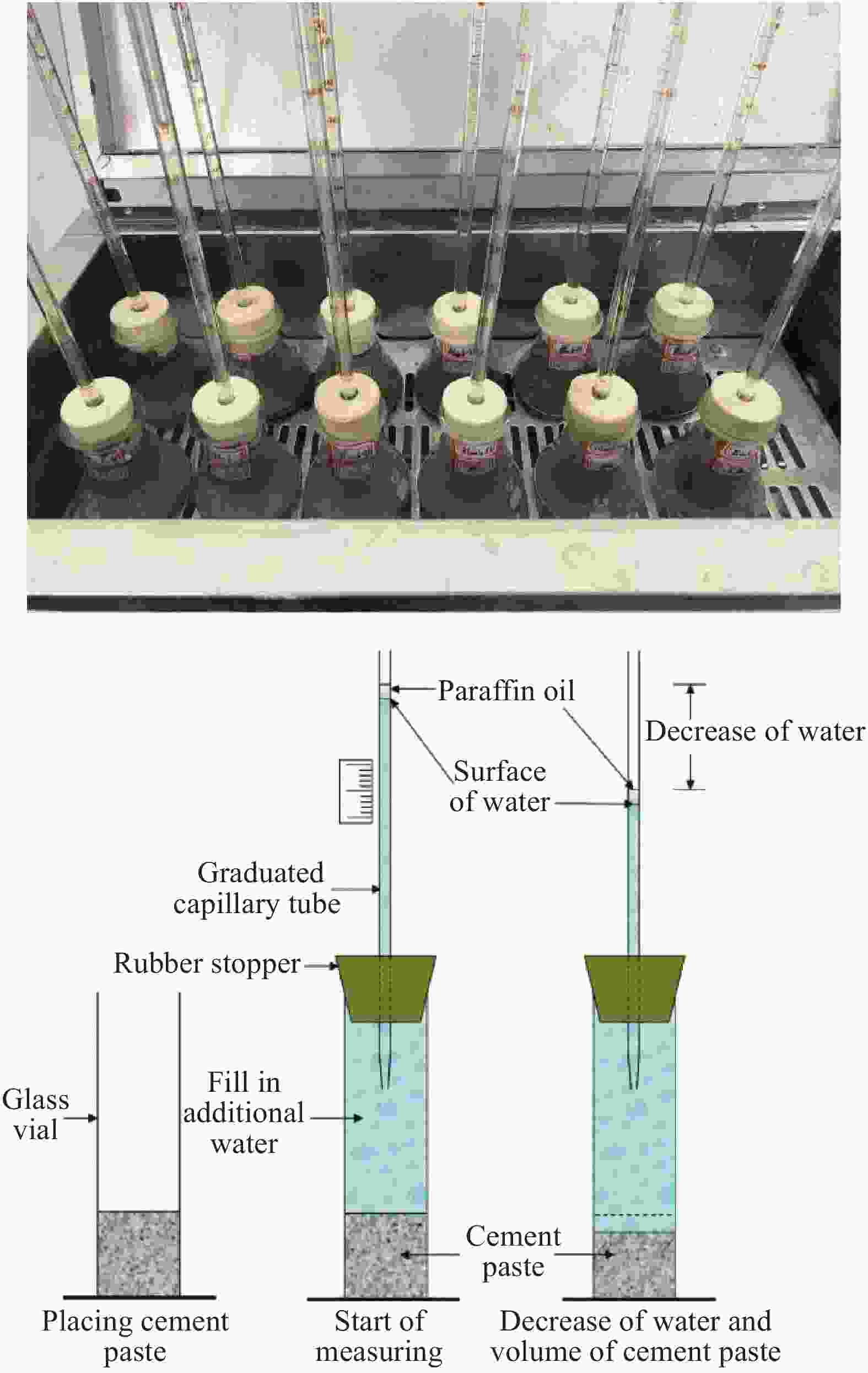
Chemical shrinkage behavior and prediction model of cement-based composite paste with the addition of graphene oxide
-
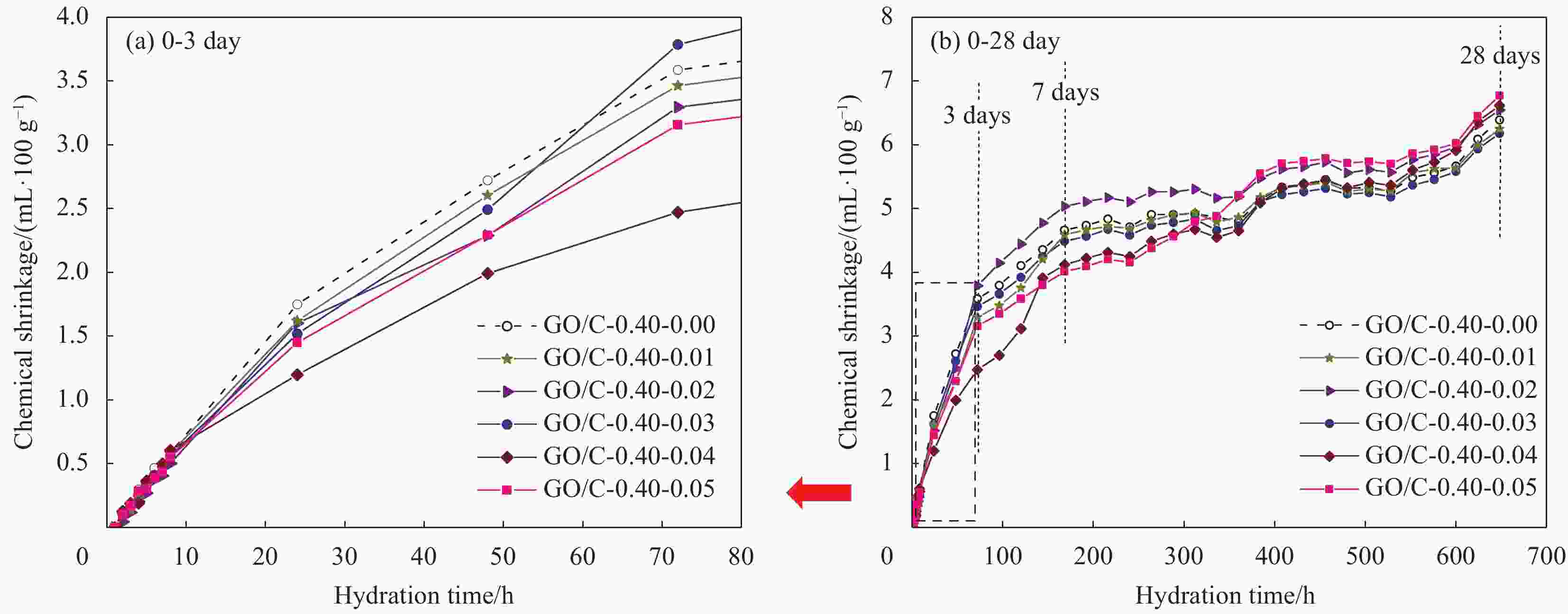
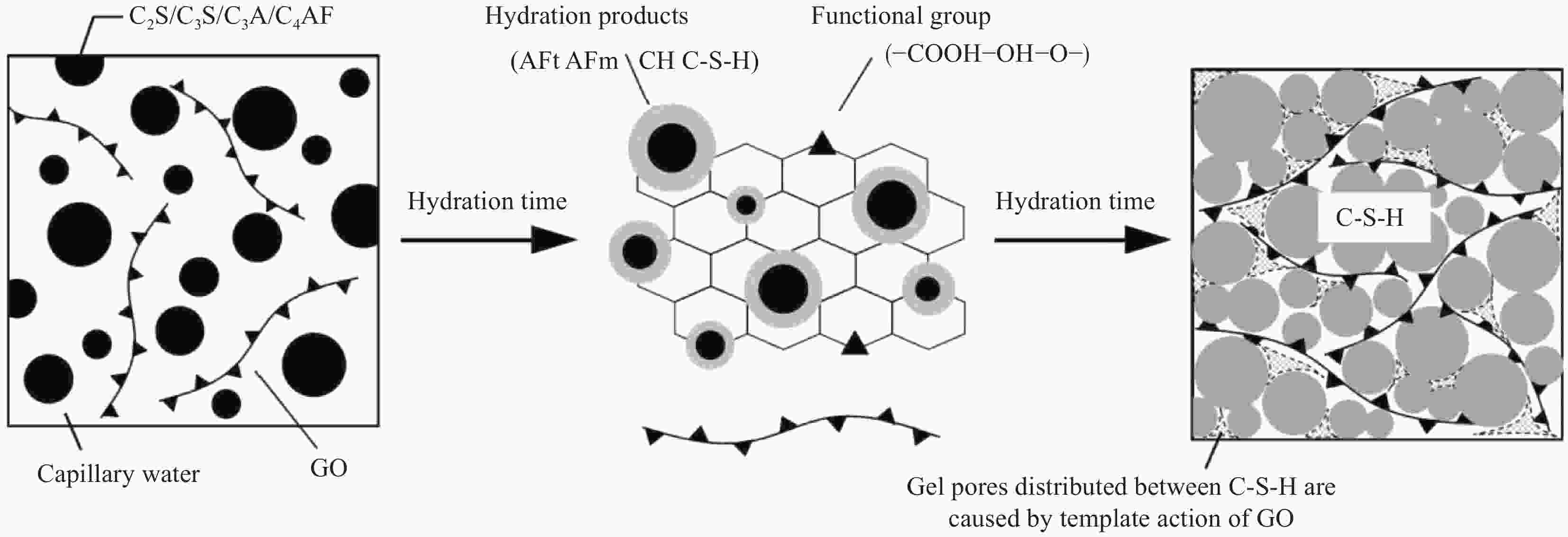
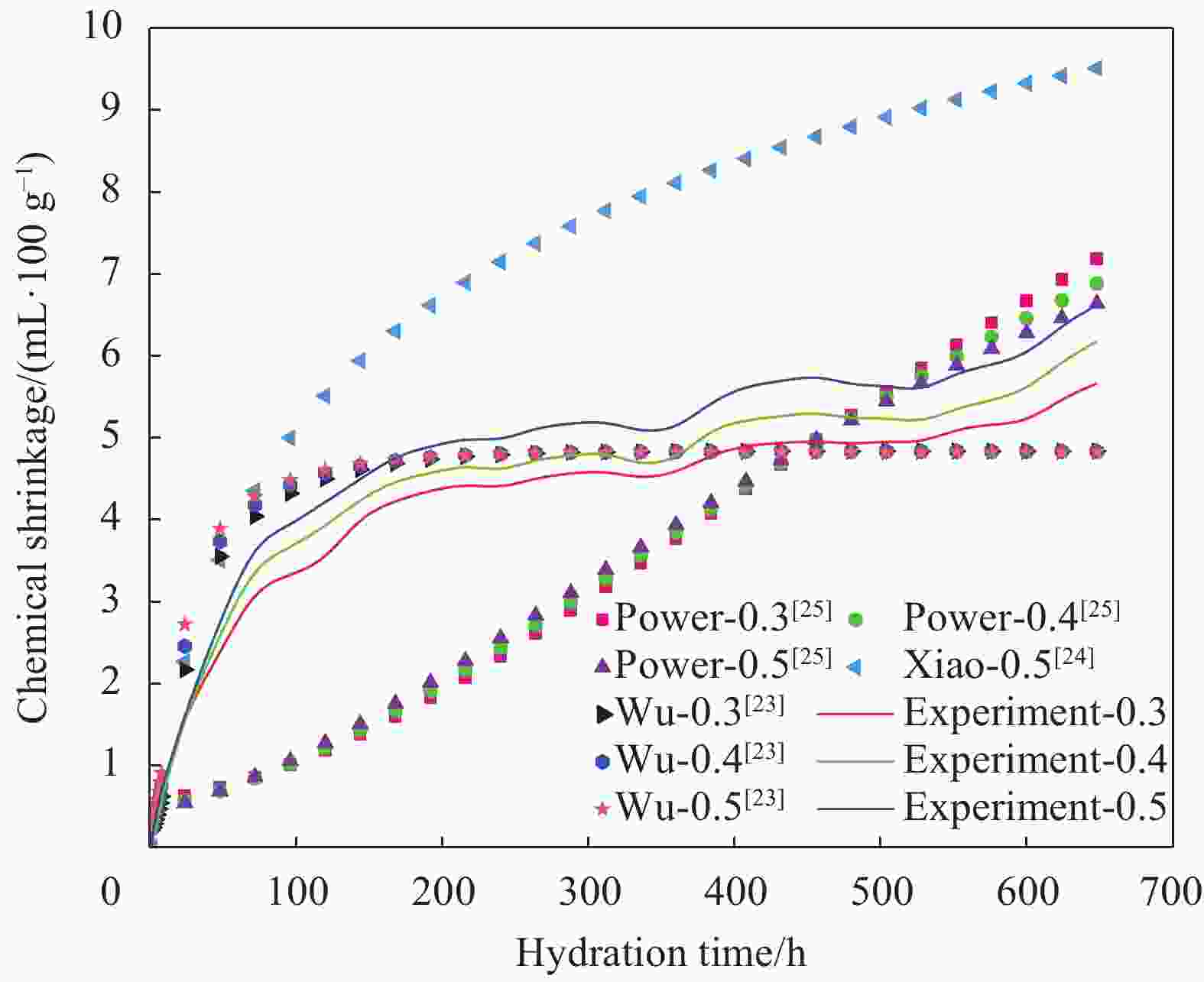
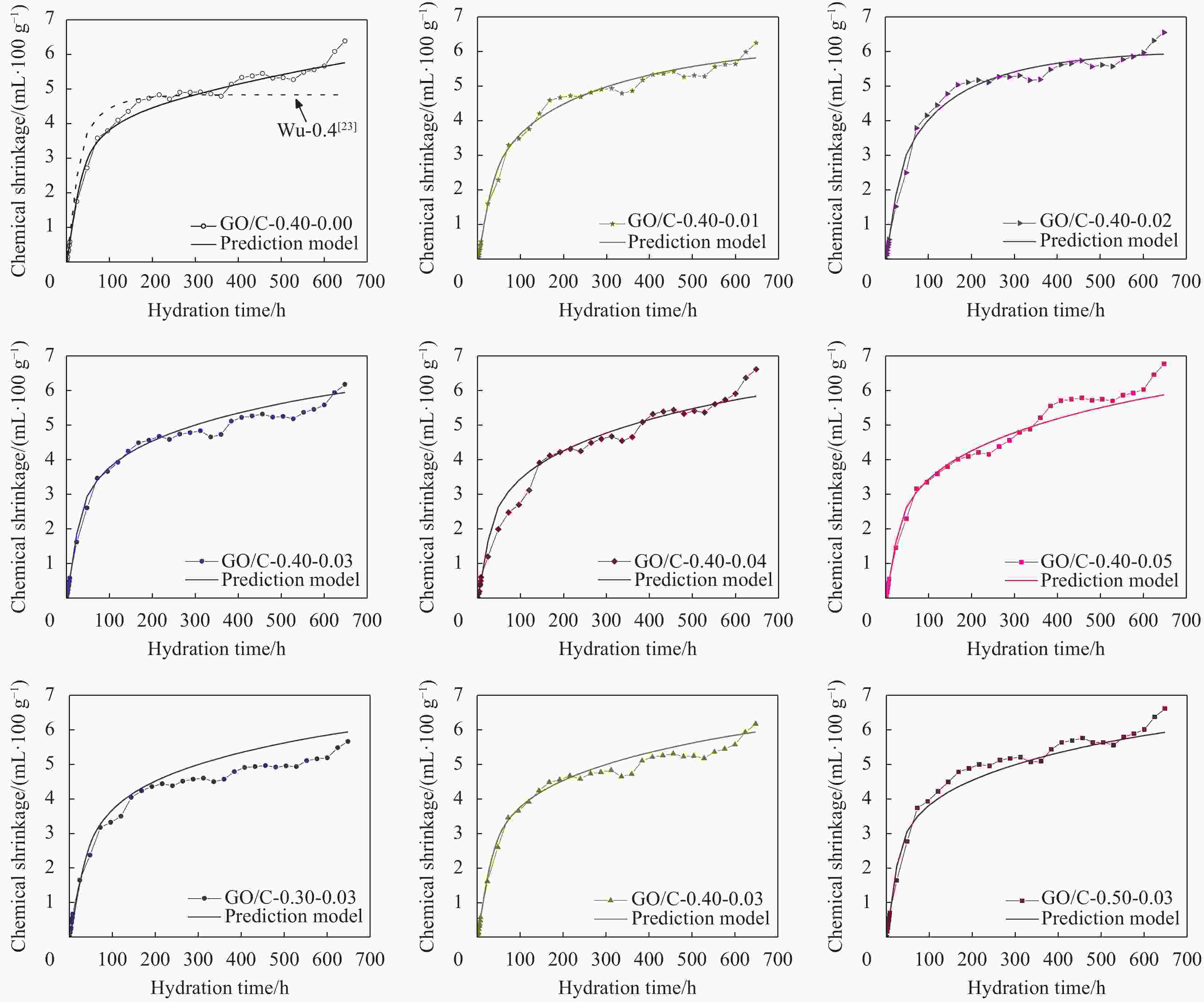
摘要: 为了揭示氧化石墨烯/水泥复合净浆(GO/C)的化学收缩特性,采用体积法对不同水灰比(0.3、0.4、0.5)和不同氧化石墨烯(GO)质量分数(0wt%、0.01wt%、0.02wt%、0.03wt%、0.04wt%、0.05wt%)的水泥复合净浆试件化学收缩进行了测定。试验结果显示:随着水灰比的增加,GO/C化学收缩显著增大。同水灰比条件下掺有氧化石墨烯的GO/C试件前期化学收缩较普通水泥净浆有所下降,当GO质量分数为0.04wt%时,收缩值达到最小;在后期水化中,GO/C试件的化学收缩增长速度明显快于普通水泥净浆。分析表明,GO对水泥净浆孔结构的调控作用和对水化产物氢氧化钙晶体的键合行为是其影响水泥净浆化学收缩性能的主要原因。同时,通过收缩模型与试验值对比发现,现有的普通水泥化学收缩模型无法精准预测GO/C的化学收缩情况,因此,为考虑GO的影响,试验在吴浪模型的基础上,引入
$K(\xi , t)$ 函数作为影响参数,并通过曲线拟合得到其具体表达式,从而建立起适用于GO/C化学收缩的预测模型。Abstract: In order to reveal the chemical shrinkage characteristics of graphene oxide cement composite paste (GO/C), the chemical shrinkage of cement composite paste with different water-cement ratio (0.3, 0.4, 0.5) and different mass fractions of graphene oxide (GO) (0wt%, 0.01wt%, 0.02wt%, 0.03wt%, 0.04wt% and 0.05wt%). The results show that the chemical shrinkage of GO/C is significantly increased with an increase in water-cement ratio. At the same water-cement ratio, the early chemical shrinkage of GO/C is lower than that of ordinary cement paste, and when the mass fraction of GO is 0.04wt%, the shrinkage value reaches the minimum. However, the growth rate of chemical shrinkage of GO/C specimens is obviously faster than that of ordinary cement paste in later hydration process. The analysis shows that the influence of GO on the chemical shrinkage of cement paste is mainly attributed to the regulation of GO on the pore structure of cement paste and the bonding behavior of GO with calcium hydroxide crystal. Furthermore, based on the comparison results of existing shrinkage models, it is found that the chemical shrinkage models of ordinary cement cannot accurately predict the chemical shrinkage of GO/C. Therefore, in order to consider the effect of GO,$K(\xi , t)$ function is introduced as an influence parameter on the basis of Wu Lang model, and its specific expression is obtained by curve fitting. Finally, a prediction model suitable for GO/C chemical shrinkage was established. -
表 1 水泥的主要技术指标
Table 1. The properties of cement.
Mass fraction/wt% Chemical analysis Density/
(g·cm−3)Specific
surface area/
(m2·kg−1)Compressive
strength at
28 days/MPaFlexural
strength at
28 days/MPaSiO2 Al2O3 Fe2O3 CaO MgO SO3 Loss of ignition 22.53 4.42 2.06 62.71 3.55 0.35 2.84 3.09 345 48.3 7.6 表 2 氧化石墨烯(GO)元素分析
Table 2. Elemental analysis of graphene oxide (GO)
Element mass fraction/wt% Carbon Hydrogen Nitrogen Sulphur Oxygen 49-56 0-1 0-1 0-2 41-50 表 3 水泥复合净浆配合比
Table 3. Mix proportions (by mass) of cement composite pastes
Group Number Water-cement
ratio ($w $/c)Dosage of
GO/wt%Ⅰ GO/C-0.30-0.03 0.30 0.03 GO/C-0.40-0.03 0.40 0.03 GO/C-0.50-0.03 0.50 0.03 Ⅱ GO/C-0.40-0.00 0.40 0.00 GO/C-0.40-0.01 0.40 0.01 GO/C-0.40-0.02 0.40 0.02 GO/C-0.40-0.03 0.40 0.03 GO/C-0.40-0.04 0.40 0.04 GO/C-0.40-0.05 0.40 0.05 表 4 四种矿物成分在不同水灰比环境下的水化参数
Table 4. Hydration parameters of four mineral components under different water cement ratios
Mineral component $w/c$ $ {\tau }_{X}({T}_{0})$ $m$ ${\alpha _{X{\rm{,0}}}}$ ${E_{{\rm{a}}X}}$ ${\alpha _X}(t)$ C3S 0.3 13.5 1.86 0.02 37.39 ${\alpha _{{{\rm{C}}_{\rm{3}}}{\rm{S}}}}(t) = {\rm{1}}{\rm{.02 - }}\exp ( - {({\rm{0}}{\rm{.0398(}}t - {t_{X,0}}))^{{\rm{1}}{\rm{.86}}}})$ 0.4 12.7 1.78 ${\alpha _{{{\rm{C}}_{\rm{3}}}{\rm{S}}}}(t) = {\rm{1}}{\rm{.02 - }}\exp ( - {({\rm{0}}{\rm{.0442(}}t - {t_{X,0}}))^{{\rm{1}}{\rm{.78}}}})$ 0.5 11.9 1.72 ${\alpha _{{{\rm{C}}_{\rm{3}}}{\rm{S}}}}(t) = {\rm{1}}{\rm{.02 - }}\exp ( - {({\rm{0}}{\rm{.0489(}}t - {t_{X,0}}))^{{\rm{1}}{\rm{.72}}}})$ C2S 0.3 71.2 1.1 0 20.78 ${\alpha _{{{\rm{C}}_{\rm{2}}}{\rm{S}}}}(t) = {\rm{1}}{\rm{.00 - }}\exp ( - {({\rm{0}}{\rm{.0128(}}t - {t_{X,0}}))^{{\rm{1}}{\rm{.10}}}})$ 0.4 65.3 1.04 ${\alpha _{{{\rm{C}}_{\rm{2}}}{\rm{S}}}}(t) = {\rm{1}}{\rm{.00 - }}\exp ( - {({\rm{0}}{\rm{.0147(}}t - {t_{X,0}}))^{{\rm{1}}{\rm{.04}}}})$ 0.5 60.9 0.96 ${\alpha _{{{\rm{C}}_{\rm{2}}}{\rm{S}}}}(t) = {\rm{1}}{\rm{.00 - }}\exp ( - {({\rm{0}}{\rm{.0171(}}t - {t_{X,0}}))^{{\rm{0}}{\rm{.96}}}})$ C3A 0.3 57.7 1.14 0.04 35.71 ${\alpha _{{{\rm{C}}_{\rm{3}}}{\rm{A}}}}(t) = {\rm{1}}{\rm{.04 - }}\exp ( - {({\rm{0}}{\rm{.0152(}}t - {t_{X,0}}))^{{\rm{1}}{\rm{.14}}}})$ 0.4 53.4 1.06 ${\alpha _{{{\rm{C}}_{\rm{3}}}{\rm{A}}}}(t) = {\rm{1}}{\rm{.04 - }}\exp ( - {({\rm{0}}{\rm{.0177(}}t - {t_{X,0}}))^{{\rm{1}}{\rm{.06}}}})$ 0.5 49.2 1.00 ${\alpha _{{{\rm{C}}_{\rm{3}}}{\rm{A}}}}(t) = {\rm{1}}{\rm{.04 - }}\exp ( - {({\rm{0}}{\rm{.0203(}}t - {t_{X,0}}))^{{\rm{1}}{\rm{.00}}}})$ C4AF 0.3 27.0 2.44 0.40 34.90 ${\alpha _{{{\rm{C}}_{\rm{4}}}{\rm{AF}}}}(t) = {\rm{1}}{\rm{.40 - }}\exp ( - {({\rm{0}}{\rm{.0152(}}t - {t_{X,0}}))^{{\rm{2}}{\rm{.44}}}})$ 0.4 23.9 2.38 ${\alpha _{{{\rm{C}}_{\rm{4}}}{\rm{AF}}}}(t) = {\rm{1}}{\rm{.40 - }}\exp ( - {({\rm{0}}{\rm{.0176(}}t - {t_{{\rm{X,0}}}}))^{{\rm{2}}{\rm{.38}}}})$ 0.5 21.4 2.30 ${\alpha _{{{\rm{C}}_{\rm{4}}}{\rm{AF}}}}(t) = {\rm{1}}{\rm{.40 - }}\exp ( - {({\rm{0}}{\rm{.0203(}}t - {t_{X,0}}))^{{\rm{2}}{\rm{.30}}}})$ Notes: τX(T0)—Characteristic time of the reaction at a constant reference temperature T0 = 293 K; m—Exponent that defines the reaction order; αX,0 and αX(t)—Degrees of hydration threshold of clinker X at the end of the dissolution period and the time of t, respectively; EaX—Activation energy of the chemical reaction for the clinker X. 表 5
$K(\xi ,t)$ 函数拟合结果Table 5. Fitting results of
$K(\xi ,t)$ Number $\xi /{\rm{wt}}\% $ Fitted values R-square ${K_0}$ ${A_1}$ $a$ ${A_2}$ $b$ K-0.00 0.00 1.647 1.450 0.594 0.869 0.001 0.9625 K-0.01 0.01 1.296 1.082 0.459 0.648 0.003 0.9758 K-0.02 0.02 1.247 1.504 0.789 0.565 0.005 0.9600 K-0.03 0.03 1.419 1.248 0.559 0.697 0.002 0.9710 K-0.04 0.04 1.423 1.340 0.813 0.791 0.002 0.9102 K-0.05 0.05 1.436 1.386 0.809 0.811 0.002 0.9541 Notes: ξ—Mass fraction of GO; K0, A1, a, A2 and b—Parameters of $K(\xi ,t)$. -
[1] WHITING D A, DETWILER R J, LAGERGREN E S. Cracking tendency and drying shrinkage of silica fume concrete for bridge deck applications[J]. Materials Journal,2000,97(1):71-77. [2] 周文芳. 复合水泥净浆化学收缩影响因素研究[D]. 长沙: 长沙理工大学, 2013.ZHOU Wenfang. Research on influencing factors of chemical shrinkage of composite cement paste[D]. Changsha: Changsha University of Science and Technology, 2013(in Chinese). [3] BELKOWITZ J S, BELKOWITZ W L B, MOSER R D, et al. The influence of nano silica size and surface area on phase development, chemical shrinkage and compressive strength of cement composites[J]. //Nanotechnology in Construction: Proceedings of NICOM5, Berlin: Springer, 2015: 207-212. [4] 陈瑜, 邓怡帆, 钱益想. 掺无机纳米矿粉水泥复合净浆的化学收缩与自收缩[J]. 硅酸盐通报, 2016, 35(9):2710-2716.CHEN Yu, DENG Yifan, QIAN Yixiang. Chemical and autogenous shrinkages of cement-based composite paste with the addition of inorganic nano-powders[J]. Bulletin of the Chinese Ceramic Society,2016,35(9):2710-2716(in Chinese). [5] 何静涛, 陈瑜, 邹成. 掺纳米颗粒水泥净浆化学收缩试验研究[J]. 交通科技与经济, 2017, 19(6):65-70.HE Jingtao, CHEN Yu, ZOU Cheng. Experimental study on chemical shrinkage of cement paste withnano-particles[J]. Technology & Economy In Areas of Communications,2017,19(6):65-70(in Chinese). [6] ZHOU J, ZHENG K, LIU Z, et al. Chemical effect of nano-alumina on early-age hydration of Portland cement[J]. Cement and Concrete Research,2019,116:159-167. doi: 10.1016/j.cemconres.2018.11.007 [7] WANG J, CHENG Y, YUAN L, et al. Effect of nano-silica on chemical and volume shrinkage of cement-based composites[J]. Construction and Building Materials,2020,247:118529. doi: 10.1016/j.conbuildmat.2020.118529 [8] PENG H, GE Y, CAI C S, et al. Mechanical properties and microstructure of graphene oxide cement-based compo-sites[J]. Construction and Building Materials,2019,194:102-109. doi: 10.1016/j.conbuildmat.2018.10.234 [9] LV S, MA Y, QIU C, et al. Effect of graphene oxide nanosheets of microstructure and mechanical properties of cement composites[J]. Construction and Building Materials,2013,49:121-127. doi: 10.1016/j.conbuildmat.2013.08.022 [10] LI W, LI X, CHEN S J, et al. Effects of graphene oxide on early-age hydration and electrical resistivity of Portland cement paste[J]. Construction and Building Materials,2017,136:506-514. doi: 10.1016/j.conbuildmat.2017.01.066 [11] YANG H, MONASTERIO M, CUI H, et al. Experimental study of the effects of graphene oxide on microstructure and properties of cement paste composite[J]. Composites Part A: Applied Science and Manufacturing,2017,102:263-272. doi: 10.1016/j.compositesa.2017.07.022 [12] LI W, LI X, CHEN S J, et al. Effects of nanoalumina and graphene oxide on early-age hydration and mechanical properties of cement paste[J]. Journal of Materials in Civil Engineering,2017,29(9):04017087. doi: 10.1061/(ASCE)MT.1943-5533.0001926 [13] GONG K, PAN Z, KORAYEM A H, et al. Reinforcing effects of graphene oxide on portland cement paste[J]. Journal of Materials in Civil Engineering,2015,27(2):A4014010. [14] HORSZCZARUK E, MIJOWSKA E, KALENCZUK R J, et al. Nanocomposite of cement/graphene oxide–Impact on hydration kinetics and Young’s modulus[J]. Construction and Building Materials,2015,78:234-242. doi: 10.1016/j.conbuildmat.2014.12.009 [15] 王琴, 李时雨, 王健, 等. 氧化石墨烯对水泥水化进程及其主要水化产物的影响[J]. 硅酸盐学报, 2018(2):163-172.WANG Qin, LI Shiyu, WANG Jian, et al. Effect of graphene oxide on hydration process and main hydration products of cement[J]. Journal of the American Ceramic Society,2018(2):163-172(in Chinese). [16] PAN Z, HE L, QIU L, et al. Mechanical properties and microstructure of a graphene oxide-cement composite[J]. Cement and Concrete Composites,2015,58:140-147. doi: 10.1016/j.cemconcomp.2015.02.001 [17] LV S, LIU J, SUN T, et al. Effect of GO nanosheets on shapes of cement hydration crystals and their formation process[J]. Construction and Building Materials,2014,64:231-239. doi: 10.1016/j.conbuildmat.2014.04.061 [18] LI X, KORAYEM A H, LI C, et al. Incorporation of graphene oxide and silica fume into cement paste: A study of dispersion and compressive strength[J]. Construction and Building Materials,2016,123:327-335. doi: 10.1016/j.conbuildmat.2016.07.022 [19] American Society for Testing and Materials. Standard test method for chemical shrinkage of hydraulic cement paste: ASTM C1608—17[S]. West Conshohocken, United States: American Society for Testing and Materials, 2017. [20] 严捍东. 废渣特性及其多元复合对水泥基材料高性能的贡献与机理[D]. 南京: 东南大学, 2001.YAN Handong. Contributions and mechanisms of waste slags characteristics and their multiple combination on high performance of cement basted materials[D]. Nanjing: Southeast University, 2001(in Chinese). [21] 吕生华, 刘晶晶, 邱超超, 等. 纳米氧化石墨烯增强增韧水泥基复合材料的微观结构及作用机理[J]. 功能材料, 2014(4):4084-4089. doi: 10.3969/j.issn.1001-9731.2014.04.018LV Shenghua, LIU Jingjing, QIU Chaochao, et al. Microstructure and mechanism of reinforced and toughened cement composites by nanographene oxide[J]. Journal of Function Material,2014(4):4084-4089(in Chinese). doi: 10.3969/j.issn.1001-9731.2014.04.018 [22] 沈洋. 硅灰对硬化水泥浆体早期收缩的影响[J]. 建筑材料学报, 2002, 5(4):375-378. doi: 10.3969/j.issn.1007-9629.2002.04.014SHEN Yang. Effect of the silica fume on the early shrinkage of hardened cement paste[J]. Journal of Build Materials,2002,5(4):375-378(in Chinese). doi: 10.3969/j.issn.1007-9629.2002.04.014 [23] 吴浪, 宋固全, 王信刚. 基于各相矿物组成含量的水泥早龄期化学收缩预测研究[J]. 混凝土, 2010(10):84-86.WU Lang, SONG Guquan, WANG Xin’gang. Cement chemical shrinkage prediction model research based on mineral composition content of each phase at early age[J]. Concrete,2010(10):84-86(in Chinese). [24] 肖开涛, 杨华全, 董芸. 水泥的化学收缩研究[J]. 长江科学院院报, 2008, 25(1):73-75.XIAO Kaitao, YANG Huaquan, DONG Yun. Study on chemical shrinkage of cement[J]. Journal of Yangtze River Scientific Research Institute,2008,25(1):73-75(in Chinese). [25] POWERS T C, BROWNYARD T L. Studies of the physical properties of hardened portland cement paste[J], Journal Proceedings, Bulletin 22, Research Laboratories of the Portland Cement Association, 1946, 43(9): 469-504. [26] BERNARD O, ULM F J, LEMARCHAND E. A multiscale micromechanics-hydration model for the early-age elastic properties of cement-based materials[J]. Cement and Concrete Research,2003,33(9):1293-1309. doi: 10.1016/S0008-8846(03)00039-5 [27] BERLINER R, POPOVICI M, HERWIG K W, et al. Quasielastic neutron scattering study of the effect of water-to-cement ratio on the hydration kinetics of tricalcium silicate[J]. Cement and Concrete Research,1998,28(2):231-243. doi: 10.1016/S0008-8846(97)00260-3 -








 下载:
下载: