length l/mm 2.290 2.290 2.290 2.750 2.750 2.750 4.500 4.500 4.500 Honeycomb core height hc/mm 3.000 3.000 3.000 5.000 5.000 5.000 10.000 10.000 10.000 Panel thickness h/mm 1.000 1.200 1.400 1.000 1.200 1.400 1.000 1.200 1.400 Thermal conductivity of sandwich
structure k/(W·m−1·K−1)Experimental average 0.218 0.222 0.219 0.214 0.213 0.212 0.204 0.199 0.202 Theoretical value 0.180 0.166 0.167 0.164 0.182 0.165 0.171 0.161 0.150 Error/% 17.4 25.2 23.8 23.3 14.6 22.1 16.1 19.0 25.7 3.2 面板厚度对蜂窝夹层材料导热系数的影响
本实验所采用的蜂窝夹层材料下面板厚度均为0.3 mm,因此主要分析上面板厚度变化对材料导热系数的影响。将蜂窝芯高度为3 mm的蜂窝夹层材料设定为A1组;蜂窝芯高度为5 mm的蜂窝夹层材料设定为A2组;蜂窝芯高度为10 mm的蜂窝夹层材料设定为A3组。面板厚度与蜂窝夹层材料导热系数之间的关系如图9所示,其中A1组夹层材料的导热系数实验值分别为0.218、0.222、0.219;A2组夹层材料的导热系数实验值分别为0.214、0.213、0.212;A3组夹层材料的导热系数实验值分别为0.204、0.199、0.202。每组中蜂窝夹层材料导热系数最多相差2.51%,因此面板厚度的变化对整个蜂窝夹层材料导热系数的影响不大。
将公式(9)化简可得:k=1h1/k1h+hc/keh+h3/k3h。由于面板导热系数与面板厚度相关,其中上面板厚度h1的变化值与蜂窝夹层材料的总厚度h至少相差一个数量级,因此随着上面板厚度h1的增加,h1/h、hc/keh、h3/k3h的数值变化不大,而不同厚度上面板的导热系数k1处于同一数量级,因此材料导热系数k的数值变动幅度不大,面板厚度不是导热系数的主要影响因素。
不同厚度蜂窝夹层材料的隔热性能见表6。可以看出,随着蜂窝夹层材料厚度的增加,同一时间,夹层板背面平均温度降低,垂直温度差变大,材料的导热性能下降,隔热性能有所增加。
3.3 蜂窝芯孔隙率、比表面积对蜂窝夹层材料导热系数的影响
孔隙率作为影响多孔材料导热系数的重要参数,其对蜂窝夹层材料导热系数的影响不可忽略。蜂窝芯材料的结构示意图如图10所示。在该蜂窝夹层结构复合材料中,由于蜂窝芯结构的特殊性,该材料内部存在多个密闭的空腔,因此孔隙率指的是蜂窝芯体中空腔的体积与蜂窝芯体总体积之比。
表 6 蜂窝夹层材料的隔热性能Table 6. Thermal insulation properties of honeycomb sandwich materialsHeight of
honeycomb
core/mmPorosity/
%Specific surface
area/(m2·m−3)Thermal
conductivity/
(W·m−1·K−1)Flame
temperature/℃Burn time/s 60 120 180 Average
temperature/℃Average
temperature/℃Average
temperature/℃3 82 2259.035 0.222 950±20 150.79 214.47 250.62 5 85 1085.938 0.213 135.30 190.48 221.69 10 91 508.823 0.199 125.69 150.95 194.48 对单个蜂窝元而言,蜂窝单元空腔的体积计算公式为
V0=3√32l2mhc (13) 其中:lm是蜂窝边长的内侧长度(mm);hc是蜂窝芯的高度(mm)。
单个蜂窝元总体积的计算公式为
V=3√32l2hc (14) 综合式(13)和式(14),孔隙率表示为
P=V0V=(lml)2×100% (15) 比表面积是物体的表面积与体积的比值。对单个蜂窝元而言,其表面积的计算公式为
S=6(lhc+lmhc+(l+lm)Δ) (16) 综合式(14)和式(16),单个蜂窝元的比表面积表示为
Sv=SV=4√33(1l+lml2+Δlhc+lmΔl2hc) (17) 通过式(15)计算出不同高度蜂窝芯体的孔隙率。在计算蜂窝芯体的比表面积时,由于蜂窝芯粘连导致部分单个蜂窝元不存在外表面,因此其表面积发生改变,根据实际蜂窝芯体内、外表面积计算比表面积,如表7所示。
表 7 不同高度蜂窝芯体孔隙率与比表面积Table 7. Porosity and specific surface area of honeycomb cores with different heightshc/mm l/mm lm/mm Δ/mm P/% Sv/(m2·m−3) 3.000 2.290 2.078 0.183 82 2259.035 5.000 2.750 2.538 0.183 85 1085.938 10.000 4.500 4.288 0.183 91 508.823 Notes: P—Porosity of the honeycomb core; Sv—Specific surface area of the honeycomb core. 将上面板厚度为0.7 mm的蜂窝夹层材料设定为B1组,将上面板厚度为0.9 mm的蜂窝夹层材料设定为B2组,将上面板厚度为1.1 mm的蜂窝夹层材料设定为B3组。B1组夹层材料的导热系数实验值分别为0.218、0.214、0.204;B2组夹层材料的导热系数实验值分别为0.22、0.213、0.199;B3组夹层材料的导热系数实验值分别为0.219、0.212、0.202。每组中蜂窝芯夹层材料导热系数最少相差8.42%,因此蜂窝芯夹层材料导热系数的主要影响因素是蜂窝芯。由图11、图12可知,蜂窝夹层材料的孔隙率与其导热系数成反比关系,孔隙率的增加导致材料的导热系数下降。蜂窝夹层材料的比表面积与其导热系数呈正比关系,随着比表面积的增加,材料的导热系数也随之增加。
在考虑蜂窝芯体材料的热传导和热辐射对蜂窝芯体材料传热性能的影响时,蜂窝壁作为蜂窝芯体在热传导中起主要作用,其传热能力受到孔隙率的影响。当孔隙率增加时,蜂窝芯层体积在整个蜂窝芯体积中的占比减少,导致蜂窝壁固体传热能力减弱。
同时,蜂窝芯体材料热辐射能力受其比表面积的影响。当比表面积增加,蜂窝胞壁的表面积在整个蜂窝芯体积中所占的比例增大,辐射面积增大。根据热辐射公式Q=σεAT4,其中:σ是斯特潘-玻尔兹曼常数;ε是表面反射率;T是表面温度;三者均为定值,因此辐射面积A的增加会直接导致其热辐射能力增强[29]。
为进一步分析蜂窝芯内两种传热路径对该蜂窝夹层结构复合材料有效导热系数的贡献,设定蜂窝芯体热辐射导热系数为kr,热传导导热系数为kf1,根据计算公式kr=4ξσT3avghc,kf1=kfΔA/A,分别计算蜂窝芯体热辐射和热传导的有效导热系数,将其除以蜂窝夹层复合材料有效导热系数作为其对应的贡献比例,其中每个蜂窝芯厚度中有3组数据,取3组数据中各自贡献比例的平均值作为最终结果,如图13所示。
随着蜂窝芯高度的增加,热传导和热辐射对蜂窝夹层材料的贡献比例发生了显著变化。其中热传导的贡献从41.4%下降到30.1%,而热辐射的贡献则从19.7%上升到43.6%。这一变化表明,在蜂窝芯高度增加的情况下,热辐射逐渐成为蜂窝夹层材料热量传递的主要方式。
从热传导和热辐射的物理机制来看,热传导是固体材料内部粒子通过振动和碰撞来传递热量的过程。当蜂窝芯高度较低时,由于固体材料的直接接触和紧密排列,热传导是主要的热量传递方式。但随着蜂窝芯高度的增加,固体材料之间的接触减少,导致热传导路径变长,热传导效率降低。
热辐射是物体由于温度而发出的电磁波能量的传递。热辐射的强度与物体表面温度的4次方成正比,即Q∝T4。由于热源通常位于一侧,随着热量向另一侧传递,随着蜂窝芯高度的增加,温度梯度会更加明显,从而增强热辐射的贡献。此外,热辐射不需要物质媒介,因此在固体材料接触减少的情况下,热辐射成为一种更为有效的热量传递方式。
在设计和优化蜂窝芯结构时,需要考虑热传递机制的变化,当蜂窝芯高度控制在5 mm以下时,可以通过减小蜂窝芯体的体积来削弱材料的导热性能;当蜂窝芯高度超出5 mm时,可以向蜂窝芯内填充绝缘物体或采用圆形蜂窝芯子来减少辐射面积,达到降低材料导热性能的目的。
4. 结 论
本文选取4种不同厚度的面板与3种不同厚度的蜂窝芯组成蜂窝夹层材料开展试验,具体结论如下:
(1)基于Swan-Pittman半经验公式和Fourier定律,建立蜂窝夹层结构传热理论模型。利用有限元软件仿真得到蜂窝芯体的平均温度,进行蜂窝夹层材料导热系数的理论值计算,并与试验平均值进行比较,发现室温情况下不同厚度蜂窝夹层材料导热系数的理论值与试验平均值吻合度较高,该理论模型适用于树脂基复合材料蜂窝夹层结构板;
(2)面板厚度对蜂窝夹层材料导热系数的影响可以忽略,蜂窝芯是影响材料导热系数的主要因素。蜂窝夹层材料的孔隙率与其导热系数成反比关系,比表面积与其导热系数呈正比关系。随着蜂窝芯高度增加,热辐射取代热传导逐渐成为蜂窝芯内部热量传递的主要方式;
(3)在蜂窝结构的设计中,蜂窝芯高度对材料的导热性能有着显著的影响。当蜂窝芯高度在5 mm以下时,可以通过减小蜂窝芯体的体积来削弱材料的导热性能;当蜂窝芯高度超过5 mm时,热辐射成为主要的热量传递方式,可以向蜂窝芯内填充绝缘物体或采用圆形蜂窝芯子来减少辐射面积,达到降低材料导热性能的目的。
-
图 3 g-C3N4纳米片(a)、WS2纳米片(b)和3-WS2/g-C3N4异质结(c)的SEM图像,g-C3N4纳米片(d)、WS2纳米片(e)和3-WS2/g-C3N4异质结(f)的TEM图像,以及g-C3N4纳米片(g)、WS2纳米片(h)和3-WS2/g-C3N4异质结(i)的HRTEM图像
Figure 3. SEM images of g-C3N4 nanosheets (a), WS2 nanosheets (b), 3-WS2/g-C3N4 heterojunction (c); TEM images of g-C3N4 nanosheets (d), WS2 nanosheets (e), 3-WS2/g-C3N4 heterojunction (f); HRTEM images of g-C3N4 nanosheets (g), WS2 nanosheets (h), 3-WS2/g-C3N4 heterojunction (i)
表 1 不同WS2/石墨相碳化氮(g-C3N4)g-C3N4异质结样品中g-C3N4和WS2的质量
Table 1 Mass of graphite phase nitrogen carbide(g-C3N4) and WS2 in different WS2/g-C3N4 heterojunction samples
Sample 1-WS2/g-C3N4 2-WS2/g-C3N4 3-WS2/g-C3N4 4-WS2/g-C3N4 5-WS2/g-C3N4 WS2/g 0.025 0.050 0.075 0.100 0.125 g-C3N4/g 1 1 1 1 1 -
[1] 天工. 我国能源领域对外开放不断扩大[J]. 天然气工业, 2018, 295(5):124. TIAN Gong. Energy field of China is opening wider to the outside world[J]. Natural Gas Industry,2018,295(5):124(in Chinese).
[2] 马园媛, 赵岐. 煤气化技术的现状及发展趋势[J]. 化工管理, 2018, 487(21):202. DOI: 10.3969/j.issn.1008-4800.2018.21.156 MA Yuanyuan, ZHAO Qi. Status quo and develop ment trend of coal gasification technology[J]. Chemical Enterprise Management,2018,487(21):202(in Chinese). DOI: 10.3969/j.issn.1008-4800.2018.21.156
[3] 毛晨旭. 超临界水煤气化工艺动力循环设计与性能分析[D]. 大连: 大连理工大学, 2018. MAO Chenxu. Design and performance analysis of power cycle with process of coal gasification in supercritical water[D]. Dalian: Dalian University of Technology, 2018(in Chinese).
[4] BOUVIER-MULLER J, ALLAIN C, ENJALBERT F, et al. Somatic cell count-based selection reduces susceptibility to energy shortage during early lactation in a sheep model[J]. Journal of Dairy Science,2018,101(3):2248-2259. DOI: 10.3168/jds.2017-13479
[5] WANG Q, LU B, DOU X, et al. Distribution network voltage control based on coordinated optimization of PV and air-conditioning[J]. International Journal of Photoenergy,2018,2018:1-7.
[6] PAUL K K, SREEKANTH N, BIROJU R K, et al. Solar light driven photoelectrocatalytic hydrogen evolution and dye degradation by metal-free few-layer MoS2 nanoflower/TiO2 (B) nanobelts heterostructure[J]. Solar Energy Materials and Solar Cells,2018,185:364-374. DOI: 10.1016/j.solmat.2018.05.056
[7] CROMWELL E F, STOLOW A, VRAKKING M J J, et al. Dynamics of ethylene photodissociation from rovibrational and translational energy distributions of H2 products[J]. Journal of Chemical Physics,1992,97(6):4029-4040. DOI: 10.1063/1.462942
[8] BRUNGER M J, BUCKMAN S J, NEWMAN D S, et al. Elastic scattering and rovibrational excitation of H2 by low-energy electrons[J]. Journal of Physics B,1991,24(6):1435-1448. DOI: 10.1088/0953-4075/24/6/027
[9] KESSON S E, SMITH I E. TiO2 content and the shoshonite and alkaline associations[J]. Nature,1972,236(68):110-111.
[10] WILLIAMS G, SEGER B, KAMAT P V. TiO2-graphene nanocomposites. UV-assisted photocatalytic reduction of graphene oxide[J]. Acs Nano,2008,2(7):1487. DOI: 10.1021/nn800251f
[11] GILJA V, KATANI Z, KREHULA L K, et al. Efficiency of TiO2 catalyst supported by modified waste fly ash during photodegradation of RR45 dye[J]. Science & Engineering of Composite Materials,2019,26(1):292-300.
[12] LI H, NA L, MING W, et al. Synthesis of novel and stable g-C3N4-Bi2WO6hybrid nanocomposites and their enhanced photocatalytic activity under visible light irradiation[J]. Royal Society Open Science,2018,5(3):171419. DOI: 10.1098/rsos.171419
[13] LI C, SUN Z, ZHANG W, et al. Highly efficient g-C3N4/TiO2/kaolinite composite with novel three-dimensional structure and enhanced visible light responding ability towards ciprofloxacin and S. aureus[J]. Applied Catalysis B: Environmental,2018,220:272-282. DOI: 10.1016/j.apcatb.2017.08.044
[14] ZHANG S, GAO H, HUANG Y, et al. Ultrathin g-C3N4 nanosheets coupled with amorphous Cu-doped FeOOH nanoclusters as 2D/0D heterogeneous catalysts for water remediation[J]. Environmental Science: Nano,2018,5:1179-1190. DOI: 10.1039/C8EN00124C
[15] MA Y, LIU E, HU X, et al. A simple process to prepare few-layer g-C3N4 nanosheets with enhanced photocatalytic activities[J]. Applied Surface Science,2015,358:246-251. DOI: 10.1016/j.apsusc.2015.08.174
[16] WU Y N, MIAO H, FAN S, et al. Determination of 23 β2-agonists and 5 β-blockers in animal muscle by high performance liquid chromatography-linear ion trap mass spectrometry[J]. Science China Chemistry,2010,53(4):832-840. DOI: 10.1007/s11426-010-0071-6
[17] JIA J, DU X, LIU E, et al. Highly efficient and stable Au/Bi2MoO6/Bi2WO6 heterostructure with enhanced photocatalytic activity for NO gas removal under visible light irradiation[J]. Journal of Physics D: Applied Physics,2017,50(14):145103. DOI: 10.1088/1361-6463/aa60e3
[18] CHU J, HAN X, YU Z, et al. Highly efficient visible-light-driven photocatalytic hydrogen production on CdS/Cu7S4/g-C3N4 ternary heterostructures[J]. Acs Applied Materials & Interfaces,2018,10(24):20404-20411.
[19] WANG X, MAEDA, KAZUHIKO, et al. Polymer semiconductors for artificial photosynthesis: Hydrogen evolution by mesoporous graphitic carbon nitride with visible light[J]. Journal of the American Chemical Society,2009,131(5):1680-1681. DOI: 10.1021/ja809307s
[20] WANG X, CHEN X, THOMAS A, et al. Metal-containing carbon nitride compounds: A new functional organic-metal hybrid material[J]. Advanced Materials,2010,21(16):1609-1612.
[21] DONG G, HO W, WANG C. Selective photocatalytic N2 fixation dependent on g-C3N4 induced by nitrogen vacancies[J]. Journal of Materials Chemistry A,2015,3(46):23435-23441. DOI: 10.1039/C5TA06540B
[22] LOU J, DONG G, ZHU Y, et al. Switching of semiconducting behavior from n-type to p-type induced high photocatalytic NO removal activity in g-C3N4[J]. Applied Catalysis B: Environmental,2017,214(10):46-56.
[23] JIAGUO Y, JIMMY C, MITCH K, et al. Effects of acidic and basic hydrolysis catalysts on the photocatalytic activity and microstructures of bimodal mesoporous titania[J]. Journal of Catalysis,2003,217(1):69-78.
[24] XIANG Q, YU J, JARONIEC M. Preparation and enhanced visible-light photocatalytic H2-production activity of graphene/C3N4 composites[J]. Journal of Physical Chemistry C,2011,115(15):7355-7363. DOI: 10.1021/jp200953k
-





 下载:
下载: