Size effect of nano SiO2 microspheres in PMMA gel polymer electrolyte and its application in all-solid-state electrochromic devices
-
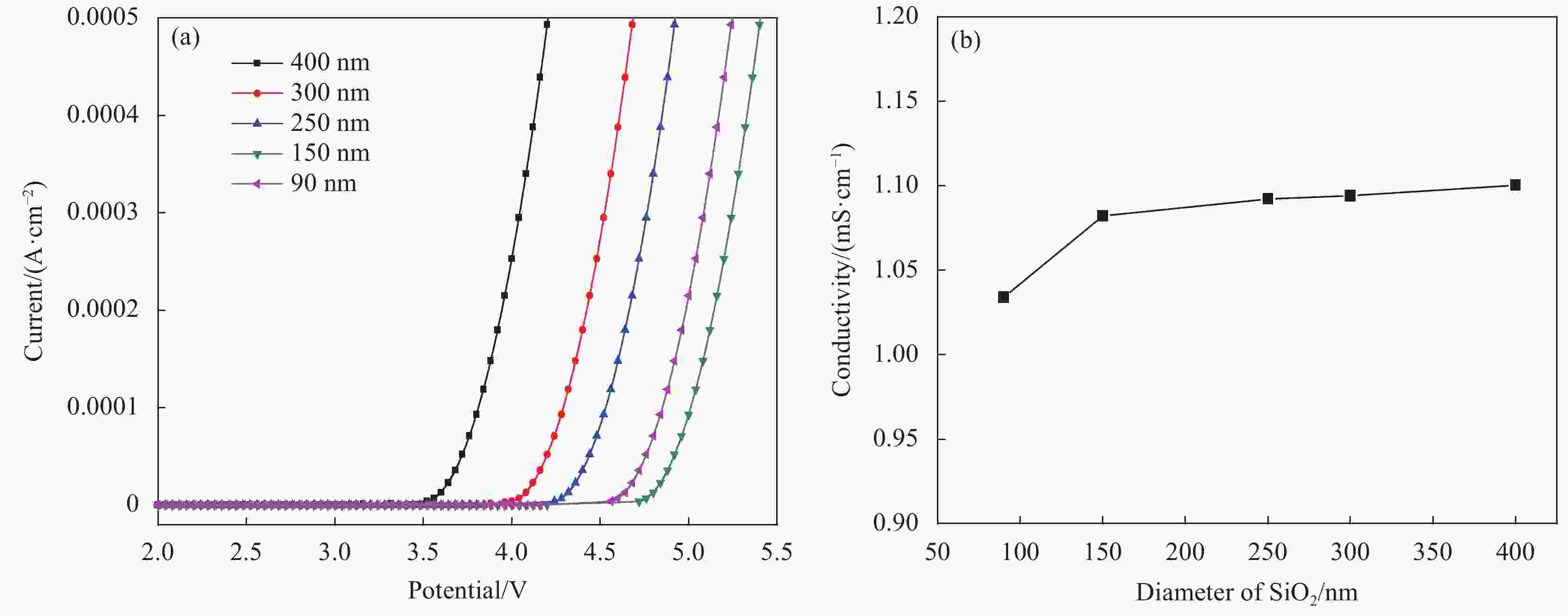
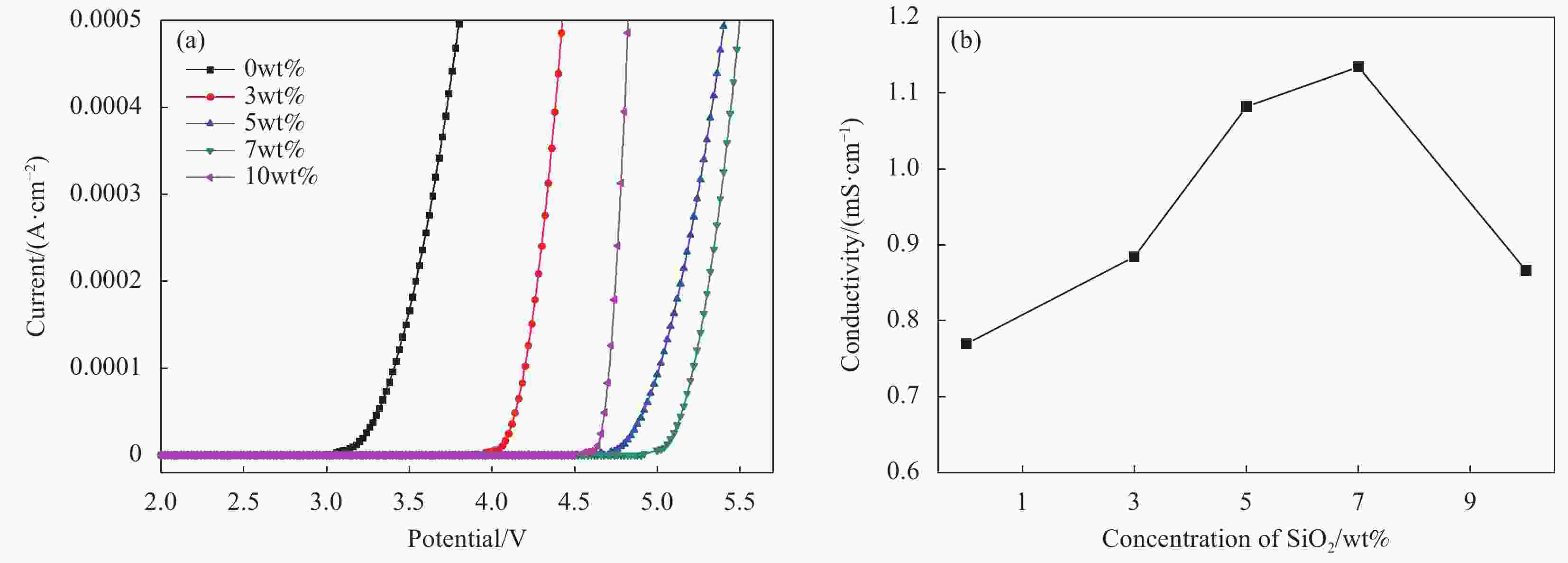
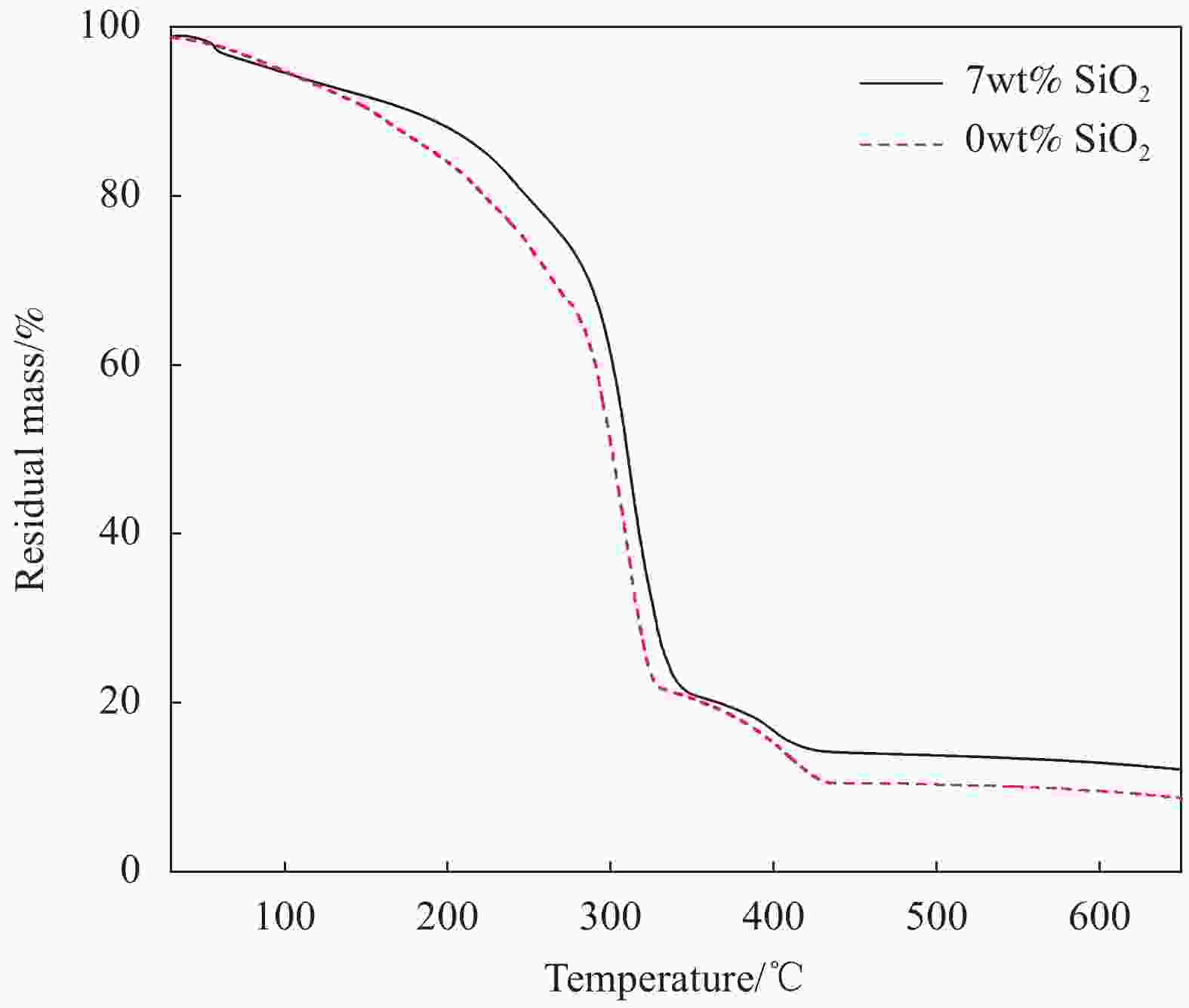
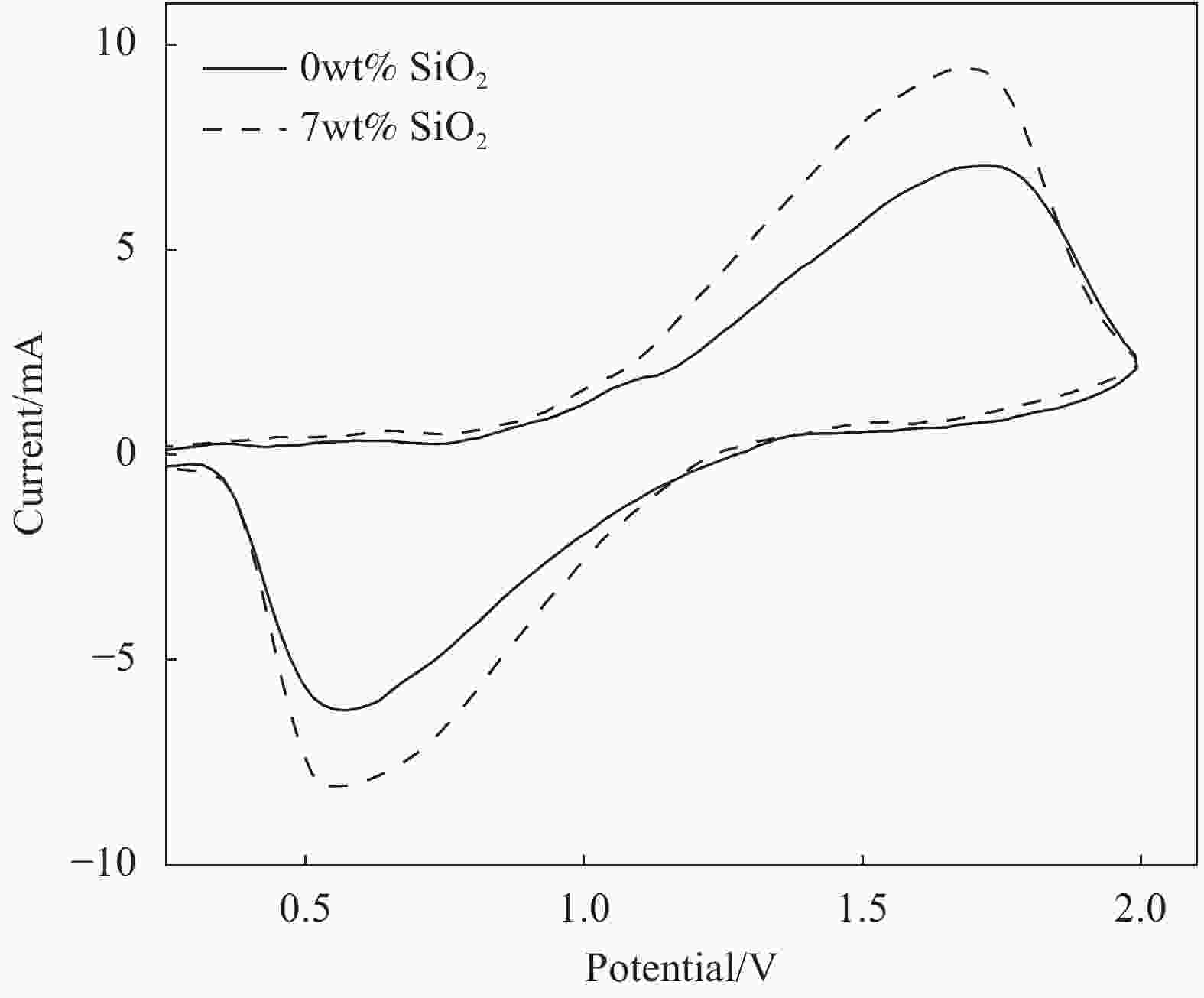
摘要: 本文采用Stöber法制备了不同粒径纳米SiO2微球,将其加入到聚甲基丙烯酸甲酯(PMMA)基凝胶聚合物电解质中,研究了纳米SiO2的尺寸效应及其对全固态电致变色器件性能的影响。研究发现,正硅酸乙酯和乙醇的体积比影响所得纳米SiO2微球粒径大小,乙醇所占体积比例越大,制备的纳米SiO2微球粒径越小。在PMMA基凝胶聚合物电解质中,当纳米SiO2微球用量相同时,电解质的电化学窗口随着纳米SiO2微球粒径的减小先增大后减小,离子电导率随纳米SiO2微球粒径的增大而增大,在纳米SiO2微球粒径为150 nm时电化学窗口达到最大,离子电导率的增速也变得不明显。当粒径为150 nm的纳米SiO2微球含量为7wt%时,PMMA凝胶电解质的电化学窗口为4.8 V,电导率为1.13 mS/cm。以该电解质组装结构为:导电玻璃(ITO)‖SiO2/碳酸丙烯脂(PC)-LiClO4/PMMA‖金属-超分子聚合物‖ITO的透射式全固态电致变色器件,所得器件可在淡绿色和深蓝色之间变色,对比度高达60.1%,且器件的稳定性得以明显提高。Abstract: In this paper, nano SiO2 microspheres with different particle sizes were prepared by the Stöber method, which were added to polymethyl methacrylate (PMMA)-based gel polymer electrolytes, and the size effect of nano SiO2 microspheres and its effect on the performance of all solid-state electrochromic devices were studied. The results show that the volume ratio of ethyl orthosilicate and ethanol affects the particle size of the obtained nano SiO2 microspheres. The larger the volume ratio, the smaller the particle size of the prepared nano SiO2 microspheres. In the PMMA-based gel polymer electrolyte, when nano SiO2 microspheres content is the same, the electrochemical window of the electrolyte first increases and then decreases with the decrease of nano SiO2 microspheres particle size, and the ionic conductivity increases with the increase of its particle size. The electrochemical window reached its maximum at 150 nm of nano SiO2 microspheres, where the increase in ionic conductivity became insignificant. Then, 150 nm SiO2 is added to PMMA gel electrolyte with a content of 7wt%, which has the electrochemical window of 4.8 V and the conductivity of 1.13 mS/cm. Besides, the electrolyte was used to assembly transmissive all-solid-state electrochromic device with a structure: Electrically conductive glass (ITO)‖SiO2/propylene carbonate (PC)-LiClO4/PMMA‖metal-supramolecular polymer‖ITO. The obtained device can change color between light green and dark blue with a high contrast of 60.1%, and the device stability is significantly improved.
-
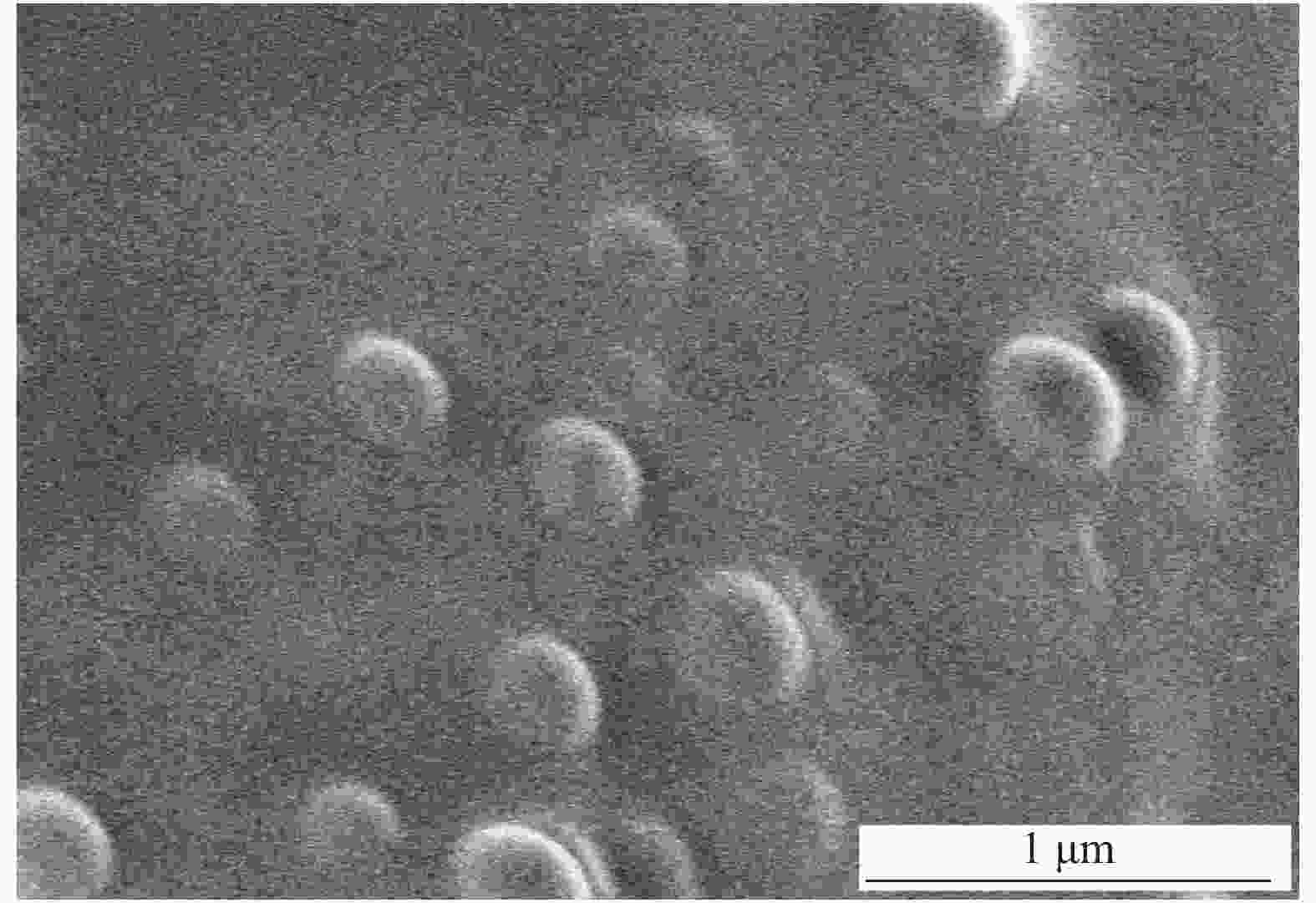
图 1 不同粒径SiO2的SEM图像: (a)正硅酸乙酯∶乙醇=3∶10; (b) 正硅酸乙酯∶乙醇=3∶30; (c) 正硅酸乙酯∶乙醇=3∶50;(d) 正硅酸乙酯∶乙醇=3∶60; (e) 正硅酸乙酯∶乙醇=3∶70 (体积比)
Figure 1. SEM images of SiO2 with different diameters: (a) Ethyl orthosilicate∶ ethanol=3∶10; (b) Ethyl orthosilicate∶ ethanol=3∶30; (c) Ethyl orthosilicate∶ ethanol=3∶50; (d) Ethyl orthosilicate∶ ethanol=3∶60; (e) Ethyl orthosilicate∶ ethanol=3∶70 (Volume ratio)
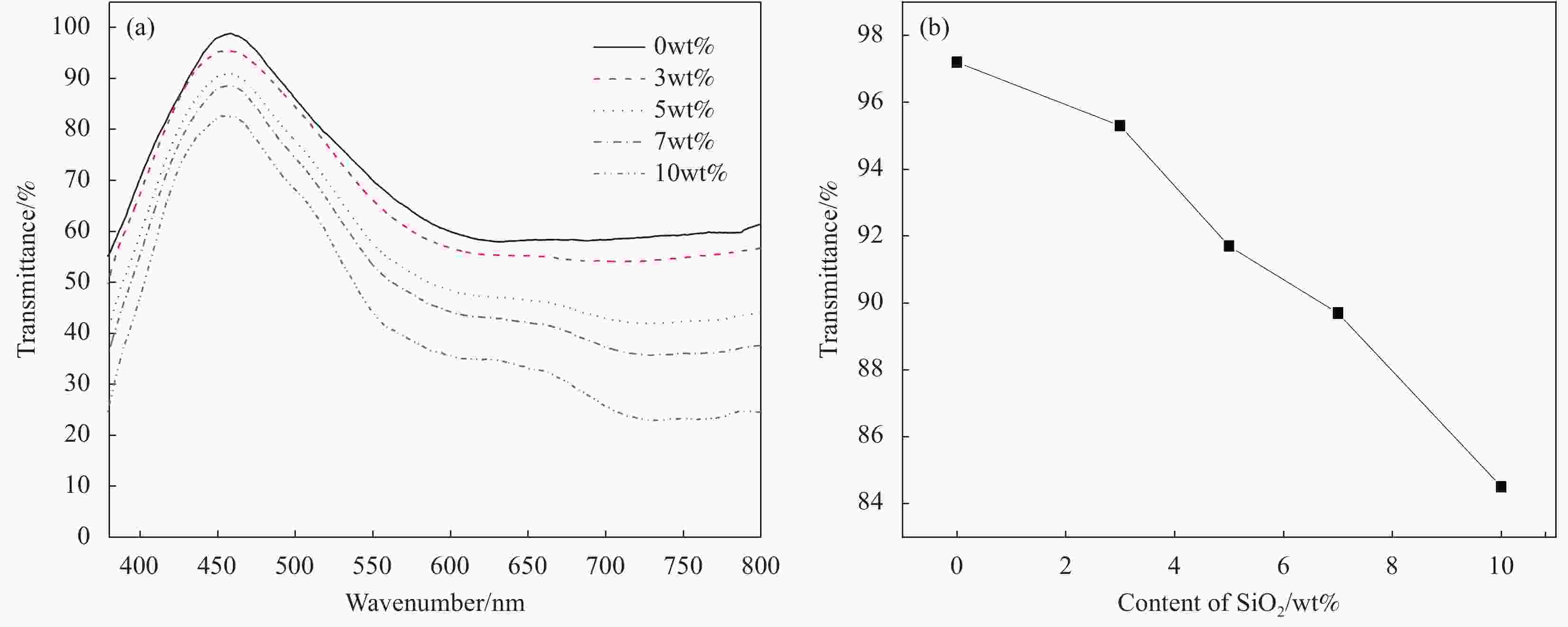
图 5 不同150 nm SiO2含量对SiO2/PMMA复合凝胶聚合物电解质可见光透过率的影响: (a)不同SiO2含量的复合凝胶聚合物电解质的透过率曲线; (b) SiO2含量与最大透过率关系
Figure 5. Effect of 150 nm SiO2 with different contents on visible light transmittance of SiO2/PMMA composite gel polymer electrolytes: (a) Transmittance curves of composite gel polymer electrolyte with different SiO2 contents; (b) Relationship between SiO2 content and maximum transmittance
表 1 乙醇浓度变化与SiO2粒径大小的关系
Table 1. Relationship between variation of reaction conditions and diameter of SiO2
TEOS/mL C2H5OH/mL NH3•H2O/mL H2O/mL Diameter of SiO2/nm 30 100 15 50 400 30 300 15 50 300 30 500 15 50 250 30 600 15 50 150 30 700 15 50 90 Note: TEOS—Ethyl orthosilicate. -
[1] HUANG Q Q, SONG J X, GAO Y, et al. Supremely elastic gel polymer electrolyte enables a reliable electrode structure for silicon-based anodes[J]. Nature Communications,2019,10:1-7. [2] 许晓雄, 邱志军, 官亦标, 等. 全固态锂电池技术的研究现状与展望[J]. 储能科学与技术, 2013, 2(4):331-341. doi: 10.3969/j.issn.2095-4239.2013.04.001XU Xiaoxiong, QIU Zhijun, GUAN Yibiao,et al. Research status and prospects of all solid-state lithium battery technology[J]. Energy Storage Science and Technology,2013,2(4):331-341(in Chinese). doi: 10.3969/j.issn.2095-4239.2013.04.001 [3] CROCE F, APPETECCHI G B, PERSI L, et al. Nanocomposite polymer electrolytes for lithium batteries[J]. Nature,1998,394:456-458. [4] DRIS N H, RAHMAN M M, WANG J Z, et al. Microporous gel polymer electrolytes for lithiumrechargeable batteryappliction[J]. Journal of Power Sources,2012,201:294-300. [5] 赵世勇. 锂离子电池用聚合物凝胶电解质研究进展[J]. 电池工业, 2014, 19(1):35-40. doi: 10.3969/j.issn.1008-7923.2014.01.008ZHAO Shiyong. Research progress of polymer gel electrolytes for lithium ionbatteries[J]. Battery Industry,2014,19(1):35-40(in Chinese). doi: 10.3969/j.issn.1008-7923.2014.01.008 [6] WANG W, ALEXANDRIDIS P. Composite polymer electrolytes: Nanoparticles affect structure and properties[J]. Polymers,2016,8(11):387. [7] NGAI K S, RAMESH S, RAMESH K, et al. A review of polymer electrolytes: Fundamental, approaches and applications[J]. Ionics,2016,22(8):1259-1279. [8] ZHUZ Q, HONG M L, GUO D S, et al. All-solid-state lithium organic battery with composite polymer electrolyte and pillar quinone cathode[J]. Journal of the American Chemical Society,2014,136(47):16461-16464. [9] 樊小伟, 梁小平. 电致变色玻璃用聚合物凝胶电解质改性研究进展[J]. 建材世界, 2017, 38(2):7-9.FAN Xiaowei, LIANG Xiaoping. Research progress of polymer gel electrolyte modification for electrochromic glass[J]. Building Materials World,2017,38(2):7-9(in Chinese). [10] RIBERIRO R, SILVA G G, MOHALLEM N D S. A comparison of ionic conductivity, thermal behavior and morphology in two polymer-LiI-LiAl5O8 composite polymerelectrolytes[J]. Electrochim Acta,2001,46(10-11):1679-1686. [11] PASQUIER A D, WARREN P C, CULVER D, et al. Plastic PVDF-HFP electrolyte laminates prepared by a phase-inversion process[J]. Solid State Ionics,2000,135(1-4):249-257. [12] KIL E H, CHOI K H, HA H J, et al. Imprintable, bendable, and shape-conformable polymer electrolytes for versatile-shaped lithium-ion batteries[J]. Advanced Materials,2013,25(10):1395-1400. [13] 张榜, 李昕, 龚䶮, 等. 金属超分子聚合物基柔性电致变色器件的制备及稳定性探索[J]. 北京服装学院学报, 2018, 38(4):1-8.ZHANG Bang, LI Xin, GONG Yan, LIU Jiguang, PANG Yali, ZHANG Yuqing, LUO Shengli, ZHANG Yuqun. Preparation and stability exploration of metal supramolecular polymer-based flexible electrochromic devices[J]. Journal of Beijing Institute of Fashion Technology,2018,38(4):1-8(in Chinese). [14] STÖBER W, FINK A, BOHN E. Controlled growth of monodisperse silica spheres in the micron size range[J]. Journal of Colloid & Interface Science,1968,26(1):62-69. [15] WU S L, MENG J Y, LI X, et al. Metallo-supramolecular polymer based on bis-2, 2': 6, 2"-terpyridine with high electrochromic performances[C]//IOP Conference Series: Materials Science and Engineering. IOP Publishing, 2018. [16] 赵丽, 余家国, 程蓓, 等. 单分散SiO2球形颗粒的制备与形成机制[J]. 化学学报, 2003, 61(4):562-566. doi: 10.3321/j.issn:0567-7351.2003.04.020ZHAO Li, YU Jiaguo, CHENG Bei, et al. Preparation and formation mechanism of monodispersed spherical silica particles[J]. Acta Chimica Sinica,2003,61(4):562-566(in Chinese). doi: 10.3321/j.issn:0567-7351.2003.04.020 [17] 符远翔, 孙艳辉, 葛杏心. 单分散纳米SiO2的制备与表征[J]. 硅酸盐通报, 2008, 27(1):154-159.FU Yuanxiang, SUN Yanhui, GE Xingxin. Preparation and characterization of monodispersed nano-silica[J]. Bulletin of the Chinese Ceramic Society,2008,27(1):154-159(in Chinese). [18] 颜建民, 张裕卿. 氨基化修饰介孔SiO2用于疏水性药物的负载和释放[J]. 化学工业与工程, 2017, 34(3):43-49.YAN Jianmin, ZHANG Yuqing. Amino-modified mesoporous silica for loading and release of hydrophobic drugs[J]. Chemical Industry and Engineering,2017,34(3):43-49(in Chinese). [19] SOLARAJAN A K, MURUGADOSS V, ANGAIAH S. High performance electrospun PVDF-HFP/SiO2 nanocomposite membrane electrolyte for Li-ion capacitors[J]. Journal of Applied Polymer Science,2017,134(32):45177. -






 下载:
下载: