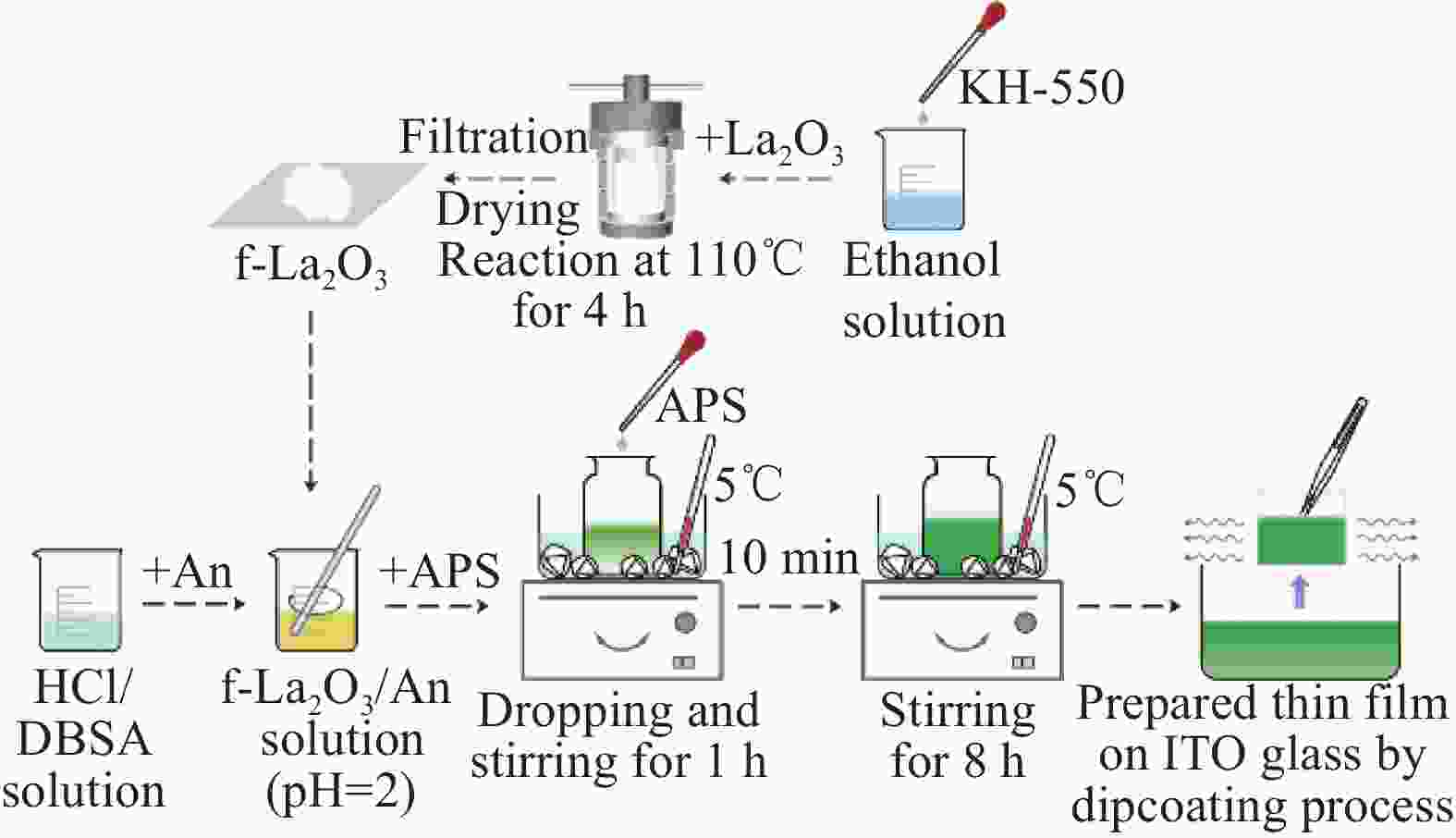
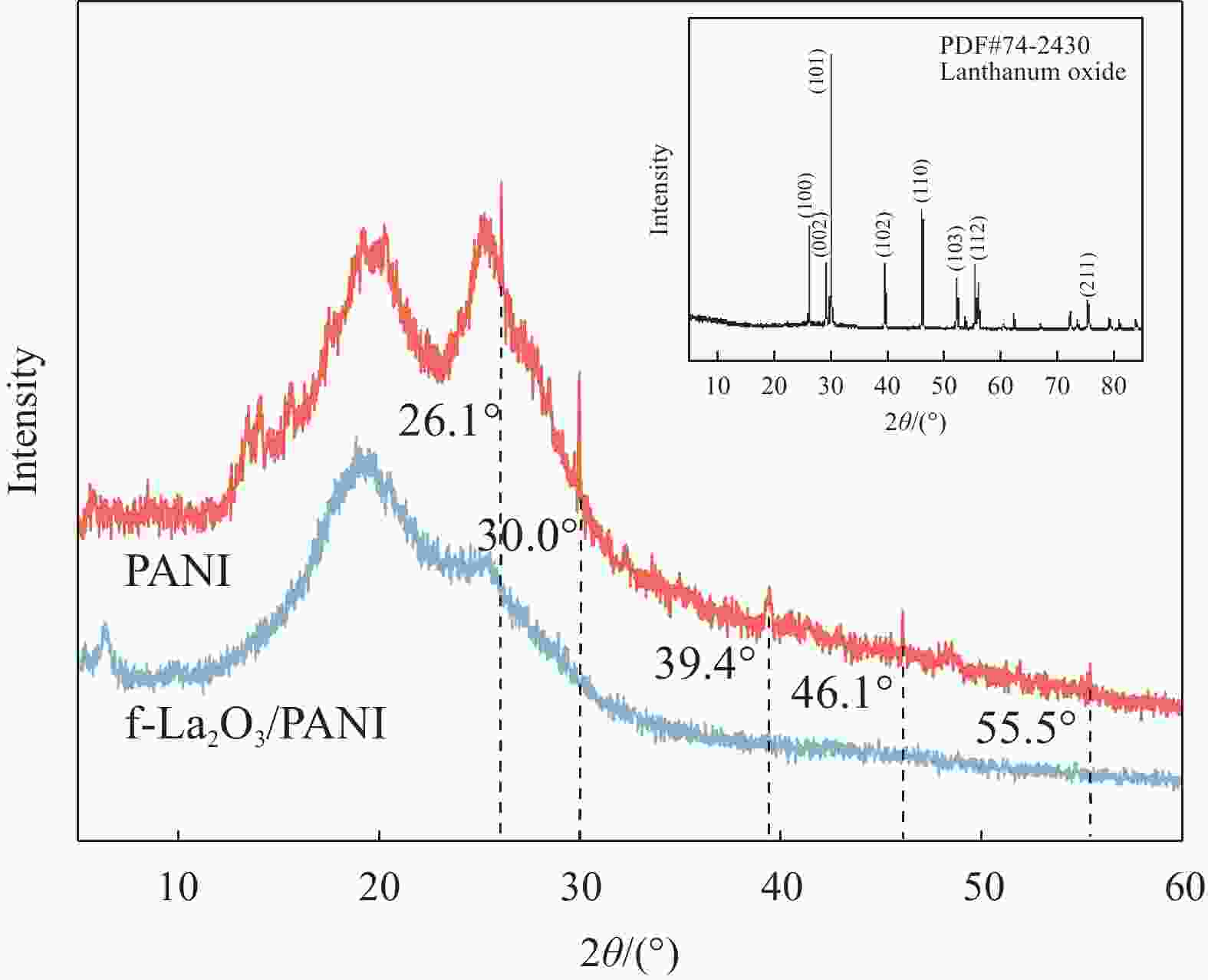
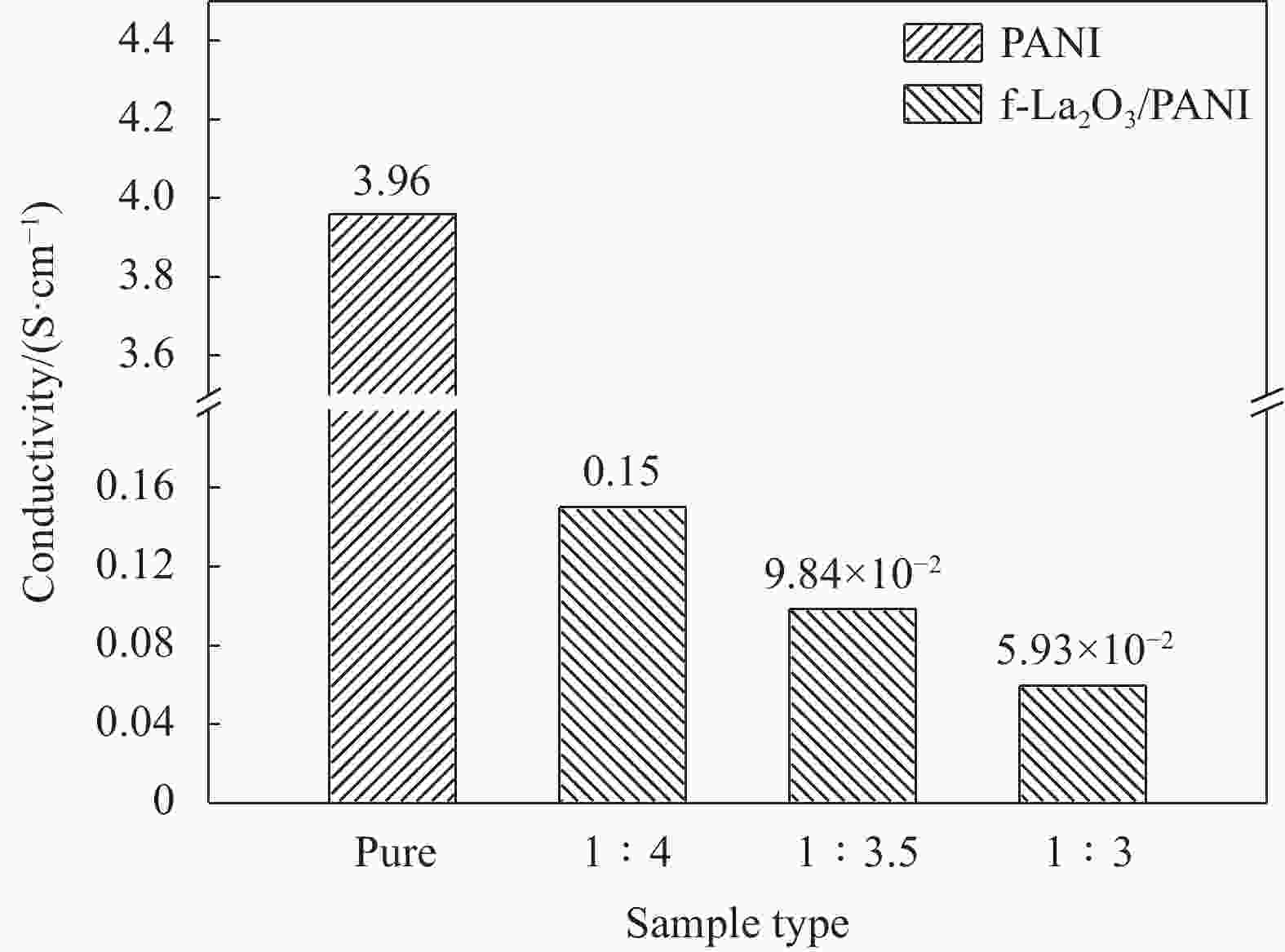
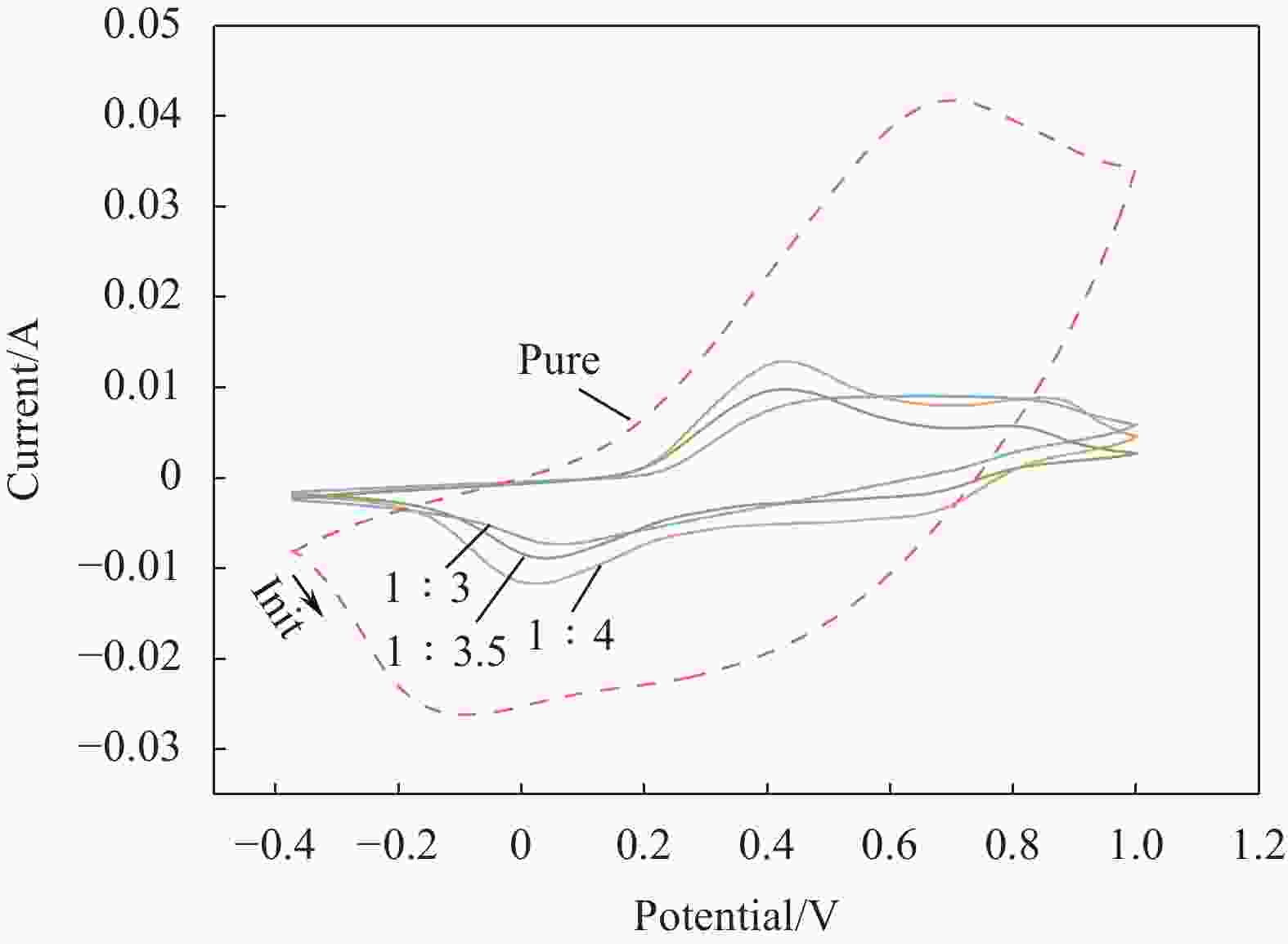
Preparation and characterization of electrochromic thin films based on fast response f-La2O3/PANI
-
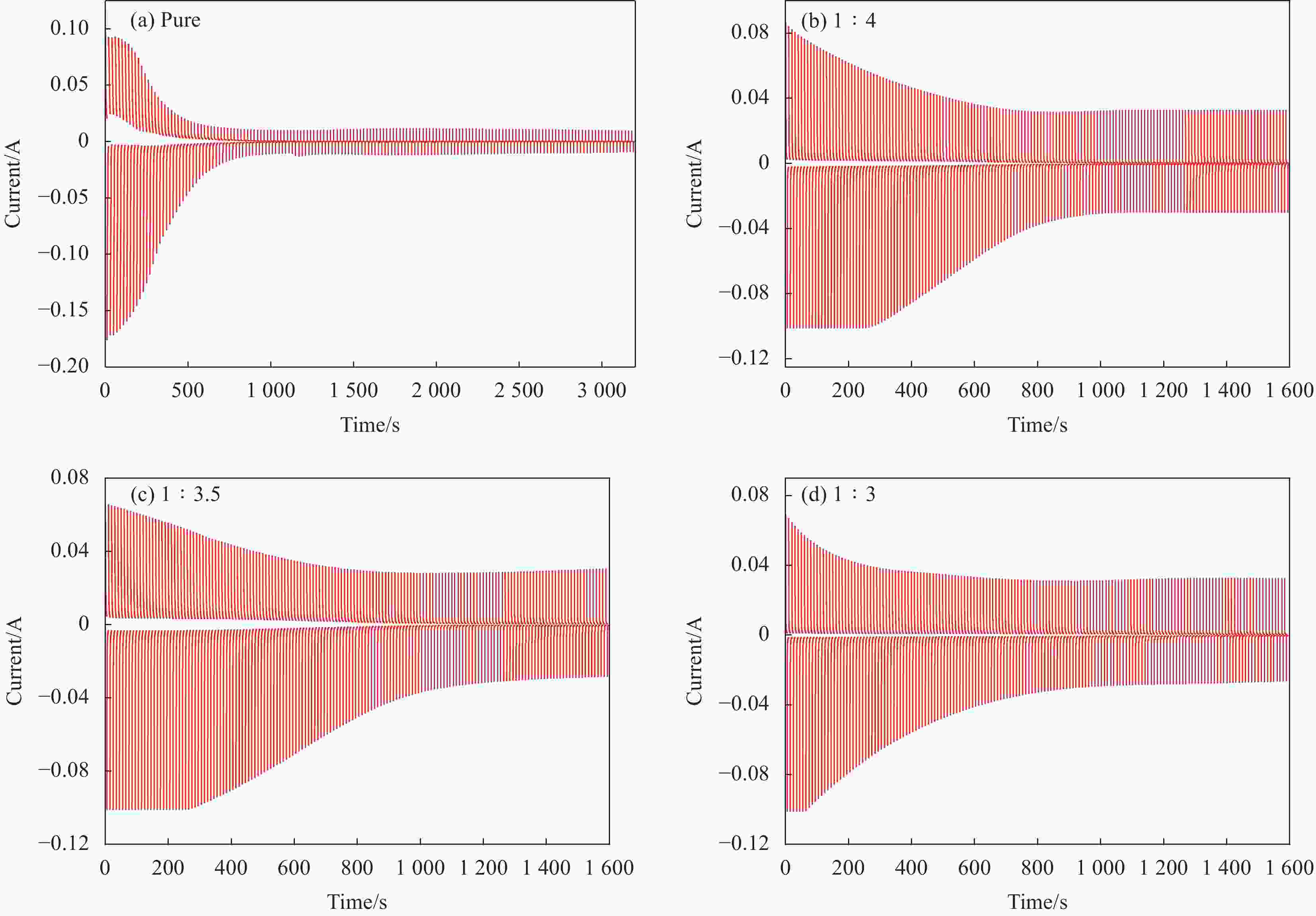
摘要: 基于电致变色薄膜在军事伪装、建筑节能、汽车等工业领域具有重要研发前景,研究通过硅烷偶联剂KH-550对氧化镧(La2O3)表面进行修饰(f-La2O3),并与导电聚合物聚苯胺(PANI)复合制备f-La2O3/PANI电致变色材料。利用XRD、FTIR、SEM-EDS、UV-vis、电化学工作站对f-La2O3/PANI电极与纯PANI电极进行对比分析,重点研究f-La2O3掺入量对PANI结构形貌、电化学性能及电致变色性能的影响。研究结果表明:f-La2O3的掺入使PANI纤维有向细小化方向变化的趋势,其复合材料较纯PANI具有更高的结晶度和分子链取向;f-La2O3会打破PANI的网络交联结构导致复合材料的电化学性能有所降低,但可以加快PANI质子化和脱质子化的转变进程,并有效抑制薄膜电致变色过程中PANI的氧化降解;当镧胺比1∶3.5时,f-La2O3/PANI复合材料的电致变色性能达到最佳,波长570 nm处的着色效率(CE)为22.81 cm2·C−1,褪色、着色响应时间(τb/τc)分别为1.29 s、1.33 s,经320次着褪色循环后薄膜电化学活性保持在初始的50%左右。Abstract: Based on the fact that electrochromic films have important research and development prospects in military camouflage, building energy saving, automobiles and other industrial fields, the surface of La2O3 was modified by silane coupling agent KH-550 and compounded with conductive polymer polyaniline (PANI) to prepare f-La2O3/PANI electrochromic material. Comparative analysis of composite electrode and pure PANI electrode by XRD, FTIR, SEM-EDS, UV-vis, and electrochemical workstation. The effect of f-La2O3 addition on the structure, morphology, electrochemical properties and electrochromic properties of PANI was investigated. The results show that the incorporation of f-La2O3 makes the PANI fibers tend to be smaller, and the composite material has higher crystallinity and molecular chain orientation than pure PANI; f-La2O3 will break the network cross-linking structure of PANI, resulting in a decrease in the electrochemical performance of the composite, but it can accelerate the transition process of PANI protonation and deprotonation, and effectively inhibit the oxidative degradation of PANI during the electrochromic process of the film; When f-La2O3/aniline molar ratio is 1∶3.5, the electrochromic performance of f-La2O3/PANI composite reaches the best, the coloration efficiency (CE) at 570 nm is 22.81 cm2·C−1, and the fading and coloring response time (τb, τc) are 1.29 s and 1.33 s, respectively. After 320 cycles of fading, the electrochemical activity of the thin films remains about 50% of the initial value.
-
Key words:
- lanthanum oxide /
- polyaniline /
- electrochromic /
- response time /
- cycling stability
-
图 5 PANI与f-La2O3/PANI薄膜表面和截面的SEM图像:(a) 功能化前后的La2O3;(b) 纯 PANI ;f-La2O3/An摩尔比1∶4 (c)、1∶3.5 (含局部Mapping图) (d)、1∶3 (e)
Figure 5. SEM images of the surface and cross-section of PANI and f-La2O3/PANI films: (a) La2O3 before and after functionalization; (b) Pure PANI ; f-La2O3/An molar ratio1∶4 (c), 1∶3.5 (with local Mapping images) (d), 1∶3 (e)
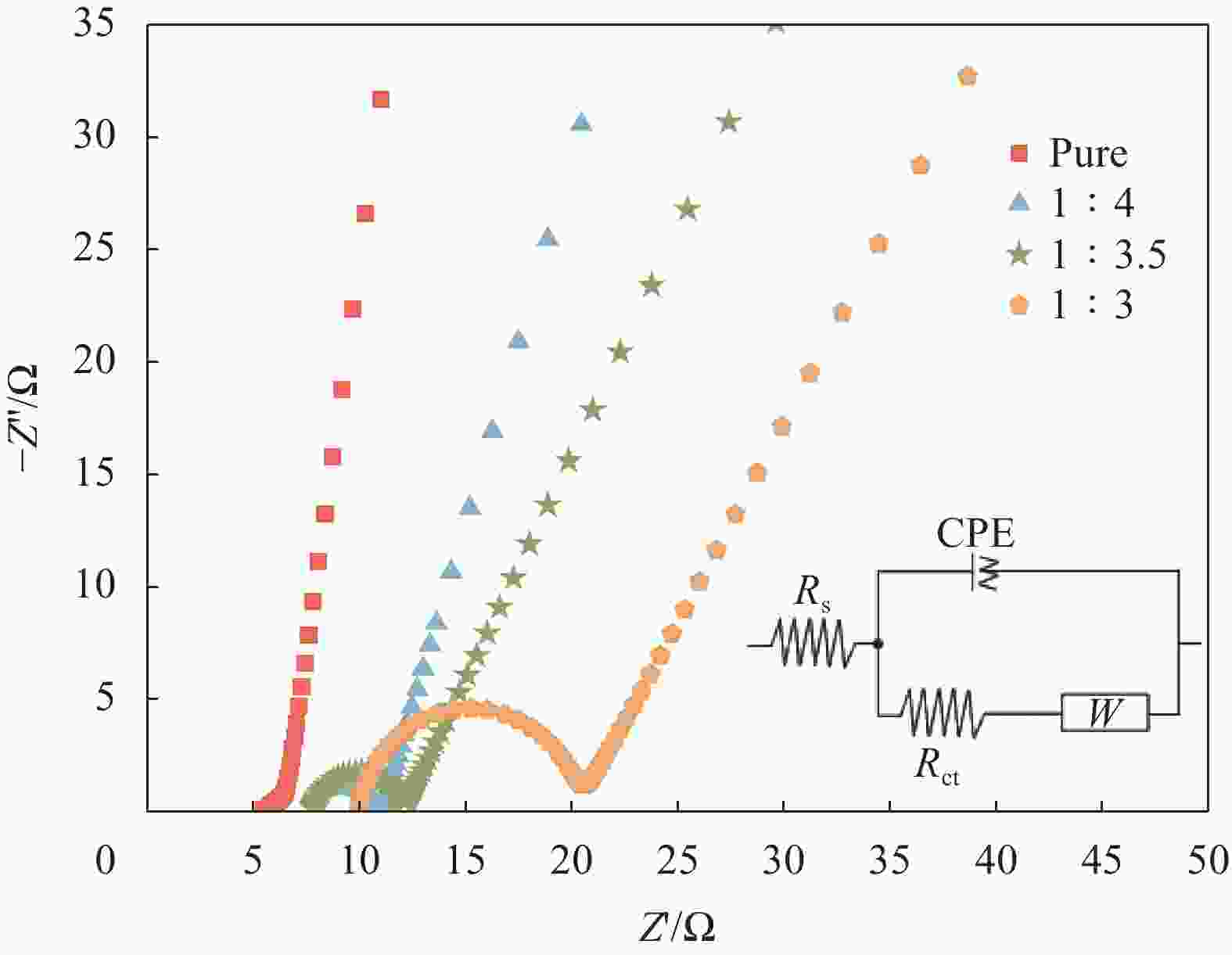
图 8 PANI和f-La2O3/PANI薄膜电极(f-La2O3/An摩尔比1∶4、1∶3.5、1∶3)的能奎斯特曲线和等效电路图
Figure 8. Nyquist curves and equivalent circuit diagram of PANI and f-La2O3/PANI films electrode (f-La2O3/An molar ratio 1∶4, 1∶3.5, 1∶3)
Z'—Real part of impedance; Z''—Imaginary part of impedance; Rs—Solution resistance; Rct—Charge transfer resistance; CPE—Nonideal capacitance; W—Warburg Impendance
表 1 功能化La2O3(f-La2O3)/聚苯胺(PANI)复合材料的原料配比设计
Table 1. Raw material ratio design of functionalization-La2O3 (f-La2O3)/polyaniline (PANI) composite material
Molar ratio
f-La2O3∶AnAn/g f-La2O3/g DBSA/g HCl/g APS/g Pure 4.66 0.00 1.41 5.7 11.4 1∶4 4.66 4.00 1.41 5.7 11.4 1∶3.5 4.66 4.65 1.41 5.7 11.4 1∶3 4.66 5.43 1.41 5.7 11.4 Notes: An—Aniline; DBSA—Dodecylbenzenesulphonic acid; APS—Ammonium persulphate. 表 2 570 nm处PANI和f-La2O3/PANI薄膜的着色效率相关参数
Table 2. Parameters related to coloring efficiency of PANI and f-La2O3/PANI films at 570 nm
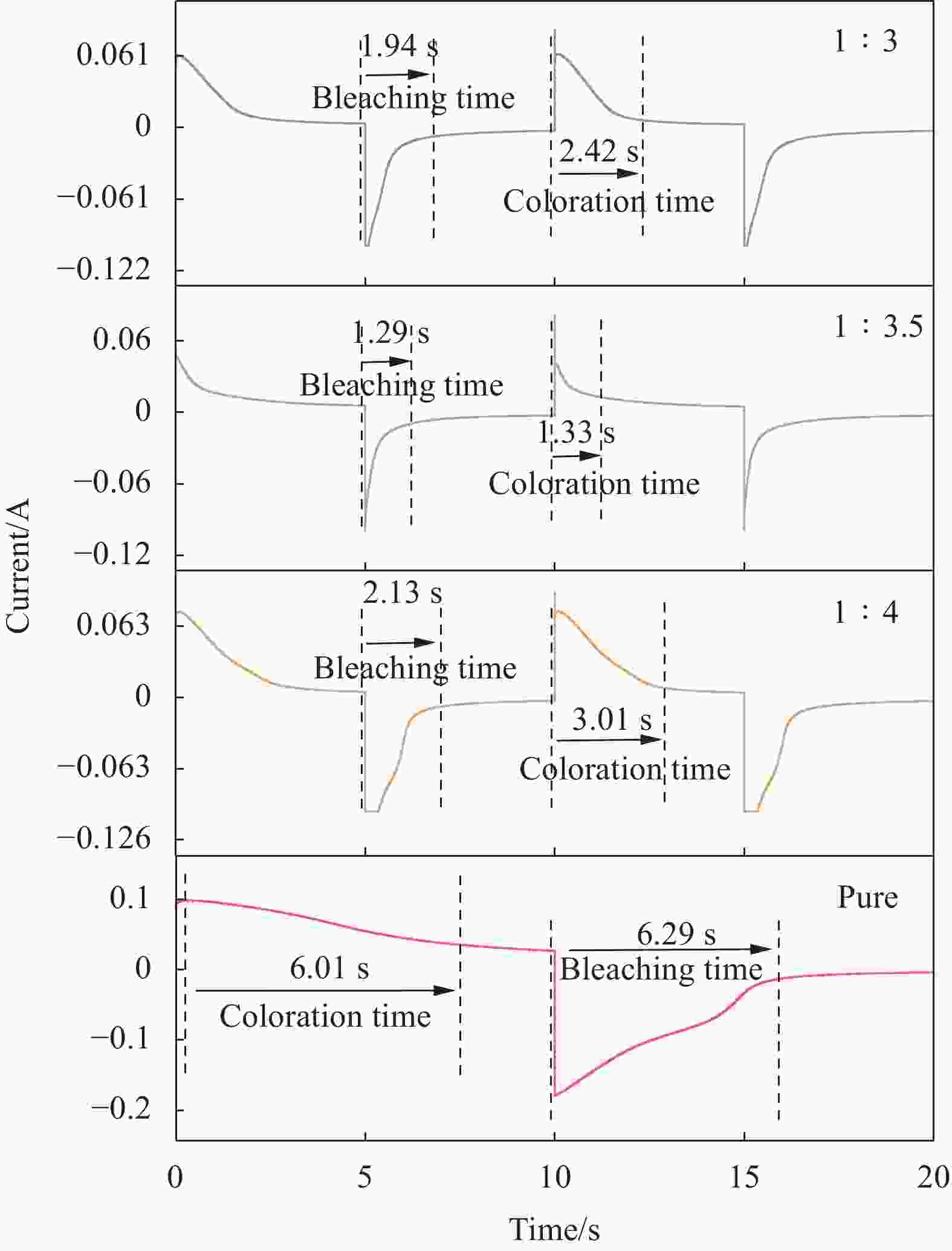
Sample Tb/% Tc/% τb/τc/s ΔOD Qin/(C·cm−2) CE(λ)/(cm2·C−1) Type Molar ratio La2O3∶An PANI — 83.0 48.4 6.29/6.01 0.23 0.1820 1.29 f-La2O3/PANI 1∶4 73.1 47.5 2.13/3.01 0.19 0.0438 4.27 1∶3.5 62.8 35.8 1.29/1.33 0.24 0.0107 22.81 1∶3 60.9 39.9 1.94/2.42 0.18 0.0275 6.68 Notes: Tb/Tc—Faded/colored state film transmittance; τb/τc—Faded/colored state film response time; ΔOD—Optical density change; Qin—Charge injection rate; CE(λ)—Coloration efficiency; λ—Wavelength; When the voltage is −0.4 V, it is in the faded state, and +0.6 V is in the colored state; S=2.52 cm2. -
[1] KORENT A, SODERŽNIK K Ž, ŠTURM S, et al. A correlative study of polyaniline electropolymerization and its electrochromic behavior[J]. Journal of the Electrochemical Society,2020,167(10):106504. doi: 10.1149/1945-7111/ab9929 [2] 张翔, 李文杰, 李森然, 等. 自供能电致变色器件研究进展[J]. 复合材料学报, 2021, 38(6): 1724-1733.ZHANG Xiang, LI Wenjie, LI Senran, et al. Research process in self-powered electrochromic devices[J]. Acta Materiae Compositae Sinica, 2021, 38(6): 1724-1733(in chinese). [3] KELLY F M, MEUNIER L, COCHRANE C, et al. Polyaniline: Application as solid state electrochromic in a flexible textile display[J]. Displays,2013,34(1):1-7. doi: 10.1016/j.displa.2012.10.001 [4] WANG Y, GONG Z, ZENG Y, et al. High-properties electrochromic device based on TiO2@graphene/prussian blue core-shell nanostructures[J]. Chemical Engineering Journal,2022,431:134066. doi: 10.1016/j.cej.2021.134066 [5] JAROSZ T, GEBKA K, STOLARCZYK A, et al. Transparent to black electrochromism—The “holy grail” of organic optoelectronics[J]. Polymers,2019,11(2):273. doi: 10.3390/polym11020273 [6] WANG H, YAO C J, NIE H J, et al. Recent progress in integrated functional electrochromic energy storage devices[J]. Journal of Materials Chemistry C,2020,8(44):15507-15525. doi: 10.1039/D0TC03934A [7] ZHOU S, WANG S, ZHOU S, et al. An electrochromic supercapacitor based on an MOF derived hierarchical-porous NiO film[J]. Nanoscale,2020,12(16):8934-8941. doi: 10.1039/D0NR01152E [8] WANG Z, WANG X, CONG S, et al. Fusing electrochromic technology with other advanced technologies: A new roadmap for future development[J]. Materials Science and Engineering: R: Reports,2020,140:100524. doi: 10.1016/j.mser.2019.100524 [9] CABUK M, GÜNDÜZ B. Controlling the optical properties of polyaniline doped by boric acid particles by changing their doping agent and initiator concentration[J]. Applied Surface Science,2017,424:345-351. doi: 10.1016/j.apsusc.2017.03.010 [10] VAN N T, DO H H, TRUNG T Q, et al. Stable and multicolored electrochromic device based on polyaniline-tungsten oxide hybrid thin film[J]. Journal of Alloys and Compounds,2021,882:160718. doi: 10.1016/j.jallcom.2021.160718 [11] ZHANG L, LI D, LI X, et al. Further explore on the behaviors of IR electrochromism of a double layer constructed by proton acid-doped polyaniline film and ITO layer[J]. Dyes and Pigments,2019,170:107570. doi: 10.1016/j.dyepig.2019.107570 [12] ZHANG S, CHEN S, HU F, et al. Spray-processable, large-area, patterned and all-solid-state electrochromic device based on silica/polyaniline nanocomposites[J]. Solar Energy Materials and Solar Cells,2019,200:109951. doi: 10.1016/j.solmat.2019.109951 [13] LIU D Y, SUI G X, BHATTACHARYYA D. Synthesis and characterisation of nanocellulose-based polyaniline conducting films[J]. Composites Science and Technology,2014,99:31-36. doi: 10.1016/j.compscitech.2014.05.001 [14] BHADRA S, KHASTGIR D, SINGHA N K, et al. Progress in preparation, processing and applications of polyaniline[J]. Progress in Polymer Science,2009,34(8):783-810. doi: 10.1016/j.progpolymsci.2009.04.003 [15] PENG H, YAN B, JIANG M, et al. A coral-like polyaniline/barium titanate nanocomposite electrode with double electric polarization for electrochromic energy storage applications[J]. Journal of Materials Chemistry A,2021,9(3):1669-1677. doi: 10.1039/D0TA08263E [16] JAMDEGNI M, KAUR A. Highly efficient dark to transparent electrochromic electrode with charge storing ability based on polyaniline and functionalized nickel oxide composite linked through a binding agent[J]. Electrochimica Acta,2020,331:135359. doi: 10.1016/j.electacta.2019.135359 [17] NWANYA A C, JAFTA C J, EJIKEME P M, et al. Electrochromic and electrochemical capacitive properties of tungsten oxide and its polyaniline nanocomposite films obtained by chemical bath deposition method[J]. Electrochimica Acta,2014,128:218-225. doi: 10.1016/j.electacta.2013.10.002 [18] XIONG S, WANG R, LI S, et al. Electrochromic behaviors of water-soluble polyaniline with covalently bonded acetyl ferrocene[J]. Journal of Electronic Materials,2018,47(7):3974-3982. doi: 10.1007/s11664-018-6281-y [19] JAMDEGNI M, KAUR-GHUMAAN S, KAUR A. Study of polyaniline and functionalized ZnO composite film linked through a binding agent for efficient and stable electrochromic applications[J]. Electrochimica Acta,2017,252:578-588. doi: 10.1016/j.electacta.2017.08.144 [20] POUGET J P, JOZEFOWICZ M E, EPSTEIN A J, et al. X-ray structure of polyaniline[J]. Macromolecules,1991,24(3):779-789. doi: 10.1021/ma00003a022 [21] DINARI M, MOMENI M M, GOUDARZIRAD M. Dye-sensitized solar cells based on nanocomposite of polyaniline/graphene quantum dots[J]. Journal of Materials Science,2016,51(6):2964-2971. doi: 10.1007/s10853-015-9605-9 [22] HE Y. Synthesis of polyaniline/nano-CeO2 composite microspheres via a solid-stabilized emulsion route[J]. Materials Chemistry and Physics,2005,92(1):134-137. doi: 10.1016/j.matchemphys.2005.01.033 [23] 张云浩, 翟兰兰, 王彦, 等. 硅烷偶联剂KH-570表面改性纳米SiO2[J]. 材料科学与工程学报, 2012, 30(5):752-756.ZHANG Yunhao, ZHAI Lanlan, WANG Yan, et al. Surface modification of nano-SiO2 by silane coupling agent 3-(methacryloyloxy)propyltrimethoxysilane[J]. Journal of Materials Science & Engineering,2012,30(5):752-756(in Chinese). [24] NOROUZIAN R S, LAKOURAJ M M. Polyaniline-thiacalix[4]arene metallopolymer, self-doped, and externally doped conductive polymers[J]. Progress in Organic Coatings,2020,146:105731. doi: 10.1016/j.porgcoat.2020.105731 [25] MARTINS J C, NETO J C M, PASSOS R R, et al. Electrochemical behavior of polyaniline: A study by electrochemical impedance spectroscopy (EIS) in low-frequency[J]. Solid State Ionics,2020,346:115198. doi: 10.1016/j.ssi.2019.115198 [26] ZHANG S, SUN G, HE Y, et al. Preparation, characterization, and electrochromic properties of nanocellulose-based polyaniline nanocomposite films[J]. ACS Applied Materials & Interfaces,2017,9(19):16426-16434. [27] GU H, GUO C, ZHANG S, et al. Highly efficient, near-infrared and visible light modulated electrochromic devices based on polyoxometalates and W18O49 nanowires[J]. ACS Nano,2018,12(1):559-567. doi: 10.1021/acsnano.7b07360 -






 下载:
下载: