Preparation and performance of graphene oxide/ZIF-7 composites
-
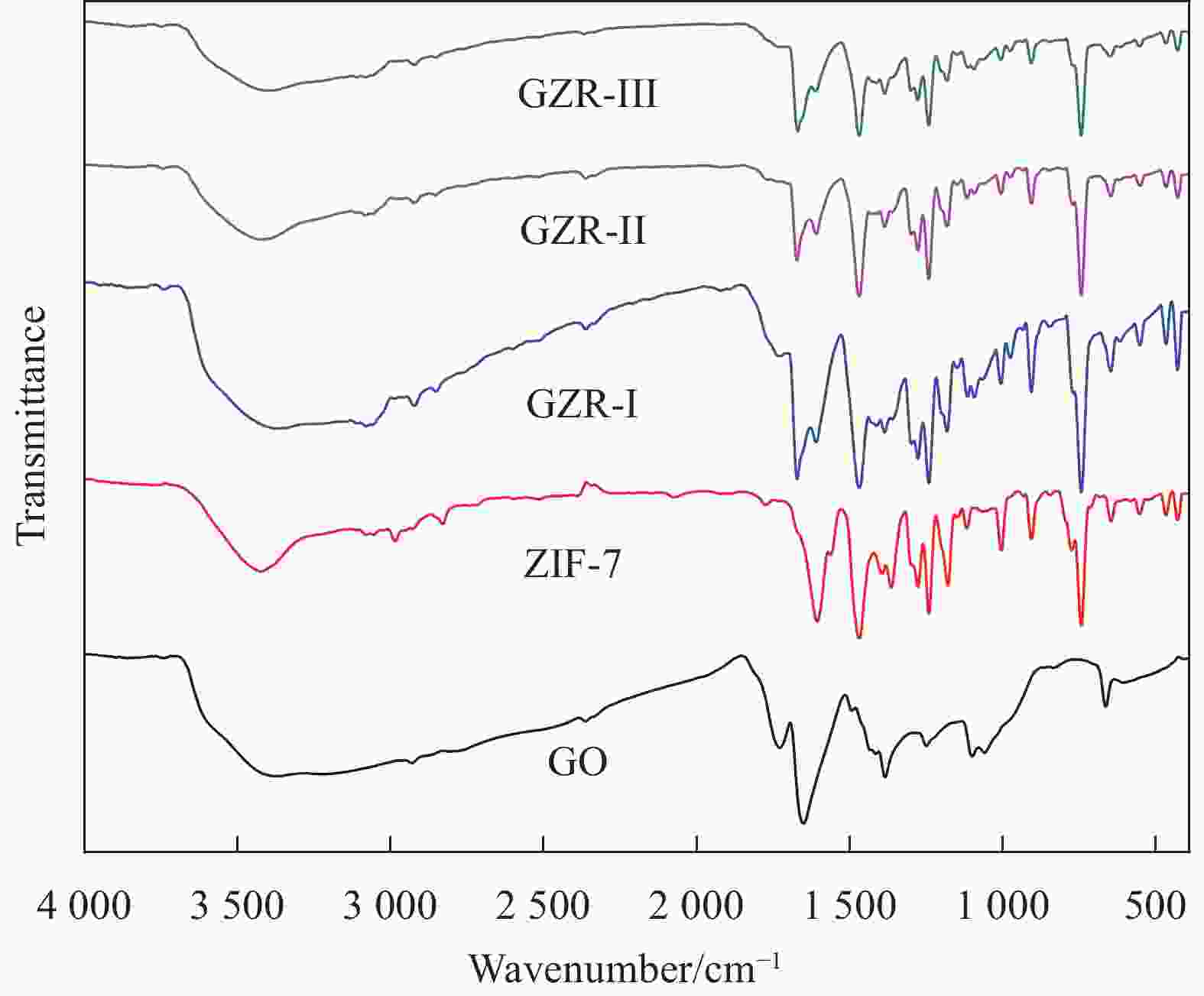
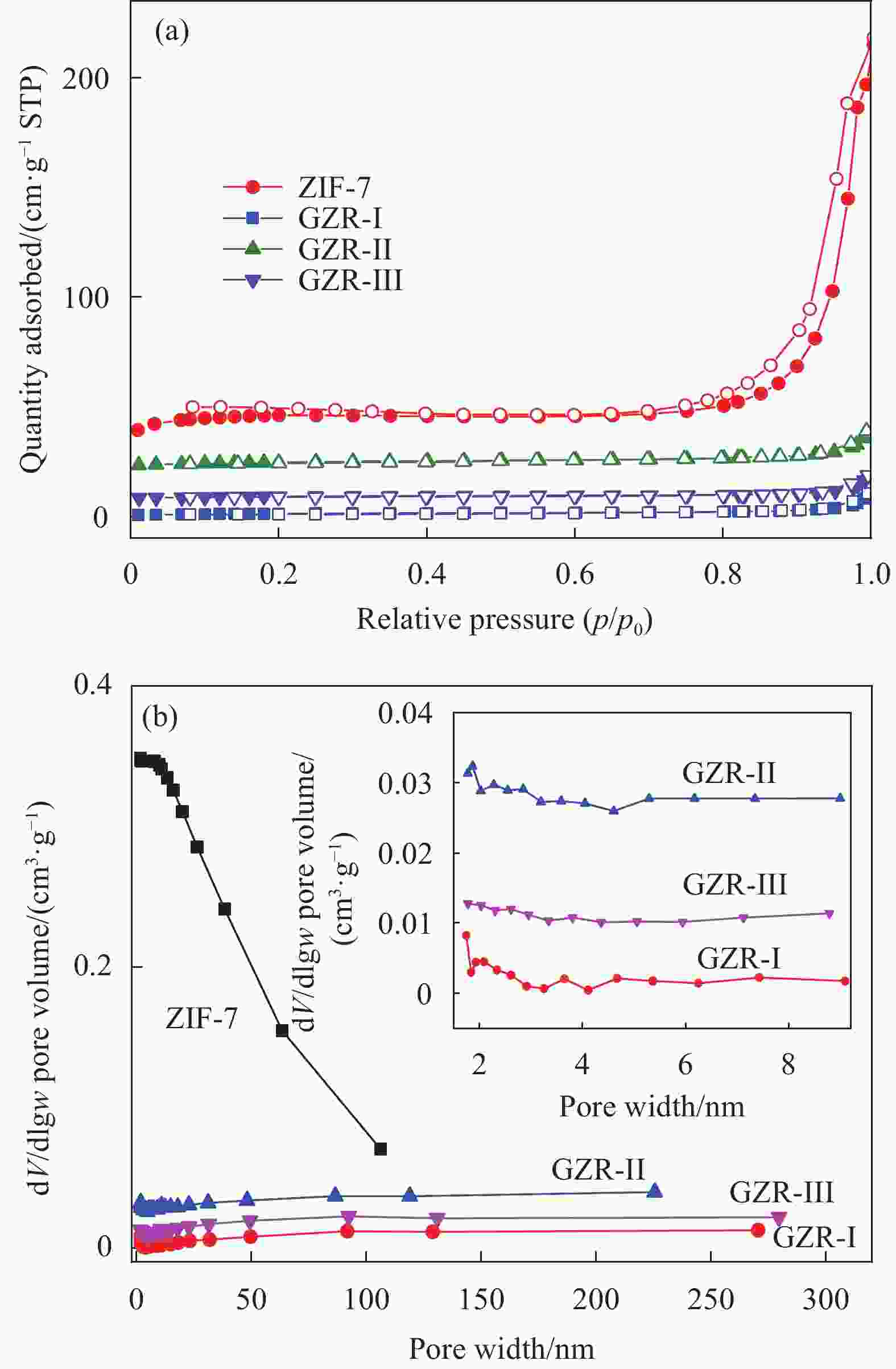
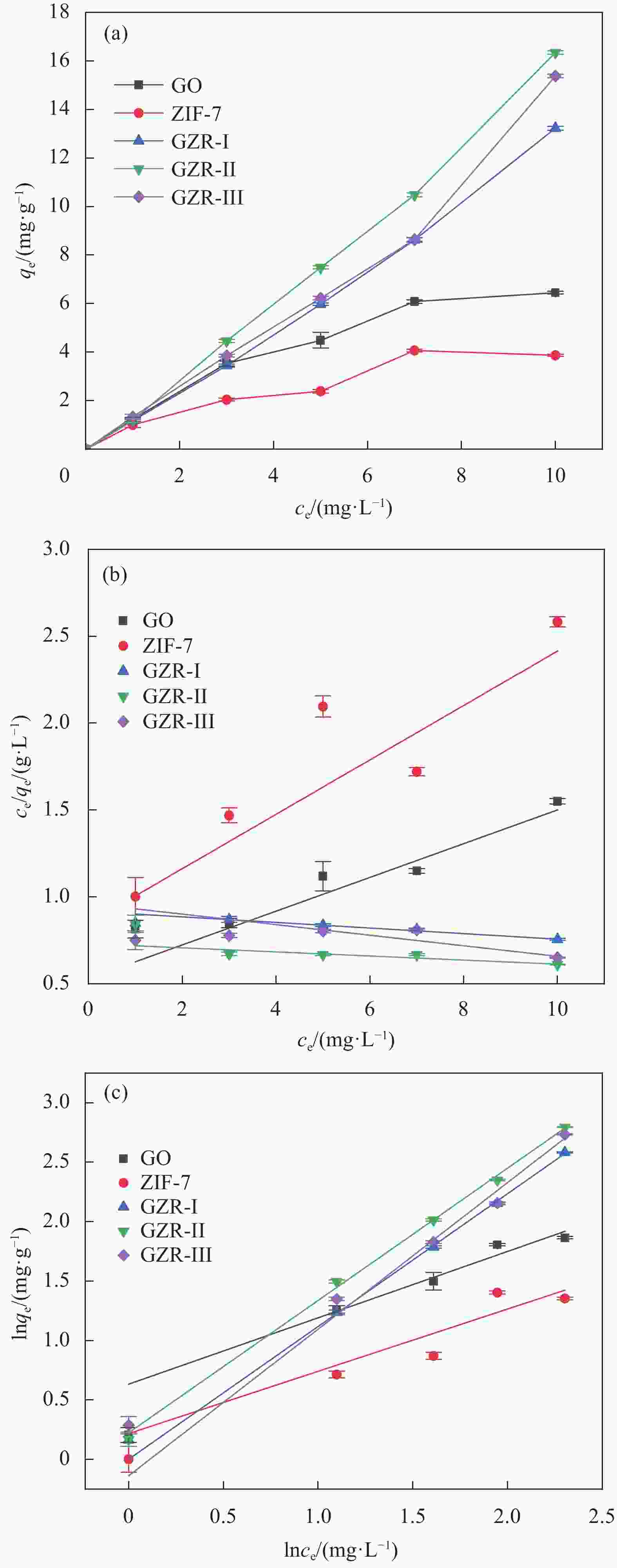
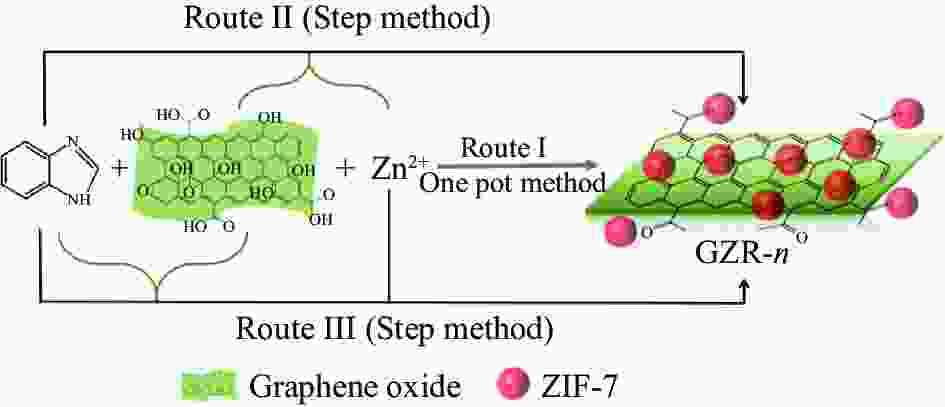
摘要: 本文采用3种合成路线实现了ZIF-7在氧化石墨烯(GO)上的原位生长,并通过PXRD、FTIR、SEM、TEM和N2吸附-脱附等对氧化石墨烯/ZIF-7复合材料(GO/ZIF-7,简写为GZR-n,其中n 代 表合成路线 I、II、III)进行了表征。研究了合成路线对ZIF-7晶体在GO上的原位生长、结晶度、微观形貌和孔径大小的影响。结果显示:通过3种合成路线均可实现ZIF-7晶体在GO表面及其片层间的原位生长,且其结晶度明显增强,部分ZIF-7晶体被GO包裹。ZIF-7晶体形貌和粒径大小在GZR-n上的原位生长受到合成路线的影响,其中,ZIF-7晶体在GZR-II中呈50 nm的纳米球状颗粒,在GZR-I和GZR-III中则形成200 nm的规则多面体。进一步研究了ZIF-7、GO和GZR-n在不同极性溶剂中的分散性能及其对有机染料的吸附性能和动力学行为。GZR-n在甲醇和氯仿中表现出良好的分散性。与ZIF-7晶体相比,GZR-I、GZR-II和GZR-III对亚甲基蓝的吸附能力分别提高了226%、302%和278%,动力学模拟结果显示,GZR-II和GZR-III对亚甲基蓝的吸附属于化学吸附,而GZR-I对亚甲基蓝的吸附属于物理吸附。Abstract: ZIF-7 crystals were in situ grown on graphene oxide (GO) by three synthetic routes, and the resulting graphene oxide/ZIF-7 composites (GZR-n) were characterized by PXRD, FTIR, SEM, TEM, and N2 isothermal adsorption-desorption. The effects of the synthetic routes on the growth, crystallinity, microscopic morphology and pore size of ZIF-7 crystals on GO were investigated. ZIF-7 crystals were grown on the surface and sheet of GO by three synthetic routes. The crystallinity of ZIF-7 crystals on GZR-n was significantly enhanced and some were wrapped by GO. The shape and size of ZIF-7 crystals growing on GZR-n were modulated by the synthesis routes. In particular, the ZIF-7 crystals are spherical particle with a diameter of 50 nm in GZR-II. For GZR-I and GZR-III, the ZIF-7 crystals are regular polyhedron with a size of 200 nm. Additional, their dispersion properties in solvents, adsorption properties and kinetic simulations for organic dyes were explored. GZR-n show good dispersion in methanol and chloroform. Compared with ZIF-7 crystals, the adsorption capacities of GZR-I, GZR-II and GZR-III for methylene blue are increased by 226%, 302% and 278%, respectively. The kinetic simulations indicate that the adsorption of GZR-II and GZR-III for methylene blue are chemisorption and that of GZR-I is physical adsorption.
-
Key words:
- graphene oxide /
- metal organic framework /
- ZIF-7 /
- composite materials /
- dye adsorption
-
表 1 GZR-n的制备
Table 1. Experimental of GZR-n
GZR-na Zn(NO3)2·6H2O/g BIM/g Route GZR-I 0.32 0.79 I GZR-II 0.80 1.99 II GZR-III 0.32 0.79 III Notes: GZR-na were prepared in N, N-dimethylformamide (DMF) dispersion of graphene oxide by three routes (The molar ratio of Zn2+ to benzimidazole was kept at 4∶25); BIM—Benzimidazole. 表 2 GZR-n对亚甲基蓝(MB)、甲基橙(MO)、孔雀石绿(MG)的平衡吸附容量qe
Table 2. Equilibrium adsorption capacity qe of methylene blue (MB), methyl orange (MO) and malachite green (MG) onto GZR-n
Adsorption qe/(mg·g−1) MB MO MG GO 6.45 9.16 11.29 ZIF-7 3.88 5.56 7.01 GZR-I 13.22 11.90 11.47 GZR-II 16.34 14.61 15.76 GZR-III 15.34 13.48 11.77 表 3 GO、ZIF-7和GZR-n对MB的吸附动力学参数
Table 3. Adsorption kinetic parameters of GO, ZIF-7 and GZR-n for MB
Sample GO ZIF-7 GZR-I GZR-II GZR-III Experimental qe/(mg·g−1) 6.45 3.88 13.22 16.34 15.34 Calculated qe/(mg·g−1) 6.38 3.77 12.99 16.34 16.07 Pseudo
first-order kinetick1/min 0.869 0.745 0.737 0.841 0.707 R2 0.960 0.799 0.951 0.848 0.890 Pseudo second-order kinetic k2/(g·mg−1·min−1) 0.066 0.085 0.008 0.003 0.003 R2 0.994 0.979 0.939 0.905 0.895 Notes: R2—Fitting constant; k1, k2—Adsorption kinetics constant. 表 4 GO、ZIF-7和GZR-n对MB的吸附等温线参数
Table 4. Adsorption isotherm parameters of GO, ZIF-7 and GZR-n for MB
Sample Langmuir adsorption isotherm Freundlich adsorption isotherm R2 KF 1/n R2 GO 0.9379 1.884 0.5581 0.8673 ZIF-7 0.7481 1.2432 0.5238 0.7171 GZR-I 0.9843 1.002 1.1167 0.9983 GZR-II 0.8566 1.252 1.1127 0.9972 GZR-III 0.7705 1.1466 1.2329 0.9741 Notes: KF—Freundlich constant; 1/n—Freundlich isotherm deviates from linearity. -
[1] CHENG P, WANG C H, KANETI Y V, et al. Practical MOF nanoarchitectonics: New strategies for enhancing the processability of MOFs for practical applications[J]. Langmuir,2020,36(16):4231-4249. doi: 10.1021/acs.langmuir.0c00236 [2] LI S Z, HUO F W. Metal-organic framework composites: From fundamentals to applications[J]. Nanoscale,2015,7(17):7482-7501. doi: 10.1039/C5NR00518C [3] WANG P, LI X H, ZHANG P, et al. Transitional MOFs: Exposing metal sites with porosity for enhancing catalytic reaction performance[J]. ACS Applied Materials & Interfaces,2020,12(21):23968-23975. [4] XU J, LIU J, LI Z, et al. Optimized synthesis of Zr(IV) metal organic frameworks (MOFs-808) for efficient hydrogen storage[J]. New Journal of Chemistry,2019,43(10):4092-4099. doi: 10.1039/C8NJ06362A [5] LI C, HE N P, ZHAO X Z, et al. Chitosan/ZIF-8 composite beads fabricated by in situ growth of MOFs crystals on chitosan beads for CO2 adsorption[J]. ChemistrySelect,2022,7(4):e202103927. doi: 10.1002/slct.202103927 [6] LIAN X, YAN B. Phosphonate MOFs composite as off-on fluorescent sensor for detecting purine metabolite uric acid and diagnosing hyperuricuria[J]. Inorganic Chemistry,2017,56(12):6802-6808. doi: 10.1021/acs.inorgchem.6b03009 [7] 李禹红, 乔瑶雨, 李超, 等. ZIF-8@PDMAPMA复合材料的构筑及其性能研究[J]. 高分子学报, 2021, 52(9):1174-1183. doi: 10.11777/j.issn1000-3304.2021.21041LI Yuhong, QIAO Yaoyu, LI Chao, et al. Fabrication and properties of ZIF-8@PDMAPMA composite materials[J]. Acta Polymerica Sinica,2021,52(9):1174-1183(in Chinese). doi: 10.11777/j.issn1000-3304.2021.21041 [8] BEN T, LU C J, PEI C Y, et al. Polymer-supported and free-standing metal-organic framework membrane[J]. Chemistry—A European Journal,2012,18(33):10250-10253. doi: 10.1002/chem.201201574 [9] FAN L L, XUE M, KANG Z X, et al. Electrospinning technology applied in zeolitic imidazolate framework membrane synthesis[J]. Journal of Materials Chemistry,2012,22(48):25272-25276. doi: 10.1039/c2jm35401b [10] AGUADO S, CANIVET J, FARRUSSENG D. Facile shaping of an imidazolate-based MOF on ceramic beads for adsorption and catalytic applications[J]. Chemical Communications,2010,46(42):7999-8001. doi: 10.1039/c0cc02045a [11] LEE H A, MA Y F, ZHOU F, et al. Material-independent surface chemistry beyond polydopamine coating[J]. Accounts of Chemical Research,2019,52(3):704-713. doi: 10.1021/acs.accounts.8b00583 [12] SHANG L, YU H J, HUANG X, et al. Well-dispersed ZIF-derived Co, N-co-doped carbon nanoframes through mesoporous-silica-protected calcination as efficient oxygen reduction electrocatalysts[J]. Advanced Materials,2016,28(8):1668-1674. doi: 10.1002/adma.201505045 [13] CHEN L Y, XU Q. Metal-organic framework composites for catalysis[J]. Matter,2019,1(1):57-89. doi: 10.1016/j.matt.2019.05.018 [14] CHEN Z L, WU R B, LIU Y, et al. Ultrafine Co nanoparticles encapsulated in carbon-nanotubes-grafted graphene sheets as advanced electrocatalysts for the hydrogen evolution reaction[J]. Advanced Materials,2018,30(30):1802011. doi: 10.1002/adma.201802011 [15] HE N P, LI C, ZHAO X Z, et al. The lamellar MOFs@polymer networks hybrids fabricated in reversed microemulsion for efficient CO2 capture[J]. Polymers Advanced Technologies,2021,33(3):750-759. [16] TEPLENSKY M H, FANTHAM M, POUDEL C, et al. A highly porous metal-organic framework system to deliver payloads for gene knockdown[J]. Chem,2019,5(11):2926-2941. doi: 10.1016/j.chempr.2019.08.015 [17] YANG S L, KARVE V V, JUSTIN A, et al. Enhancing MOF performance through the introduction of polymer guests[J]. Coordination Chemistry Reviews,2021,427:213525. doi: 10.1016/j.ccr.2020.213525 [18] 乔瑶雨, 张学辉, 赵晓竹, 等. 石墨烯/金属-有机框架复合材料制备及其应用[J]. 化学进展, 2022, 34(5):1181-1190.QIAO Yaoyu, ZHANG Xuehui, ZHAO Xiaozhu, et al. Preparation and application of graphene/metal-organic frameworks composites[J]. Progress in Chemistry,2022,34(5):1181-1190(in Chinese). [19] GEIM A K, NOVOSELOV K S. The rise of graphene[J]. Nature Materials,2007,6:183-191. doi: 10.1038/nmat1849 [20] QIAO Y Y, HE N P, ZHANG X H, et al. In situ growth of MOF crystals to synthesize a graphene oxide/ZIF-7 gel with enhanced adsorption capacity for methylene blue[J]. New Journal of Chemistry,2022,46(29):14103-14111. doi: 10.1039/D2NJ02293A [21] TKACHEV S V, BUSLAEVA E Y, NAUMKIN A V, et al. Reduced graphene oxide[J]. Inorganic Materials,2012,48(8):796-802. doi: 10.1134/S0020168512080158 [22] PARK S, AN J, JUNG I, et al. Colloidal suspensions of highly reduced graphene oxide in a wide variety of organic solvents[J]. Nano Letters,2009,9(4):1593-1597. doi: 10.1021/nl803798y [23] KUMAR G, MASRAM D T. Sustainable synthesis of MOF-5@GO nanocomposites for efficient removal of rhodamine B from water[J]. ACS Omega,2021,6(14):9587-9599. doi: 10.1021/acsomega.1c00143 [24] ZHENG Y, ZHENG S S, XUE H G, et al. Metal-organic frameworks/graphene-based materials: Preparations and applications[J]. Advanced Functional Materials,2018,28(47):1804950. [25] PARK J S, GOO N I, KIM D E. Mechanism of DNA adsorption and desorption on graphene oxide[J]. Langmuir,2014,30(42):12587-12595. doi: 10.1021/la503401d [26] CAI W X, LEE T, LEE M, et al. Thermal structural transitions and carbon dioxide adsorption properties of zeolitic imidazolate framework-7 (ZIF-7)[J]. Journal of the American Chemical Society,2014,136(22):7961-7971. doi: 10.1021/ja5016298 [27] XIAO T, LIU D X. Progress in the synthesis, properties and applications of ZIF-7 and its derivatives[J]. Materials Today Energy,2019,14:100357. doi: 10.1016/j.mtener.2019.100357 [28] ZHENG J, CHENG C, FANG W J, et al. Surfactant-free synthesis of a Fe3O4@ZIF-8 core-shell heterostructure for adsorption of methylene blue[J]. CrystEngComm,2014,16(19):3960-3964. doi: 10.1039/c3ce42648c [29] PETIT C, BURRESS J, BANDOSZ T J. The synthesis and characterization of copper-based metal-organic framework/graphite oxide composites[J]. Carbon,2011,49(2):563-572. doi: 10.1016/j.carbon.2010.09.059 [30] LI S S, DAI J, YAN Q, et al. Effect of zeolitic imidazole framework (ZIFs) shells of core-shell microspheres on adsorption of Roselle red dye from water[J]. Inorganic Chemistry Communications,2018,97:113-118. doi: 10.1016/j.inoche.2018.09.015 [31] MARCANO D C, KOSYNKIN D V, BERLIN J M, et al. Improved synthesis of graphene oxide[J]. ACS Nano,2010,4(8):4806-4814. doi: 10.1021/nn1006368 [32] STOCK N, BISWAS S. Synthesis of metal-organic frameworks (MOFs): Routes to various MOF topologies, morphologies, and composites[J]. Chemical Reviews,2012,112(2):933-969. doi: 10.1021/cr200304e [33] KANG C H, LIN Y F, HUANG Y S, et al. Synthesis of ZIF-7/chitosan mixed-matrix membranes with improved separation performance of water/ethanol mixtures[J]. Journal of Membrane Science,2013,438:105-111. doi: 10.1016/j.memsci.2013.03.028 [34] YANG Q X, LU R, REN S S, et al. Three dimensional reduced graphene oxide/ZIF-67 aerogel: Effective removal cationic and anionic dyes from water[J]. Chemical Engineering Journal,2018,348:202-211. doi: 10.1016/j.cej.2018.04.176 [35] RATTANA, CHAIYAKUN S, WITIT-ANUN N, et al. Preparation and characterization of graphene oxide nanosheets[J]. Procedia Engineering,2012,32:759-764. doi: 10.1016/j.proeng.2012.02.009 [36] STRANKOWSKI M, WŁODARCZYK D, PISZCZYK Ł, et al. Polyurethane nanocomposites containing reduced graphene oxide, FTIR, Raman, and XRD studies[J]. Journal of Spectroscopy,2016,2016:7520741. doi: 10.1155/2016/7520741 [37] HUANG A S, LIU Q, WANG N Y, et al. Bicontinuous zeolitic imidazolate framework ZIF-8@GO membrane with enhanced hydrogen selectivity[J]. Journal of the American Chemical Society,2014,136(42):14686-14689. doi: 10.1021/ja5083602 [38] SAHIN F, TOPUZ B, KALIPCILAR H. Synthesis of ZIF-7, ZIF-8, ZIF-67 and ZIF-L from recycled mother liquors[J]. Microporous and Mesoporous Materials,2018,261:259-267. doi: 10.1016/j.micromeso.2017.11.020 [39] JIANG D N, CHEN M, WANG H, et al. The application of different typological and structural MOFs-based materials for the dyes adsorption[J]. Coordination Chemistry Reviews,2019,380:471-483. doi: 10.1016/j.ccr.2018.11.002 [40] GUO H Y, JIAO T F, ZHANG Q R, et al. Preparation of graphene oxide-based hydrogels as efficient dye adsorbents for wastewater treatment[J]. Nanoscale Research Letters,2015,10(1):272. doi: 10.1186/s11671-015-0931-2 [41] HASAN Z, JHUNG S H. Removal of hazardous organics from water using metal-organic frameworks (MOFs): Plausible mechanisms for selective adsorptions[J]. Journal of Hazardous Materials,2015,283:329-339. doi: 10.1016/j.jhazmat.2014.09.046 [42] JABBARI V, VELETA J M, ZAREI-CHALESHTORI M, et al. Green synthesis of magnetic MOF@GO and MOF@CNT hybrid nanocomposites with high adsorption capacity towards organic pollutants[J]. Chemical Engineering Journal,2016,304:774-783. doi: 10.1016/j.cej.2016.06.034 -





 下载:
下载: