ZIF-8-SiO2 and adsorption on U(VI)
-
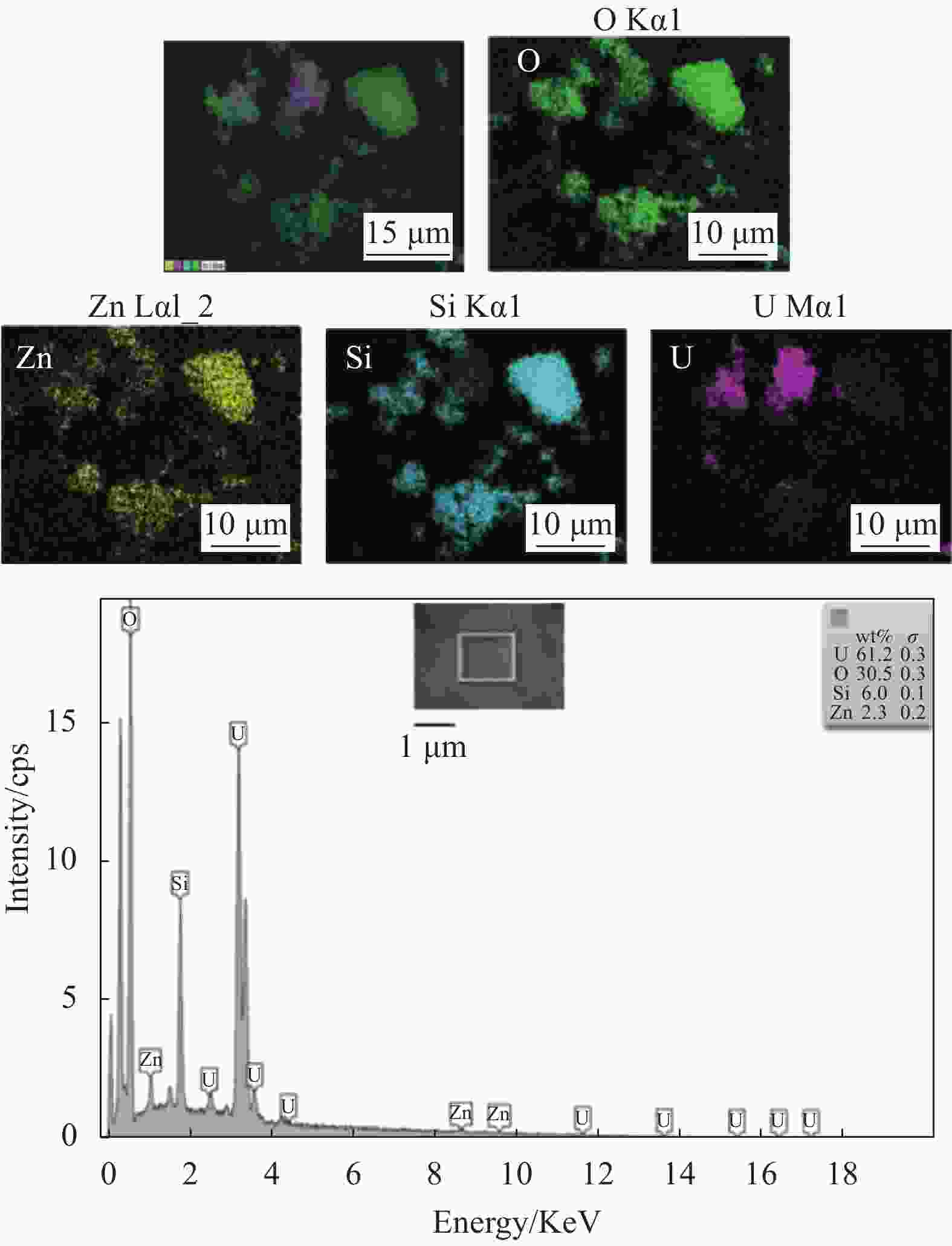
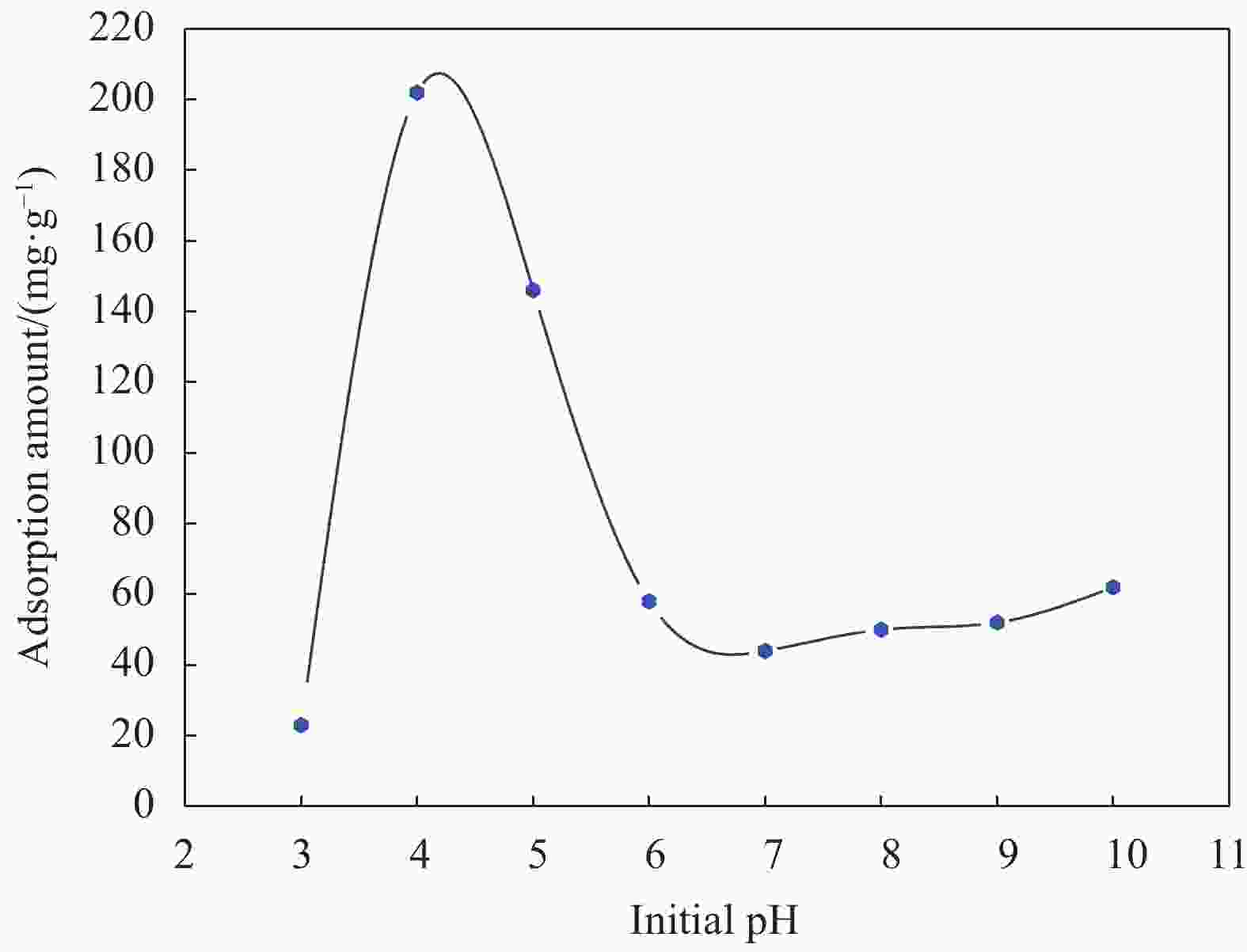
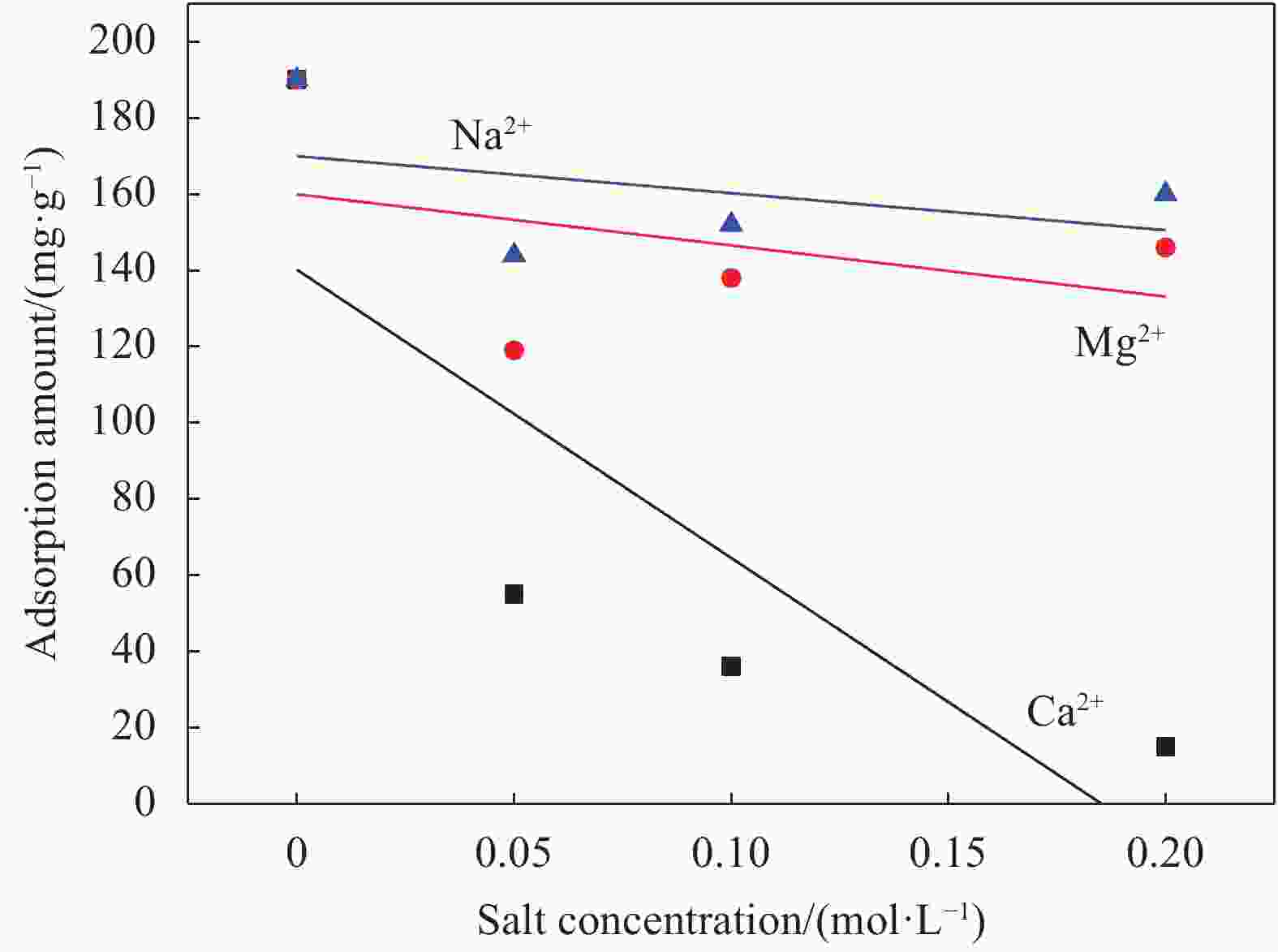
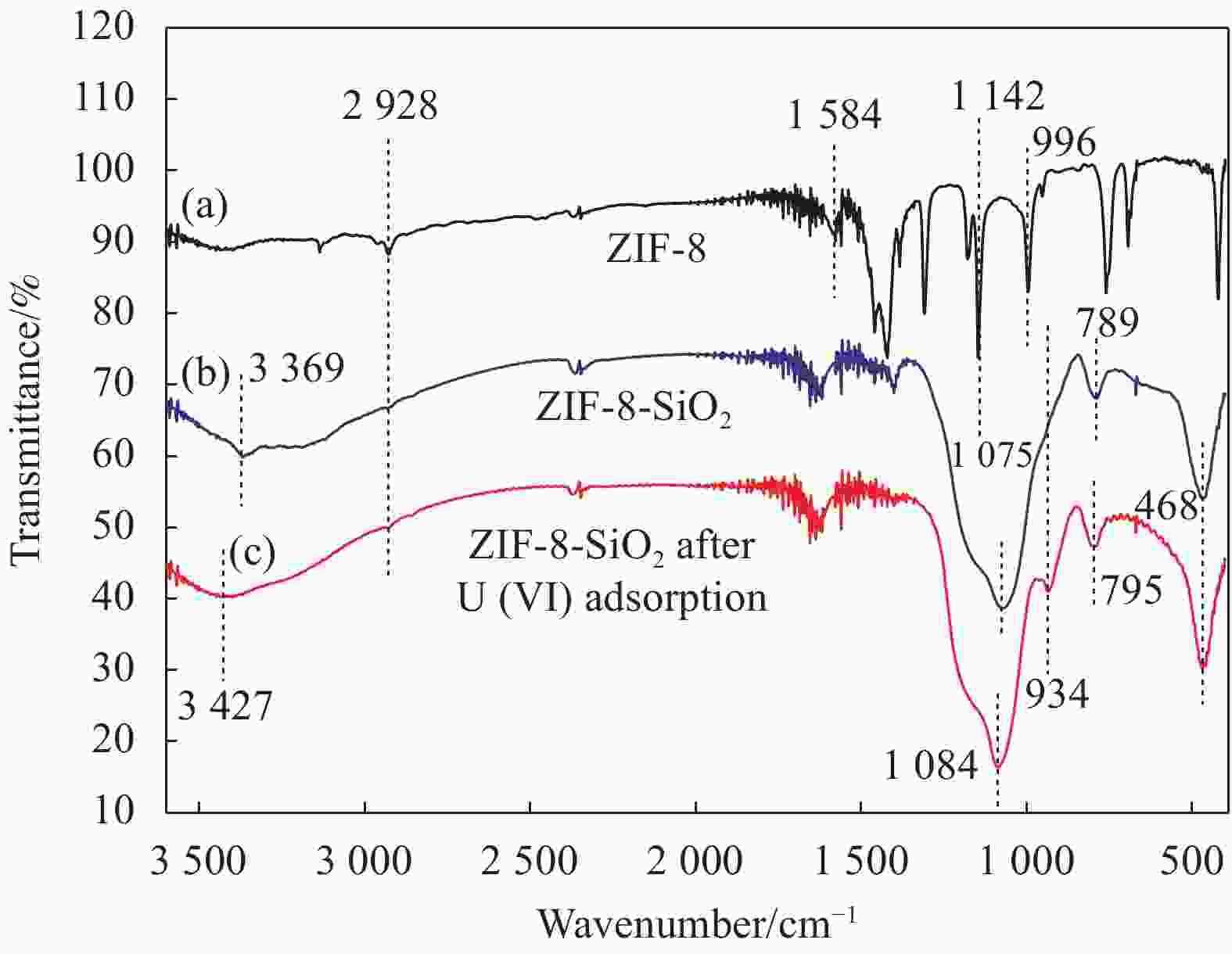
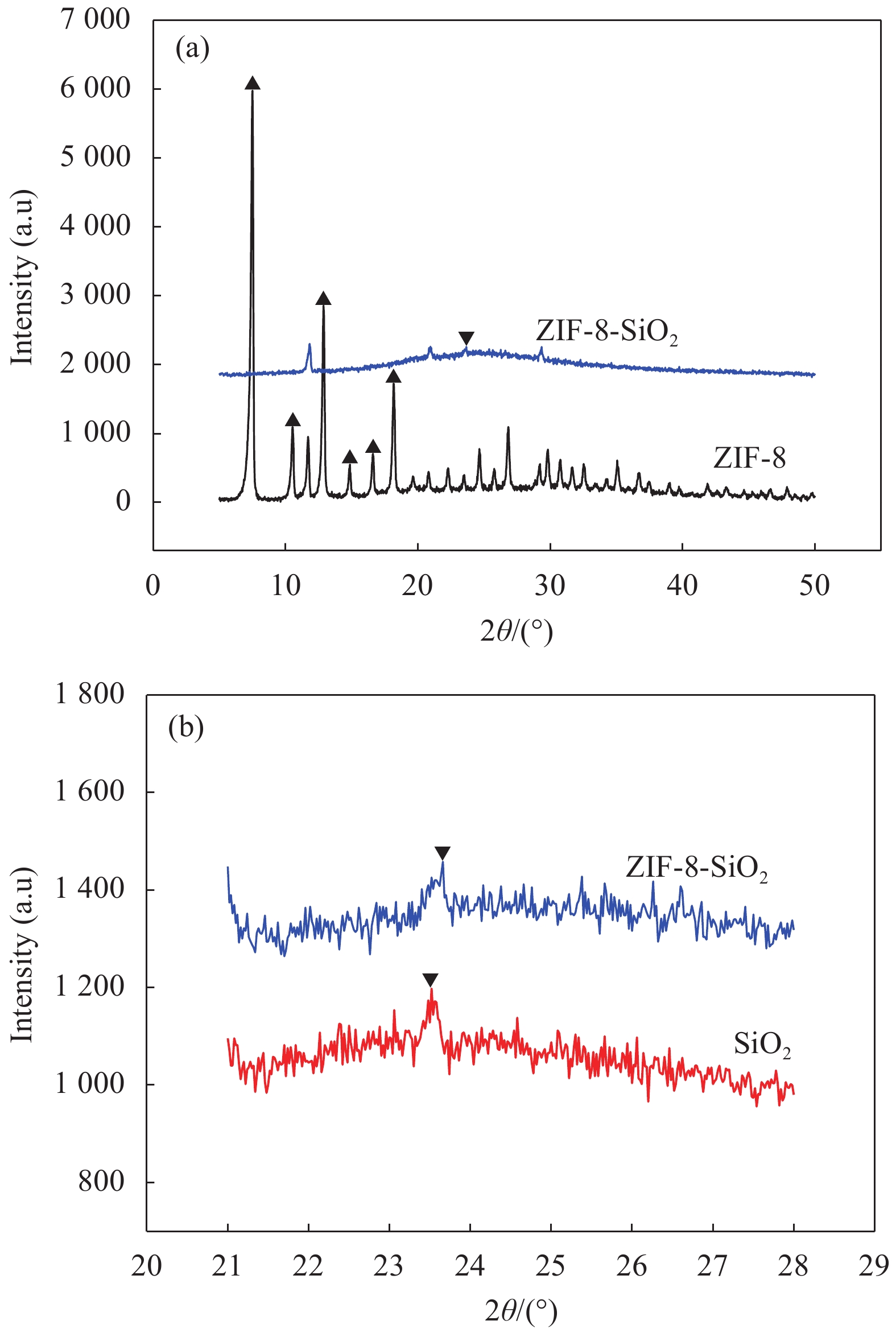
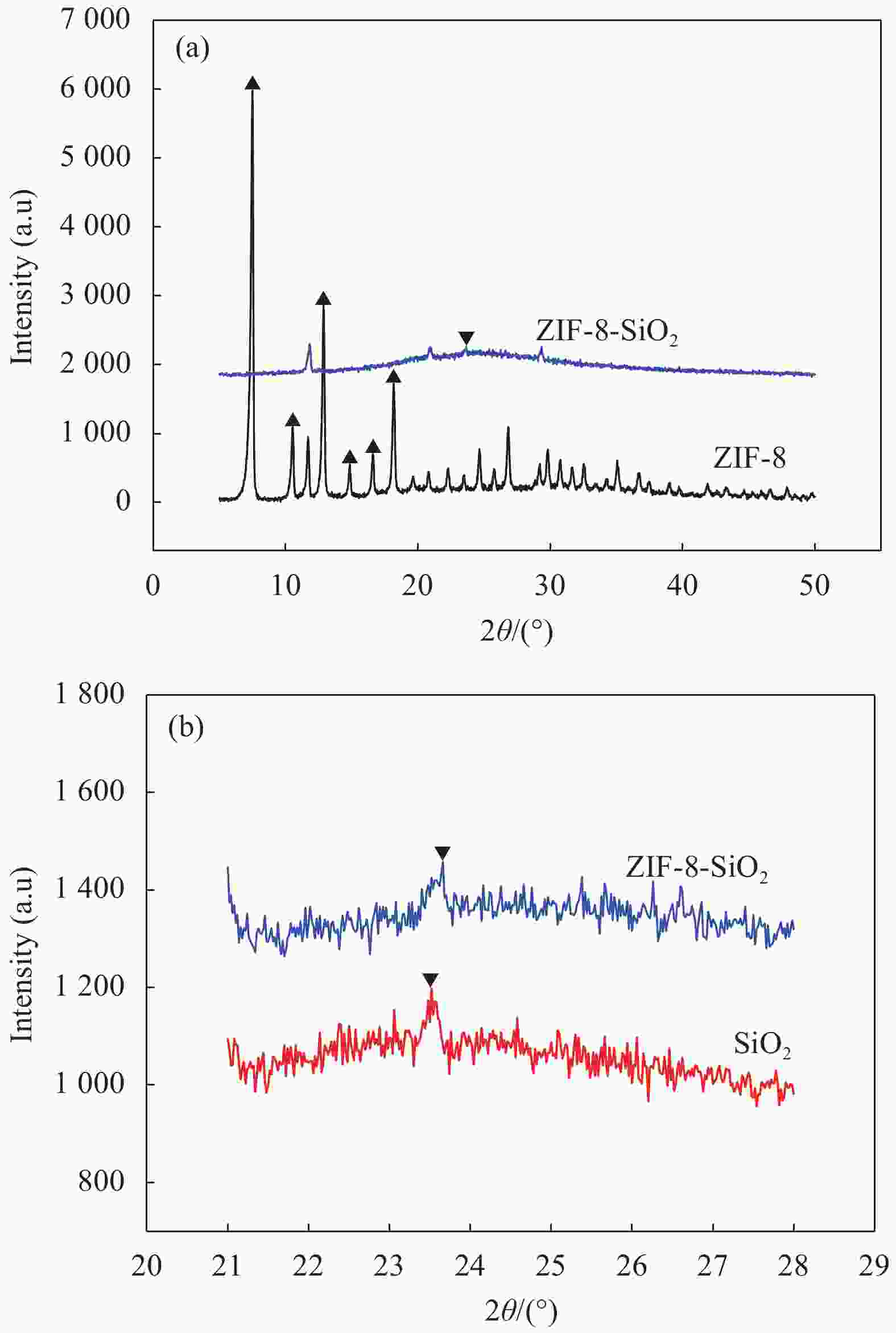
摘要: 以沸石咪唑酯骨架结构材料ZIF-8及硅酸四乙酯(TEOS)为原料制备ZIF-8-SiO2复合材料,并采用XRD、SEM、EDS等方法对ZIF-8-SiO2的结构及吸附U(VI)前后的形貌进行表征,结果表明ZIF-8-SiO2成功制备且对U(VI)具有良好的吸附作用。以静态吸附实验,分别考察了pH值、时间、温度、溶液初始铀浓度、盐浓度及超高压环境等对ZIF-8-SiO2吸附性能的影响。由实验结果可得,在初始浓度为80 mg·L−1时,25℃下ZIF-8-SiO2对U(VI)的最大实际吸附量为498 mg·g−1,根据Langmuir模型拟合结果分析得出,ZIF-8-SiO2对U(VI)的理论吸附量最高可达678.5 mg·g−1,且在200~500 MPa范围内,压强越高越有利于吸附。通过FTIR、XPS等方法对ZIF-8-SiO2吸附铀酰离子前后的结构进行分析,探究该吸附过程中可能存在的吸附机制。
-
关键词:
- ZIF-8-SiO2 /
- 复合材料 /
- 铀处理 /
- 吸附法 /
- 高压模拟
Abstract: Zeolite imidazole ester framework structure material ZIF-8 and TEOS were used to prepare ZIF-8-SiO2 composite, the structure of ZIF-8-SiO2 and the morphology before and after adsorption of U(VI) were characterized by XRD, SEM, EDS. And the results show that ZIF-8-SiO2 is successfully prepared and has a good adsorption effect on U(VI). The effects of pH value, time, temperature, initial uranium concentration and salt concentration on the adsorption performance of ZIF-8-SiO2 were investigated by static adsorption experiments. According to the experimental results, when the initial concentration is 80 mg·g−1, the maximum actual adsorption capacity of ZIF-8-SiO2 on U(VI) at 25℃ is 498 mg·g−1, and according to the analysis of Langmuir model fitting results, the theoretical adsorption capacity of the material on U(VI) is up to 678.5 mg·g−1. And in the range of 200~500 MPa, the higher the pressure, the better the adsorption. FTIR and XPS were used to characterize the structure of the material before and after uranylion adsorption, to explore the possible adsorption mechanism during the adsorption process.-
Key words:
- ZIF-8-SiO2 /
- composite materials /
- uranium processing /
- adsorption method /
- high pressure simulation
-
表 1 25℃下 ZIF-8-SiO2对U(VI)吸附的Langmuir及Freundlich模型拟合参数
Table 1. Langmuir and Freundlich model parameters for adsorption of U(VI) by ZIF-8-SiO2 at 25℃
Sample parameters ZIF-8-SiO2 Langmuir Cs,max/(mg·g−1) 678.5 b/(L·mg−1) 0.01232 R2 0.9889 Freundlich KF/(mg·g−1) 20.63 1/n 0.6267 R2 0.9700 Notes: Cs,max—Maximum adsorption capacity of ZIF-8-SiO2 for U(VI); b—Fitting parameters of Langmuir adsorption model; KF—Fitting parameters of Freundlich adsorption model; n—Linear factor or a potential energy nonuniformity factor, reflecting the heterogeneous energy of the adsorption surface; R2—Coefficient of association. 表 2 ZIF-8-SiO2对U(VI)吸附动力学拟合相关参数
Table 2. Pseudo-first and pseudo second order kinetics parameters for U(VI) removal by ZIF-8-SiO2
Parameter 25℃ 45℃ Pseudo-first order K1/(L·mg−1) 0.01039 0.03427 R2 0.9849 0.8681 Pseudo-second order K2/(g·min−1·mg−1) 5.847×10−5 2.311×10−4 R2 0.9954 0.9519 Notes: K1, K2—Adsorption rate constant; R2—Coefficient of association. 表 3 ZIF-8-SiO2对U(VI)吸附的热力学参数
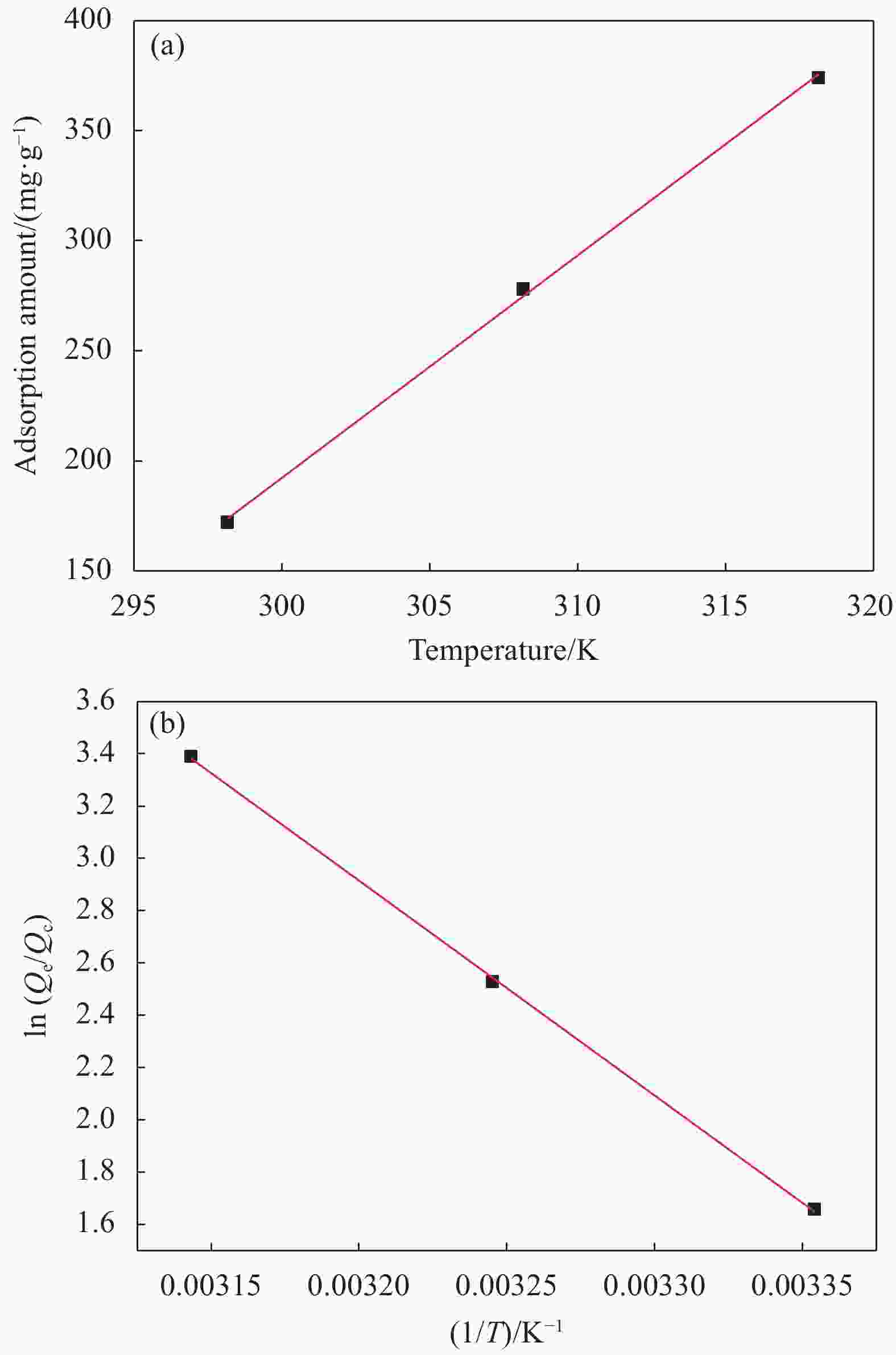
Table 3. Thermodynamic parameters of adsorption of ZIF-8-SiO2 on U(VI)
ZIF-8-SiO2 ΔH/(kJ·mol−1) 68.33 ΔS/(J·mol−1·K−1) 242.9 ΔG/(kJ·mol−1),298.15 K −4.088 ΔG/(kJ·mol−1),308.15 K −6.517 ΔG/(kJ·mol−1),318.15 K −8.946 Ea/(kJ·mol−1) 54.19 Notes: ΔH—Standard enthalpy change; ΔS—Standard entropy change; ΔG—Gibbs free energy change; Ea—Activation energy of adsorption. -
[1] LINDNER H, SCHNEIDER E. Review of cost estimates for uranium recovery from seawater[J]. Energy Economics,2015,49:9-22. doi: 10.1016/j.eneco.2015.01.016 [2] 王国华, 杨思芹, 周耀辉, 等. 生物还原法修复铀污染地下水的研究进展[J]. 环境科学与技术, 2019, 42(8):47-53.WANG G H, YANG S Q, ZHOU Y H, et al. Research progress on the bioremediation of groundwater polluted by uranium via bio-reduction[J]. Environmental Science & Technology,2019,42(8):47-53(in Chinese). [3] 敖浚轩, 徐晓, 李玉娜, 等. 海水提铀研究进展[J]. 辐射研究与辐射工艺学报, 2019, 37(2):3-28.AO J X, XU X, LI Y N, et al. Research progress in uranium extraction from seawater[J]. Journal of Radiation Research and Radiation Processing,2019,37(2):3-28(in Chinese). [4] WOJCIECHOWSKI A. The U-232 in thorium cycle[J]. Progress in Nuclear Energy,2018,106:204-214. doi: 10.1016/j.pnucene.2018.03.011 [5] 王双, 冉永红, 李娟, 等. 铀的生殖毒性效应及作用机制研究进展[J]. 局解手术学杂志, 2019, 28(1):86-90.WANG S, RAN Y H, LI J, et al. Advances in reproductive toxicity of uranium and its mechanism[J]. Journal of Regional Anatomy and Operative Surgery,2019,28(1):86-90(in Chinese). [6] CAO H, CHEN J, ZHANG J, et al. Heavy metals in rice and garden vegetables and their potential health risks to inhabitants in the vicinity of an industrial zone in Jiangsu, China[J]. Journal of Environmental Sciences-china,2010,22(11):1792-1799. doi: 10.1016/S1001-0742(09)60321-1 [7] HARIBALA, HU B, WANG C, et al. Assessment of radioactive materials and heavy metals in the surface soil around uranium mining area of Tongliao, China[J]. Ecotoxicology and Environmental Safety,2016,130:185-192. doi: 10.1016/j.ecoenv.2016.04.002 [8] 魏广芝, 徐乐昌. 低浓度含铀废水的处理技术及其研究进展[J]. 铀矿冶, 2007, 26(2):90-95. doi: 10.3969/j.issn.1000-8063.2007.02.007WEI G Z, XU L C. Treatment technology of low concentration uranium-bearing wastewater and its research progress[J]. Uranium Mining and Metallurgy,2007,26(2):90-95(in Chinese). doi: 10.3969/j.issn.1000-8063.2007.02.007 [9] FENG M L, SARMA D, QI X H, et al. Efficient removal and recovery of uranium by a layered organic-inorganic hybrid thiostannate[J]. Journal of the American Chemical Society,2016,138(38):12578-12585. doi: 10.1021/jacs.6b07351 [10] WANG Y S, CHEN Y T, LIU C, et al. Preparation of porous magnesium oxide foam and study on its enrichment of uranium[J]. Journal of Nuclear Materials,2018,504:166-175. doi: 10.1016/j.jnucmat.2018.03.041 [11] 宋莉芳, 夏慧芸, 陈华鑫, 等. 中孔金属有机骨架材料的制备与应用[J]. 化学进展, 2014, 26(7):1131-1142.SONG L F, XIA H Y, CHEN H X, et al. Preparation and application of mesoporous metal-organic frameworks[J]. Progress in chemistry,2014,26(7):1131-1142(in Chinese). [12] WANG C, LIU X, KESER DEMIR N, et al. Applications of water stable metal-organic frameworks[J]. Chemical Society Reviews,2016,45(18):5107-5134. doi: 10.1039/C6CS00362A [13] WU Y H, PANG H W, YAO W, et al. Synthesis of rod-like metal-organic framework (MOF-5) nanomaterial for efficient removal of U(VI): batch experiments and spectroscopy study[J]. Science Bulletin,2018,63(13):831-839. doi: 10.1016/j.scib.2018.05.021 [14] ZHANG Y, CHEN R, LIU Q, et al. Polypyrrole modified FeO-loaded graphene oxide for enrichment of uranium(VI) from simulated seawater[J]. Dalton Transactions,2018,47:12984-12992. doi: 10.1039/C8DT02819B [15] LIU F T, SONG S S, CHENG G, et al. MIL-101(Cr) metal-organic framework functionalized with tetraethylenepentamine for potential removal of Uranium (VI) from waste water[J]. Adsorption Science and Technology,2018,36(7-8):1550-1567. doi: 10.1177/0263617418789516 [16] 张晋维, 李平, 张馨凝, 等. 水稳定性金属有机框架材料的水吸附性质与应用[J]. 化学学报, 2020, 78(7):597-612. doi: 10.6023/A20050153ZHANG J W, LI P, ZHANG X N, et al. Water adsorption properties and applications of stable metal-organic frameworks[J]. Acta chimca sinica,2020,78(7):597-612(in Chinese). doi: 10.6023/A20050153 [17] 张蕾蕾, 唐安琪, 王章慧, 等. 以ZIF-8为模板制备聚多巴胺/聚乙二醇复合纳米胶囊[J]. 功能高分子学报, 2018, 31(6):546-552.ZHANG L L, TANG A Q, WANG Z H, et al. Preparation of polydopamine/poly (ethylene glycol) composite nanocapsules with ZIF-8 nanoparticles as templates[J]. Journal of Functional Polymers,2018,31(6):546-552(in Chinese). [18] XU X, WANG X, LIU M, et al. ZIF/SiO2 core-shell microsphere extraction coupled with LC–MS/MS for the quantita-tive analysis of four plant growth regulators in navel oranges[J]. Journal of Separation Science,2018,41(18). doi: 10.1002/jssc.201800286 [19] HAMEED B H, TAN I A W, AHMAD A L. Adsorption isotherm, kinetic modeling and mechanism of 2, 4, 6-trichlorophenol on coconut husk-based activated carbon[J]. Chemical Engineering Journal,2008,144(2):235-244. doi: 10.1016/j.cej.2008.01.028 [20] WU C, LIU Q, CHEN R, et al. Fabrication of ZIF-8@SiO2 micro/nano hierarchical superhydrophobic surface on AZ31 magnesium alloy with impressive corrosion resistance and abrasion resistance[J]. ACS Appllied Mater Interfaces,2017,9(12):11106-11115. doi: 10.1021/acsami.6b16848 [21] 王彦惠, 成建峰, 赵玉婷, 等. Fe3O4/SiO2-NH2磁性复合纳米材料对铀(VI)的吸附性能[J]. 环境化学, 2019, 38(9):2149-2158. doi: 10.7524/j.issn.0254-6108.2019011003WANG Y H, CHENG J F, ZHAO Y T, et al. Adsorption of Fe3O4@SiO2-NH2 magnetic composite nanomaterials on Uranium (VI)[J]. Environmental Chemistry,2019,38(9):2149-2158(in Chinese). doi: 10.7524/j.issn.0254-6108.2019011003 [22] LIU R, ZHANG W, CHEN Y T, et al. Uranium (VI) adsorption by copper and copper/iron bimetallic central MOFs[J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects,2020,587:124334. doi: 10.1016/j.colsurfa.2019.124334 [23] 许成, 张炜, 陈元涛, 等. UiO-66/UiO-66-PYDC的制备及其对UO22+吸附性研究[J]. 环境科学学报, 2018, 38(11):4350-4359.XU C, ZHANG W, CHEN Y T, et al. Preparation of UiO-66/UiO-66-PYDC and their absorptive properties on uranyl ions[J]. Acta Scientiae Circumstantiae,2018,38(11):4350-4359(in Chinese). [24] 王芳, 张辉, 戴仲然, 等. 磁性核壳CoFe2O4@SiO2@PIL-AO复合材料的制备及其吸附U(VI)性能研究[J]. 环境科学学报, 2018, 38(9):3605-3613.WANG F, ZHANG H, DAI Z R, et al. Preparation of magnetic yolk/shell CoFe2O4@SiO2@PIL-AO composite material and its adsorption behavior for U(VI)[J]. Acta Scientiae Circumstantiae,2018,38(9):3605-3613(in Chinese). [25] 林晨曦, 冯洋, 李晴, 等. 动力学、热力学和活化能在花生壳吸附铅离子实验中的应用[J]. 化学教育, 2020, 41(8):53-57.LIN C X, FENG Y, LI Q, et al. Application of kinetics, therodynamics and activation energy in adsorption of Pb2+ by peanut shell[J]. Chinese Journal of Chemical Education,2020,41(8):53-57(in Chinese). [26] 宋艳晖, 钱春园, 张托弟, 等. 三维凹凸棒石-氧化石墨烯的制备及其吸附性能[J]. 非金属矿, 2020, 43(2):9-12. doi: 10.3969/j.issn.1000-8098.2020.02.003SONG Y H, QIAN C Y, ZHANG T D, et al. Preparation and adsorption properties of 3D attapulgite-graphene oxide[J]. Non-Metallic Mines,2020,43(2):9-12(in Chinese). doi: 10.3969/j.issn.1000-8098.2020.02.003 [27] KUMAR K S, DAHMS H, WON E, et al. Microalgae-A promising tool for heavy metal remediation[J]. Ecotoxicology and Environmental Safety,2015:329-352. [28] ZHU L, SUN Y L, SONG L J, et al. Dihydroxy bezladely derivatives functionalized mesoporous silica SBA-15 for the sorption of U(VI)[J]. Journal of Radioanalytical and Nuclear Chemistry,2016,310:125-137. doi: 10.1007/s10967-016-4779-4 -





 下载:
下载: